Sulfuryl Fluoride: Rethinking Its Role, Risks, and Future
Historical Development
Sulfuryl fluoride’s story started back in the early 1950s, when researchers were searching for alternatives to methyl bromide. The world was waking up to the dangerous effects of ozone-depleting chemicals, and fumigation practices sat right at the crossroads of necessity and environmental responsibility. The early development grew out of hard lessons: dealing with insect infestations in lumber and agriculture without harming the planet. Scientists struggled to balance efficacy with ecological impact, and while sulfuryl fluoride arrived on the scene as a new hope, its journey has been far from straightforward.
Product Overview
Known in the field as a quick-acting, gas-phase pesticide, sulfuryl fluoride gets sold and used under names like Vikane. Its major selling points have always revolved around its ability to knock out tough pests—everything from bed bugs to termites. Users favor it partly because it penetrates deep into materials like wood and concrete, so hiding pests don’t stand much of a chance. Unlike the earlier era of heavy, lingering residues, this chemical usually leaves behind little to clean up, and that tends to fit the frantic schedules of commercial operators and homeowners alike.
Physical & Chemical Properties
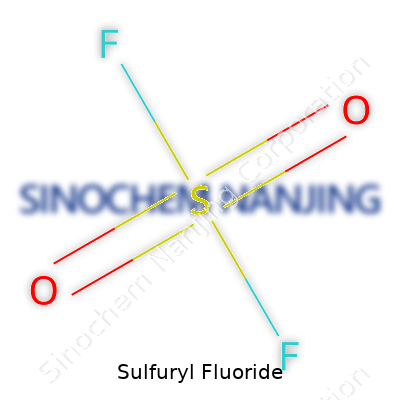
At room temperature, sulfuryl fluoride shows up as a colorless, odorless gas. Technicians handling it know that, even at low concentrations, it can be dangerous—breathing it in accidentally can land someone in serious trouble. Its chemical formula, SO2F2, reflects its makeup, and it boils at a chilly minus 55 degrees Celsius. You won’t find it dissolving easily in water, but it mixes readily with air. The molecule’s stability works both as an asset and a liability: it lingers long enough to do its job, but that same durability creates downstream issues.
Technical Specifications & Labeling
Labels matter a great deal with sulfuryl fluoride. Governments across the world treat its distribution with care, demanding clear-cut directions and strict hazard warnings. The packaging favors cylinders or pressurized containers, and compliance officers look for highly visible dye markings on tanks or containers. Manufacturers don’t have much room for error—misreading instructions or cutting corners on labeling can lead to fines or, worse, serious accidents. It’s not difficult to see why. The stakes get higher any time a material can threaten operators and bystanders in seconds.
Preparation Method
Industry production often runs through the reaction of sulfur dioxide and chlorine with fluorides such as calcium fluoride or sodium fluoride. Facilities that equip themselves to handle these processes must keep tight control on venting, containment, and temperature regulation. From practical experience, this step isn’t something just any operator can wing—it’s a dance of skill, equipment, and caution, where the smallest slip can trigger toxic gas leaks or equipment failure.
Chemical Reactions & Modifications
Chemists regard sulfuryl fluoride as both reactive and stubborn. Given a strong enough base or nucleophile, operators can drive it to make salts like fluorosulfates. Most day-to-day uses don’t call for these transformations, but academic labs sometimes explore ways to tweak its reactivity for niche industrial syntheses. Any new reactions bring their own hazards and waste products, so anything beyond regulated uses calls for a good reason and a strong risk management plan.
Synonyms & Product Names
Regulars in pest control and fumigation circles use a handful of names for sulfuryl fluoride. Among them: Vikane, Dowfume, and sulfuryl difluoride. Each one points to the same core chemical, even if they hint at industry quirks and regional preferences. Knowing these synonyms keeps operators, buyers, and regulators on the same page—a small but important piece in avoiding hazardous mix-ups.
Safety & Operational Standards
There’s no ignoring the safety issues tied to sulfuryl fluoride. Training sits at the heart of safe use, but it only works when operators stay vigilant. The gas’s lack of color and odor trips up even experienced applicators, turning simple jobs into risky ones if detection equipment gets skipped or fails. Regulations rightly require careful site preparation, thorough sealing of structures, and reliable aeration before reentry. Workplaces opt for continuous monitoring—portable sensors, breathing apparatus, proper signage—because memories of past poisonings run deep in the professional community. Everybody in the chain, from producers to end-users, has to treat these protocols as non-negotiable.
Application Area
Sulfuryl fluoride holds its ground as the weapon of choice for structural fumigation. Pest control operators reach for it in homes, museums, food processing warehouses, and mills. The main edge comes from its ability to crush all stages of insect life cycles, including eggs, which can outwit many less aggressive pesticides. People who remember losing priceless antique furniture or business-critical grain supplies to infestations tend to appreciate its power. Yet, all that efficiency brings controversy: environmentalists flag its role as a greenhouse gas, researchers monitor its breakdown in real-world conditions, and regulators weigh its impact on human health.
Research & Development
Research on sulfuryl fluoride hasn’t stood still. Labs keep looking for ways to reduce uncertainty around environmental fate and breakdown products. In recent years, studies tuned in to atmospheric chemistry, tracking the compound as it drifts, reacts with sunlight, or interacts with surfaces. Innovators work on detection technology, like portable sensors or real-time analytics, so field teams can spot leaks quicker and clear buildings with greater confidence. There’s also a push to discover molecular tweaks that could turn this compound into something less persistent, less likely to add to climate change. Progress runs uneven, with regulations and funding helping shape each step forward.
