Tert-Butyl Isocyanate: A Sharp Lens on Its Role in Modern Chemistry
Historical Development
Back in the mid-20th century as chemists explored organic synthesis, a subset of isocyanates started making waves in labs, shaping industries far beyond the basic roots of urethane chemistry. Tert-Butyl isocyanate entered the scene as researchers needed more selective, less hazardous alternatives to methyl or ethyl isocyanate, which had already developed a notorious reputation for their volatility and toxicity. The t-butyl group brought a touch of balance to the family, offering reactivity where it counted but dialed back on some of the riskier side effects. It started in pharmaceutical labs, found its place among intermediates for drugs, and grew into a versatile role in organic synthesis as years passed. The real turning point came as demand for targeted, high-yielding reactions intensified, making tert-butyl isocyanate a staple on chemical shelves where high standards and worker safety could not be compromised without consequences.
Product Overview, Physical & Chemical Properties
Tert-butyl isocyanate (C5H9NO) stands out with its clear, colorless to pale yellow liquid appearance. What you get is something volatile, with a boiling point sitting around 76–78°C under normal atmospheric pressure and a density just at 0.87 g/cm³. The strong odor signals its reactive isocyanate group, which always underscores a need for caution in handling. Tert-butyl isocyanate dissolves easily in most organic solvents but keeps itself well out of water—it reacts strongly, a reminder of the powerful chemistry lurking behind its simple formula. It resists decomposition at room temperature, but under heat or open flame you'll experience a rapid breakdown and the violent release of toxic gases. Another strength lies with its reactivity pattern: the t-butyl group adds bulk, slowing down hydrolysis and making this compound more controllable than its nastier cousins without giving up what synthesis requires.
Technical Specifications & Labeling
In practice, purity matters. Laboratories and industry suppliers deliver tert-butyl isocyanate in sealed bottles or drums featuring tamper-proof closures, purged with inert gas to avoid the humidity of the air—a single careless moment allows it to absorb moisture, taking on water with reactive force. Spec sheets for reliable batches mark purity above 98%, minimal acid content (less than 0.01%), a controlled isocyanate content, and absence of stabilizers unless specially requested. Labels demand clarity—UN numbers, hazard codes for both inhalation and skin contact, and strict “Keep Away From Water” warnings. Emergency procedures stay front and center on every shipment, not just for compliance, but because you only need one mishap to see why those warnings earn respect in real lab work.
Preparation Method
Producing tert-butyl isocyanate comes down to creating a safe, straightforward pathway. Traditionally, it originated from the reaction of tert-butylamine with phosgene, a process managed in closed systems under rigorous environmental controls. Chemists moved toward more responsible approaches over the last two decades, seeking phosgene substitutes that reduce risk. Newer routes such as the Curtius rearrangement of tert-butyl azide, or the decomposition of tert-butyl carbamates using dehydrating agents, took over as green chemistry gained a foothold. Each pathway hinges on temperature control, dry conditions, and scrupulous gas handling—not only for product yield, but to prevent unplanned exposure. Not just a textbook reaction, every step links to ethical practice because waste phosgene or isocyanate runoff remains as dangerous in 2024 as it did fifty years back.
Chemical Reactions & Modifications
Chemists value tert-butyl isocyanate for its knack to work as a partner in a host of synthetic schemes. Its isocyanate group drives reaction with alcohols, amines, and water, forging carbamates, ureas, and amides with a level of control harder to maintain with smaller isocyanates. The bulky tert-butyl tail offers steric shielding, steering reaction outcomes when regioselectivity or suppression of side-products is crucial—vital in both laboratory optimization trials and at scale in active pharmaceutical ingredient production. Tert-butyl isocyanate can undergo further transformation, such as hydrolysis to the corresponding amine, or reduction to produce tert-butylamine selectively. It works as a blocking group for isocyanate chemistry, protecting target molecules during staged synthesis or complex molecule assembly, which matters a great deal in medicinal chemistry where precision marks the difference between a successful clinical candidate and a wasted run.
Synonyms & Product Names
Out in the wild, tert-butyl isocyanate goes by several aliases. Suppliers might list it as 2-methyl-2-propyl isocyanate, t-BuNCO, or simply t-butyl isocyanate. It shows up in safety data sheets and procurement lists under its CAS number, 1609-86-5, which offers the certainty that you’re getting the exact material—something buyers lean on because similar sounding compounds often bring very different risks and reactivity profiles. Naming conventions may vary, but the chemistry stays the same, keeping things straightforward for professionals who rely on clear identification during stock checks or incident reports.
