Tetrachlorophthalic Anhydride: A Down-to-Earth Look at a Versatile Chemical
Historical Development
Tetrachlorophthalic anhydride came into the limelight through the persistent efforts to develop flame retardants and synthetic resins during the rapid industrial expansion after World War II. The chemical industry recognized its value when searching for ingredients that could stand up to high temperatures and corrosive conditions in the 1950s. Researchers at the time faced growing demand from electrical, paints, and plastics sectors, all looking for something more reliable, and tetrachlorophthalic anhydride provided a crucial answer. Its introduction followed earlier work with simpler phthalic anhydrides, but this molecule’s four chlorine atoms gave it much stronger fire resistance and chemical reactivity, opening up new pathways in industrial organic chemistry.
Product Overview
In practice, tetrachlorophthalic anhydride offers chemists and manufacturers a highly efficient building block. It stands out among similar anhydrides for its ability to impart strong fire resistance to whatever it’s added to. It arrived as a white to pale yellow crystalline solid, easy to handle in bulk and with a reputation for both versatility and potency. Its adoption by producers of resins and flame-retardant plastics soon followed. This wide appeal comes from the molecule’s structure and from lessons learned using other, less effective additives in heavy-duty composites, coatings, and insulation materials.
Physical & Chemical Properties
With a melting point typically around 260 °C and a boiling point above 370 °C, tetrachlorophthalic anhydride routinely handles demanding environments. The crystalline powder dissolves in organic solvents such as acetone, offering flexibility in industrial preparation methods. Its four chlorine atoms ring the aromatic structure, which leads to strong electronegativity and excellent resistance to both acids and bases. Its density and solid-state stability make it a go-to for applications where long-term durability matters, particularly in electrical insulation, specialty coatings, and fire barriers in construction.
Technical Specifications & Labeling
Standard industry packaging typically comes in airtight, corrosion-resistant drums, with clear GHS (Globally Harmonized System) labeling signaling its irritant properties and the need for PPE during handling. CAS number 117-08-8 provides traceability in procurement channels. Suppliers often ship it with certificates covering purity, moisture content, and trace contaminants, demanded by quality managers who expect to meet tight regulatory standards. Strict adherence to these specifications helps users avoid unwanted impurities that could jeopardize end-product reliability and workplace safety.
Preparation Method
Manufacturers prepare tetrachlorophthalic anhydride by chlorinating phthalic anhydride, usually in the presence of catalysts. Over time, refinements in reactor design and purification techniques have made this process less energy-intensive and more consistent. In practice, the focus lies on keeping the process clean, minimizing formation of polychlorinated byproducts, and recycling leftover materials to cut down on waste. The adoption of closed systems and careful scraping away of contaminated deposits helps ensure a steady, high-quality yield with lower environmental impact compared to older batch processes.
Chemical Reactions & Modifications
Chemists value tetrachlorophthalic anhydride for its aggressive reactivity with nucleophiles—especially in synthesizing resins and polymers for specialty applications. It easily reacts with alcohols and amines to form corresponding esters and imides, serving as the backbone for strong, flame-resistant compounds. Its electron-withdrawing groups give rise to derivatives that withstand high voltages and thermal loads. Those working in R&D often build on its molecular foundation when targeting new performance coatings and adhesives that won’t degrade under stress.
Synonyms & Product Names
On technical datasheets and packaging, this compound goes by several aliases: TCPA, tetrachloro-1,2-benzenedicarboxylic anhydride, and 3,4,5,6-tetrachlorophthalic anhydride. Older reports sometimes list “chlorinated phthalic anhydride.” These synonyms help researchers navigate patent databases, supply chains, and regulatory filings, where consistent naming keeps procurement and documentation flawless across different languages and industrial standards.
