Tetraethylammonium Perfluorooctanesulfonate: Reflecting on Its Past, Present, and What’s Next
From Laboratory Curiosity to a Hot Topic
Looking back, the path of Tetraethylammonium Perfluorooctanesulfonate (TEA PFOS) hasn't exactly hugged the headlines like plastics or pharmaceuticals, but those who track developments in advanced chemical solutions have noticed its growing role. Decades ago, researchers playing with tetraethylammonium compounds likely didn’t expect to see this cation paired with a perfluorinated sulfonate anion. The perfluorooctanesulfonate structure, a legacy from the early days of fluorochemical exploration, brought chemical stability and unique surface activity. Blending this with the well-recognized TEA cation introduced a new family of salts with a mixture of traditional use in phase transfer and the environmental baggage that comes with robust fluorinated chains. Once the spotlight turned toward persistent chemicals, the industry needed to think beyond utility and start asking, what kind of shadow does this compound cast?
Unlocking the Character of TEA PFOS
As a chemical, TEA PFOS stands out thanks to its long, perfluorinated tail paired with the quarternary ammonium head. Its looks are deceiving—what appears just like another lab salt turns out to be resistant to acids, bases, heat, light, and almost anything else we routinely throw at molecules. Its surface-active properties give it a strong hand in areas such as electrochemistry and specialty coatings. Users count on its high thermal stability, and the compound doesn’t break down easily, even with prolonged exposure to harsh conditions. It dissolves well in polar solvents, a feature that opens the door for solutions in electrochemical devices, liquid chromatography, and even unconventional uses in oil and gas. This physical and chemical stubbornness, the same trait that makes it so useful, also makes it a long-term guest in soil and water—a fact that has rung alarms in environmental circles.
Preparation and Reactions: A Tightrope Walk
Synthesizing TEA PFOS isn’t rocket science, but it isn’t without its headaches. Researchers usually start with perfluorooctanesulfonic acid, a molecule already carrying some heavy environmental baggage. Neutralizing this acid with tetraethylammonium hydroxide or a similar base yields the desired salt. Each step needs careful attention because a slip can send hazardous substances into waste streams. Once produced, TEA PFOS resists most common chemical attacks, even standing tall against oxidative stress and hydrolysis. Its reactivity centers on exchange reactions, and it takes a pretty aggressive approach to nudge the perfluorinated tail off its anchor. From a research perspective, this low reactivity poses a challenge since developing less-persistent alternatives or breaking down waste requires more than brute force.
From Labels to the Lab: Understanding Risks and Standards
Bringing a bag of TEA PFOS into the lab isn’t trivial for those grounded in safety and responsibility. Its labeling in most jurisdictions has shifted over time, keeping pace with a growing understanding of perfluorinated compounds. What once passed as a specialized lab reagent now carries warnings about persistence, environmental impact, and health. Instructions remind chemists that gloves, goggles, and fume hoods are the bare minimum; nobody slings this stuff around like table salt. The real problem really starts at the disposal stage. No industrial incinerator and certainly no municipal landfill can truly put a dent in the molecular stability of a perfluorooctanesulfonate. Regulatory agencies link TEA PFOS to tough compliance standards, demanding tight record-keeping, traceability, and, sometimes, outright replacement.
Where TEA PFOS Finds Its Place
In practice, those reaching for TEA PFOS often belong to specialized corners of science and engineering. Where the situation calls for a highly persistent, conductive, or surface-modifying agent, this compound pops up: certain fuel cell electrolytes, surfactants for hard-to-wet surfaces, or in experimental studies of ion exchange. Some research teams use it to sharpen selectivity in electrochemical applications. Reports document its use in separation science, particularly as an additive in liquid chromatography for separating complex organic mixtures. Each new use drags along a new set of safety considerations, reflecting not just the advantages but the consequences as the molecule moves from lab to waste bin—or worse, into the open environment.
