Tetrafluorohydrazine: A Deep Dive
Historical Development
Tetrafluorohydrazine hardly jumps out at most people, even those peering into the corners of modern chemistry. Chemists began looking at this compound with serious eyes as the world’s interest in fluorine chemistry soared right after the Second World War. Back then, the push for new rocket fuels and the natural curiosity about nitrogen-fluorine chemistry led folks at places like DuPont and university labs to run early syntheses and collect what had been largely overlooked up to that point. Over the next several decades, research groups working on fluorinated materials unraveled more practical aspects of the molecule’s reactivity—sometimes out of pure scientific curiosity, sometimes driven by defense projects, where the compound’s energetic properties came under scrutiny. Today, tetrafluorohydrazine still rides on that legacy of applied curiosity and risk-taking research.
Product Overview
Ask people in the specialty gas industry about N2F4—the shorthand for tetrafluorohydrazine—and a certain caution flashes across their faces. The gas isn’t something you stick in a standard lecture-hall demonstration. Instead, it finds its way to places where people demand something different: strong oxidizing power, radical chemistry, or high-energy alternatives. It’s a serious player in niche fertilizer, rocket propulsion, and special synthesis applications, rarely joining the daily routine of most labs. The purchases trickle in from defense contractors or academic researchers rather than bulk buyers, and packaging demands careful handling—heavy steel cylinders, strict documentation, and delivery under watchful eyes.
Physical & Chemical Properties
Tetrafluorohydrazine comes as a colorless, toxic gas near room temperature. The vapor condenses into a pale green liquid as temperatures drop below its boiling point—just above 50°C under standard pressure. It carries a biting, acrid odor that reminds seasoned chemists of the risks. Its molecular weight, about 104 g/mol, puts it in the league of heavier industrial gases. In terms of structure, two nitrogen atoms bridge with four fluorine atoms, creating a staggered geometry that influences its chemical behavior. Its reactivity stems from those nitrogen-fluorine bonds, which store energy and break apart under the right conditions. Solubility in water barely registers, but fluorinated solvents pick it up more easily. Thermal stability remains questionable—the stuff decomposes above 75°C, spitting out nitrogen, fluorine, and dangerous byproducts.
Technical Specifications & Labeling
Handling tetrafluorohydrazine means trusting the label. The United Nations designates it as UN 2418, and it falls under hazard class 2.3 (toxic gases). Shipping must clearly display those diamond warning symbols. Each cylinder label lists purity (typically above 98% for research use), total volatile impurities, and manufacturing lot traceability. Regulatory documentation follows it everywhere—Safety Data Sheets (SDS), shipping manifests, and compliance certifications are as much a part of the purchase as the gas itself. The purest batches support electronic-grade demands. No one in the business mistakes this for an off-the-shelf commodity.
Preparation Method
Most commercial production of tetrafluorohydrazine traces back to the oxidative fluorination of hydrazine or hydrazine derivatives. Large fluorine plants typically prepare the gas in controlled reactors lined with nickel or Monel to hold back the brutally reactive fluorine. Chemists run elemental fluorine over anhydrous hydrazine, keeping the temperature cool enough not to lose everything to runaway thermal decomposition. The gas emerges contaminated with unwanted byproducts—hydrazine, dinitrogen difluoride, and traces of HF—so cold traps and careful distillation step in to collect the cleanest fraction. Smaller labs sometimes take nitrogen trifluoride and coax it through a secondary reduction, but industry mainly sticks with the direct fluorination recipe.
Chemical Reactions & Modifications
Tetrafluorohydrazine isn’t the most cooperative reagent, but its radical-based chemistry allows for some fascinating transformations. Researchers discovered decades ago that it reacts violently with strong nucleophiles—think alkali metals or even water under the wrong circumstances—at room temperature or lower. It acts as a powerful fluorinating agent, transferring its fluorine atoms to metals, organics, and even silicon-based frameworks. High-energy chemists harness its ability to form nitrenes and related species, which go on to build more exotic molecules. Under UV or thermal activation, N2F4 splits to form nitrogen trifluoride (NF3) and reactive intermediates; that side reaction has sparked more than one accident in academic labs. The molecule’s capacity for both adding and subtracting fluorines has kept it in the toolkit of those building new molecules for electronics or energetic materials.
