Tert-Octylamine: A Critical Look at Its Journey, Properties, and Future
Historical Development of Tert-Octylamine
Chemists have sought out branched-chain aliphatic amines for specialized applications since the mid 20th century. Tert-Octylamine gained a foothold as crude oil refining ramped up demand for novel surfactants and extractants. Researchers worked out the fundamental reactions behind its synthesis from simple petrochemical feedstocks through careful observation of amination protocols. Synthetic methods matured in tandem with the chemical industry, favoring production pathways that could scale efficiently, such as reductive amination of tertiary octyl halides or oxo chemicals. Over the years, engineers ramped up output and consistency, meeting the strict purity requirements demanded by processors in everything from metal extraction to chemical synthesis.
Product Overview
Tert-Octylamine serves as a specialized amine, falling squarely into the category of higher aliphatic amines. Its structure, characterized by heavy branching around the nitrogen center, makes it quite different from linear cousins like n-octylamine. In many chemical plants, this molecule finds use as an intermediate—its non-polar hydrocarbon tail grants it unique properties among amines. While it gets less attention compared to shorter-chained or aromatic amines, its value reveals itself in extraction chemistry, corrosion inhibition, and surfactant design. Producers typically offer the compound in technical grade liquids or, less frequently, in solid states adjusted by temperature and storage conditions.
Physical & Chemical Properties
Tert-Octylamine presents as a colorless, oily liquid at room temperature. Its boiling point sits higher than most linear amines, reflecting the bulky tert-octyl group. The odor, sharp and characteristic of simple amines, signals exposure. With a density slightly below that of water and limited solubility in aqueous systems, it tends to float and form distinct phases. While its melting point allows for liquid handling at most ambient climates, the real interest lies in its basicity and reactivity. A strong nucleophile due to the electron-donating alkyl arms, tert-octylamine reacts with acids, alkylating agents, and often features in phase transfer catalysis. It resists rapid oxidation, giving it a certain resilience under harsh process conditions.
Technical Specifications & Labeling
Manufacturers list tert-octylamine under CAS number 6160-26-1, outlining parameters such as purity exceeding 98%, color index, nitrogen content, and specific gravity. Labels warn of caustic potential, route of entry for toxicity, and flammability. Product data sheets highlight recommended storage conditions, emphasizing cool, well-ventilated environments, and tightly sealed containers to protect from moisture. Hazard symbols and detailed handling instructions reflect a commitment to safety rooted in years of global transport and industrial usage guidelines.
Preparation Method
Industrial synthesis of tert-octylamine draws from robust alkylation chemistry. The process often begins with isooctene or tert-octanol, undergoing conversion to tert-octyl chloride via chlorination. This precursor reacts with ammonia or a suitable amine under controlled conditions, favoring the formation of tert-octylamine with minimal side products. Older methods made use of high-pressure hydrogenation on aldehydes or oximes, but modern routes rely mostly on catalytic amination, which scales efficiently and generates fewer byproducts. Purification involves distillation and, sometimes, additional filtration to remove residual catalyst or other organic impurities.
Chemical Reactions & Modifications
Tert-octylamine enters a variety of synthetic reactions. As a nucleophile, it participates in alkylation and acylation, forming quaternary ammonium salts and amides. The bulk of the tert-octyl group influences reaction pathways, steering selectivity. In aqueous solutions, its behavior as a base allows for neutralization with acids, creating ammonium salts valuable in phase transfer processes. Its incorporation as a ligand in transition metal catalysis continues to rise. Chemists modify the molecule through oxidation, or by attaching functional groups at the nitrogen center, expanding its use in surfactant chemistry and polymer modification.
Synonyms & Product Names
Across the industry, tert-octylamine is known by several synonyms, notably 2,4,4-Trimethyl-2-pentylamine and TOMA. Suppliers shipping globally may use alternate trade names reflecting formulations tailored for extraction, solvent, or agrochemical markets. On material safety data sheets, these alternate names often appear alongside structural formulas and registry numbers, clarifying identity and reducing confusion with structurally related amines.