Digging Into Toxicity and Environmental Burdens
For as long as perfluorinated compounds have been in the public eye, their toxicity has ridden shotgun. Research on TEA PFOS reveals persistence in both the environment and living systems. Studies indicate that perfluorooctanesulfonate anions accumulate in water, soil, and even animal tissues, resisting metabolic breakdown. Both chronic and acute exposures tie back to developmental, immune, and liver impacts in animal studies. The tetraethylammonium cation, for its part, has documented biological activity and can affect nerve signaling in high enough doses. No serious researcher shrugs off the combined toxicity risk. The real frustration emerges from the stubbornness of these molecules: even advanced water treatment barely dent their concentration, so environmental buildup continues almost unchecked. In places where PFOS contamination has already reached drinking water, local communities face years—sometimes decades—of expensive clean-up and health monitoring.
Labeling, Synonyms, and Staying Clear-Headed
In journals and chemical catalogs, TEA PFOS travels under a host of synonyms: tetraethylammonium perfluorooctane sulfonate, TEA-PFOS, and others. Synonyms keep chemists on their toes, especially when researching regulations or toxicity data. As I’ve learned over years chasing down cross-referenced compounds, missing an alternate name sometimes means missing a regulatory restriction or a research finding buried under a less-common alias. One thing doesn’t change: every label, no matter how it spells out the compound, waves a clear red flag for careful handling and responsible disposal.
The Push for Better, Safer Uses—And Alternatives
Peer-reviewed publications now overflow with efforts to untangle society from the grip of persistent PFOS derivatives. As years roll by, I see more researchers working on breaking down these compounds, seeking out new, less persistent surfactants or ionic liquids, and devising methods for selective removal from wastewater. Some advances hold water, at least in controlled lab conditions, but no one has discovered a drop-in replacement that checks every performance box without raising a new concern. Regulation has stepped in, not always seamlessly, and industry feels pressure to phase out stubbornly persistent chemicals whenever viable. For every promising tweak, there’s a long wait for real-world validation and for downstream effects to become clear. The science community needs both sharper tools and broader policies, with unrelenting public scrutiny to keep environmental and public health in focus.
Looking Forward: Managing Legacies and Making Progress
Staring past the present, TEA PFOS serves as a stark reminder that molecules built for indestructibility don’t just solve scientific problems—they create new ones. Where there’s exposure, there’s risk, and no clever chemistry sidesteps responsibility. Industry can’t wish away the long half-lives. Regulators can only drag enforcement so far without buy-in from researchers, producers, and users who really grok the problem. Better detection, better methods for destruction, and new classes of less persistent compounds top the wishlist for labs and lawmakers who understand just how sticky this problem can be. Conversations between chemists, toxicologists, environmental scientists, and the public remain vital for making informed, equitable decisions. We don’t always get to choose the legacies chemicals leave behind, but we do get a say in how hard we fight to deal with them.
What is Tetraethylammonium Perfluorooctanesulfonate used for?
What It Does and Where It Goes
Tetraethylammonium perfluorooctanesulfonate sounds like a mouthful, but it’s not just a tongue-twister. This chemical shows up in labs, factories, and even in some products you wouldn’t expect. Most often, researchers and manufacturers turn to it when they need a strong surfactant—a substance that lowers surface tension between liquids or a liquid and a solid. That means it can help keep things from separating, or let materials spread out more easily, which gives it value in making things like specialty coatings and in electrochemical processes.
People who work in advanced chemistry or battery technology know that tetraethylammonium perfluorooctanesulfonate helps stabilize certain reactions. For example, during the development of new battery designs, this chemical can boost ion transport—a critical property for how well a battery holds and delivers power. My time working in a university research lab made it clear that without reliable surfactants like this one, some experiments barely got off the ground.
The Fluorinated Problem No One Loves
The trouble with compounds containing perfluorooctanesulfonate—the “PFOS” family—shows up in headlines over water contamination or lasting pollution. While tetraethylammonium perfluorooctanesulfonate is useful, the carbon-fluorine bonds at its core resist breaking down. The same properties that make it great for chemical stability or resistance to heat and water also mean it can stick around in the environment a very long time. In fact, scientists have measured these compounds in groundwater and even remote wildlife, raising serious eyebrows about their lasting footprint.
Public health researchers worry because PFOS compounds can build up in living tissues, including humans. Studies in animals suggest these chemicals can affect liver function, reproductive systems, and maybe even increase cancer risk. To support this, the Environmental Protection Agency in the U.S. and similar agencies abroad keep pushing for stronger regulations and more careful monitoring. I remember covering a story on local fish advisories, where even tiny concentrations of PFOS forced warnings against eating what came out of some rivers.