Synonyms & Product Names
Trade and technical literature use a handful of names for this stuff: tetrafluorohydrazine, perfluorohydrazine, and disazotetrafluoride all point back to N2F4. Some older military and space documents refer to it as “tetrafluoroazodine,” but that’s rare outside government circles. Chemical suppliers stick to the formal IUPAC name and the four-litter chemical shorthand. No matter the name, the hazard warnings stay the same.
Safety & Operational Standards
Few gases command as much caution in the lab and field as tetrafluorohydrazine. Its acute toxicity rivals the most unforgiving chemicals—exposure damages the lungs, skin, and eyes in seconds, and inhaling even small concentrations can cause severe respiratory distress. Every protocol starts with engineering controls—glove boxes, ventilated fume hoods, explosion-proof fittings. Operators wear multi-layered personal protective equipment: face shields, heavy-duty gloves, and splash-resistant gowns. Gas detection monitors constantly sample the area, tuned to pick up fugitive emissions before they reach harmful levels. Regulators demand full traceability from manufacturer through delivery and possession. Emergency protocols require on-site antidotes and rapid evacuation routes. The industry remembers the handful of major incidents—leaks at research facilities, accidental venting during transport—and builds extra layers of redundancy into every procedure. For all its promise, the chemical lets its user know: mistakes come at a high price.
Application Area
Its old reputations in rocketry and explosives research still pop up occasionally, mainly in the development of high-thrust propulsion and chemical lasers. More recently, electronics manufacturers use tetrafluorohydrazine as a specialty etchant and precursor in the growing world of semiconductors, where high-purity fluorine sources matter. Academic researchers exploring the boundaries of high-energy chemistry turn to this gas when building new classes of nitrogen-fluorine macrocycles, polynitrogens, or energetic intermolecular complexes. Surface chemists sometimes choose it for controlled fluorination of materials, seeking new properties in polymers or metals. A handful of niche suppliers stock it for the rare customer chasing next-generation energetic materials, but most of the demand stays locked behind security clearances and strict export controls.
Research & Development
Work on tetrafluorohydrazine keeps moving, not always under the spotlight. Teams in industrial labs and major universities run studies into controlled decomposition, seeking ways to harness its reactivity without stumbling into runaway exotherms. Others explore its role as an intermediate in making metal fluorides or advanced ceramic coatings. The biggest technical barriers remain its toxicity, its thermal instability, and the sheer cost of producing it under safe conditions. More theoretical chemists continue to model its radical chemistry and compare notes with bench chemists, trying to predict where the next big breakthrough lurks. A few collaborations with defense sectors still back research into more stable storage and safer handling, but those reports rarely make the public circuit. Research groups keep close tabs on advances, knowing that any improvement in containment, catalysis, or reactivity opens the door to new science and technology—and to larger-scale applications, if the safety record keeps pace.
Toxicity Research
A close look at the medical and environmental data shows why health researchers take N2F4 seriously. Exposure at concentrations as low as 10 ppm triggers eye and respiratory distress, and prolonged contact can leave scarring or permanent tissue damage. Its decomposed products, including nitrogen oxides and fluorine, only pile on the risk. Chronic effects stay poorly characterized due to limited exposure data—almost all interaction with the compound occurs under tight controls—but the available animal models show significant risk to the lungs and central nervous system. No antidote exists; treatment means removing the victim from exposure, aggressive decontamination, and acute symptom management. Environmental fate studies remain few: the molecule decomposes or dilutes quickly in open air, especially in ultraviolet sunlight, but accidental releases near populated areas could spell disaster. Regulatory agencies treat it with the same care they give more famous chemical threats—from permit paperwork to disposal, every step ties back to the core understanding: safety first, always.
