Tetrahydrofurfurylamine: A Deep Dive
Historical Development
Chemists began paying attention to tetrahydrofurfurylamine in the mid-20th century, right alongside the growing interest in furan derivatives. Back in those days, the food industry searched for new solvents, and the pharmaceutical sector craved gentle building blocks that wouldn’t break down too quickly. Scientists explored furfural’s many pathways; one led to the reduction and amination process that gives rise to tetrahydrofurfurylamine. By the 1970s, labs in both Europe and the United States worked out how to streamline its production. Fast forward to the present, this compound no longer hides on the fringes of specialty chemicals. Folks from agrochemistry to fine chemicals recognize its solid utility and reliability. Commercial catalogs list it as a staple, not a specialty.
Product Overview
Tetrahydrofurfurylamine steps forward as a colorless, low-viscosity liquid. It carries a faint, ammoniacal note in its smell, a reminder of its aliphatic amine roots. At first glance, many would underestimate this simple molecule; yet, applications in pharmaceuticals, fuel, water treatment, and synthesis speak for themselves. Packing both a cyclic tetrahydrofuran ring and a primary amine group, this chemical bridges solubility in water and compatibility with organic solvents—an unusual combination that helps it shine as a versatile intermediate.
Physical & Chemical Properties
Tetrahydrofurfurylamine has a molecular formula of C5H11NO and a molecular weight just over 101 grams per mole. It boils at around 179°C under atmospheric pressure. Because of its polarity, it mixes well with water, alcohols, and many ethers, but it doesn’t get along with most hydrocarbons. The amine group gives it a slightly basic character, so it picks up protons easily — this makes for straightforward salt formation, which is useful in purification. The compound stays stable under normal conditions, though with strong oxidizers or acids, reactivity creeps up. Handling becomes easier compared to more volatile amines, thanks to its higher boiling point and lower vapor pressure.
Technical Specifications & Labeling
Lab and plant operators often encounter tetrahydrofurfurylamine under specifications like >99% purity for use in fine chemical synthesis, and 98% or technical grades for bulk industrial requirements. Labels must display its UN number (2734) for transport purposes, a clear hazard diamond indicating flammable liquid and corrosive attributes, and the standard GHS pictograms. Responsible suppliers include batch traceability, water content (commonly under 0.5%), and a certificate of analysis upon request. In Europe, safety data sheets conform to REACH regulations, dictating both safety and traceability.
Preparation Method
Manufacturers typically synthesize tetrahydrofurfurylamine starting with tetrahydrofurfuryl alcohol, itself made from furfural by hydrogenation. The next major step, amination, involves reacting the alcohol with ammonia under pressure using hydrogen and a solid catalyst like ruthenium or nickel. Some methods use reductive amination routes, where the alcohol, ammonia, and hydrogen combine in a single reactor. These approaches favor high yields, especially under carefully controlled temperature and pressure. Scaling up this reaction means controlling by-products such as secondary amines, and catalyst fouling becomes a pain point that operators monitor closely.
Chemical Reactions & Modifications
Chemists use tetrahydrofurfurylamine for more than just basic amine chemistry. Its primary amine group opens doors to N-alkylation, acylation, and formation of Schiff bases—common steps toward building pharmaceuticals and specialty polymers. The cyclic ether brings hydrophilicity and a hint of rigidity, which helps in tuning the physical properties of reaction products. Oxidation and N-derivatization also attract interest for generating surfactants, detergents, or drug candidates. More advanced work even employs the amine as a chiral auxiliary, capitalizing on the tetrahydrofuran ring's stereochemistry for asymmetric synthesis.
Synonyms & Product Names
You might spot this chemical under names like THFA, tetrahydro-2-furylamine, or 2-(Aminomethyl)tetrahydrofuran in various supply catalogs. Pharmaceutical researchers sometimes list it as Furfurylamine, tetrahydro-, though this risks confusion with the unsaturated analog. CAS number 2682-49-7 clears up any ambiguity for regulatory and purchasing needs. Producers globally choose brand names and technical grade tags, but the core identity traces back to this small family of synonyms.