Industry Challenge: Usefulness Fighting with Hazards
Anyone making electronics, semiconductors, or laboratory chemicals faces tough decisions. Tetraethylammonium perfluorooctanesulfonate delivers performance they can’t always match using less persistent chemicals. For manufacturers, switching to something safer takes investment, and there’s no guarantee the safer option will do the job as well. Many companies now invest in research to find new surfactants that perform under tough conditions without leaving behind hazardous residues. Some are even partnering with research universities or green chemistry startups to speed things along.
From my perspective, the biggest push needs to come from both ends—manufacturers looking for safer alternatives and governments or consumers demanding accountability. Clear labeling and transparency help, but real change follows when pollution costs get factored into business decisions. For anyone curious about safer practices, some labs already use alternative surfactants, and more often than not, they share data publicly to help others follow suit.
What We Can Do
Working in science taught me the value of both innovation and responsibility. Tetraethylammonium perfluorooctanesulfonate brings benefits that matter to industry and research, but it also carries risks that spread far beyond the lab. Supporting more research into alternatives, holding industries to higher standards, and making sure regular people hear about these chemicals—these steps make a real difference. We need solutions that respect human health and the world outside the factory gates.
Is Tetraethylammonium Perfluorooctanesulfonate hazardous or toxic?
Why This Chemical Raises Concerns
Tetraethylammonium perfluorooctanesulfonate, a mouthful for anyone, looks like a technical detail buried in scientific paperwork, but its stories stretch well past the lab bench. Working in environmental advocacy, I’ve seen how substances like this slip through the cracks until their footprints show up in drinking water or soil. Tetraethylammonium by itself turns up in research settings as a nerve-blocker. Then it’s paired with perfluorooctanesulfonate, part of the infamous family known as PFOS compounds—one of those persistent environmental pollutants nobody wants hanging around.
Human Health Issues
PFOS compounds, recognized for their persistence, caught the attention of toxicologists years ago. Studies link them to problems like immune system suppression, thyroid disruption, higher cholesterol, and even some cancers. In my neighborhood, parents ask me if better water filters help. My answer: filters only do so much, since these chemicals resist breaking down and stay in blood for years. Tetraethylammonium perfluorooctanesulfonate combines the toxic legacy of both its components. Its long-term effects aren’t totally mapped out, but regulators sound alarms even before all the data appears because histories of related chemicals set a troubling precedent. China, the US, and the European Union all list PFOS as a contaminant of concern. People living near plants that used or manufactured PFOS compounds report higher health risks in their families; these stories keep cropping up in environmental health offices and local clinics.
Environmental Impact
The trouble with perfluoroalkyl substances is they stick around—water, soil, even rainfall can carry traces to wild places. As a science educator in my city, I met kids who liked fishing local ponds, but we’ve had to tell them certain spots aren’t safe anymore because of bioaccumulation in fish and small mammals. Toxicity affects aquatic life too. Algae stop growing, invertebrates start dying off, and sometimes birds and mammals higher up the food chain show illnesses linked to these very chemicals. Once these substances get into rivers and lakes, no easy fix exists. Conventional treatment facilities don’t filter them all out, and the chemical structure holds up against sunlight, microbes, or time.
Policy and Solutions
Communities, including mine, push for testing—not just at waste sites, but in routine water safety checks. Public pressure led some cities to upgrade water systems with advanced treatments like activated carbon and reverse osmosis. This helps, but it doesn’t solve the release problem. Stronger chemical safety laws make a difference too. The European Chemicals Agency added PFOS to its strict list of restricted substances. In the US, EPA targets industrial discharge and aims to ban certain uses.
Innovation offers hope. New filter materials and destruction methods pop up in scientific journals every few months, including techniques using high heat or electrochemical cells to break these molecules apart. Still, efforts need funding and political will. Listening to affected families motivates action. People want clean air and water, not technical jargon or chemical loopholes. Addressing these substances isn’t just about reading reports—it means making communities safer by demanding transparency and science-based policies in everything from industrial permits to kitchen faucets.