Future Prospects
Demand for specialized fluorinated reagents likely won’t disappear, especially as industries chase more energetic materials or more precise patterns in semiconductor manufacturing. Improvements in onboard monitoring, safer containment vessels, and automated handling promise to widen the molecule’s industrial footprint, provided regulatory approval keeps up. If researchers succeed in developing less toxic analogs or in stabilizing the gas in storage and transport, the range of uses for N2F4 might grow beyond its current confines. New computational techniques keep picking at the theoretical chemistry, trying to tease out untapped potential or novel applications. Entrepreneurs and veteran chemical makers alike keep their ears open—recognizing opportunity in the tricky world of specialty chemicals means balancing innovation with the responsibility to keep workers and communities safe.
What are the main uses of Tetrafluorohydrazine?
Chemical Processing and Semiconductor Applications
Not every chemical makes headlines or features in classrooms, but tetrafluorohydrazine keeps some very advanced technology humming along. Factories making semiconductor chips count on it. Its unique chemical structure allows engineers to use it during etching steps. One important point: tetrafluorohydrazine can break down into reactive intermediates under the right conditions, and those fragments help carve intricate circuits onto chips. No one pulls the latest graphics card or smartphone out of the box without benefiting from such precise etching chemistry.
My time in electronics assembly introduced me to just how picky semiconductor manufacturing can be. In cleanrooms, workers protect wafers worth thousands of dollars, and purity takes center stage. Tetrafluorohydrazine’s volatile nature means it’s less likely to leave unwanted residues, which matters when dealing with microscopic features. If impurities sneak in, entire batches may end up scrapped. So, quality control covers everything from the source material to exhaust treatment, ensuring people and the planet stay safe.
Rocket Propellants and Aerospace Engineering
Rocket engineers also find tetrafluorohydrazine attractive. Its high-energy bonds release significant thrust paired with the right fuel. The NASA community has reported on its role as an oxidizing agent in experimental rocket systems or as a part of bipropellant combinations. Unlike classic fuels such as hydrazine, adding fluorine kicks reaction energies higher, leading to greater performance. With space launches, every kilo matters, and using energetic chemicals gives more boost for less mass. Handling those chemicals takes training, as exposure presents clear hazards, but in controlled environments, they help reach new heights—literally.
Safety protocols around these chemicals remain strict. Years ago, I visited a facility where scientists showed off protective gear and outlined multiple containment barriers. You cannot shortcut here—spills or leaks pose trouble not only for the person nearby but also for air and groundwater. Keeping workers healthy demands a culture of respect for procedures, daily maintenance, and leak detection. It’s not glamorous, but it keeps launches on schedule and crews out of the ER.
Specialty Synthesis and Advanced Materials Research
Specialty labs experiment with tetrafluorohydrazine as a building block for new molecules. Looking at academic journal archives, chemists incorporate it to introduce fluorine atoms into larger organic molecules. Such fluorinated molecules often pop up in pharmaceuticals, crop protection, or polymers built to handle harsh environments. A stronger carbon-fluorine bond resists heat and chemical attack, so researchers keep exploring fluorine sources for next-generation materials.
From my university days, I remember how introducing new reagents can sometimes turn run-of-the-mill substances into something groundbreaking. Take Teflon, for instance. Broad interest in fluorinated substances follows the search for durable yet lightweight coatings, non-stick applications, or new drugs with precise biological effects. Getting regulatory approval takes time, especially with chemicals known for toxicity risks, so transparent reporting and peer-reviewed safety studies earn trust, meeting E-E-A-T guidelines valued by scientists and the public alike.
Addressing Environmental and Safety Concerns
It’s impossible to ignore the downsides. Tetrafluorohydrazine and its byproducts linger in air or water if not managed correctly. Groups like OSHA and the EPA track these chemicals, listing potential health issues from prolonged exposure. Installing scrubbers, testing workplace air, and training staff protects both workers and communities. Cutting emissions comes down to investment: up-to-date ventilation, regular inspections, and fast response drills.