Safety & Operational Standards
Direct skin contact with tetrahydrofurfurylamine causes irritation, and inhalation of vapors triggers throat and lung discomfort. Personal experience with this compound drove home the importance of nitrile gloves and good eye protection—missing either creates unnecessary risk. Standard protocol calls for keeping it in tightly sealed containers, away from acids, oxidizers, and ignition sources. Splash goggles and chemical fume hoods keep exposure in check. Spills, if they occur, ask for inert absorbents and careful cleanup, given the liquid’s potential to damage plastics and paints. Waste streams containing this amine get routed to specialist disposal in accordance with U.S. EPA guidelines or similar local standards. Training staff remains non-negotiable for any high-turnover facility handling amines regularly.
Application Area
This amine acts as a building block in pharmaceutical manufacturing, where chemists use it to introduce hydrophilic side chains in active pharmaceutical ingredient (API) candidates. Agrochemical labs rely on it to modify pesticide precursors, aiming to boost stability and environmental breakdown. Resin formulators include it as a curing agent for epoxy and polyurethane systems, particularly where water resistance and flexibility go hand-in-hand. In water treatment, tetrahydrofurfurylamine chelates and binds metal ions, helping cleanse wastewater streams from industrial plants. More specialized uses include surfactant production, fuel additive research, and the synthesis of biodegradable polymers. Over the past decade, I’ve seen R&D scaleups focus on niche applications where water solubility meets strong base strength, especially for "green" alternatives.
Research & Development
R&D teams have carved out interesting directions using tetrahydrofurfurylamine, especially in biologically active fine chemicals and smart polymers. Researchers use its amine group to prepare functionalized resins for chromatography, enhancing selectivity in protein purification. Biotech start-ups favor derivatives based on this molecule for medical diagnostics and drug delivery, noting the balance between hydrophilicity and synthetic flexibility. Publications out of major universities show new derivatives finding use in catalysts for asymmetric reactions. In industrial chemistry, process engineers target safer catalytic methods, reducing hazardous waste compared to older batch processes using harsh acids. Academic partnership programs push the boundaries of catalytic hydrogenation, aiming to boost yields from renewable feedstocks like hemicellulose, tying into long-term goals for sustainable chemical manufacturing.
Toxicity Research
Research into the toxicity of tetrahydrofurfurylamine shows moderate irritancy to eyes and skin, but low acute oral toxicity in rodent studies. Chronic exposure tests suggest potential for reversible liver stress at high doses, although standard workplace exposure limits—0.5 ppm in many jurisdictions—sit well below this threshold. Some studies probe for mutagenic effects but haven’t turned up any strong red flags. Nonetheless, gaps persist in eco-toxicological data, especially regarding long-term effects in aquatic settings. Regulators in Europe and North America watch this space closely, especially as more industries seek to adopt the chemical as a bio-based alternative. In my own lab circles, any exposure triggers medical review regardless of severity—early intervention matters more than waiting for symptoms.
Future Prospects
Looking ahead, demand for tetrahydrofurfurylamine is poised to climb alongside the growth in green chemistry and sustainable manufacturing. Synthetic routes based on renewable starting materials line up with both investor and regulatory priorities. Advances in catalysis, as documented in recent patent filings, promise higher efficiency and lower emissions during large-scale production. Application areas are primed to expand, with biopolymer research and drug development leading the charge. The focus on safer, less toxic alternatives for surfactants and coatings means further study around biodegradability and environmental impact will guide both innovation and regulation. Ultimately, future years will see tighter integration of this compound both in process design for cleaner synthesis and in end-use applications built with environmental responsibility in mind.
What is Tetrahydrofurfurylamine used for?
Getting Acquainted with Tetrahydrofurfurylamine
Tetrahydrofurfurylamine rarely turns up in household conversations, but it plays a role in many products that touch our lives. This compound, often shortened to THFA, carries a bit of a tongue-twister name but brings real, tangible benefits to the table. A colorless liquid with a mild odor, THFA belongs to a group of chemicals known as amines—compounds frequently found in lotions, cleaning solutions, and, believe it or not, some medicine manufacturing lines.
Why Manufacturers Reach for THFA
Working in a research lab exposed me to a wide world of solvents and intermediates. THFA stood out because companies love using it as a building block for bigger, more complex molecules. Its ability to dissolve a broad range of substances gives chemical engineers a reliable tool for reactions that might stall with other choices. Paints and coatings end up smoother, adhesives gain strength, and electronics manufacturers get the purity they expect, all in part because they trusted THFA in the first place.
