Tert-Butyl Bromoacetate: A Closer Look from Lab Bench to Industry
Historical Development
Tert-butyl bromoacetate didn’t just appear overnight. Early synthetic chemists found value in alkyl halides for building blocks, and soon enough, tert-butyl esters started making an appearance in research-driven labs. It’s not just a story about a niche reagent; this compound grew out of necessity as organic and medicinal chemistry demanded more selective protecting groups and efficient routes to complex molecules. Since the late twentieth century, it’s become common for modern laboratories to stock tert-butyl bromoacetate, nudged by the need for flexible routes in esterification and further modifications. Experienced chemists can recall a time when purities varied wildly and technical specs included far more ambiguity than what’s found in carefully labeled bottles today.
Product Overview
Tert-butyl bromoacetate serves as an alkylating agent. It usually comes as a colorless to pale yellow liquid and holds a special place in synthetic planning for pharmaceuticals, agrochemicals, and specialized materials. Chemists reach for this reagent because bromoacetates are highly reactive, and the tert-butyl ester gives a unique handle for further transformations. Whether in research or production, this compound fills a gap that other esters and halides can’t always address, especially where mild deprotection or tuneable reactivity is needed.
Physical & Chemical Properties
This liquid’s molecular formula is C6H11BrO2, and it tips the scales at 195.05 g/mol. Volatility and density inform how it’s handled: boiling point sits around 146–148°C, with a density near 1.258 g/cm³ at 20°C. It’s got a faintly sweet, penetrating odor typical of organobromides, although seasoned chemists will agree that inhalation is unwise, no matter how faint. Miscibility trends—immiscible with water, freely soluble in common organic solvents like dichloromethane or chloroform—set it up for use in a range of transformations from simple alkylations to more exotic coupling reactions. It reacts strongly with nucleophiles, particularly those containing sulfur, oxygen, or nitrogen atoms.
Technical Specifications & Labeling
Modern suppliers list tert-butyl bromoacetate at purities of 97% or higher, which once felt out of reach. Labels provide more than a CAS number (5292-43-3); hazard warnings, UN number (UN 1760 for corrosive liquids), and recommended storage details—cool, dry, away from incompatible substances like strong bases or nucleophiles—stand front and center. Clear guidance for personal protective equipment—nitrile gloves, safety goggles, fume hoods—reminds users of regulatory compliance and good lab practice, echoing lessons learned from the rare minor lab accident. Manufacturers often list assay method, impurities limits, water content (ideally well below 0.5%), and storage recommendations as a standard part of their technical data sheets.
Preparation Method
Lab-scale prep typically involves reaction of tert-butyl alcohol with bromoacetyl bromide or direct esterification of bromoacetic acid with isobutylene under acid catalysis. For those who prefer a milder approach, coupling bromoacetic acid and tert-butanol using dicyclohexylcarbodiimide and catalytic acid brings strong yields, sidestepping the rougher world of direct halogenation. Each route carries trade-offs—dealing with bromoacetyl bromide means stinging fumes and more intensive ventilation, but the method brings decent yields and keeps purification manageable. Industrial setups often use continuous flow techniques, where mixing, reaction control, and product isolation happen far from the hands of operators, reducing risk and improving output consistency.
Chemical Reactions & Modifications
This ester acts as an electrophilic partner in a host of alkylation reactions. Nucleophilic substitution produces glycine derivatives, useful for peptide synthesis and in medicinal chemistry to build non-standard amino acids. The tert-butyl group resists a lot of reaction conditions, only cleaving under the right acidic circumstances like trifluoroacetic acid or hydrogen chloride in organic solvents. This trait lets chemists introduce and then later remove the protecting group, often at late stages, trimming synthetic routes and minimizing unwanted side products. Modifications can also include transformation into the corresponding acid or amide, thanks to the bromine atom’s reactivity in nucleophilic substitution. Many industrial and pharmaceutical organizations actively explore new catalysts and greener methods to make these transformations more sustainable.
Synonyms & Product Names
Other monikers show up in literature, including tert-butyl 2-bromoacetate, 2-bromoacetic acid tert-butyl ester, as well as GC and NMR references using abbreviations like tBuBrAc or TBBAc. In catalog listings, researchers might spot the name bromoacetic acid 1,1-dimethylethyl ester, or simply BAcOBu-t. Each name reflects a slightly different slice of the compound’s background, but the structure stays consistent regardless of the label.