Safety & Operational Standards
Direct experience with isocyanates teaches that no shortcuts exist in handling this class. Tert-butyl isocyanate causes skin, eye, and respiratory irritation; even brief unprotected exposure runs the risk of serious medical emergencies. Engineering controls in modern facilities put ventilation first—fume hoods, sealed handling systems, and respirators for workers running larger scale fermentation or chemical synthesis. Emergency showers and eyewash stations stand ready at arm’s reach in every well-run lab. Training sticks as the cornerstone. Personnel learn about immediate decontamination, proper use of gloves and goggles, and the first signs of trouble. For spills, procedures focus on immediate evacuation, localized cleanup by trained crews, and prompt notification of environmental hazards. Waste handling steers clear of water sources and involves dedicated incineration or specialized chemical neutralization. It's not just compliance; those measures mean real people go home safe at the end of each shift.
Application Area
Organic synthesis drives most of the demand for tert-butyl isocyanate. Pharmaceutical companies put it to work as a building block for the synthesis of ureas and carbamates, where selectivity and yield often shape the bottom line just as much as regulatory approval. Agrochemical developers look to it for controlled pesticide intermediates, offering active components that require a fine balance between stability and reactivity. In small-scale research, it steps in as a blocking reagent, a synthon for heterocycles, or a selective modifier for polymer production. Material science teams leverage its functional group to introduce specific cross-linking in polyurethane development, targeting niche markets like automotive coatings, insulation, and advanced foams that set the difference between commodity goods and high-performing specialty products.
Research & Development
Innovation cycles in organic chemistry keep tert-butyl isocyanate relevant. As scientists chase more sustainable, less hazardous synthesis, this compound works into developing one-pot processes and supports newer catalytic systems for greener, more diverse product lines. Drug discovery teams continue to modify its structure, searching for derivatives with altered biological properties—sometimes reducing toxicity, other times fine-tuning pharmacokinetics. In teaching labs, the relatively manageable risk and reliable reactivity of tert-butyl isocyanate make it ideal for instructing students about carbon-nitrogen bond formation, safe handling, and data analysis. Research on process intensification, including microreactor approaches, promises higher throughput with smaller footprints and less waste, showing how established chemicals like tert-butyl isocyanate adapt to new scientific frontiers. Funding agencies keep an eye on projects aiming to use less hazardous reagents altogether but acknowledge that compounds with strong track records and clear benefits still have a place at the bench—provided safety standards stay uncompromised.
Toxicity Research
Toxicologists have tracked isocyanate dangers for decades, and tert-butyl isocyanate sits right there with its relatives. Its acute symptoms—skin rashes, burning eyes, throat discomfort—show up quickly with improper use. Chronic exposure risks touch on asthma-like symptoms, lung sensitization, and immune system complications. Inhalation at high concentrations may cause delayed pulmonary edema, and even low-level exposure triggers allergies over time. These facts aren’t lost on the industry; regulatory policies push for periodic monitoring of exposed workers, air quality control, and real-time detection tools. Testing with animal models has shown moderate acute oral toxicity, but less concern for bioaccumulation or carcinogenicity compared to aromatic isocyanates. Environmental data urges careful storage and handling since runoff or spills threaten aquatic life, drawing more attention from environmental health agencies. The body of research supports smart use—good controls, clear risk communication, and constant vigilance in any operation involving this or any isocyanate.
Future Prospects
Interest in tert-butyl isocyanate remains strong as industries look for reliable, predictable partners for complex synthesis. Market trends suggest demand will stay steady or even rise as pharmaceutical and specialty materials work continues its upward curve. On the scientific side, teams are probing safer, solvent-free reactions and looking for even less hazardous alternatives, but the unique properties of tert-butyl isocyanate—balanced reactivity, manageable risk—keep it firmly in use. More attention focuses on digital process control, incorporating sensors to monitor reaction conditions and reduce human error. Regulatory scrutiny will probably tighten, reflecting lessons from past industrial accidents, but responsible preparation and clever application mean this compound is set to adapt rather than disappear. Conversations about greener chemistry will persist, and tert-butyl isocyanate will keep holding a place at the table, as the chemical industry prioritizes not only reactivity and cost but the broader impact on people and the environment.
What is Tert-Butyl Isocyanate used for?