The Hidden Role of THFA in Clean Energy and Agriculture
Biofuels grab headlines, but not many people realize how important ingredients like THFA are in this shift. Chemists turn to it as an intermediate while developing cleaner-burning fuels from plant materials instead of fossil resources. It helps bridge the gap between crude sugars and final products like renewable diesel. Farmers looking for environmentally-friendly pesticides might encounter formulations improved with THFA, which lends stability and effectiveness. I've read research studies showing THFA-based solutions can decrease the harsh chemical load on fields and help keep local waterways cleaner.
A Key Player in Pharmaceuticals and Medical Research
During my time collaborating with pharma startups, the push for smaller, safer, and more efficient chemical processes came up again and again. Companies put THFA to work as a reagent—a helper molecule—on the road to new medicines. Its structure lets researchers fine-tune reactions that create painkillers, antibiotics, and even cancer drugs. In these labs, small changes in chemical choice ripple out to influence drug purity, availability, and patient health.
Environmental and Safety Concerns
Reliance on THFA asks for some caution, especially outside tightly controlled plants. Safety data points to mild skin irritation and a need for proper ventilation. Accidental spills can disrupt aquatic habitats, so waste handling can’t fall through the cracks. Government agencies such as the EPA publish guidelines for workplace safety and waste disposal, reinforcing the need for strong safety protocols. In my experience, the best labs run regular training sessions and checks, making sure nobody skips essential steps with substances like THFA.
What Comes Next for Tetrahydrofurfurylamine
Industries with a taste for sustainability keep looking for ways to recycle or substitute traditional chemicals. Companies investing in green chemistry investigate whether bio-based routes to THFA can cut down on fossil fuel use. Some startups pilot fermentation processes using agricultural leftovers, opening a pathway to lower emissions and supporting rural economies in the process.
Seeking Balance Between Use and Responsibility
The story of THFA often goes unseen, yet its impact runs deep—fueling industrial progress without always drawing public attention. We start to see that familiar tension between benefits and risks. Science keeps evolving, and companies refine how they use, store, and dispose of it. People working in labs or on production floors, like myself, notice the difference these choices make day in and day out.
What is the chemical structure of Tetrahydrofurfurylamine?
Getting Familiar with Tetrahydrofurfurylamine
Tetrahydrofurfurylamine isn’t a household name, but in lab settings or chemical industries, it crops up more often than most people realize. The molecule comes from furfural, which itself starts out in things like oats and corn cobs. Picture chemicals not as mysterious entities, but as building blocks that, like the bricks in a house, shape a lot of the materials and goods that show up around us.
A Closer Look at the Structure
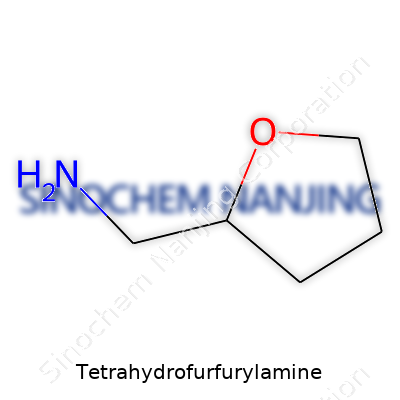
Tetrahydrofurfurylamine carries a backbone called tetrahydrofuran, a five-membered ring with four carbons and one oxygen. Think of it a bit like a doughnut made of atoms, the oxygen atom lending it some unique chemical personality. Tacked onto this ring is an amine group — a nitrogen atom swaddled with two hydrogen atoms. This gives the compound the core structure of 2-(aminomethyl)tetrahydrofuran. The amine group isn’t just an afterthought. Its presence shapes how this molecule behaves and what it can take part in chemically.
In a world guided by practical uses, Tetrahydrofurfurylamine’s chemical structure drives its performance. The five-membered ring delivers stability, resisting harsh breakdowns. The nitrogen group, on the flip side, is reactive enough to take part in forming new bonds. These features help manufacturers prefer it when making certain polymers, resins, or specialty chemicals.
Why Does the Structure Matter?
A little chemistry from school days comes in handy here. Organic compounds show different behavior depending on how their basic parts are arranged. Tetrahydrofurfurylamine’s ring keeps it flexible but not too floppy. It doesn’t evaporate at room temperature, protecting workers from sudden strong odors in a factory. People who handle industrial cleaners, coatings, or drugs have good reason to care about attributes like these. Scientists rely on the structure’s stability in lab reactions, giving them a repeatable, reliable platform to build on.