Safety & Operational Standards
Working with tert-butyl bromoacetate calls for up-to-date safety protocols. Exposure to skin, eyes, or respiratory tract causes immediate irritation and, in some cases, lasting harm. Standard operating procedures enforce chemical-resistant gloves and double-check for splash potential, since even a small spill can linger in the memory (and on the skin). Laboratories and pilot facilities keep this material tightly controlled in ventilated spaces, backed up by local exhaust fitting and easily accessible eyewash stations. Good waste disposal practices make a difference: halogenated organic waste must stay separate from regular organic solvents to avoid violent reactivity or hazardous byproducts. Employers train staff on spill control and first aid, not as a bureaucratic box-tick but out of a real understanding of the risks.
Application Area
Pharmaceutical chemistry leans heavily on tert-butyl bromoacetate for introducing protected glycine or other building blocks in peptide synthesis. The compound’s reactivity matters most in routes where mild conditions extend the shelf-life of sensitive intermediates. When drug discovery jumps off the page to the pilot plant, synthetic methods using this ester make scale-up smoother and less hazardous compared to less selective reagents. Agricultural researchers find it handy for making pro-drugs or novel herbicidal scaffolds that need a reliable, removable protecting group. Material scientists use it for surface functionalization of polymers and preparing customized resins, making the connection between academic synthesis and industrial application clearer than ever.
Research & Development
Ongoing research circles around better synthetic routes, often with greener chemistry in mind. As environmental regulation tightens, organizations pivot to solventless methods or flow chemistry to cut down hazardous byproducts. Research teams publish new catalytic systems that lower requirements for strong acids or expensive reagents. Analysts in industry track improvements in purification, pushing for lower residual bromine or byproducts. In university and industrial innovation centers, scholars tinker with new reactions using this compound to increase the suite of derivatives accessible for pharma, diagnostics, or performance materials.
Toxicity Research
Batch data and animal studies indicate moderate acute toxicity. Inhalation or skin exposure leads to burns and mucous membrane irritation. Chronic or repeated exposure brings risks of liver or kidney impacts, nudging safety officers to reinforce protective clothing and emergency protocols. Some groups investigate longer-term environmental persistence and effects on aquatic life, given the growing attention on trace contaminants in wastewater. Regulatory filings, especially under REACH in Europe or TSCA in the United States, demand tight data on biodegradability and breakdown profile. Not only does this help shape workplace protections, but it also influences acceptable release limits and handling requirements downstream.
Future Prospects
Green chemistry stands poised to change the way tert-butyl bromoacetate crops up in supply chains. Researchers pursue milder, safer alkylation techniques and look for renewable feedstocks. The chemical industry now invests in more automated, continuous processes, trimming down direct contact and improving control over emissions. As demand increases for new peptide medicines, high-performance materials, and custom agrochemicals, the reach of these improvements only widens. Environmental responsibility and safety get more attention now than ever before. This means the future for tert-butyl bromoacetate involves more than just new applications but safer and more sustainable ways to keep using a compound with a proven record.
What is the chemical formula of Tert-Butyl Bromoacetate?
Chemical Formula and Structure
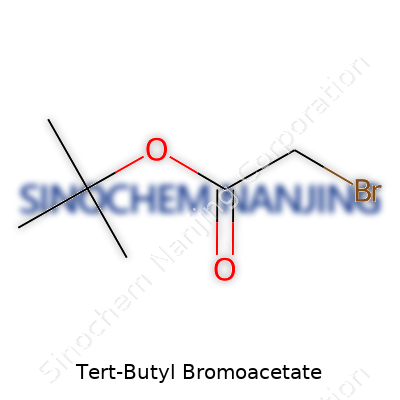
Tert-Butyl Bromoacetate uses the chemical formula C6H11BrO2. That formula doesn’t tell the whole story, though. Each part has a job: six carbon atoms, eleven hydrogen atoms, one bromine atom, and two oxygen atoms come together in one distinctive molecule. Scientists use the shorthand O=C(OCC(C)(C)C)CBr to show its structure. That means the molecule has both a tert-butyl group and a bromoacetate piece, which gives it its name and properties. The tert-butyl group provides bulk, while bromoacetate brings reactivity. Each of those pieces changes how the compound works in a lab setting.
