Tetracyanoethylene: A Deep Dive Into a Pioneering Chemical Compound
Historical Development
Chemists first pulled tetracyanoethylene, or TCNE, out of the test tube back in the late 1950s. The research world was hungry for new organic molecules with sharp electron-withdrawing abilities. The arrival of TCNE wasn’t just another chemical on the shelf; it gave rise to a flood of exploration in charge-transfer chemistry, organic electronics, and materials development. From the start, investigators realized TCNE didn’t behave like most run-of-the-mill organic compounds. It quickly became a go-to molecule for exploring molecular conductivity and electron-accepting properties. This compound shaped entire fields, especially in organic superconductors and molecular magnets, which grew out of that mid-century heyday.
Product Overview
TCNE stands out as a crystalline, colorless to pale yellow solid with a sharp, slightly unpleasant smell. Its symmetrical, compact structure hints at why it draws attention in fields demanding reliability and high reactivity. As an electron acceptor, TCNE frequently lands in advanced organic synthesis, solar cell development, and cutting-edge polymer chemistry. Chemists, engineers, and researchers see TCNE as a building block, a test case for new ideas, and as a bridge between bench-top discovery and scaled-up industrial innovation.
Physical & Chemical Properties
The solid form has a melting point hovering around 200 degrees Celsius. Dense packing in the crystal lattice hints at its low vapor pressure and durable stability under dry, inert conditions. TCNE comes alive under the right triggers—it dissolves well in polar organic solvents; throw it into acetonitrile or dichloromethane, and you get quick dissolution. That’s a sign of its strong dipole forces and high reactivity. Results from studies show that TCNE’s four cyano groups magnetically pull electrons, giving it a knack for forming charge-transfer complexes. That rare mix of thermal stability and strong oxidizing power makes it stand apart from typical organic chemicals. In practice, chemists choose TCNE for work that demands certainty and strength at the molecular level.
Technical Specifications & Labeling
Commercial TCNE arrives tagged with its name, synonyms, and key hazard warnings. Reliable suppliers detail purity (often above 98%), batch traceability, and date-coded packaging. Labels cover molecular formula (C6N4), CAS registry number 670-54-2, and warn about irritant properties. Handling recommendations, such as storage in tightly sealed containers under nitrogen, mirror its sensitivity to moisture and light. These handling protocols protect both research work and the health and safety of workers.
Preparation Method
Labs typically synthesize TCNE through the direct reaction of malononitrile with chlorine or bromine under controlled temperature and solvent conditions. The process forms an ideal blend of yield and purity. Industrial setups refine steps like slow halogen addition and rigorous removal of byproducts to coax out maximum product per kilo of raw material. Careful distillation and crystallization follow, yielding the unmistakable crystalline solid. The method fits small-batch, high-value scenarios—reflecting the tightrope of purity and material cost that drives the specialty chemicals market.
Chemical Reactions & Modifications
TCNE’s structure leans into cycloaddition or nucleophilic addition reactions, forming charge-transfer adducts that scientists tap for organic electronics and sensors. The reactivity stems from the electron-deficient double bond and the four hungry cyano groups. Reducing TCNE leads to salts with strong electron-acceptor properties. Pair it with electron-rich aromatics, and chemists see vivid color shifts—evidence of instant charge transfer. Specialized derivatives come out of targeted functionalization, building materials for advanced electronics. This chemistry offers a toolkit for those pushing boundaries in crystal engineering and organic semiconductors.
Synonyms & Product Names
Besides the formal tetracyanoethylene, the chemical world knows it as TCNE, ethene-1,1,2,2-tetracarbonitrile, and by its registry numbers in trade catalogs. Researchers swap these names depending on their task, but every label refers to the same robust molecule. In the annals of organic chemistry literature, those shorthand nicknames tell a story about the wide reach of TCNE across projects, labs, and scientific generations.