The amine group allows this molecule to react with acids to form stable salts or surfactants, which is a fancy word for compounds that help oils and water mix. This simple shift in arrangement nudges it into new roles, from playing a part in rust removers and detergents to serving as an intermediate for pharmaceutical synthesis.
Safety and Sustainable Choices
Relying on chemicals sourced from renewable feedstocks shapes industry attitudes for the better. Tetrahydrofurfurylamine traces its lineage to furfural, a bio-based chemical. That means less dependence on fossil fuels and more sustainable practices, when farms and forests are responsibly managed.
Like most amines, it’s not risk free. People handling it for the first time might notice irritation to skin or airways. Proper gloves and good ventilation aren’t just recommendations; they’re non-negotiable for folks who care about health in the workplace. More companies track these risks using real-world data. The switch to personal protective equipment isn’t normally a burden; it’s simply about staying clear-headed and safe, and it makes for fewer emergency room trips.
Solutions and Smarter Use
Research keeps unpacking ways to push for greener, safer, and more effective uses for chemicals like Tetrahydrofurfurylamine. Choosing renewable sources for the raw materials and tightening up training around handling mean fewer accidents and a lighter impact on the environment. Researchers in green chemistry push for processes that pull less waste and draw from less toxic methods. Chemists and engineers want to get more utility from every molecule—fewer leftovers, less exposure risk, longer shelf life.
Staying curious about these structures connects everyday users, workers, and innovators in a long line of problem-solvers. Whether it’s designing better antifreeze, safer cleaners, or new medicines, knowing what shapes a simple molecule like Tetrahydrofurfurylamine helps everyone work smarter and safer.
Is Tetrahydrofurfurylamine hazardous or toxic?
Looking at Real Risks, Not Just Labels
People see a chemical name like tetrahydrofurfurylamine and questions start swirling. There’s a gut-level concern about what it could do to you. This stuff lands in laboratories, chemical plants, and sometimes specialty cleaners, so it matters to get the facts right. Safety means more than a label — it’s about understanding the real exposures workers and the public might have, not just reading hazard warnings out of a safety manual.
So, What Actually Happens if You Touch or Breathe It?
I’ve seen the Material Safety Data Sheets handed around job sites. For tetrahydrofurfurylamine, the word “irritant” comes up a lot. If you spill it on your skin or splash it in your eyes, it stings and burns, and you’ll want to rinse thoroughly. It can leave your skin red or peeling after a longer exposure. Breathe in strong fumes and it catches in your throat, making your nose run or your lungs ache. There are cases of nausea and headaches if there’s a big spill in a closed space. People whose jobs kept them working around open containers talked about dry skin and irritated eyes by the end of a shift.
The Science Behind Toxicity
Acute toxicity numbers are one way chemists measure risk. Tests show that tetrahydrofurfurylamine scores low for lethal doses in rats — so, swallowing a little won’t knock you down fast, but nobody healthy should be mixing up a glass of the stuff. It’s not classified as carcinogenic, mutagenic, or a reproductive hazard by any major agency at this point. The real trouble starts if you keep getting it on your skin week after week. Reports show some folks develop a sensitivity over time, with gradually worse rashes and even allergic reactions. That’s how a chemical like this can sneak up on you: not as a single catastrophe, but as a daily hassle that gets overlooked until someone can’t work anymore.
Handling It Without Fuss — But With Smarts
It makes sense to compare this amine to more notorious chemicals in the same workplaces. Ammonia: deadly in high doses. Acetone: lights up with a spark and dries skin fast. Tetrahydrofurfurylamine doesn’t explode, but it lingers on desks and gloves — and forget to wear goggles or a respirator, it’ll remind you with a cough.
I always found that common sense and training trump fear. Aprons, gloves, and good goggles knock down most of the day-to-day risks. Plenty of fresh air or working with a vented fume hood keeps the air safe. I’ve washed chemical splashes off my arms in a hurry before and felt lucky that eye washes and emergency showers were close by. Chemical handling can never turn into an afterthought in any plant or lab.
Better Info and Policies Improve Safety
Hazard ratings come from actual tests and worker reports, and regulations around the world differ, but most agree this is a chemical that needs protections in place. I’d push for more regular skin checks on folks in frequent contact, just like how some facilities already do with solvents and acids. Updated training, clear signage, and better spill clean-up kits go a long way. In the end, solid science and first-hand experience keep people safer than a scary name ever could.