Why the Formula Matters in Science
The formula tells chemists exactly what they’re working with. In organic synthesis, even a small change in a chemical’s formula can trip up a reaction and send it sideways. Tert-Butyl Bromoacetate gets used for specific syntheses—especially when making amino acids, peptides, and pharmaceuticals. It brings the bromo group to the table, which acts as a leaving group during reactions. That opens the door to new carbon-carbon or carbon-nitrogen bonds. The tert-butyl part acts like a shield, making the molecule more selective and reducing side reactions.
Applications and Impact
The pharmaceutical industry depends on precision. Small error in a formula can create big issues in downstream products. Tert-Butyl Bromoacetate helps people create building blocks for medications and biological research. It doesn’t just stay on the shelf; researchers buy it and use it every day in their labs. It speeds up the creation of drug candidates and supports the hunt for new therapies. My own time in a university lab taught me this: ignore a compound’s formula, and you may waste days repealing failed tests. Checking and double-checking the formula avoids expensive mistakes—something graduate students and professionals alike learn fast.
Risks and Responsible Handling
Chemicals deserve respect. Tert-Butyl Bromoacetate brings hazards, mostly because it contains bromine. If misused, it can irritate skin, eyes, and the respiratory tract. It can also react explosively with strong bases or metals. Safety data sheets—always read them—spell out all the risks. Doctors and researchers use gloves, lab coats, and keep a fume hood humming any time they uncap a bottle of this stuff. Following strict storage and disposal protocols helps avoid spills and exposures. Knowledge plus preparation equals safety. I learned that lesson early on, after watching a minor lab spill clear out a whole floor. Harmless compounds don’t exist, only careful chemists.
Room for Improvement
In science, new solutions show up all the time. Some researchers look for alternatives because of environmental concerns tied to halogenated reagents. Green chemistry offers another path by looking for non-brominated alternatives that do the same job without the extra hazards. Universities and companies encourage researching less toxic options, which can help reduce the burden on waste disposal systems. Progress often comes from people willing to ask what works better, safer, and cleaner.
Science Demands Details
For anyone working in synthesis, the chemical formula where each atom sits means the difference between a breakthrough and a failure. Tert-Butyl Bromoacetate demonstrates this. Its structure and formula direct research and production, affecting everything from drug development to materials science. Behind every bottle stands more than a label; there’s a story about precision, care, and pursuit of better answers.
What are the main applications or uses of Tert-Butyl Bromoacetate?
What Drives Its Demand in Research and Industry?
Tert-Butyl Bromoacetate rarely shows up in a grocery store conversation, but step into a chemistry lab or a specialty manufacturing plant, and you’ll hear about it pretty quickly. I remember my own days in academic research, standing at the bench and watching reactions kick off as soon as a fresh bottle of this reagent hit the table. Its taut structure—a bromoacetate core protected by a tert-butyl group—makes it a favorite for getting reactions to go in just the direction you want.
Tool for Organic Synthesis
The backbone of any chemical lab is its set of building blocks. Tert-Butyl Bromoacetate holds special value for anyone working on building molecules from the ground up. Chemists often use it to install tert-butoxycarbonyl (Boc) protected carboxymethyl groups in larger molecules. It brings a high level of reliability for producing intermediates that need further tweaking, so many patents for pharmaceuticals mention this compound in route descriptions. The synthesis of amino acids often leans on this reagent, making it easier to add specific groups or modify the structure in controlled steps. Creating α-amino acids often starts with this very molecule.
Role in Making Pharmaceuticals
In drug discovery and pilot-scale development, few molecules see use as often as Tert-Butyl Bromoacetate. It plays a major role in constructing active pharmaceutical ingredients (APIs) where selective protection and alkylation make or break success. For example, complex cancer drugs need delicate steps in their assembly, and this reagent shows up more than once in the patent literature for precisely this reason. Large chemical manufacturers rely on tert-butyl bromoacetate as a key intermediate for producing antiviral and antibacterial agents, among others. The trusted nature of this substance means entire batches can hinge on its purity and reliable reactivity.
Peptide Synthesis and Protecting Groups
In the world of peptides, protecting groups serve as traffic directors. Tert-Butyl Bromoacetate lets chemists install the tert-butyl ester group on carboxylic acids, which shields the acid from unwanted reactions until the final steps. This selective masking method saves a lot of frustration, especially in multi-step syntheses where cross-reactions can easily derail a project. Fast-paced biotech companies and academic research teams regularly add tert-butyl bromoacetate to their toolbox for this reason.