What is the chemical structure and formula of Tetraethylammonium Perfluorooctanesulfonate?
Making Sense of the Chemistry
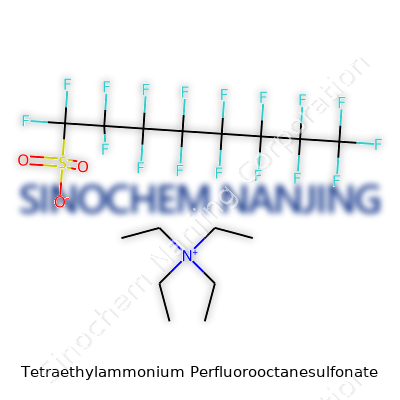
Anyone curious about the science behind powerful surfactants will run into compounds like Tetraethylammonium Perfluorooctanesulfonate. So what is this mouthful of a molecule? Let’s break it into pieces. Tetraethylammonium refers to a positively charged ammonium ion sporting four ethyl groups (each one a two-carbon chain). Its formula looks like (C2H5)4N+. This “tetraethyl” cation behaves a bit like a chemical Lego, plugging into a variety of negatively charged partners.
Here, that partner is Perfluorooctanesulfonate, a giant among anions, with the formula C8F17SO3-. “Perfluoro” says that every hydrogen on an octane chain (eight carbons in a row) has vanished, replaced by fluorine. That makes the chain bombproof against acids, bases, and even heat. The sulfonate end, -SO3-, gives the molecule its water-loving tail, while the fluorinated chain repels water like nothing else.
Packing the Ions Together
Put the positive tetraethylammonium together with the negative perfluorooctanesulfonate and you get the salt: (C2H5)4N+ C8F17SO3-. The chemical structure itself stretches out like most ionic compounds. The two ions don’t form a fancy covalent bond, but rather stack against each other thanks to their opposite charges.
Working with perfluorooctanesulfonate derivatives sparks a lot of controversy. The compound’s unique chemical stability doesn’t just mean high performance in industrial applications. That same stubbornness leaves it sitting in the environment for years. Unlike table salt or sugar, these molecules refuse to be chewed up by microbes. Research out of the US Environmental Protection Agency and several European agencies points to perfluorinated compounds as persistent organic pollutants, linked to real health risks once they build up in soil, water, or living tissues.
Why Know the Structure?
This isn’t just academic knowledge. If you work around chemicals like these, knowing the structure and the formula offers real-world value. The fluorinated chain explains why stains, grease, or even water beads up and wipes away when these molecules get involved. That’s useful in firefighting foams or surface treatments, but risky if you don’t want your groundwater contaminated.
Experience tells me that resolving the risks baked into the structure doesn’t start with fear or hype or with outlawing all fluorochemicals overnight. It comes from visibility and clear labeling, so downstream users can actually see what they’re dealing with. Transparency ties into trust. The alternatives—moving toward shorter perfluorinated chains or non-fluorinated surfactants—are picking up steam. Still, none can beat the performance we get from the perfluorinated heavy hitters—yet.
Regulators took notice years ago. Many industries have started working to avoid or replace perfluorooctanesulfonate-based surfactants. Safer disposal, rigorous testing, and even the search for biodegradable versions all rest on understanding the root structure and formula of the compound. That step often makes the difference between trusting chemistry for good and cleaning up after steep mistakes.
How should Tetraethylammonium Perfluorooctanesulfonate be stored and handled?
Why Proper Storage and Handling Matter
Anyone working near chemicals recognizes the edge that comes with Tetraethylammonium Perfluorooctanesulfonate (TEA-PFOS). This compound, used in niche research and industry, sits under global scrutiny because of the persistent environmental and health questions around PFOS substances. From my time running a modest chemical stockroom, forgetting best practices, even for a few minutes, can lead to spillages or exposure nobody wants to deal with—direct contact with PFOS materials raises red flags for both human health and the environment.
Smart Storage Steps
Good storage starts with sealing. TEA-PFOS comes in powdered or crystalline forms, creating dust that travels. Leaving containers open doesn’t just threaten sample integrity; it puts maintenance staff and coworkers at risk. Containers should always be airtight, thick enough to resist accidental knocks, and clearly labeled. In my experience, labels should sit on both the lid and the container body, bold enough for anyone to see. Facilities focusing on safety lock chemicals in cool, dry storerooms far from sunlight, which prevents unwanted reactions and keeps humidity away. Places that cut corners with ventilation almost always regret it later, since proper airflow keeps buildup of dangerous gases or dust in check.