Safety & Operational Standards
Handling TCNE demands old-school lab discipline and modern chemical hygiene routines. Direct skin exposure, even to solids or dust, causes irritation. There’s also a risk of inhaling airborne particles, especially in poorly ventilated spaces. Strict protocols—fume hoods, gloves, goggles—aren’t optional. Standard operating procedures mandate storage away from acids, bases, and oxidizers. Emergency guidelines require prompt washing and medical advice for exposure. Waste goes into labeled, sealed containers for chemical incineration or professional disposal. These measures keep teams safe and support public trust in chemical research.
Application Area
TCNE doesn’t hide in a single corner of science or technology. Early research into organic superconductors depended on its electron-acceptor power. In recent decades, solar energy research leaned on TCNE to boost efficiency and longevity in organic photovoltaic cells. Analytical labs use TCNE for titrating electron-rich samples, proving its value as a diagnostic reagent. Turn to high-end polymer research, and there’s TCNE at the root of new conductive and magnetic materials. It even influences molecular sensing and high-stakes forensic trace detection. Any domain that relies on fast electron transfer finds a home for TCNE.
Research & Development
New directions in materials science push the limits of what TCNE-based molecules can do. Scientists fine-tune the electronic properties of TCNE adducts, hoping to unlock breakthroughs in flexible electronics and next-generation batteries. The chemical’s sensitivity lets developers design sensors that respond instantly to environmental changes. Discovery isn’t limited to a handful of tech labs—universities and startups worldwide dive into TCNE’s possibilities, competing to patent new uses in light-sensitive films, bioelectronics, and environmental monitoring technologies. Sharing data at conferences and in open-access journals drives that creative race and keeps the whole field moving forward.
Toxicity Research
Toxicologists study TCNE with clear priorities: understand risks, set exposure limits, and prevent health incidents. Test data on TCNE point to moderate acute toxicity by ingestion or inhalation. Researchers flag its irritant effects on skin, eyes, and mucous membranes. Animal studies feed into workplace exposure guidelines and regulatory limits. Medical teams use this data to plan emergency care protocols. Environmental fate grabs attention as well—research crews monitor any breakdown products to track persistence and potential long-term risks outside the lab. This focus on safe handling and risk management underpins all enterprise using TCNE.
Future Prospects
Looking forward, TCNE sits at the intersection of chemistry innovation and tech progress. As demands rise for green energy, efficient electronics, and advanced sensor systems, markets signal a hunger for TCNE-based technologies. Researchers experiment with molecular tweaks to unlock new electronic behaviors and boost charge mobility. Sustainability concerns inspire greener synthesis methods, cutting waste and toxic byproducts at every stage. The momentum isn’t slowing. Industry analysts track patents, new commercial launches, and academic breakthroughs, all mapping out a future where TCNE continues to spin up fresh ideas and practical solutions for scientific and technological challenges.
What is Tetracyanoethylene used for?
Beyond the Lab: Where TCNE Matters
Tetracyanoethylene, usually called TCNE, doesn’t pop up in daily conversation. Most people haven’t heard about it unless their job or curiosity brings them into the world of chemistry. Yet, this small molecule has built a strong reputation in research labs and more than a few production facilities. I remember talking with a grad student who called TCNE "good trouble"—brilliant in the right hands, tricky if treated carelessly. Its story proves that powerful tools sometimes take the quietest forms.
A Magnet for Electrons
TCNE draws attention because it grabs and holds electrons better than almost any molecule its size. Chemists call this “electron-accepting ability.” In my early years working on organic materials, we used TCNE to test how easily new compounds would give up their electrons. That one trait fuels a range of real-world uses, especially in electronics. In organic semiconductors and organic solar cells, TCNE changes how electricity flows through other molecules, tuning their behavior for faster, smarter circuits. Engineers count on this molecule to probe and enhance materials, especially for flexible and printable electronics, where every electron’s journey counts.
The Bond Breaker and Maker
TCNE jumps into action as a building block for bigger, more complex molecules—especially in pharmaceuticals and dyes. It takes part in what chemists call cycloaddition reactions, connecting carbon atoms in ways that open doors for new drugs and pigments. The first time I handled TCNE in a reaction, the color change was instant—deep red to black—proof of how reactive, and sometimes dramatic, this molecule can be. In my experience, few other chemicals drive these transformations with such efficiency or precision. Drug researchers lean on TCNE to push past roadblocks in making cancer therapies and high-performance imaging agents.