Safety & Operational Standards
Working with tetrachlorophthalic anhydride means paying close attention to safety protocols due to its irritant nature, particularly for skin, eyes, and mucous membranes. Engineering controls, such as sealed process equipment and local exhaust ventilation, go a long way toward keeping exposure levels down in the workplace. Strict PPE rules—gloves, goggles, and chemical-resistant clothing—aren’t up for debate. Companies push for rigorous training and emergency plans so workers know exactly how to respond to spills or accidental exposures. International guidelines demand regular workplace monitoring and comprehensive safety data sheets available where the compound gets stored or processed.
Application Area
Tetrachlorophthalic anhydride finds its primary use in the plastics and resins sector, showing up in the formulation of specialty polyesters and alkyd resins. The electrical industry counts on it to boost safety in insulation materials, printed circuit board laminates, and wire enamels. The paint and coatings field values it for its contribution to intumescent and fire-retardant layers, which protect structural steel and other building materials from intense heat. Some agricultural chemicals rely on its reactivity, setting it apart from other, less effective ingredients that lack this level of resistance to thermal degradation.
Research & Development
R&D teams continue probing into new frontiers for tetrachlorophthalic anhydride. Researchers in polymer science routinely investigate modified formulations to knock down costs, cut emissions, and meet tougher green chemistry standards. Environmental scientists study how this compound breaks down in soil and water, while engineers work to optimize synthesis with next-generation catalysts that slash waste. The push for high-performance, environmentally friendly fire retardants means this compound still draws plenty of attention in labs and startup companies.
Toxicity Research
Studies indicate that tetrachlorophthalic anhydride can irritate skin, respiratory tract, and eyes, leading to firm action in workplace hygiene and storage. Animal testing and limited epidemiological data help paint a picture that chronic, high-level exposure could affect organ systems, though industry groups remain focused on prevention, not just treatment. Over the years, the trend points toward safer production, clearer labeling, and improved reuse or neutralization methods for effluents and residues. The chemical's persistence in the environment and possible bioaccumulation risks push operators to step up monitoring and remediation efforts, often under the watchful eyes of regulatory agencies.
Future Prospects
Looking ahead, demand for materials that blend top-notch fire resistance with environmental accountability will likely keep tetrachlorophthalic anhydride in the mix. As green chemistry gains traction, producers work on new formulations and process tweaks to minimize toxicity, cut carbon footprints, and improve recycling rates. The spread of electronics into cars, homes, and infrastructure raises the stakes for safety, pushing more research into safe handling, alternative synthesis, and less hazardous substitutes. But for now, tetrachlorophthalic anhydride offers a reminder that chemistry’s progress often rests on balancing risk, performance, and practical safeguards.
What are the main uses of Tetrachlorophthalic Anhydride?
Chemistry Behind Factory Floors
Tetrachlorophthalic anhydride often turns up wherever chemists want things that last or resist heat. Factory managers know this chemical for its special role in making resins and coatings much tougher than before. Talk to someone in the world of industrial paints or electrical insulators, and this tricky-to-pronounce compound comes up almost every time. My uncle spent years as a technician in a circuit board plant; he'd say a resin without the right additives crumbles fast under heat, and he's right—this molecule keeps those boards running longer and cuts down costly failures.
Fire-Resistant Materials Done Right
Look at buildings, trains, or planes, and you’ll see strict rules about what burns and what doesn’t. Fires spread fast with the wrong ingredients. Watch a sample test for electrical cable insulation, and you can tell the difference as soon as manufacturers swap in additives like tetrachlorophthalic anhydride. Its role as a flame-retardant agent isn't marketing hype—studies underline that this compound slows burning, prevents smoke, and stops that sticky, melting drip from wires, which once caused a scary short-circuit in our old kitchen. For people working around hot, crowded equipment, the peace of mind matters more than specs on a paper.
Boosting Plastics and Polymers
Plastic makers rarely stick with the basics. They’re always pushing for lighter, longer-lasting parts that don’t break the bank. Adding tetrachlorophthalic anhydride toughens up products that have to withstand abuse. In car interiors, seat foam, carpet backings, and some PVC pipes, this chemical makes things less likely to deform or discolor. You wouldn’t notice it day-to-day, but a soccer parent who’s cleaned juice spills or sun-melted toys from their SUV may owe some thanks to the compound’s staying power.