Safety & Operational Standards
Operators handling tert-octylamine follow rigorous standards shaped by occupational health research. The amine can cause skin burns, respiratory distress, and serious eye injury on exposure. Facilities invest in proper storage, including explosion-proof ventilation and chemical spill management systems. Protective clothing, gloves, and goggles remain mandatory. Many countries require emergency eyewash stations and rapid spill containment near storage areas. Disposal protocols treat tert-octylamine as hazardous waste, ensuring incineration or chemical neutralization. Safety data keep evolving, with new studies on acute and chronic exposure influencing workplace practices.
Application Area
The versatility of tert-octylamine finds it a place in industries from mining to consumer products. In hydrometallurgy, it extracts rare metals from ore leachates, pairing with organic acids to pull nickel, cobalt, and copper from crowded solutions. In oil refineries, the amine acts as a corrosion inhibitor, safeguarding pipelines and reactors during crude oil processing. Surfactant manufacturers tweak its alkyl structure for customized detergents and dispersants. In specialty chemicals, its reactivity provides the backbone for the synthesis of agrochemical intermediates and ingredients for textile softeners. Even the pharmaceutical sector keeps tabs on derivative molecules with similar backbones, looking for novel bioactive agents.
Research & Development
Recent research explores greener synthesis routes, reducing environmental impact tied to petrochemical amine production. Teams have trialed biocatalysts and solvent-free reactors. The search for less toxic extraction agents in metals recovery circles back to tert-octylamine, as its tuning potential outpaces simpler amines. Analytical chemists use advanced chromatography and spectroscopy equipment to refine purity and quantify trace contaminants. Universities have probed the amine’s behavior in novel solvent systems and evaluated its compatibility with sustainable processes, drawing in funding from state energy and technology agencies.
Toxicity Research
Occupational medicine keeps a close watch on the toxicity profile of tert-octylamine. Acute inhalation leads to irritation and, in some cases, chemical burns to the respiratory tract. Skin absorption has prompted more studies in controlled exposure labs, exploring both short-term and repeated exposure effects. Animal tests highlight neurotoxicity and delayed systemic impacts if exposures exceed recommended levels. Regulatory authorities, guided by fresh toxicology data, tighten permissible exposure limits as a result. Environmental impact assessments measure persistence and breakdown products in soil and water, raising questions about handling spills near sensitive ecosystems.
Future Prospects
Tert-octylamine’s future ties closely to shifts in resource extraction, green chemistry, and regulatory change. With global transitions towards cleaner energy and recycling critical metals, demand for effective, selective extractants looks set to increase. Advances in catalysis and polymer chemistry unlock new uses tied to the molecule’s distinctive shape and reactivity. Stringent controls on emissions and waste disposal drive further innovation, spurring industries to invest in safer production and handling technologies. Beyond its traditional applications, synthetic biologists and process chemists eye the backbone for next-generation materials, seeking to reconstruct its synthesis with renewable feedstocks or engineer biodegradable analogs for cleaner manufacturing chains.
What is Tert-Octylamine used for?
Where Tert-Octylamine Shows Up in Modern Life
Tert-Octylamine rarely makes headlines, yet its fingerprints are all over the chemical world. In daily life, most people never hear about this compound, but its use matters, especially in how things run behind the scenes. I remember helping a friend who managed water treatment for a small town. He had to figure out which additives worked best to keep the water system clean without damaging the equipment. Tert-Octylamine came up on his shortlist. It plays a role in what chemists call “floatation agents.” In plain terms, it helps separate out unwanted stuff in ore processing and wastewater treatment. It clings to certain minerals and lets others fall away. This simple ability saves companies time, money, and keeps some pretty nasty materials out of our rivers.
The Oily Side: Making Lubricants Work Properly
I spent a summer at a machine shop, where metal parts moved non-stop and heat built up quickly. Lubricants held everything together and prevented meltdown. Tert-Octylamine finds its way into lubricants as a chemical building block. It grabs onto acids in oil, keeping the machinery from corroding. There’s more: it also stops foaming. If you’ve watched oil fizz, you know how bubbles mess up bearings and moving parts. This chemical flattens those bubbles fast, helping engines and turbines run longer before breaking down. Safe and efficient machines mean fewer replacements, lower costs, and less downtime.