Cracking Codes: Analytical Uses
Scientists need ways to tell which molecules lie in a mix, and how they behave. TCNE partners up with other molecules, often changing colors when it reacts—a gift for anyone running a test. I remember my mentor showing me a classic trick: add TCNE to a mysterious solution. If the color changed, you just learned something useful. Chemical sensing kits and forensic tools sometimes rely on TCNE’s signature reactions to flag hidden ingredients, such as industrial solvents or illegal drugs.
An Eye on the Future: Energy and Materials
Interest in TCNE keeps growing as energy technology pushes forward. Research teams are testing TCNE-based compounds in the search for better batteries and supercapacitors. The goal: pack more energy in less space using organic molecules instead of rare metals. There’s a real chance, with continued work, that TCNE could help build rechargeable power packs light enough for wearables or medical implants. The demand for safe, efficient energy storage cuts across business, health, and personal electronics, and the hunt for new materials points back to molecules like TCNE.
Handling Risks, Building Trust
TCNE’s strengths come with responsibilities. As someone who’s used it, I know the molecule deserves respect: it can irritate skin, eyes, and lungs if handled carelessly. Proper training and ventilation protect workers in labs and on production lines. Following strict safety procedures matches both common sense and best practices. Companies with reputations for quality always lay out clear guidelines when handling reactive chemicals like TCNE. Reliability in chemical sourcing and transparency about risks protect both employees and end users.
Connecting the Dots
TCNE proves how a little-known chemical can quietly support progress across electronics, medicine, and industrial research. Its uses matter not just for science, but for anyone who benefits from better energy storage, faster computers, or safer drug manufacturing. Focusing on safety, training, and research sets the stage for new breakthroughs, keeping innovation grounded in real experience.
What are the safety precautions when handling Tetracyanoethylene?
A Closer Look at the Hazards
Tetracyanoethylene, or TCNE, isn’t something you bump into every day unless you’re working in a research lab or a chemical plant. It’s a strong electron acceptor, used for making certain organic compounds, semiconductors, dyes, and pesticides. This stuff doesn’t play around — one whiff or splash causes serious problems. The bitter almond smell gives a hint: it’s no friend to your lungs or skin. I’ve been part of a small lab team that once handled TCNE, and we treated it with the kind of respect reserved for a live wire.
Why Taking Precautions Really Matters
TCNE burns skin and eyes, damages your lungs, and doesn’t even take much to do it. Some chemicals irritate if you spend hours with open bottles, but TCNE gives no room for mistakes. The National Institute for Occupational Safety and Health (NIOSH) flags it as a chemical that can cause delayed lung injury. Spilled on the skin, it soaks in and poisons tissue. Breathing it hits the nervous system. In my experience, trust in goggles and a lab coat only goes so far; the whole setup needs airtight discipline.
Barriers That Work — Not Just Gloves
Rubber gloves never hurt, but regular lab gloves don’t always cut it. Heavy-duty nitrile or butyl gloves form the first shield. Anything less, TCNE finds its way through. Splash goggles block accidents, but the best defense pairs those with a face shield. I’ve seen one small droplet splatter on a bare arm during my student days, and the rash made an impression nobody forgot. No one should ever work with TCNE without a fresh change of gloves, goggles, lab coats, and a solid pair of sleeves under those coats.
Breathing Fresh Air Is a Top Priority
Forget fume hoods, and you’re flirting with disaster. TCNE vapors float up fast, and standard ventilation doesn’t cut them. I’ll never forget a professor who insisted on double-checking every exhaust before starting work. Chemical-resistant aprons, splash guards, and approved respirators fill in the gaps. If the job gets messy or large-scale, respirators with organic vapor cartridges beat out simple face masks every time. In my years in academic labs, we used fit-tested respirators when shifting more than a few grams.