Pushing Boundaries in Dye and Pigment Sectors
Behind every bold color on a piece of plastic or fabric, there’s a slew of chemical tweaks to avoid fading or breaking down. Tetrachlorophthalic anhydride stands out for its ability to anchor pigments within a polymer structure. Companies turning out the bright, fade-resistant colors used in traffic cones, warning signs, or safety gear, rely on chemicals like this to make sure performance matches expectation.
Looming Concerns and Better Practices
No one in my family has fond memories of old labs with open solvents or bare hands; it’s just risky. The health side of working with this substance needs respect. Researchers and safety agencies have documented its strong odor and its knack for causing skin and lung irritation. Plants see tighter gloves, better masks, and sealed processes than ever. It’s not paranoia—Chlorinated chemicals can stick around in the environment, and we’ve all seen news about local waterways near chemical sites. The industry must stick with routine exposure checks, filter exhausts, and better training. As a country, we can push for more sustainable variants without losing out on safety or quality—chemists are already testing new formulas, but the old compound keeps showing its value, especially where performance still trumps cost.
What is the chemical formula and structure of Tetrachlorophthalic Anhydride?
Understanding the Core Details
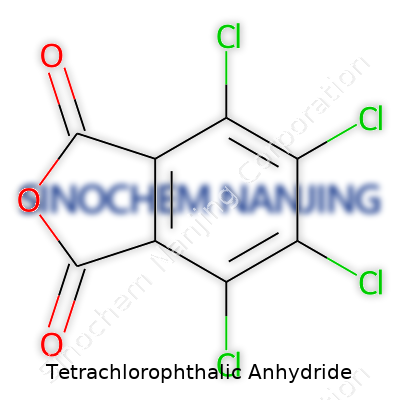
Tetrachlorophthalic anhydride is a tongue-twister, but its chemical formula is straightforward: C8Cl4O3. The name says a lot about the molecule. This isn’t an obscure compound found only in high-level research; it's a key building block in several industries. One look at the structure and you’ll see why it matters. Picture a pair of hexagons, fused at two corners—an aromatic benzene core that’s been chlorinated. Four chlorine atoms attach to the ring, replacing four hydrogens, altering both the molecule's reactivity and bulkiness. Then, the anhydride part finishes things off. Two carbonyl (C=O) groups join up with an oxygen, wrapping into a five-membered ring at the edge of the aromatic system. In chemist speak, it’s a derivative of phthalic anhydride, but with extra chlorines packing a punch.
Why the Structure Matters in Real-World Uses
It’s not just about drawing molecules on paper. Those chlorine atoms make the anhydride much more resistant to heat and chemical breakdown. In practice, this lets manufacturers use it to produce flame-retardant resins and high-performance plastics. I remember in an old laboratory job, catching a whiff of the pungent solid as we cracked open a shipment. Workers often see it as a fine white or yellowish powder that demands respect—heavy-duty gloves and a good fume hood. With C8Cl4O3 in polyester resins or insulation materials, products gain durability in harsh environments.
The Science That Backs Up Its Importance
The chlorinated phthalic system acts like a double-edged sword. High reactivity makes it valuable, but its persistence in the environment can present challenges. Data from government toxicity databases, such as the U.S. Environmental Protection Agency, show concerns about chlorinated aromatic compounds and their tendency to linger in water systems. Some reports link careless disposal of these compounds to ecological issues, from fish kills to groundwater risk.
Safety and Environmental Solutions
Handling such a chemical doesn’t feel abstract if you’re the one donning the respirator. I’ve witnessed the difference made by clear labeling and properly maintained extraction fans during chemical processing. Responsible companies often implement closed production systems. Spills or open-air releases can spread dust and vapors far from the original source, so capturing and neutralizing waste streams is essential. Government regulation shapes much of this practice, but it really comes down to the people on the ground. Those using tetrachlorophthalic anhydride, especially in composites and paints, tend to invest in training to prevent health hazards—skin and airway irritation being the top risks.