Pushing Ahead in Pesticides and Herbicides
Out on the farm, crop protection rests on chemistry. Pesticides and herbicides often use Tert-Octylamine to help active ingredients dissolve in water. This amine supports even spraying and better sticking to leaves. I grew up around fields where weeds overran crops because the spray just slid off the plants. Formulators now use Tert-Octylamine to improve the mix, so less chemical ends up wasted, and more of it does the job. That helps keep yields high and chemical run-off low.
Handling Safety and Sustainability
With so many uses, safety stays at the top of the list. Studies in both Europe and North America show that Tert-Octylamine can irritate skin or eyes if handled poorly. Factories include ventilation and gloves when using it, following rules from OSHA and REACH. Education about handling these chemicals has grown, but there’s room for improvement. More investment in training and personal protective equipment could bring workplace accidents down further.
Big chemical companies now look at longer-term impacts, too. There are ongoing reviews about how this compound breaks down in nature and if it builds up in fish or soil. Some labs are testing greener alternatives for certain uses, like plant-based amines that promise a lighter touch on ecosystems. Investing in research like this could balance industry needs with environmental responsibility.
Why Tert-Octylamine Matters
While consumers rarely see its name, Tert-Octylamine backs up technology in mining, lubrication, and agriculture. If everyone along the supply chain does their part—chemists picking safer formulas, regulators setting smart limits, and workers following safety plans—the benefits can outweigh the risks. We depend on the hidden chemistry inside everyday systems far more than most would guess.
What is the chemical formula of Tert-Octylamine?
What’s Behind C8H19N?
Tert-octylamine comes with a formula that looks simple if you see it on a page: C8H19N. But there’s a lot going on behind that string of letters and numbers. If you’ve ever spent time in a chemistry class or worked with industrial chemicals, you know formulas show more than just building blocks—they give clues about a molecule’s behavior, how it reacts, and why people use it.
Everyday Impact, Industry Backbone
The amine group in tert-octylamine lets manufacturers use it as a building block in everything from agrochemicals to flotation solutions in mining. Its branched structure lowers its melting point and changes how it interacts with solvents, compared to straight-chain amines. I remember a colleague in the specialty chemicals business talking about how tert-octylamine helps separate valuable minerals from ore, speeding up a process that once took days. In some factories, it works almost like a traffic controller, making sure the right chemicals meet up at the right time.
Why the Structure Matters
The “tert” in tert-octylamine means the nitrogen attaches to a central carbon, not the end of a chain. That may sound like chemistry jargon, but that one change shifts a whole range of properties. For instance, it gives the molecule a bit more resistance to breakdown by microbes and sunlight. I’ve talked to plant managers who pick tert-octylamine for its consistency in harsh production environments. When a process runs around the clock, reliability carries a lot of weight.
Safety Stays in the Spotlight
With that many hydrogens packed into a small molecule, and a reactive nitrogen on board, safety never leaves the conversation. In my early days, a senior technician drilled into me that even simpler amines can irritate eyes and lungs if you don’t treat them with respect. Tert-octylamine can trigger allergic reactions in sensitive workers and reacts with acids to form potentially hazardous compounds. Every storage drum should come with clear guidance—good ventilation, chemical gloves, goggles. Regulations such as REACH in Europe require users to track how much comes in and goes out and make sure nobody faces unnecessary risks.
Finding Greener Paths
Companies continue searching for ways to reduce the environmental impact of nitrogen-based chemicals. Seeing tert-octylamine in this context, some plant operators are moving to closed-loop recovery systems, capturing vapors and recycling waste. In my experience, investing in these upgrades pays off long-term, not just in regulatory compliance but in well-being for people who work with these chemicals every day. Younger engineers push hard for safer alternatives and better operations, recognizing that switching to greener solvents or less volatile amines can make a measurable difference for both the environment and the bottom line.
Why It Matters Right Now
Every raw material touches the lives of thousands, sometimes millions, of people. While C8H19N is just a formula to some, it represents a chain of decisions, impacts, and responsibilities. Understanding that, and asking hard questions about efficiency, safety, and sustainability, matters as much as nailing the formula on a chemistry test. The chemical industry changes fast, but the basics—like putting people and safety first—never go out of style.
Is Tert-Octylamine hazardous or toxic?