Storage and Cleanup: The Unsung Routine
TCNE belongs in tightly sealed glass or compatible plastic containers, away from acids and bases. One slip-up mixing TCNE with strong bases can cause runaway reactions, and the resulting cyanide byproducts can be deadly. Only trained teams handle cleanup, using materials like vermiculite for spills and making sure not to toss waste into regular trash bins. Every spill gets reported and documented. Eyes and lungs can’t be replaced, so we always choose caution over speed — and it’s worth it every time.
Building a Culture of Caution
Respect for chemicals grows with clear rules and good habits. Regular training on chemical risks makes a bigger difference than a stack of warning posters. New folks shadow veterans and pick up the right way to do things — we watch out for each other’s gear and don’t cut corners. Keeping antidotes and emergency eyewash stations close at hand can end up saving someone’s sight, or life. Everyone on the team carries an understanding that safety isn’t bureaucracy, but real protection from chemicals that don’t forgive mistakes.
What is the chemical structure and formula of Tetracyanoethylene?
The Story Behind the Structure
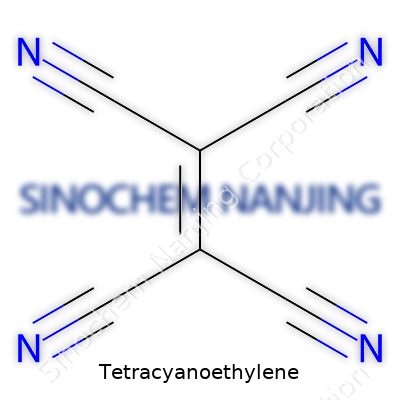
Ask anyone who’s worked in an organic chemistry lab about tetracyanoethylene, and you’ll probably get a knowing nod. This molecule, formula C6N4, turns up in some pretty interesting places. It’s not just the name that’s a mouthful—it’s the arrangement of atoms. The structure features two carbon atoms double-bonded at the center, flanked on each corner by a cyano group (that’s –C≡N for those new to chemistry lingo). In the shorthand of chemists, it looks like this: NC–C(CN)=C(CN)–CN. Think of it as a tight, flat framework, with every end packed with electronegativity, courtesy of the cyano groups.
Electrons on the Move
Tetracyanoethylene doesn’t crop up in everyday conversation, but it should. Saturated with those nitrogen-packed cyano groups, this molecule acts as a strong electron acceptor. That means if you ever need to pull electrons away from a substance—maybe to trigger a reaction, to make a new organic conductor, or to invent new dyes—tetracyanoethylene is high on the list of candidates. This property makes it essential in materials science and research that pushes the boundaries of electronics or synthetic chemistry.
The Importance of Structure in Real-Life Chemistry
With the structure and formula laid out, it’s tempting to move on. But real-world experience says there’s more at stake. Tetracyanoethylene’s flat symmetry and electronegativity give it unique reactivity. One key reason: those cyano groups lay a sort of chemical trap for other molecules. In my own college lab, mixing it with reactive species led to dramatic color changes—almost always a sign of deep electronic shifts. That’s no parlor trick; it’s the basis for creating new charge-transfer compounds that underpin organic electronics and sensors.
Hazards, Handling, and Responsibility
The appeal of tetracyanoethylene in advanced chemistry has a flip side. One touch on bare skin feels like an accident waiting to happen. Being reactive isn’t just great for making new compounds; it signals danger for careless handling. Anyone preparing or using this chemical should trust only glass or Teflon tools and go all-in on protective gear. The dangers might not always get center stage, but they stick with anyone who’s worked around high-energy organics.
Broader Impacts and Solutions
Why care about a molecule like this? In today’s push for better batteries, greener electronics, and smarter medical devices, electron-accepting compounds shape the entire field. Without molecules like tetracyanoethylene, progress in organic semiconductors slows to a crawl. Still, chemists need to stay sharp—safe handling protocols separate innovative science from a trip to the emergency room. Institutions could step up by mandating more practical training and revamping chemical storage standards. The structure might be simple on paper, but responsible use always calls for real-world experience and a well-stocked safety cabinet.