Ongoing Work
Safer alternatives remain a priority. Some newer research pushes toward less-persistent flame retardants or recyclable alternatives. For now, though, the molecular structure of tetrachlorophthalic anhydride keeps it locked into certain practical needs where heat and chemical resilience outweigh the drawbacks.
What safety precautions should be taken when handling Tetrachlorophthalic Anhydride?
Respecting the Risks
Tetrachlorophthalic anhydride sounds like something out of a chemist’s nightmare. For the rest of us just handling chemicals at work or in an academic lab, knowing exactly what we're getting into matters. This stuff comes as a white crystalline powder, but looks tell only part of the story. It irritates eyes, skin, and destroys peace of mind if it’s inhaled. I remember the headache that hit me the first week I started in an organic chemistry lab—turns out it related to improper ventilation. I learned that day: anytime you can smell something strong in a lab, assume it isn’t good for your lungs.
The CDC lists respiratory symptoms like coughing and shortness of breath after exposure. In serious cases, sneaky chemicals produce delayed symptoms that show up hours later. Chemical manufacturers include strong warnings for a reason. One mishap can put you out for weeks or cause permanent harm. It pays to go beyond the bare minimum and make sure your processes protect everyone—even on a rushed day.
The Basics: Containment and Protection
A fume hood stands as your best friend with any anhydride. If your face is above the beaker, move. Keep that glass sash between you and the powder. Moving it around outside of a fume hood even for what feels like a quick weigh-out is where trouble starts. A good dust mask never matches what a certified chemical respirator does. For my own peace of mind, I double-check cartridge specs before relying on a respirator.
Gloves should be non-negotiable. Nitrile or neoprene hold up best. Vinyl tears fast, especially if you’re wrangling powder from a stiff jar. I’ve caught myself once or twice starting a task without gloves—the sting of that mistake sticks. With all chemicals, but especially compounds like this, eye protection matters. Safety glasses or better yet, a real face shield. Just one splash turns a twenty-dollar lab session into an ER visit.
Training, Storage, and Dealing With Spills
Work only with folks who know the cleanup plan. Most incidents I’ve seen came from someone skipping training or brushing off the risk. Keep Safety Data Sheets (SDS) where everyone can reach them—and make sure people read those sheets not just once, but every few months. Proper labeling prevents confusion; unmarked containers invite mistakes.
Storage seems boring until you’re searching for the right lid with gloved, powdery hands. Keep tetrachlorophthalic anhydride away from water, strong bases, or acids. It slowly chews through some plastics, so glass or compatible plastics do a better job. Tightly sealed containers block moisture and accidental spills. I still remember the time a box of old chemicals had lids loosen up—the room stank for days. Waste should never get tossed in regular trash. Use a designated chemical waste bin, and coordinate regular pickups with hazmat-trained teams.
Long-Term Health and Real Accountability
Repeated exposure piles up. Even if somebody feels fine after a small spill, sensitive lungs or skin pick up the effects with time. Medical checkups give early warning before problems get bad. Supervising teams need more than a checklist—they require real understanding.
Good science and production mean more than results on paper—they mean people finish the day in the same health they started. For anyone working with chemicals, I recommend being that person who speaks up, who reminds coworkers about respirators or double-checking hoods before starting work. In science and industry, that quiet insistence saves more than inconvenience—it saves lives.
How should Tetrachlorophthalic Anhydride be stored?
Why Storage Matters More Than You Think
Storing chemicals like Tetrachlorophthalic Anhydride isn’t just a box-checking exercise for regulatory compliance. Over the years, I’ve seen what happens when safety gets overlooked—a neighbor’s small business in my town almost lost its entire stock because a volatile container triggered a chain reaction in an overcrowded storeroom. These lessons stick because the risks are real: exposure can damage skin and eyes, inhalation brings lung irritation, and improper storage risks fires or chemical spills. Factories, research labs, and even small workshops rely on sound practices to save lives, protect workers, and keep insurance costs from skyrocketing.