A Closer Look at Tert-Butyl Isocyanate
Tert-butyl isocyanate doesn’t show up much in everyday conversations, even among folks who like science. You won't run into it on a stroll through the grocery store. This chemical mostly keeps to the quiet corners of industrial labs and research spaces. Yet, its role ripples through several sectors, and the choices made about it have effects beyond those four lab walls.
How It Earns Its Place
Most of the people who handle tert-butyl isocyanate work in organic synthesis. That means building molecules you can’t dig out of the ground or harvest from a plant or animal. Synthesis lets brainy people tweak the natural world and create new things—pharmaceuticals, pesticides, specialty plastics, coatings, and adhesives. Tert-butyl isocyanate acts as a tool to make specific parts of these molecules, working especially well for introducing a tert-butyl carbamoyl group.
If you have a painkiller that targets one tricky receptor, you’re probably looking at a product that got there with help from some stubbornly obscure chemicals. Tert-butyl isocyanate can help dial in the right shape of a drug molecule, control how slowly it breaks down in the body, or make sure it performs only its intended job. People who have needed a precise medicine—maybe for cancer or a rare infection—directly benefit from these building blocks, even if they never see a container of the raw chemicals.
Why it Matters for More than Just Chemists
The world doesn’t run without these chemical tools. While a lot of chemicals do similar jobs, replacing tert-butyl isocyanate isn’t always a snap. Chemists like it for how cleanly it reacts and how reliably it delivers the bits they want. In factories where margins matter and quality can’t slip, shaving off even a single wasteful side reaction matters. Fewer impurities in a medicine, less environmental cleanup after a batch is finished—these payoffs touch everyone, even if you’ve never studied a reaction scheme.
Handling isocyanates demands respect. The same reactivity that helps in drugs and plastics also means accidents can get serious quickly. Safer work practices and tight control help keep risks in check. Some companies have moved to closed systems and better ventilation, but older or smaller outfits sometimes cut corners. Stories about lung issues or skin problems in workers remind us that progress shouldn’t put people in harm’s way. Researchers and safety advocates keep pushing for better protocols, easier access to protective equipment, and—where possible—safer substitutes.
Weighing Progress and Responsibility
Earning trust from the public, regulators, and the medical community takes more than just inventions. Open information, smart risk management, and commitment to worker safety tell people that the work going on in those labs matters and is worth supporting. I’ve seen companies foster this balance—listening to workers, training them, and keeping lines open to those who understand the chemistry. Turning raw materials into tomorrow’s cures or technologies should never mean turning a blind eye to the people doing the work or to those downstream.
Progress in chemistry often starts with molecules that barely get noticed outside their field. Tert-butyl isocyanate’s story is one of usefulness balanced by a duty of care—one that’s worth understanding, and one that stays relevant as the world keeps relying on chemistry to solve tough problems.
What are the safety precautions when handling Tert-Butyl Isocyanate?
Understanding the Risks Firsthand
Handling Tert-Butyl Isocyanate (TBI) means working with a chemical that isn’t shy about its hazards. If you’ve ever caught even a faint whiff from an open container, you know it packs a punch—its vapors sting the nose and eyes, even from a distance. Those who work in labs or chemical plants have seen gloves melt and nitrile break down, so no one’s kidding around with TBI. It reacts with moisture and attacks your respiratory system before you even realize you breathed too deep.
Engineering Controls Over Guesswork
Ventilation always tops the list because fumes don’t wait around for you to open a window. A chemical fume hood, not just a standard exhaust, belongs in any room where TBI is opened. Don’t trust makeshift setups or homegrown fans; proper equipment prevents late-night runs to the emergency room. I’ve seen people pay for cheapskate shortcuts with swollen eyes and persistent coughs.
Dress Like You Mean It
Standard lab coats and garden-variety gloves offer false reassurance. TBI gets through porous materials. Go for nitrile gloves—double them up if you can—and trade cotton coats for chemical-resistant aprons or sleeves. Safety goggles with side shields aren’t optional. A splash on bare skin burns right through, and even tiny droplets trigger rashes or welts. After a bad experience cleaning up a sticky spill, I never skip face protection or gloves, no matter how small the job.
Preparedness Isn’t Optional
Spills rarely announce themselves. Absorbent pads rated for organics must be on hand. Use inert materials—Vermiculite or similar—since TBI shouldn’t meet water. Pouring water on a bench contaminated with TBI gives off toxic gas, and I watched a junior tech accidentally do this just once. A spill kit belongs within arm’s reach, not locked in a distant cabinet.