Limit the Risk: Hands-on Handling
No one wants to discover their gloves weren’t up to the job. Nitrile gloves rated for chemical protection, long-sleeved lab coats, splash-proof goggles—these create the first line of defense against splashes or accidental hand-to-face contact. Even experienced handlers get complacent and drop protocols. Having worked side-by-side with new lab techs, I believe in a buddy-check system: walk through the steps out loud with a partner to catch absent-minded mistakes.
Dusting and spills happen. Simple paper towels won’t cut it. Labs I worked in kept HEPA-filtered vacuums ready for dry material and spill kits specifically built for PFOS cleanup, not just generic ones. Taking shortcuts—like sweeping—kicks hazardous particulates into the air. Used personal gear and cleaning materials go straight into chemical waste bins, never the regular trash. Neglecting this step turns minor incidents into contamination headaches that last weeks.
Mind the Regulations
Following the rules isn’t just paperwork. PFOS compounds draw attention from regulatory agencies for good reason. Disposal always follows strict local rules, sometimes involving special hazardous waste pick-up services. Unapproved dumping means big fines and lasting damage. Staying in compliance also means keeping up-to-date with training and keeping safety data sheets where everyone can see them. In my old lab, biannual drills ensured that no one forgot these steps, no matter how repetitive they seemed.
Better Habits, Safer Workplace
Checking in on stored chemicals every week gives early warning if lids loosen or labels fade. Assigning responsibility for this task beats the “someone else will get it” problem every single time. Training new staff through hands-on demonstration builds confidence, much more than dry manuals. I’ve noticed that staff who feel ownership of their workspace rarely cut corners, and near-misses drop fast as a result.
PFOS substitutes keep entering labs, but TEA-PFOS remains in use in enough places that the old lessons still matter. Listening to those with daily handling experience, supporting them with clear protocols and good equipment, keeps workplaces safe and fines off the board. That’s something every lab and warehouse can get behind.
What are the solubility and physical properties of Tetraethylammonium Perfluorooctanesulfonate?
Getting to Know the Compound
Tetraethylammonium Perfluorooctanesulfonate stands out from a lot of chemicals that cross a lab bench. This molecule brings together a perfluorinated tail and a charged ammonium group, handing it a real split personality. One half dodges water while the other side attracts it. That split creates some headaches and possibilities, depending on what you’re doing.
Water and Solvent Solubility
Its perfluorinated backbone shrugs off almost anything resembling water or traditional organic solvents. In plain terms, think of the way droplets of oil bead up on a raincoat – not mixing, no matter what. The tetraethylammonium portion drags a bit of water-sympathy into the mix, which means this salt does dissolve somewhat in water, but clumps and hazes can happen if concentrations push too high.
That odd balance pops up often in the research. Early reports show limited but definite solubility in water, more than the perfluorooctanesulfonic acid itself offers, mostly because the ammonium group gives the molecule some grip on water molecules. Yet that perfluorinated tail hates sticking around with polar solvents for long. For folks who’ve struggled with PFOS derivatives, it’s a familiar battle – slow dissolving, potential for micelle formation, always a guess on what will stay in solution over time.
It fares better in polar aprotic solvents like dimethyl sulfoxide and acetonitrile, but don't expect miracles. Ethanol or methanol can sometimes help, especially for making solutions for analytical work, but the tail will always resist full blending. The unique mix of hydrophobic and ionic nature often means special handling or sonication to coax it into solution.
Physical Properties Stand Out
The physical properties of Tetraethylammonium Perfluorooctanesulfonate draw directly from that hybrid structure. It typically presents as a white, powdery solid at room temperature, with a relatively high melting point thanks to the strong ionic interaction between the ammonium and sulfonate groups. The melting range, based on published values of similar salts, lands above 200°C. Not many working labs want to push temperatures that high, especially considering the stability of perfluoroalkyls often means thermal degradation releases perfluorinated fragments.