What Makes Tetrachlorophthalic Anhydride So Demanding
This chemical reacts strongly with water, releasing corrosive fumes. It gets under your skin—literally and figuratively—if it leaks. Standard plastic drums won’t cut it. Corrosion-resistant steel containers, kept tightly sealed, do the heavy lifting. Glass works for smaller batches in the lab, but scaling up demands tougher materials. Store it in a cool, dry place, away from anything that might leak, drip, or sweat condensation. Any moisture in the air or from a small spill spells trouble.
The Right Environment: Not Just Any Room Will Do
Experience tells me the real battle is against humidity. Tetrachlorophthalic Anhydride clumps or turns sticky in a damp environment. Ventilation helps draw away dangerous fumes if something goes wrong, but cross-breezes from open windows turn a minor accident into a neighborhood drama. Rely on exhaust fans designed for chemical fumes, and make sure ducts don’t dump vapors into public spaces. Never stack this stuff high or squeeze it onto crowded shelves. Heavy containers can tip and break, spilling contents—fixing that costs a fortune and risks lawsuits.
Separate from Incompatibles: No Chemical Soup
I learned early from a warehouse supervisor: never store oxidizers, acids, or moisture-loving powders with Tetrachlorophthalic Anhydride. Shelf space might fill up, but one careless stack-up causes chaos. Whether in a chemical supply room or a basement shelf, take a walk and physically check labels before shelving new deliveries. If possible, keep spill kits and neutralizing agents within arm’s reach. Rushing to find a spill kit in another room wastes precious minutes during an emergency.
People, Training, and Personal Protective Equipment
Workers forget, get distracted, or skip steps when routines get busy. Refresher training keeps chemical safety top of mind. Proper gloves, goggles, and breathing masks should be as familiar as toolboxes. Every single worker in the building should know the location of the emergency eyewash and shower station. Stories circulate of staff blindly searching for help while corrosive chemicals eat away at clothing. Don’t let that scenario unfold.
Moving Forward: Take Storage Seriously
Safety with Tetrachlorophthalic Anhydride starts with respect, not fear. Every safe storage area reminds us that preparation beats improvisation. Track inventories, look for leaks and cracks, and don’t gamble with shortcuts. Many accidents start with convenience edging out caution—a leaky cap here, a blocked ventilation duct there. Take the small daily steps to keep chemicals, people, and businesses safe.
What are the potential health hazards of Tetrachlorophthalic Anhydride exposure?
Breathe Deep, But Know What’s Around You
Factories, paint shops, chemical sites—many places use tetrachlorophthalic anhydride. The word sounds complicated, but the concern is simple. If this stuff gets in your lungs, eyes, or on your skin, it can give you trouble. Years ago, I worked a summer job hauling waste drums. We wore thick gloves, masks, eye protection. Nobody liked the smell or the warning stickers, but we tried not to think much about what might happen if things went wrong.
Lungs Feel It First
Get a whiff of dust or vapor, and your airways get scratchy fast. Workers exposed to airborne tetrachlorophthalic anhydride often cough or develop a sore throat. Shortness of breath, chest tightness, even asthma-like symptoms show up in some people. According to research from the National Institute for Occupational Safety and Health, this compound can sensitize lungs over time—meaning it might not bother you much at first, but repeated exposure raises your odds of developing problems. It’s not some vague risk; real folks have ended up with chronic bronchitis.
Skin and Eyes on the Front Lines
If you’ve ever spilled a chemical on your hands, you know trouble starts in seconds. This is no exception. Red, itchy skin or a painful rash develops fast after direct contact. Wash it off, and you’ll probably still feel stingy for hours. The risk of long-term skin problems increases with repeated splashes—manufacturing workers and cleaners face this hazard most. Eyes can burn, tear up, or swell shut with even a small splash or dusty breeze. More than one emergency room visit taught me that the eye flush station isn’t just a prop.