Don’t Play Hero with Personal Health
If there’s a whiff in the air, your lungs notice. Respirator cartridges suited for organic vapors protect you before you need a doctor’s help. Once symptoms appear, the damage is done. Hands-on experience says always choose overprotection—a good mask and a full-face shield trump regret. Should anyone get exposed, eyes and skin need an eyewash or safety shower. It saves hours of pain and months of recovery.
Respect Storage and Labeling
TBI breaks down quickly with moisture, so dry, sealed containers matter more than you’d expect. Never store it alongside acids, alcohols, or anything that might start a reaction. A clear label with “Danger: Toxic Vapor” keeps coworkers from reaching for the wrong bottle. Accidents happen when stockrooms double as storage for lunchboxes—keep food and drink out of chemistry areas.
Training Makes All the Difference
Reading material safety data sheets in a hurry doesn’t stick. Real training—where techs try on equipment and practice emergency procedures—makes a difference. I learned faster from senior colleagues who narrate mistakes than from dry manuals. New hires should watch demos and practice on fake spills before touching a real bottle.
Conclusion: Care Never Goes Out of Style
Safe handling of Tert-Butyl Isocyanate means treating every drop with respect. The risks aren’t just words on a label; they show up as real, lasting consequences. Every layer of protection you use buys you peace of mind, and that’s something you appreciate the most after you’ve seen things go sideways.
What is the chemical formula of Tert-Butyl Isocyanate?
Recognizing the Chemistry
Tert-Butyl isocyanate has a formula that looks simple on paper: C5H9NO. Breaking it down, you get five carbons, nine hydrogens, one nitrogen, and one oxygen. This doesn’t read as complex as some of the bigger industrial molecules, but don’t let that fool you—simple formulas like these are building blocks for some big things in chemistry and industry.
Use Cases and Real-World Impact
Tert-Butyl isocyanate isn’t a household name, but it finds its way into labs and factories that work on specialized organic syntheses. Chemists reach for it to add a specific “isocyanate” group to other molecules, which starts a chain reaction that brings out new pharmaceutical compounds and research intermediates.
Some of the most advanced medicines and crop treatment chemicals owe their existence to key reactions started with substances like this. If you’ve ever wondered how researchers design new drug candidates or protective agents for crops, it usually starts out with these small but potent chemical catalysts.
Safety Comes Before Convenience
Having spent time in academic and industrial labs, I have seen seasoned chemists handle isocyanates with a certain amount of caution. Isocyanates often cause issues if not used properly. Reactions with water, moisture in the air, or unprotected skin expose people to the risk of respiratory irritation or allergic reactions. All it takes is a tiny amount in the wrong place to spark real problems for someone’s health.
Companies train their staff rigorously on the use of personal protective gear and fume hoods. This is not bureaucracy for the sake of bureaucracy—it’s about keeping lungs and skin safe. Governments regulate storage and transport for good reason. Hidden risks in plain-looking chemicals become real very quickly if ignored.
Balancing Innovation and Responsibility
Research constantly pushes scientists to try new synthetic routes and build better products. Nobody can afford to ignore the environmental impact, either. Older manufacturing methods involving isocyanates sometimes release problematic byproducts. Today’s generation of chemical engineers and researchers keep a close eye on green chemistry protocols, reusing solvents and minimizing emissions as much as possible.
Efforts to design safer alternatives or streamline reaction processes continue daily. The push toward sustainability has forced each player in the chemical supply chain to rethink procedures, both in the lab and at the scale-up facility. Fine-tuned ventilation, closed systems for transfer, and proactive training make a real difference on safety records.
Working Toward Smarter Solutions
Tert-Butyl isocyanate’s formula—C5H9NO—looks straightforward. Using it responsibly calls for understanding more than just those letters and numbers on a chalkboard or spreadsheet. Anyone planning to work with chemicals like this needs clear protocols, sound judgment, and a willingness to double-check the basics every time.
Progress in pharmaceutics, agrochemicals, and specialty materials relies on this balance: unlocking new possibilities while guarding against old risks. The best labs never lose sight of either side of that equation.
How should Tert-Butyl Isocyanate be stored?