Density runs higher than many simple organic salts. You handle it with care, gloves and all, since the powder loves to cling to surfaces and can turn airborne when disturbed. Perfluorinated substances have a way of resisting chemical breakdown, and they don’t degrade under most routine conditions. Combine that persistence with its surfactant ability—dragged straight from the perfluorooctyl chain—and it ends up accumulating anywhere it’s used.
Why It Matters and Where Solutions Lie
Anyone working with this chemical faces dual responsibility: tapping its unique properties and avoiding environmental problems. Perfluorinated chemicals build up in soil and water, resist breaking apart, and show up in places no one expects, from rivers to Arctic ice.
The real challenge sits in disposal and clean-up. Labs need tighter waste tracking, proper neutralization steps, and alternatives that break down naturally. Research into better surfactant replacements matters—a lot of promising work focuses on short-chain perfluoroalkyls, which leave a smaller footprint. As manufacturers shift their attention, regulatory guidance pushes for greener chemicals and tighter emissions controls.
Scientists should keep eyes open for new ways to reformulate or phase out these stubborn molecules. Until then, it takes tough protocols and close monitoring to use tetraethylammonium perfluorooctanesulfonate wisely. Experience teaches that strict handling, transparency, and ongoing education work best to reduce accidental release and protect both people and the planet.
| Names | |
| Preferred IUPAC name | N-ethyl-N,N,N-triethylammonium 1,1,2,2,3,3,4,4,5,5,6,6,7,7,8,8,8-heptadecafluorooctane-1-sulfonate |
| Other names |
TEA-PFOS Tetraethylammonium perfluorooctane sulfonate |
| Pronunciation | /ˌtɛtrəˌɛθaɪl.əˈmoʊniəm ˌpɜːrˌflʊə.roʊˌɒkˌteɪnˈsʌl.fəˌneɪt/ |
| Identifiers | |
| CAS Number | 56773-42-3 |
| 3D model (JSmol) | `3DModel: "C(CN(CC)(CC)CC)(CC)(CC)N+(C)(C)(C)C.C(C(C(C(C(C(F)(F)S(=O)(=O)O)(F)F)(F)F)(F)F)(F)F)(F)F"` |
| Beilstein Reference | 4271461 |
| ChEBI | CHEBI:132702 |
| ChEMBL | CHEMBL2106018 |
| ChemSpider | 29233304 |
| DrugBank | DB11282 |
| ECHA InfoCard | 20a994d2-4edc-4e0d-bddc-2f7f2418d135 |
| EC Number | 62037-72-1 |
| Gmelin Reference | 1206078 |
| KEGG | C14343 |
| MeSH | D000072632 |
| PubChem CID | 21903355 |
| RTECS number | YJ8980000 |
| UNII | 872WN8S8KZ |
| UN number | UN3464 |
| Properties | |
| Chemical formula | C8H20F17NO3S |
| Molar mass | 564.41 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 1.62 g/cm3 |
| Solubility in water | Soluble in water |
| log P | -2.2 |
| Acidity (pKa) | -3.3 |
| Refractive index (nD) | 1.353 |
| Viscosity | 12 cP (20 °C) |
| Dipole moment | 1.98 D |
| Thermochemistry | |
| Std enthalpy of formation (ΔfH⦵298) | Tetraethylammonium Perfluorooctanesulfonate: ΔfH⦵298 = -1732 kJ/mol |
| Pharmacology | |
| ATC code | C10AX11 |
| Hazards | |
| Main hazards | Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS06,GHS08 |
| Signal word | Warning |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P261, P264, P271, P273, P280, P302+P352, P305+P351+P338, P312, P332+P313, P337+P313, P362+P364, P391, P501 |
| NFPA 704 (fire diamond) | 2-0-0 |
| Flash point | > 100 °C |
| Lethal dose or concentration | LD50 (oral, rat) > 2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 110 mg/kg (oral, mouse) |
| NIOSH | NA сел |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 0.1 mg/m³ |
| Related compounds | |
| Related compounds |
Tetraethylammonium chloride Perfluorooctanesulfonic acid Tetramethylammonium perfluorooctanesulfonate Tetraethylammonium bromide Ammonium perfluorooctanesulfonate Sodium perfluorooctanesulfonate |