Wrapping Up a Complex Story
Tetracyanoethylene proves that a single, elegantly structured molecule carries outsized importance. Its formula, C6N4, unlocks doorways to new reactions and disruptive tech, but only for those who respect chemistry’s double-edged nature. Working with it highlights both the promise and the discipline at the heart of discovery.
How should Tetracyanoethylene be stored?
Why Tetracyanoethylene Demands Respect
Tetracyanoethylene stands out in any lab or industrial setting. It’s a strong electrophile, highly reactive, and doesn’t hesitate to cause trouble if handled without proper thought. For anyone who’s come across its sharp, acrid odor or heard stories about its sensitivity to moisture, it’s easy to see why careful storage isn’t just about following rules— it's about protecting people and the workplace.
Understanding the Risks
This compound reacts fiercely with water and forms cyanide compounds that threaten health. In my early research days, a jar of Tetracyanoethylene sat on a shelf, far from traffic, wrapped in caution tape; that precaution paid off. Moisture sneaks in through unsealed containers. Any slip opens the door to toxic gases that invade the workspace and linger. Several chemistry journals highlight accidental releases from poorly sealed glassware or when humidity creeps above safe levels. In countries with muggy summers, storage failures have forced costly cleanups.
Keeping Things Safe
Safety for this kind of chemical starts with the container. Glass with airtight, ground-glass stoppers stands above cheap plastics. Plastic breathes over time, and even supposedly tough grades warp. Glass won’t react or let vapors pass. The next step is a dry cabinet, fitted with desiccants. Silica gel or phosphorus pentoxide packets change color as they absorb moisture, helping spot leaks. Modern storage cabinets come with built-in humidity sensors, and it’s smart to log readings daily. A jar tucked alongside calcium chloride performs better than any lazy “room temperature” label.
A refrigerator serves well, as long as it’s not a frost-free model. Those cycle moisture through the compartment, defeating the point. An old-school, dedicated scientific fridge, marked clearly, does the job. Ventilation in the storage room cuts danger further, diluting any accidental vapor releases. I’ve seen too many labs stack chemicals in dingy corners, forgetting that Tetracyanoethylene reacts not only with water but also with basic substances. No cleaning agents or incompatible solvents belong near its vault.
Clear labels matter too. Mark the hazard phrases, add the date the container was opened, and alert others to necessary gloves and eye protection. Memory can fade faster than one thinks, but bold reminders keep routines sharp. Locks or badge systems keep unauthorized hands away. Whether a large plant or teaching lab, it’s easy to assume everyone knows what’s dangerous until someone learns the hard way.
Getting Real on Disposal
Disposal forms part of storage. Most spills happen when folk try to pour leftovers into generic waste or let them gather dust. Partnering with accredited hazardous waste specialists closes this loop. Staying up-to-date with local regulations pays off too. In my area, local fire marshals run periodic checks, and they expect clear logs and tidy chemical inventories.
Taking short cuts with Tetracyanoethylene stores up trouble. Reliable containers, dryness, accurate records, and exclusive access form the practical basics. Simple habits keep this tricky compound from turning into a horror story.
Is Tetracyanoethylene hazardous to health or the environment?
A Quick Dive into Tetracyanoethylene
I’ve come across a lot of folks who haven’t heard of tetracyanoethylene, but chemists know it as a sharp-smelling, yellow solid with a knack for making complicated molecules. The chemical stands out for its high reactivity, earned from its four cyanide groups. It finds its way into labs as a reagent, but some manufacturers have an eye on it for electronics and specialty materials. Still, what do people outside the lab need to know about tetracyanoethylene — especially when it escapes test tubes?
Where Health Worries Crop Up
The structure of tetracyanoethylene isn’t just there for show. Those cyanide groups spell trouble. If someone breathes in the dust or gets it on their skin, it can cause acute toxicity, ranging from nausea to breathing problems. The small molecules can slip through skin or get into the lungs, where the body eventually breaks them down and releases cyanide. Cyanide doesn’t stick around in the body, but its short-term effects pack a punch: headaches, dizziness, at high levels even convulsions or death. There’s no shortage of old case reports or animal studies linking acute cyanide exposure to serious health risks.