Digging Down: What Happens Inside the Body
Not many people think about their liver or kidneys at work, but studies show this chemical finds its way inside with enough exposure. Animal research points to possible liver and kidney damage at high doses. The data for long-term, low-dose effects is thin, but nobody’s eager to take chances. Some tests link the compound to possible reproductive harm—something rarely mentioned on job site posters, but important for families planning their future.
Staying Safe on the Job
Personal experience and data agree on one thing: safety gear matters. No mask? More coughing. No gloves? More burns. The Occupational Safety and Health Administration sets limits on airborne exposure for a reason. Regular air monitoring and proper ventilation beat out wishful thinking every time. I learned the hard way that shortcuts add up over weeks and months, often at the body’s expense.
Better Workplaces, Healthier Lives
Sticking to safety protocols keeps people out of trouble. Training helps workers notice problems before health issues sink in. Managers swapping out outdated processes for safer alternatives can lower risks for everyone. Workers speaking up about headaches, rashes, or trouble breathing push employers to improve conditions. Governments, too, have a role—they inspect, enforce, and fund research. No one expects every plant to turn high-tech overnight, but every step helps.
What’s At Stake
Talking openly about chemical hazards means fewer surprises and better health for workers, families, and communities nearby. Tetrachlorophthalic anhydride is just one of many chemicals on everyone’s radar, but it reminds us not to brush aside health warnings for the sake of short-term convenience. A cleaner, safer workplace pays off for all of us down the line.
| Names | |
| Preferred IUPAC name | 3,4,5,6-Tetrachloroisobenzofuran-1,3-dione |
| Other names |
TCPA 1,2,4,5-Tetrachlorobenzene-1,2-dicarboxylic anhydride Tetrachlorophthalic acid anhydride |
| Pronunciation | /ˌtɛtrəˌklɔːrəˈfθælɪk ænˈhaɪdraɪd/ |
| Identifiers | |
| CAS Number | 117-08-8 |
| Beilstein Reference | 135923 |
| ChEBI | CHEBI:34610 |
| ChEMBL | CHEMBL194485 |
| ChemSpider | 10176 |
| DrugBank | DB16640 |
| ECHA InfoCard | 19f8f93e-702f-465e-8371-4e7768402851 |
| EC Number | 204-118-5 |
| Gmelin Reference | 9307 |
| KEGG | C19271 |
| MeSH | D014180 |
| PubChem CID | 8599 |
| RTECS number | TP9625000 |
| UNII | 6C67VMD3R2 |
| UN number | UN3344 |
| Properties | |
| Chemical formula | C8Cl4O3 |
| Molar mass | 345.89 g/mol |
| Appearance | White crystalline powder |
| Odor | Pungent |
| Density | 1.8 g/cm3 |
| Solubility in water | Slightly soluble |
| log P | 2.96 |
| Vapor pressure | 0.000065 mmHg (25°C) |
| Acidity (pKa) | 1.00 |
| Basicity (pKb) | pKb: 12.8 |
| Magnetic susceptibility (χ) | -94.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.676 |
| Viscosity | 1.09 mPa·s (66°C) |
| Dipole moment | 2.61 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 370.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -587.8 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1763 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Toxic if inhaled, causes severe skin burns and eye damage, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS07, GHS08 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H334, H335, H351, H373 |
| Precautionary statements | Precautionary statements: P261, P264, P271, P272, P273, P280, P302+P352, P304+P340, P305+P351+P338, P310, P312, P321, P333+P313, P362+P364, P405, P501 |
| NFPA 704 (fire diamond) | 3-1-2-W |
| Autoignition temperature | 400°C |
| Lethal dose or concentration | LD50 oral rat 2300 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 2500 mg/kg |
| NIOSH | TI0175000 |
| PEL (Permissible) | PEL: 1 mg/m3 |
| REL (Recommended) | REL: 0.2 mg/m³ |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Phthalic anhydride Tetrachlorophthalic acid Tetrabromophthalic anhydride Hexachlorophthalic anhydride Chlorendic anhydride Chlorophthalic anhydride |