Understanding Tert-Octylamine
Tert-octylamine belongs to a family of amines that help drive big chunks of chemical production, especially in factories working with metals, rubber, and cleaning agents. Factories value it for its ability to pull certain minerals out of ores and for making other chemical ingredients. Most people never run into tert-octylamine at home or on a walk, so the danger mostly affects those who handle it for a living.
Hazards You Won’t See on the Label
Some might think that if a chemical isn’t in every household cleaner, it can’t be that dangerous. Fact is, tert-octylamine causes direct harm. The fumes irritate the nose, throat, and eyes. Even small splashes or vapors trigger red, itchy skin or burning eyes. Swallowing-accidental or not-can bring serious nausea and stomach pain. Larger exposures can even make people feel dizzy or shaky. These aren’t vague, hypothetical dangers—they come straight from incident reports in real chemical plants. The European Chemicals Agency documents fire hazards and makes it clear: there’s a risk, even at room temperature, because it can give off dangerous vapors.
Long-Term Health Impacts
Many people focus on the splash risks, but that’s not the whole story. The Centers for Disease Control tracked workers who spent years around chemicals like tert-octylamine. They found that repeated contact can make skin conditions chronic and tough to treat. There’s also evidence from animal studies that tert-octylamine damages the liver and affects blood. While no direct proof links it to cancer in people, I’ve read enough warnings from toxicologists to know it’s smarter not to take chances. Chemicals that irritate tissues and cause cell changes can become a problem down the road.
Environmental Concerns
Factories don’t keep all the byproducts inside. Spills or careless dumping leak into soil or waterways. Tert-octylamine sticks around in the environment instead of breaking down quickly. Aquatic life—fish, tiny freshwater insects—react badly to even trace amounts. A recent European risk assessment flagged its persistence and toxicity to aquatic life. It’s part of why rules keep getting stricter about what goes down the drain in factories across Europe, the U.S., and East Asia.
No Substitute for Good Controls
Engineering can lower risk. That means sealing up pipes, keeping workers out of contact with any splashing, and using scrubbers or exhaust fans in factories where vapor comes off equipment. Protective gear—proper gloves, sturdy goggles—still matters. Decades ago, I saw too many places skip these steps in a hurry to meet quotas. Shortcuts always ended up costing more when someone got hurt or sick. Training helps most when leadership takes health as seriously as production numbers.
Looking Ahead: Safer Alternatives and Better Oversight
Tert-octylamine isn’t the only option out there. Some companies have started switching to amines that break down faster in the environment and bring less risk to workers. Still, change always meets resistance, mainly from folks worried about cost or reliable supply. Strong oversight, both from government inspectors and from within a company’s own safety teams, keeps old habits from creeping back. Chemicals like tert-octylamine remind us why clear labeling, easy-to-find safety sheets, and honest training sessions remain crucial—everybody deserves a safe place to work, whether in a small workshop or a sprawling industrial plant.
What are the storage and handling requirements for Tert-Octylamine?
Keeping It Safe in Real-World Conditions
Anyone who has ever worked around chemicals like Tert-Octylamine understands that storage isn't just about clearing a shelf in the warehouse. This amine comes with some real risks. It carries a strong ammonia-like odor, can irritate skin and eyes, and vapor exposure has a habit of sneaking up on you if you don’t manage airflow. For people on the factory floor, comfort actually means fewer headaches and less skin trouble, so solid storage methods have an impact on day-to-day life, not just abstract safety scores.
Simple mistakes create expensive cleanups and even injury. Tert-Octylamine needs a cool, dry area with enough ventilation so vapors never have a chance to collect. Fires can start easily because this amine can ignite when temperatures climb too high, especially near sparks or open flames. I learned in my first month in a lab not to cut corners—labeling matters, and open containers turn a minor job into a hazardous one. A clearly marked drum, sealed tight and away from oxidizers, saves everyone time and second guessing during inspections and even emergencies.
Handling Practices That Actually Work
Tert-Octylamine’s tendency to corrode or eat through less robust materials catches new teams off guard. Metal containers resist this, but regular checks for leaks or corrosion keep things from spiraling. Deliveries sometimes arrive in drums that look fine but show rust on the bottom edge—a detail field techs spot before transferring anything to storage. The lesson: trust the experienced handlers, and always check before moving or decanting.