Nobody Wants a Spill on This One
Few chemicals make lab workers tense up quite like tert-butyl isocyanate. Years back as a junior chemist, I learned the hard way that it can react with water in a blink. A clumsy cap, a hint of humidity, and suddenly there’s a smell in the air that means trouble. I have spent extra hours sorting out cleanup—and chasing the headaches that followed. No one forgets their first bad experience with an isocyanate in the wild.
Why Tert-Butyl Isocyanate Demands Attention
This stuff brings some serious hazards. It reacts violently with moisture or even gentle warmth, producing gases that push pressure up in sealed bottles—and not the pleasant kind. Even a normal room, if left unchecked, spells risk for rupture. Vapors can irritate eyes, throat, and lungs. Gloves and goggles, sure, but it all comes down to the container and the shelf where it sits.
Safe Storage: Lessons from Real Life
Storing tert-butyl isocyanate always starts with airtight bottles. I learned early that only sealed glass with Teflon-lined caps stands up to this compound’s vapor. Metal drums, loose screw-tops, or weak seals simply cannot block its restless urge to react. On the shelf, humidity matters too. In my current lab, we use a tightly managed desiccator and silica packs.
Room choice plays a huge role. This isn’t a bottle for the open storage room next to the coffee machine or windowsill. A cool, dry, well-ventilated chemical cabinet—ideally explosion-proof—provides something close to peace of mind. Flammables sit far away. Any acetone or acids find their own home, because isocyanates tend to pick fights with all the wrong neighbors.
Labeling: The Details Save Lives
More than once, I have seen a tired colleague grab the wrong bottle from a crowded shelf. Clear, unambiguous labels and hazard warnings speak louder than lectures. All secondary containers need the same attention. If a team member feels uncertain about a label, that’s a failure upstream. Training and written reminders on proper labeling have become a weekly habit where I work.
Handling Emergencies Before They Begin
In my experience, every chemical accident tells a story about shortcuts. Emergency supplies should not hide under stacks of boxes—spill kits, absorbent materials, and proper neutralizers need to be within easy reach, not tucked away behind mop buckets. Lab mates practice drills for leaks or unexpected reactions. Real preparedness shows up in how fast a team reacts, not in how boldly it pretends disaster can’t happen.
Building a Culture That Outlasts One Person
Storage practices don’t stay strong through rules alone—they pass from worker to worker. My first mentor in chemical safety liked to say, “You don’t store a compound. You store consequences.” That stuck. Training new faces, walking through storage areas, and actually talking about near-misses make a world of difference. Policies matter, but stories and lived experiences keep people alert and respectful toward what sits on those shelves. Tert-butyl isocyanate doesn’t forgive those who forget.
What are the potential hazards of Tert-Butyl Isocyanate exposure?
Knowing the Risks: What Tert-Butyl Isocyanate Can Do
Some chemicals create risks that outlast the lab and linger through supply chains. Tert-Butyl Isocyanate gets used for its role in making pharmaceuticals, pesticides, and specialty coatings. People working with this compound face a long list of health warnings, and it’s not just about direct exposure—those risks can ripple out to others nearby.
If you have ever visited a plant or a lab where volatile chemicals are handled, the air can sting the nose a bit, even through the filters. Tert-Butyl Isocyanate lets out a sharp, pungent odor, but its real damage starts deeper. Breathing it in can irritate the nose, throat, and—most dangerously—the lungs. Shortness of breath and coughing show up quickly, and folks with asthma or allergies suffer even more. Some cases can spiral quickly to pulmonary edema. Even a brief lapse in ventilation or a loose valve can unleash that risk.
Skin and Eye Dangers
Everyone working with this compound should keep gloves and safety glasses as a second skin. Splashes rarely play by the rules. Tert-Butyl Isocyanate causes skin burns and severe eye injury. Simple contact brings redness, pain, and sometimes chemical burns that don’t heal in a hurry. If even a drop lands in the eye, blindness becomes a real threat. From personal experience, routine becomes the enemy—people tend to let their guard down on routine tasks, not just big spills.
Long-Term Exposure: What Slow Poisoning Looks Like
Frequent contact multiplies the risks. Over months or years, workers can develop occupational asthma, skin sensitization, and sometimes more subtle health issues that doctors struggle to link back to the factory. Even small leaks or poorly cleaned spills stick around, hiding in soft plastics or clothing. Over time, even short-term symptoms can harden into chronic health problems. Families sometimes feel the impact too—people unknowingly carry traces home on their clothes.