Materials Science and Specialty Chemicals
Its reach goes beyond pharma. In the field of specialty polymers and materials science, researchers count on this reagent to build complex architectures with controlled branching or specific end-group functionality. Materials that end up in everything from drug delivery vehicles to specialty coatings often start with a molecule like this. Its structure makes it a versatile component for creating reactive sites or introducing new functional groups onto otherwise unreactive backbones.
Safe Handling and Sustainable Practices
Lab safety officers remind everyone that chemical reagents with halogen and ester groups deserve respect. Tert-Butyl Bromoacetate calls for gloves, goggles, and a fume hood, especially during scale-up. Reputable labs prioritize supplier quality and eco-conscious waste management, limiting unnecessary exposure and following green chemistry principles. More companies invest in training and safer, more efficient processes, pushing for both productivity and workplace health. The rise of sustainable supply chains and stricter regulations means chemical firms can’t skip these steps anymore.
Real Progress Through Practical Use
When you watch a molecule like Tert-Butyl Bromoacetate in action, you see how the right tools shape the future of drug discovery, materials engineering, and research as a whole. Its legacy grows every time a new medicine, material, or scientific breakthrough traces back to a careful reaction, powered by one small bottle in the hands of a skilled chemist.
What is the proper storage condition for Tert-Butyl Bromoacetate?
Handling Chemicals Like You Mean It
Thinking about working with Tert-Butyl Bromoacetate leaves no room for winging it. The stuff has a reputation: strong odor, skin and eye irritant, flashes up if you drop the ball on proper handling, and the classic, loves to break down in light and heat. I’ve been around enough organic labs to know shortcuts don’t pay. Tidy habits save skin, time, and research budgets.
Why Conditions Matter
Set a bottle of Tert-Butyl Bromoacetate on a sun-soaked bench and, over time, sticky residue and a harsh stink take over. Worse, the actual compound drifts away from spec—no chemist has patience for ruined yield or weird side reactions. Light breaks bonds. Heat stirs up volatility. Forget to seal the lid, and moisture creeps in, building up unwanted acids that turn your hard-won compound to junk.
Best Storage Habits—Proven in Real Labs
Use a tight-sealing amber glass bottle for any quantity you plan to keep. Amber glass cuts ultraviolet and blue light, keeping breakdown to a minimum. A screw cap with a solid liner, not those flimsy snap-on types, keeps out humid air and laboratory fumes. Keep the bottle in a dry, flame-free spot. Air-conditioned storage rooms tend to hover around 20–25°C, but for longer-term storage or if local summers get steamy, refrigerator space (around 2–8°C) gets the edge. Not a standard kitchen fridge—with chemical odors, dedicated flammable or chemical fridges earn their keep.
Tert-Butyl Bromoacetate rarely wins beauty contests for stability. Humidity triggers hydrolysis. Bromine-based compounds can release gas if handled rough or opened too much. Flammability ratings signal: don’t stash this near open flames, hotplates, or reactive metals like sodium or potassium. Most glassware fails at the hands of carelessness. Spills can happen, and once the skin tingling starts, you’ll wish you had gloves—so nitrile gloves and splash goggles matter, always.
Facts Back Up Good Habits
It’s not just preference or rumors. The Sigma-Aldrich and Alfa Aesar chemical catalogs state Tert-Butyl Bromoacetate wants darkness, cool conditions, and a tight cap. They didn’t print those protocols for nothing. Some suppliers toss molecular sieves into the bottle to bite off that last bit of moisture; smart move, especially if you can’t run chillers everywhere. Data sheets show decomposition temps; the further you keep the chemical from that line, the longer it lasts.
Better Storage, Better Outcomes
Even if you’re only prepping small batches of protected glycine or peptoid segments, skip the rush: label your container by date, keep it above the lab floor, and far from acid cabinets or base baths. Make a habit of checking containers for leaks or discoloration—brown streaks or vinegar smells say it’s time to order fresh material. Dump used or degraded Tert-Butyl Bromoacetate following hazardous waste rules. Tossing it down the sink looses more than a stink; it crosses a line in both safety and compliance.
How to Do It Right
Success in the lab means knowing what sits in your bottle and keeping it right from start to finish. A controlled, cool, and light-shielded place, with containers that close tight, reduces surprises and upholds all those hours of prep work. It’s what professionals—and anyone who values clean results or their own well-being—choose every day.
What are the safety precautions when handling Tert-Butyl Bromoacetate?