Solutions begin with honest record-keeping and clear communication between plant operators, regulators, and local residents. By keeping channels open, communities feel safer—the right to know what’s in your backyard matters to everyone. Adapting production to safer substitutes, whenever possible, also helps, but for now, industries keep a watchful eye on both the benefits and risks of tetrafluorohydrazine. As technology and safety mechanisms improve, so does the future for people and the environments they call home.
What safety precautions should be taken when handling Tetrafluorohydrazine?
Getting Real About the Risks
Tetrafluorohydrazine isn’t just a tough name to pronounce; it’s a tough chemical to work around. This compound packs some serious punch. Researchers have called out how its volatility and toxicity make it a major concern for tech and manufacturing circles, especially in labs and industries pushing the edge with electronics or rocket propellants. I’ve seen firsthand how a quick mistake with reactive gases can turn a normal workday into a scramble for the safety shower.
Why Small Missteps Can Lead to Big Problems
A lot of folks hear warnings about dangerous chemicals and assume a little care goes a long way. With tetrafluorohydrazine, skin contact or inhalation can mean real medical emergencies. A splash on an unprotected hand might bring on burns or worse. Even small amounts in the air can irritate the lungs or worse, topple someone over if ventilation doesn’t keep up.
Regulators in places like the US and Europe set limits for airborne exposure, and inspectors keep a sharp eye on leaks in facilities where this stuff gets handled. This isn’t just about ticking boxes; without the right safeguards, the fallout lands on workers, not just profits.
Gear Up—Every Time
Lab coats alone won’t handle this job. Good nitrile gloves, which meet chemical resistance standards, keep skin safe. Goggles with side shields block any wayward spray, and face shields help if lines or valves throw off a surprise burst. A full respirator, properly fitted and certified for gas and vapor protection, becomes the line between safety and a hospital trip. I’ve watched a new tech try shortcuts to save time, only to end up with a chemical burn and a lesson nobody wanted him to learn the hard way.
Containment: Keep It In, Keep People Out
Fume hoods rated for corrosive gases do a better job than just cracking a window. Workers handle all transfers and workups beneath these hoods to catch drips, fumes, and the occasional unexpected reaction. Industry reports tell us the spills that cause injuries start with people skipping simple fixes: using open beakers, ignoring tiny leaks, or running transfer hoses across walkways.
Fix and Double-Check the Basics
Sturdy containers, leak-free connections, and clear labeling cut down the biggest risks fast. I’ve always double-checked gas lines and seals—people remember the first time someone failed to catch a bad O-ring before pressurizing a cylinder. Sudden leaks, followed by a dash for the emergency stop, never look good in the incident log.
Training and Honest Communication
Anyone near tetrafluorohydrazine needs steady training, not just a binder of rules that sits in the break room. Walk-throughs of what could go wrong, paired with real drills, put the focus on what matters. Open conversations on mistakes or close calls help catch near misses before they turn into emergencies. Science backs this up—studies in workplace safety show big drops in injuries when organizations give people a voice and a little backup.
Never Slack on Storage
Cylinders of tetrafluorohydrazine stay in well-ventilated, hazardous-material-rated spaces that stay locked unless trained workers need access. No shortcuts—no “just this once” excuses. Unused containers go right back to approved storage. Even the smallest slip up, like wedging a tank in a corner to “save time,” walks hand-in-hand with disaster.
Push for Accountability and Help
If something does go wrong, fast support matters. Everyone in reach has got to know where the emergency shower, eyewash, and neutralization kits stand. Covering emergencies in drills, not just as footnotes in paperwork, creates reflexes you can count on. I’ve stood through more than one debrief that ended with sore muscles and a sharp reminder: with chemicals like tetrafluorohydrazine, only real preparation keeps people safe—faking it never works.
What is the chemical formula of Tetrafluorohydrazine?