Lab safety data sheets flag tetracyanoethylene as dangerous for a reason. They recommend gloves, eye protection, and strong ventilation. I’ve never met a chemist eager to handle it casually. It’s not just about one big spill. Even small, repeated mishandling can build up harm, especially for workers who use it all the time.
Where the Environment Picks Up the Tab
Chemicals like tetracyanoethylene don’t leave ecosystems untouched. If it spills onto soil or leaks into water, tiny organisms feel it first. Fish and insects are especially sensitive to cyanide. The Environmental Protection Agency keeps a watchful eye on cyanide compounds for this reason. Tetracyanoethylene can break down, releasing free cyanide that disrupts aquatic life by shutting down enzymes essential for energy.
Once in the wild, the compound breaks down over days to weeks, but in that window, it can travel far and hit sensitive habitats. Past incidents with related chemicals have shown that even a few grams can wipe out fish in small ponds and streams. Soil microbes, which handle decomposition, also lose effectiveness in the presence of cyanide. That means slower breakdown of organic material and a weakened ecosystem — something farmers and gardeners can notice, often in fading plant growth and unhappy roots.
A Path Toward Safer Use
The risks tied to tetracyanoethylene aren’t just technical details for specialists. Oversight and habits matter. Manufacturing plants can cut risks by keeping the chemical sealed during transport and storage. Neutralizing station runoff suits industrial settings, removing cyanide before water returns to rivers. For small-scale labs, simple fixes like glove boxes and splash guards make surprises less likely. I’ve seen labs ban all unnecessary use altogether, turning to less hazardous reagents whenever possible.
Everyone benefits when disposal firms and municipal treatment plants stay aware of these kinds of hazards. Containment, not improvisation, keeps problems from spreading. Good training, a clear understanding of routes of exposure, and a willingness to swap out risky chemicals can lead to fewer headlines about contamination or worker injuries. At every step, prevention stands as the strongest tool. Without it, both people and local environments bear the cost.
| Names | |
| Preferred IUPAC name | 2,3,3,4-Tetracyanobut-2-ene |
| Other names |
TCNE Ethene, tetracyano- Cyanoethylene, tetra- 1,1,2,2-Tetracyanoethene |
| Pronunciation | /ˌtɛtrə.saɪˌænoʊ.ɪˈθiːliːn/ |
| Identifiers | |
| CAS Number | 670-54-2 |
| Beilstein Reference | 605774 |
| ChEBI | CHEBI:52723 |
| ChEMBL | CHEMBL12380 |
| ChemSpider | 25222 |
| DrugBank | DB14625 |
| ECHA InfoCard | 100.005.477 |
| EC Number | 202-155-1 |
| Gmelin Reference | 706927 |
| KEGG | C02355 |
| MeSH | D014234 |
| PubChem CID | 11820 |
| RTECS number | KB9450000 |
| UNII | 53C1WP1557 |
| UN number | UN1760 |
| Properties | |
| Chemical formula | C6N4 |
| Molar mass | C6N4, 128.09 g/mol |
| Appearance | White to pale yellow solid |
| Odor | Odorless |
| Density | 1.21 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 0.75 |
| Vapor pressure | 0.011 mmHg (25°C) |
| Acidity (pKa) | pKa = -0.27 |
| Basicity (pKb) | 13.3 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 1.605 |
| Viscosity | 0.83 mPa·s (20 °C) |
| Dipole moment | 3.97 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 281.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 110.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1393 kJ mol⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation, suspected of causing cancer. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS06,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | Precautionary statements of Tetracyanoethylene: "P261, P264, P270, P271, P272, P273, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P311, P312, P321, P330, P337+P313, P362+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-2 |
| Flash point | 82 °C |
| Lethal dose or concentration | Lethal dose or concentration: "LD50 (oral, rat): 60 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 320 mg/kg |
| NIOSH | VQ6475000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 0.1 ppm |
| IDLH (Immediate danger) | IDLH: 25 mg/m³ |
| Related compounds | |
| Related compounds |
Fumaronitrile Maleic anhydride Chloranil Tetracyanoquinodimethane |