Why This Chemical Gets Extra Respect
Anyone who’s worked in a chemistry lab comes to respect certain compounds after a few run-ins with splashes, stings, or a wayward fume. Tert-Butyl bromoacetate lands high on that list. Its sharp, pungent odor is a reminder: this is not something to take lightly. I remember opening a bottle once and realizing immediately that goggles, gloves, and a fume hood aren't up for debate. Proper handling makes all the difference, not just for your own health, but for the folks working next to you.
What Makes Tert-Butyl Bromoacetate Risky
A small spill or splash can cause a lot of trouble. Touching the liquid leads to irritation or burns, and breathing in the vapor might trigger coughing, shortness of breath, or worse. The chemical doesn't care if you're careful most of the time. You have to be careful every single time. Scientists have traced the effects of accidental exposure, and plenty have written about damaged skin, eye injuries, or respiratory issues after ignoring basic precautions with alkyl halides like this one.
Gloves, Goggles, and Good Habits
Using sturdy, chemical-resistant gloves—think nitrile or neoprene—keeps the compound off your skin. Once, a student in my lab thought one pair of latex gloves was enough, and wound up with a burning sensation and a ruined afternoon. Learn from their mistake. Standard lab cups and cloth gloves don’t block this stuff; only thick, purpose-built gloves work. Eye protection matters even more. Regular prescription glasses leave gaps; even a tiny splash can slip through. Tight-fitting chemical splash goggles protect for real.
Don’t Turn Your Nose Up at the Fume Hood
Trying to handle Tert-Butyl bromoacetate in open air brings trouble before you notice. The fumes are sneaky, finding their way to your nose and lungs and irritating them without much warning. Even if the smell doesn’t seem strong, fumes linger at low levels—too low to notice, but enough to cause harm. A solid fume hood pulls those vapors away from your face, and keeping it running properly beats any air freshener or makeshift fan.
The Right Gear—And the Right Response
It goes beyond gloves and goggles. A lab coat that covers arms, closed-toe shoes, and long pants all help. Inexperienced folks sometimes skimp here, but a quick search online shows too many stories of ruined jeans, hospital trips, and even permanent scars. If you ever spill some Tert-Butyl bromoacetate on the skin, flush with water right away. Don’t try to rub it off. Speed and volume of water make the difference between mild irritation or a trip to urgent care.
Storage that Doesn’t Play Around
Tert-Butyl bromoacetate stays in tightly sealed bottles, in cool, well-ventilated spaces. You keep it away from heat sources, because this chemical reacts with fire in dangerous ways. I’ve seen bottles swell and leak when left in sunlight or near a hotplate. All containers get labeled clearly, no short-hand or fading ink. That means no one grabs the wrong one, even if they’re in a rush.
Spill Response and Waste Disposal
Even the best folks make mistakes. Spilling a little means reaching for the spill kit, not a wad of paper towels. Absorbent materials and neutralizing agents meant for organic chemicals work best. Once the spill is cleaned, waste goes in a sealed, labeled container for proper pickup. Never dump this compound in a drain or regular trash. Local rules lay out how to label and store chemical waste, and following those rules protects more than just lab workers.
No Shame in Asking for Help
Handling tough chemicals like this demands up-to-date training. Watching a video once in high school doesn’t cut it. I’ve run through the safety routine dozens of times in group meetings, and there’s nothing wrong with repeating instructions. It helps new lab members feel comfortable asking questions, and it keeps accidents from happening.
What is the purity grade available for Tert-Butyl Bromoacetate?
Pushing for Pure Compounds in the Lab
Working with Tert-Butyl Bromoacetate usually comes down to how clean you can get it. Purity isn’t just a buzzword—it decides if your final product actually works, or if you’ll end up running another round of chromatography, watching deadlines slip away because the starting material wasn't clean enough. Plenty of bench chemists, myself included, end up hunting for high-purity batches simply to dodge the headaches that impurities bring.
Standard Purity Ranges in the Market
Most suppliers put out Tert-Butyl Bromoacetate with a minimum purity around 97%. Some push a little higher, heading up to 98% or even 99%. These numbers don't just come from thin air; vendors back them up with GC or NMR data, and anybody ordering for a research lab is right to call and ask for a copy before spending their grant money. Purity levels in this zone suit most synthetic routes, especially in pharmaceutical settings where trace impurities can throw off spectra, introduce side reactions, or cause scale-up to stall.