Understanding the Basics
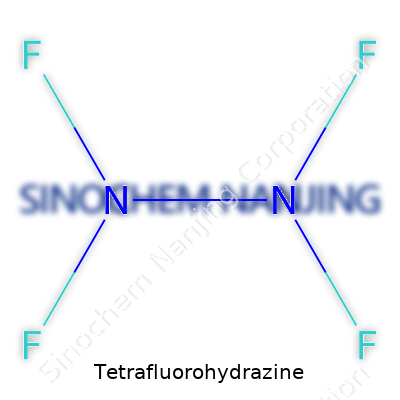
Tetrafluorohydrazine shows up in chemical conversations with the formula N2F4. This compound doesn't often enter mainstream news, but its presence is felt in certain industries and research labs. If you have friends who work in chemistry, you may have heard stories about the challenges of handling reactive or unstable molecules. Tetrafluorohydrazine has earned respect and caution for these reasons. It contains two linked nitrogen atoms, each bonded to two fluorine atoms. That's it—no extra bells, no hidden complexity in the backbone.
Real-World Applications
While this molecule sounds exotic, its properties put it on the radar for folks working with fluorine chemistry. Back in college, one lab instructor loved recounting tales of fluorine compounds—how their intense reactivity caused both breakthroughs and disastrous messes. Tetrafluorohydrazine plays a part in rocket propellants and specialty synthesis. It can release energy rapidly, which makes it valuable when control is in expert hands. The high fluorine content brings unique firefighting and chemical-resistant possibilities, though these remain more in the realm of research than in everyday products.
Safety and Environmental Concerns
No conversation about N2F4 skips over safety. Chemicals heavy in fluorine frequently create risks to people and ecosystems. Handling tetrafluorohydrazine means dealing with toxicity and the potential for violent decomposition. Hospitals and poison control centers sometimes reference incidents involving fluorine-rich compounds, ringing alarm bells in communities near manufacturing facilities. In my experience, hearing about the evacuation of several blocks because of a minor leak proved just how powerfully these gases can impact lives. Robust safety protocols stand as the frontline defense for workers and nearby residents.
Supporting Safe Innovation
Experts with years in chemical manufacturing stress the value of double-checking containment measures and emergency plans, especially for compounds as sensitive as N2F4. The chemical’s instability drives ongoing searches for safer alternatives or stabilizing additives. Some labs focus on designing derivatives with less volatility, hoping to keep the perks of strong fluorination while dialing down the hazard. A few companies invest in advanced sensors and leak detection technology, which has reduced major incidents in several facilities.
Education and Next Steps
Community education forms a critical line of defense. Local emergency planners keep residents informed about the chemicals stored and transported near their homes. Schools and youth organizations now run more hands-on science demonstrations to demystify chemistry and highlight both the rewards and risks. This connects people more directly to the realities scientists and industry workers face every day. Future progress will come from both safer lab practices and new policy debates on chemical transparency and reporting.
Accountability and Leadership
Companies dealing with tetrafluorohydrazine and similar chemicals carry the responsibility for upholding rigorous safety standards. Leadership starts with company culture, where experts predict and address failure points before accidents cost lives or damage environments. By building networks between public health officials, regulators, and industry experts, stronger oversight emerges. This approach limits not only the likelihood of runaway incidents but builds community trust in a chemical industry that many see as distant or secretive.
How should Tetrafluorohydrazine be stored?
Why Storing Tetrafluorohydrazine Demands Respect
Tetrafluorohydrazine isn’t something you leave lying around in a dusty corner. Many folks in labs or industry deal with chemicals every day, but this one stands out. I’ve seen storage mishaps, and nobody enjoys the fallout. Tetrafluorohydrazine holds a reputation for being sensitive and sometimes unpredictable. It takes a small mistake for this gas to turn dangerous.
Understanding the Risks
Conversations around this substance tend to focus on its reactivity and toxicity. Inhaling even small amounts can cause serious respiratory harm. If mixed accidentally with the wrong compounds, explosions or fires aren’t out of the question. I remember reading about a leak in a facility where poor storage led to an emergency evacuation. Not only were people put at risk, the company faced regulatory fines and clean-up nightmares.