What are the storage conditions for Tetrahydrofurfurylamine?
Understanding Tetrahydrofurfurylamine’s Quirks
Tetrahydrofurfurylamine, often used in labs and manufacturing, comes with its own set of challenges the moment it lands on your shelf. This colorless liquid, with a faint ammonia-like smell, draws water out of the air if left uncapped. Once, at a shared university lab, someone left a bottle open overnight. Next day, the amine’s makeup had shifted and the experiment went sideways. So storing it right isn’t just good housekeeping—it makes or breaks experiments, product batches, and safety standards.
Pick Your Container Wisely
This chemical reacts with acids and oxidizers, so just dumping it into the first available bottle leads to trouble. Glass with a tight-fitting cap or high-density polyethylene containers work well. That old trick of reusing soda bottles doesn’t cut it; you risk chemical leaching or even a ruptured container. Metal drums rust on contact, so those are out. Keeping your chemicals in designated, chemical-resistant vessels doesn’t just protect tetrahydrofurfurylamine; it protects everything nearby.
Temperature Isn’t Just a Number
In hot storage rooms, tetrahydrofurfurylamine evaporates faster and pressure builds up. I’ve seen staff struggle with stoppers they couldn’t twist off, all because someone forgot that wide swings in temperature stress a chemical’s stability. Storing this amine at room temperature, away from heaters, sunlight, and chillers, keeps both the liquid and its vapor right where you want them. Sticking a thermometer in the storage room gives a reality check—guesswork leads to ruined stock and wasted money.
Keep Air and Moisture Out
Humidity sneaks in by surprise. Even a quick pour without recapping lets the chemical begin absorbing water. Once, I saw beads of water forming inside what was supposed to be a dry bottle—the difference between a controlled reaction and a runaway hazard. Every time you finish, tighten the cap. Adding a desiccant packet in the storage space helps absorb stray water. Some operations use nitrogen blanketing to push out humid air, reducing contamination risks for sensitive experiments.
Label Everything and Keep Inventory Honest
Every container should stand out with a clear, precise label. Long ago, faded masking tape led our group to use the wrong amine for an important synthesis. Modern printed labels list the name, date received, and a hazard warning. A digital inventory—nothing fancy, even a spreadsheet—helps you spot forgotten or expired chemicals. Proper labeling protects both workers and any emergency crew who may one day enter your storage room.
Safe Access and Good Habits Count
Separate your amines from acids and oxidizers. Chemicals don’t care about your deadlines; if mixed up on a cluttered shelf, they react unpredictably. Shelves, trays, and spill kits create layers of protection. Good habits—like wearing gloves and goggles and washing hands after handling—should kick in the moment you enter the storage space. Skipping these steps, even for just a second, leads to burns, inhalation hazards, or worse. Posting procedures by the door keeps everyone on track, from green interns to old hands.
Disposal Done Right
Spills or waste build up over time. Pouring leftovers down the drain violates health and environmental laws, and brings hefty fines. Contract with licensed waste handlers. Make sure routine checks track leaks, shelf life, and proper storage conditions. Cutting corners leads only to bigger costs later on. Each bottle treated with care means a safer workplace and a cleaner environment.
What are the typical applications of Tetrahydrofurfurylamine in industry?
Chemical Manufacturing’s Unsung Ally
Tetrahydrofurfurylamine, often shortened to THFA, pops up in a surprising range of industrial settings. It’s got a good reason for sticking around: this liquid slips into chemical reactions a lot more smoothly than you’d expect. Its backbone comes from furfural, a plant-based material, so in some ways, it fits the growing push for more renewable feedstocks. The chemical industry likes using it as a building block. Factories rely on THFA to help form pharmaceuticals, crop chemicals, and some rather useful specialty polymers. I’ve seen how it makes reactions run cleaner or more predictable compared to some old-school amines. This saves time, and if you’re working with expensive ingredients, small efficiencies add up fast.