Using the right gloves, goggles, and long sleeves seems obvious until you meet someone who skips the basics. I have seen burns and rashes from quick splashes. A good rule in the chemical world says: treat every opening or transfer of Tert-Octylamine like it’s going to spill, so secondary containment and drip pans aren’t just overkill. Most experienced workers install spill kits nearby, and routine safety drills pay off when the rare splash lands on someone or escapes to the floor.
Addressing Hazards and Reducing Risk
Fire is the main fear. Tert-Octylamine's flash point sits at a level where a summer day or an equipment failure pushes things over the edge. Storing away from direct sunlight, away from oxidizers, acids, or strong sources of heat, reduces this risk. Too many fires have started from a forgotten lightbulb or a space heater left running on a cold morning. Investing in temperature and vapor monitoring seems expensive until weighed against an actual fire or evacuation.
Contaminated air is no small issue. Workers caught off guard by vapor can experience nausea or irritation without warning, especially in tight rooms. With enough thought given to airflow—open fans, regular exhaust checks, no slow buildup of fumes—these problems almost vanish. Companies adopting clear air monitoring with meters often catch rises in vapor before they force evacuations.
Where Improvements Lead to Fewer Accidents
Relying on checklists that track age and condition of stored Tert-Octylamine makes a difference. Strong training turns the newest staff into someone reliable during incidents. Adding routine refreshing of old stock keeps chemical degradation from altering handling needs or raising danger unexpectedly. Pairing this with strict control over who accesses these chemicals, along with regular hands-on practice using safety equipment, leads to a more predictable outcome every single shift.
I've seen that basic respect for these guidelines, along with a work culture that values routine safety talks, has brought incident reports down to almost zero in well-run plants. Humans remember stories more than rules, so sharing what went wrong last year helps everyone avoid repeating mistakes. In the end, these practical precautions keep Tert-Octylamine manageable even for busy teams.
What are the main applications and industries that use Tert-Octylamine?
Mining and Extraction
Mining outfits lean on Tert-Octylamine for its strong performance as a flotation agent. It separates valuable ores from the rock in processes like copper, molybdenum, and nickel mining. I’ve seen engineers talk about how it lifts mineral yields and trims waste. Given the high cost of raw extraction and mounting environmental rules, any edge matters. Facts back this up—industry reports mention that specific amine collectors, including Tert-Octylamine, can bump up recovery rates up to several percent points. It might sound modest, but across a giant copper mine, that’s a lot of metal. This chemical’s ability to help companies extract more resources from the same ore brings both economic and environmental relevance—less rock processed means less energy burned and less waste dumped.
Rubber and Plastics Manufacturing
Factories making rubber rely on Tert-Octylamine as an anti-scorching agent. High heat and pressure cure rubber into tires and gaskets we count on each day. If rubber cooks too fast or unevenly, finished products crack or go brittle. In my talks with plant managers, consistent batches and fewer duds mean fewer recalls and a steadier supply chain. Chemists point out Tert-Octylamine’s positive impact on the cost and longevity of conveyor belts, seals, and tires. Plastics manufacturers use it in production lines that mold everyday goods—from car bumpers to furniture parts—because it keeps reactive ingredients under control and avoids product flaws.
Oil Refining and Chemical Processing
Tert-Octylamine works as a corrosion inhibitor in oil refineries. Pipes and machinery running crude oil face heavy-duty acids that eat through metal. Based on equipment maintenance data, facilities using effective inhibitors report less downtime and reduced repair bills. Reliable operations slow down leaks and big environmental spills. In the chemical industry, this amine acts as an intermediate for surfactants and dyes. Cationic surfactants made from Tert-Octylamine show up in detergents, disinfectants, and emulsifiers. These products clean up factories, homes, and even water systems.
Pharmaceuticals and Specialty Chemicals
Drug formulators turn to Tert-Octylamine as a building block for specialty chemicals. Its molecular structure slips into the synthesis of stabilizers, agents, and reaction promoters. I’ve heard from researchers who choose this amine because it reacts cleanly, with few byproducts. Efficient reactions mean purer ingredients for medicines or cosmetic creams, which both regulators and end-users want. In chromatography, which biotech firms use to test and purify everything from vitamins to vaccines, Tert-Octylamine helps set pH and improves separation performance.