Best Storage Conditions
You want to keep tetrafluorohydrazine in sturdy, corrosion-resistant cylinders. Stainless steel or nickel-plated containers stand up to the chemical pretty well. Plain old steel or glass shouldn’t be trusted here. Store these cylinders upright. This isn’t just some bureaucratic rule—a sudden knock or tip-over can damage the valves, and leaks follow quickly.
Heat exposure escalates every hazard. Keep storage environments cool, ideally below room temperature. Dryness matters, too, since moisture spells trouble anywhere fluorine chemistry is involved. That means staying far away from damp basements or anywhere humidity runs high.
Everyday Handling and Monitoring
Put up clear labels. From personal experience, I’ve watched too many close calls with misidentified bottles and cylinders. No one should ever have to guess what’s in that tank.
Install gas detectors nearby. These devices provide early warnings if a leak sneaks up. Relying just on your nose or sight doesn’t cut it, especially with a gas you can’t always smell at dangerous concentrations. Good airflow in your storage area makes sense, too. Any mishap gets swept out and diluted before it can build up to hazardous levels.
Legal Compliance and Worker Safety
Regulators like OSHA aren’t just ticking boxes—they’re basing rules on hard lessons. Secure all storage areas against unauthorized access. Only trained people should handle these cylinders. Employers should keep safety data sheets front and center and make sure everyone updating those files knows why changes matter.
Regular cylinder inspections catch rust, dents, or worn-out valves early. If a tank starts showing its age, swap it out. It isn’t worth the gamble. I’ve seen how quickly an “it’ll be fine” attitude backfires.
Looking to the Future
Better materials for containers and smarter sensors are showing up as technology pushes forward. While those options can cost more, cutting corners here can lead to accidents, lawsuits, or worse.
For organizations with sustainability goals, proper practices around reactive chemicals matter more than ever. Dealing with hazardous waste after an incident often causes more environmental damage than just storing things safely from the start.
Smart storage routines and a healthy respect for tetrafluorohydrazine don’t just protect people—they save companies and communities from disaster.
What are the physical and chemical properties of Tetrafluorohydrazine?
Understanding Tetrafluorohydrazine
Tetrafluorohydrazine, known in chemistry circles as N2F4, is a colorless gas with a sharp, biting odor. You can almost smell the danger just from handling the bottles in a well-ventilated lab, and with good reason. This compound stores plenty of energy in the bonds between its nitrogen atoms and fluorine hitchhikers. Working with it teaches respect for the less glamorous aspects of chemistry, such as handling toxic fumes and making sure protective gear actually does its job.
Physical Properties That Matter in Real Life
N2F4 boils at just above room temperature—about 26°C (78°F)—so it doesn’t stick around as a liquid under everyday conditions. Its vapor pressure means any leaks travel fast, pushing odor and risk beyond the immediate workspace in no time. That vapor pressure is no joke: this gas will try its best to find a way out of containment if given the slightest opportunity. Its density is about three times that of air, so it hugs the ground, which increases the risk for workers who assume it’ll disperse like a lighter gas.
Electronics cleanrooms and NASA test facilities use compounds like these not just for their novelty, but because their properties fill a need. Storage infrastructure matters—a corroded valve or tired gasket quickly becomes a crisis. One lesson I’ve learned: never take shortcuts with fluorinated gases.
Chemical Behavior: Reactivity That Commands Caution
The fluorine content makes Tetrafluorohydrazine one of those compounds everyone treats with a little suspicion. Mixed with air and exposed to a spark, it decomposes explosively, filling the air with toxic byproducts like hydrogen fluoride (HF) and nitrogen oxides. Those byproducts linger long after the initial event, so emergency response plans account for staying power, not just the first wave of trouble. Fluorine likes to form strong bonds, and Tetrafluorohydrazine takes this enthusiasm to another level—it's a tough oxidizer. In organic syntheses, the compound acts as a fluorinating agent, able to rip through carbon frameworks many molecules rely on for stability.