Pharmaceuticals and Agrochemicals: Getting the Job Done
Drug synthesis is complicated work, and THFA plays a quiet supporting role. In several steps, it acts as a base or an intermediate, making it easier to attach amino groups or tweak the molecules. This lets chemists produce active pharmaceutical ingredients with fewer side reactions or byproducts. I’ve talked to colleagues working with herbicide and fungicide production who value the way it behaves in their mix tanks. The agricultural industry cares about consistency and avoiding production hiccups, so having a reliable amine source smooths out a lot of headaches. The amount of product that gets wasted goes down, and plants can keep up with demand even as targets shift each growing season.
Solvents and Coatings: Subtle Yet Significant
Some folks notice THFA because of its contribution as a specialty solvent. It dissolves a wide variety of organics, which is handy for custom coatings, inks, and adhesives. I remember a project that involved reformulating a water-resistant coating. THFA helped balance performance and process safety, since it carried less fire risk than more volatile options. You see it in electronic sealants and even advanced cleaning agents. Since it doesn’t eat away at sensitive components, device manufacturers like using it in precision electrical assembly.
Green Chemistry Push
One reason for THFA’s recent uptick links to the sustainability angle. Processes that shift from petrochemicals toward renewables lean on furfural-based inputs. Using THFA helps manufacturers keep up with regulations and the growing expectations from buyers who watch for green labels. The chemical isn’t perfect, but it proves that greener starting points do find a home in hard-nosed industry settings. I’ve noticed companies highlight this in safety data sheets or marketing, though the actual safety profile requires close attention. If your plant already uses amines, swapping THFA can sometimes trim emissions or toxic waste – a small shift, but the sum matters across an industry.
Challenges and Better Paths Forward
Tetrahydrofurfurylamine isn’t a silver bullet. Facilities still need to factor in safe handling—amines can trigger strong reactions or health effects in direct exposure. Monitoring air quality and giving workers solid training cuts down on risks. Manufacturers can also pressure suppliers to certify origins and environmental impact. Efforts in recycling solvents and tightening reaction yields push costs lower and waste streams cleaner. Specialty chemical firms experiment with using even more plant-based sources, driven both by global policy shifts and everyday cost pressures. Solutions often come from tweaks in process controls or clever engineering rather than headline-grabbing breakthroughs.
| Names | |
| Preferred IUPAC name | 2-(Aminomethyl)oxolane |
| Other names |
2-(Aminomethyl)tetrahydrofuran THFA Tetrahydro-2-furylamine Tetrahydrofuran-2-ylamine 2-Tetrahydrofurfurylamine |
| Pronunciation | /ˌtɛtrəˌhaɪdroʊˈfɜːrfjəlaɪmɪn/ |
| Identifiers | |
| CAS Number | 4296-28-4 |
| 3D model (JSmol) | `3DModel:JSmol|C4H9NO|CC1CCOC1N` |
| Beilstein Reference | 1718732 |
| ChEBI | CHEBI:53032 |
| ChEMBL | CHEMBL140186 |
| ChemSpider | 157356 |
| DrugBank | DB04116 |
| ECHA InfoCard | ECHA InfoCard: 100.041.783 |
| EC Number | 211-069-2 |
| Gmelin Reference | 62272 |
| KEGG | C06454 |
| MeSH | D02.886.300.249.500. |
| PubChem CID | 8906 |
| RTECS number | YO8400000 |
| UNII | 4PJ2Q7ZV4C |
| UN number | UN2675 |
| CompTox Dashboard (EPA) | DTXSID3054673 |
| Properties | |
| Chemical formula | C5H11NO |
| Molar mass | 89.14 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | amine-like |
| Density | 0.973 g/mL at 25 °C |
| Solubility in water | Miscible |
| log P | 0.21 |
| Vapor pressure | 0.316 hPa (at 20 °C) |
| Acidity (pKa) | 9.2 |
| Basicity (pKb) | 5.26 |
| Magnetic susceptibility (χ) | -7.71 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.454 |
| Viscosity | 12 mPa·s (25 °C) |
| Dipole moment | 1.70 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 311.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -321.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3655 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | Precautionary statements: P280, P261, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | > 74°C |
| Autoignition temperature | 215°C |
| Explosive limits | Explosive limits: 1.3–9.7% |
| Lethal dose or concentration | LD50 (oral, rat): 2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 200 mg/kg |
| NIOSH | RN 2682 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 150 mg/m³ |
| IDLH (Immediate danger) | No IDLH established. |
| Related compounds | |
| Related compounds |
Furfurylamine Tetrahydrofuran 2-Methyltetrahydrofuran Furfural Pyrrolidine |