Potential Risks and Safer Options
The workhorse uses of Tert-Octylamine don’t brush away the health and safety angles. People in plants must handle it with care, as direct exposure can irritate skin, eyes, and lungs. Regulators like OSHA and the European Chemicals Agency check its toxicity and set tough exposure limits. Although modern plants install fume hoods, gloves, and real-time monitors, older sites may lag. There’s also a push for greener replacements where possible. Chemical engineers look at surfactants from renewable feedstocks or less persistent amines to cut down on pollution.
Looking Forward
Tert-Octylamine, for all its utility, stands at a crossroads familiar to most specialty chemicals. The challenge is keeping up its benefits for mining, rubber, and other mainstays, while stepping up worker safety and reducing lasting impact on the environment. Smart investment in safer handling, process re-design, and ongoing research gives industries a safer path and opens the door to next-generation solutions that work for both people and the earth.
| Names | |
| Preferred IUPAC name | 2,4,4-Trimethylpentan-2-amine |
| Other names |
2-Ethylhexan-1-amine 2-Ethylhexylamine Octylamine, tert- 1-Amino-2-ethylhexane T-OA |
| Pronunciation | /ˌtɜːrt-ˈɒktɪl.əˌmiːn/ |
| Identifiers | |
| CAS Number | 616-34-2 |
| Beilstein Reference | 1718733 |
| ChEBI | CHEBI:148567 |
| ChEMBL | CHEMBL93116 |
| ChemSpider | 21222 |
| DrugBank | DB11256 |
| ECHA InfoCard | 03bcf9c8-ee62-46f6-b0ba-5b8d076c2d93 |
| EC Number | 204-685-9 |
| Gmelin Reference | 59098 |
| KEGG | C06217 |
| MeSH | D017150 |
| PubChem CID | 12314 |
| RTECS number | RG5425000 |
| UNII | 5DKT2IPZ6J |
| UN number | UN 2077 |
| CompTox Dashboard (EPA) | DTXSID7020185 |
| Properties | |
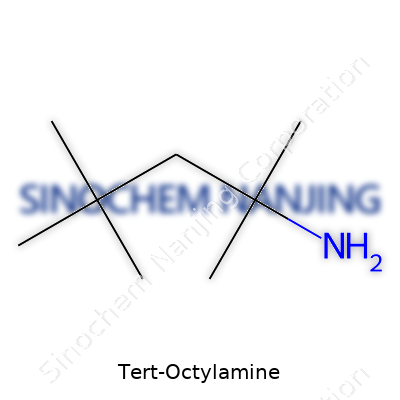
| Chemical formula | C8H19N |
| Molar mass | 129.25 g/mol |
| Appearance | Colorless to yellowish liquid |
| Odor | Ammonia-like |
| Density | 0.783 g/mL at 25 °C |
| Solubility in water | Insoluble |
| log P | 2.9 |
| Vapor pressure | 0.5 mmHg (20°C) |
| Acidity (pKa) | 10.60 |
| Basicity (pKb) | 3.34 |
| Magnetic susceptibility (χ) | -5.72×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.429 |
| Viscosity | 4.04 mPa.s (25 °C) |
| Dipole moment | 0.9169 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 253.6 J·K⁻¹·mol⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -213.20 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5442.0 kJ/mol |
| Pharmacology | |
| ATC code | N01AX13 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS05,GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H302, H314, H410 |
| Precautionary statements | H302: Harmful if swallowed. H314: Causes severe skin burns and eye damage. H411: Toxic to aquatic life with long lasting effects. |
| NFPA 704 (fire diamond) | 1-3-0-A |
| Flash point | 80 °C |
| Autoignition temperature | 280 °C |
| Explosive limits | Explosive limits: 0.9–7.0% |
| Lethal dose or concentration | LD50 oral rat 1,750 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat: 650 mg/kg |
| NIOSH | RN0150000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Tert-Octylamine: "5 mg/m³ |
| REL (Recommended) | 3 mg/m³ |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Diisobutylamine Triethylamine n-Octylamine 2-Ethylhexylamine tert-Butylamine |