Why These Properties Matter
Each year, accidents remind everyone about the risks. Fluorinated hydrazines like N2F4 aren’t just academic interests—they play key roles in aerospace, deep-cleaning electronic surfaces, and rocket research. A single misstep—forgetting to double-check a seal or underestimating the compound’s persistence—can lead to hospitalization or worse. The sense of danger never really goes away, even for veterans. Real experience has taught industry leaders to take nothing for granted, even after years of working with these materials.
Safety training with Tetrafluorohydrazine goes well beyond textbooks. Real drills using non-toxic stand-ins and real gear make a difference, giving people muscle memory if things go wrong. Detection equipment has to work in real time and alarms should never get ignored, especially with a compound so dense and unforgiving.
Paths Toward Safer Use
Smart facility design—ventilation, remote sensors, and double-containment systems—reduces risk more than just following written protocols. There’s value in keeping up with research, especially as engineering teams chase more stable alternatives or better neutralization techniques. Chemists and engineers alike realize that sometimes the right answer is avoiding high-risk chemicals when safer substitutes accomplish the same task.
| Names | |
| Preferred IUPAC name | tetrafluorodiazane |
| Other names |
Perfluorohydrazine Nitrogen tetrafluoride N2F4 |
| Pronunciation | /ˌtɛ.trəˌflʊə.rə.haɪˈdreɪziːn/ |
| Identifiers | |
| CAS Number | 10036-47-2 |
| 3D model (JSmol) | `JSmol.loadInline("data/mol:NF2NF2")` |
| Beilstein Reference | 1208952 |
| ChEBI | CHEBI:30036 |
| ChEMBL | CHEMBL1230609 |
| ChemSpider | 77126 |
| DrugBank | DB14325 |
| ECHA InfoCard | 18bbcfb7-5495-41df-bc2f-03ef8e957c01 |
| EC Number | 208-731-6 |
| Gmelin Reference | 1576 |
| KEGG | C19152 |
| MeSH | D013742 |
| PubChem CID | 9504 |
| RTECS number | YR8750000 |
| UNII | H9FAX9577A |
| UN number | UN Hazard Number: 3297 |
| Properties | |
| Chemical formula | N2F4 |
| Molar mass | 104.01 g/mol |
| Appearance | Colorless gas |
| Odor | Odorless |
| Density | 2.73 g/L at 25 °C |
| Solubility in water | slightly soluble |
| log P | 1.778 |
| Vapor pressure | 31.6 psig ( 25°C) |
| Acidity (pKa) | -2.0 |
| Basicity (pKb) | 10.3 |
| Magnetic susceptibility (χ) | -66.8·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.252 |
| Viscosity | 0.617 cP (25°C) |
| Dipole moment | 0.00 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 291.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -7.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -534 kJ mol⁻¹ |
| Hazards | |
| Main hazards | Toxic if inhaled, causes skin and eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS04, Danger |
| Pictograms | GHS04 |
| Signal word | Danger |
| Hazard statements | H280, H330 |
| Precautionary statements | P261, P280, P304+P340, P308+P313, P405, P501 |
| NFPA 704 (fire diamond) | 1-0-2-W |
| Autoignition temperature | 280 °C |
| Explosive limits | 4.5–60% |
| Lethal dose or concentration | LD50 (oral, rat): 180 mg/kg |
| LD50 (median dose) | 125 mg/kg (rat, oral) |
| NIOSH | SN2110000 |
| PEL (Permissible) | PEL: 0.01 ppm (as N₂F₄) |
| REL (Recommended) | 0.25 ppm (0.78 mg/m³) |
| IDLH (Immediate danger) | 40 ppm |
| Related compounds | |
| Related compounds |
Difluorodiazine Nitrogen difluoride Hydrazine Tetrafluorohydrazine hexafluoropropylene adduct |