I've seen plenty of cases where a “just good enough” chemical grade ruined a project. That stray 2% can mean a lot if there's an active bromide or something else reactive hiding in your flask. No one wants to explain to a supervisor why they need another order and a week to repeat the same experiment.
Why Technical Grade Doesn’t Cut It
Lower grades exist, dropping to 95% or less. These cut-corner products pop up on the market as “technical” or “industrial” grades. If purity isn’t essential, maybe to make a lower-tier intermediate for non-pharma applications, some places take the chance to save money. My own experience tells a different story—if there’s no clear certificate of analysis, you risk days lost troubleshooting. In regulated sectors like APIs or specialty materials, technical grades don’t get near the lab bench. No one wants to gamble a process on a bottle that hides what’s in that last five percent.
How Purity Impacts Quality and Safety
Purity means more than “clean.” Unwanted byproducts amp up the hazard risk, especially if they include leftover bromide, acids, or moisture. Safety teams in manufacturing settings demand full transparency since these compounds bring risks from both chemistry and regulatory angles. GMP facilities run regular audits, and they’ll reject shipments if the COA isn’t up to scratch. From my own view, running a reaction without knowing what’s in your flask defeats the whole purpose of careful science.
Making Better Choices: Supplier Tips
Finding the right supplier takes legwork. A reputable seller shows clear HPLC, GC, or NMR data and offers lots that repeat the same specs over time. Smart labs keep records on which vendors deliver reliable, consistently high-purity materials. If a batch of Tert-Butyl Bromoacetate comes in at 97% or better, most researchers can trust it—still, I always run a quick check, since a bad batch wastes more than just time.
What the Future Could Offer
Researchers deserve tighter controls and more transparency. Online tracking of lot history and digital COAs before shipping aren’t just nice extras— they’re fast becoming basic expectations. I picture a future where every bottle, no matter where it’s shipped, comes with a QR code for instant verification by GC-MS, helping chemists stay a step ahead of contamination and setbacks. Until then, real-world experience, clear data, and a touch of skepticism remain the keys for keeping projects on track.
| Names | |
| Preferred IUPAC name | 2-Bromo-2-methylpropyl 2-bromoacetate |
| Other names |
Bromoacetic acid tert-butyl ester Tert-butyl 2-bromoacetate 2-Bromoacetic acid tert-butyl ester Tert-butyl bromomethylcarboxylate Tert-butyl bromoethanoate |
| Pronunciation | /ˈtɜːrtˈbjuːtɪl ˌbroʊmoʊəˈsiːteɪt/ |
| Identifiers | |
| CAS Number | 5292-43-3 |
| Beilstein Reference | 1718739 |
| ChEBI | CHEBI:52391 |
| ChEMBL | CHEMBL1319957 |
| ChemSpider | 16819 |
| DrugBank | DB13892 |
| ECHA InfoCard | ECHA InfoCard: 100.009.791 |
| EC Number | 211-239-6 |
| Gmelin Reference | 80864 |
| KEGG | C05364 |
| MeSH | D017947 |
| PubChem CID | 75485 |
| RTECS number | CY8950000 |
| UNII | 42JZO3G76G |
| UN number | UN 3265 |
| CompTox Dashboard (EPA) | DTXSID7079650 |
| Properties | |
| Chemical formula | C6H11BrO2 |
| Molar mass | 181.06 g/mol |
| Appearance | Colorless to yellowish liquid |
| Odor | Pungent |
| Density | 1.377 g/mL at 25 °C |
| Solubility in water | Slightly soluble |
| log P | 1.84 |
| Vapor pressure | <0.1 mmHg (20°C) |
| Acidity (pKa) | 23.4 |
| Magnetic susceptibility (χ) | -6.35 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.433 |
| Viscosity | 1.234 cP (20°C) |
| Dipole moment | 3.10 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 399.8 J·mol⁻¹·K⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | ["GHS02", "GHS07"] |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P210, P261, P280, P301+P312, P305+P351+P338, P337+P313 |
| Flash point | 70 °C |
| Autoignition temperature | > 225 °C |
| Lethal dose or concentration | LD50 (oral, rat): 950 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 950 mg/kg |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 ppm |
| Related compounds | |
| Related compounds |
Ethyl bromoacetate Methyl bromoacetate Isopropyl bromoacetate Tert-butyl chloroacetate Tert-butyl iodoacetate Tert-butyl acetate Bromoacetic acid |