Combustibility and Environmental Fallout
Like most isocyanates, tert-butyl isocyanate lights up fast. Fire crews know that one mistake during storage or transport turns into toxic fumes, including carbon monoxide and nitrogen oxides. These chemicals hurt people in neighboring businesses, drivers on the road, or nearby homes. Small leaks into streams or storm drains threaten fish and wildlife, and those chemicals stick around far longer than most folks realize.
Practical Steps for Safer Work
No substitution beats basic awareness. Tight procedures, regular training, and clean habits help catch mistakes before they become disasters. Plus, regular air monitoring gives early warnings before anyone starts coughing. Proper chemical storage with working fire suppression keeps a spill from turning into a neighborhood emergency. From a management side, regular maintenance and strong communication lines keep everyone aware of the risks in real time. Wearing personal protective equipment helps, but strong systems, smart storage, and training on real-life incidents do more for safety than rules posted on the wall.
Turning a blind eye to the subtle, day-to-day risks behind tert-butyl isocyanate never pays off. Companies willing to spend on prevention and workers willing to speak up keep the worst accidents off the news. Safety culture grows from daily habits, not grand slogans.
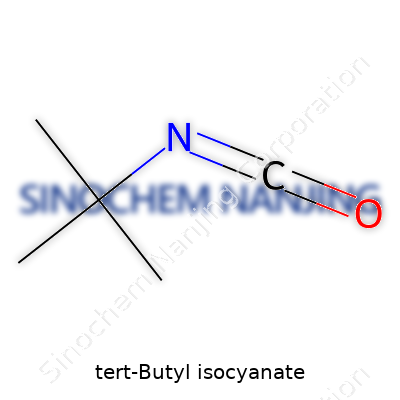
| Names | |
| Preferred IUPAC name | 2-Isocyanato-2-methylpropane |
| Other names |
Isocyanic acid, tert-butyl ester tert-Butylcarbamic acid, anhydride 1-Isocyanato-1-methylethane tert-Butylisocyanat 2-Methyl-2-propyl isocyanate |
| Pronunciation | /ˈtɜːrtˌbjuːtɪl aɪsoʊˈkaɪəneɪt/ |
| Identifiers | |
| CAS Number | 1609-86-5 |
| Beilstein Reference | 1461056 |
| ChEBI | CHEBI:141662 |
| ChEMBL | CHEMBL133207 |
| ChemSpider | 21106104 |
| DrugBank | DB04122 |
| ECHA InfoCard | ECHA InfoCard: 100.011.023 |
| EC Number | 2079-29-8 |
| Gmelin Reference | 107225 |
| KEGG | C18373 |
| MeSH | D015857 |
| PubChem CID | 12046 |
| RTECS number | YD9625000 |
| UNII | 4742LU704X |
| UN number | UN2988 |
| CompTox Dashboard (EPA) | DF7R43Z65G |
| Properties | |
| Chemical formula | C5H9NO |
| Molar mass | 101.15 g/mol |
| Appearance | Colorless liquid |
| Odor | Pungent |
| Density | 0.852 g/mL at 25 °C |
| Solubility in water | Reacts with water |
| log P | 0.97 |
| Vapor pressure | 4.2 hPa (20 °C) |
| Acidity (pKa) | 15.64 |
| Basicity (pKb) | 13.04 |
| Magnetic susceptibility (χ) | -7.74×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.357 |
| Viscosity | 0.782 cP (20°C) |
| Dipole moment | 1.3482 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 324.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -87.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4166.7 kJ/mol |
| Pharmacology | |
| ATC code | Not assigned |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS02,GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H226, H302, H311, H314, H317, H330 |
| Precautionary statements | P210, P261, P280, P304+P340, P305+P351+P338, P312, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-2-W |
| Flash point | 25 °C (77 °F; 298 K) |
| Autoignition temperature | 460 °C |
| Explosive limits | Explosive limits: 1.3–8.5% |
| Lethal dose or concentration | LD50 oral rat 640 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 640 mg/kg |
| NIOSH | NIOSH: WL3675000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Tert-Butyl Isocyanate: 0.005 ppm (ceiling) |
| REL (Recommended) | REL (Recommended): "C 5 mg/m³ |
| IDLH (Immediate danger) | IDLH: 25 ppm |
| Related compounds | |
| Related compounds |
Methyl isocyanate Ethyl isocyanate Phenyl isocyanate n-Butyl isocyanate Isopropyl isocyanate |