Toxicity Research
Most folks outside the lab don’t realize just how persistent sulfuryl fluoride can be. Toxicity research delivers a sobering reality check: the gas attacks the nervous system, and deaths have occurred when protocol gets ignored. In animals, studies saw neurotoxicity and even reproductive effects at high exposures. Possible impacts on workers prompt regulators to set exposure limits and require protective gear, sometimes down to the last minute traced on a site log. Environmental toxicologists have flagged contamination concerns for non-target organisms, especially in regions near regular or historic fumigation use.
Future Prospects
The future of sulfuryl fluoride won’t follow a simple line. On one side, a world hooked on the rapid turnover of goods and pest-free homes keeps pulling this chemical back into use. On the other, mounting evidence calls for alternatives and stronger oversight. Regulatory agencies continue tightening restrictions, looking hard at lifecycle impacts, and zeroing in on greenhouse gas contributions. Some companies now throw energy behind integrated pest management or non-chemical barriers, hoping to sidestep dilemmas presented by legacy fumigants. New research keeps inching toward better formulations, smarter delivery technologies, and even bio-based replacements. Sulfuryl fluoride’s story ultimately challenges society to weigh immediate problems against longer-term environmental trade-offs, and to stay nimble enough to adapt new tools while respecting hard-won lessons from decades of use.
What is Sulfuryl Fluoride used for?
Why Sulfuryl Fluoride Matters in Pest Management
I still remember my first encounter with sulfuryl fluoride during a home fumigation project. You walk into a house that looks untouched, yet behind closed doors, this colorless gas goes to work. Used by professionals to clear out insects and termites, sulfuryl fluoride has become a go-to solution far beyond what most homeowners realize.
This compound earned its reputation for effectiveness. Unlike older chemicals that often left residue or odor, sulfuryl fluoride doesn’t stick around in the same way. It gets released, fills the treated structure, and does its job: kills off pests like termites, bed bugs, and beetles, reaching even into cracks and corners where sprays miss. Its ability to penetrate hard-to-reach spaces is a major reason why pest control industries rely on it for whole-structure treatments.
Food Safety and Sulfuryl Fluoride
Warehouses and food industry professionals also lean on sulfuryl fluoride for a different but related reason. Bulk grains and packaged foods can quickly become a haven for pests. If you look at how food gets shipped around the world, infestations can create massive loss and waste. Fumigation with sulfuryl fluoride helps clear out problems without tainting the product or drastically altering its quality. The gas dissipates quickly under proper ventilation, which is key to meeting modern food safety standards.
Some people ask about safety, and those concerns aren't misplaced. Research from the Environmental Protection Agency has set strict guidelines on how much residue can remain on foods after treatment. Workers must take special care when handling the gas, wearing protective gear and following protocols to the letter. These steps reduce risks to both consumers and professionals, but oversight remains important.
Environmental Impact
There’s a flip side. Like many chemicals, sulfuryl fluoride doesn’t get a free pass just for being effective. It’s a greenhouse gas, and recent studies showed it sticks around in the atmosphere longer than many realized. As climate change takes a front seat, industry leaders and environmentalists have started weighing these impacts in their daily choices. Relying too much on any single chemical can backfire, so it’s worth questioning whether continued use at current rates makes sense, even if the trade-off seems small at first glance.
Seeking Alternatives and Responsible Use
Alternatives include targeted heat treatments, and integrated pest management — a fancy way to describe using a mix of methods to control pests. You might see increased investment in research into biological controls, though they can be tricky to scale. For now, sulfuryl fluoride remains in the toolbox, especially where bedrock pest problems threaten food or property.
Practically, any pest treatment plan works best with careful planning. If folks learn how to prevent infestation in the first place — sealing up cracks, reducing moisture, and storing food safely — reliance on powerful gases drops. As more information comes out about the long-term effects of different options, both professionals and homeowners would do well to stay informed and ask questions, not just trust what’s always been done.
Most people won’t come into contact with sulfuryl fluoride themselves, but its use touches our homes, our food, and the environment we share. My experience on the ground taught me that safe handling and honest answers often matter more than a magic bullet. Tools like these carry real benefits, but they demand respect and thoughtfulness in how they’re used, today and for the long run.
Is Sulfuryl Fluoride safe for humans and pets?
Understanding Sulfuryl Fluoride
Sulfuryl fluoride has taken up a role as a fumigant, especially for pest control in homes and storage facilities. Pest professionals bring it in for jobs like termite elimination. They use it because it does a thorough job, reaching deep into wood structures where bugs like to hide. It doesn't leave a mess behind, which appeals to property owners tired of chemical residues.
Health and Safety Concerns
Most people don't realize how potent sulfuryl fluoride can be. Direct exposure isn't something to take lightly. Short-term effects include coughing, shortness of breath, nausea, and sometimes dizziness. At high levels, exposure has led to nervous system symptoms or even fatalities. Pets face similar risks, and sometimes more severe ones, thanks to their smaller size and inability to recognize danger and vacate the area.
Regulators have set safety benchmarks for how much sulfuryl fluoride can be present in food and the air during and just after fumigation. Trained workers measure these levels before letting people or animals back inside. It usually takes between six and forty-eight hours before the gas clears out enough for reentry.
Experience from the Field
My neighbor hired a pest control company two years ago to deal with termites in his historic house. Pets and humans both stayed at my home for two days. The crew posted big red warning signs, locked down the house, and checked every corner with gas detectors before they let anyone back in. The company emphasized no exceptions—those rules came from hard-earned lessons in the industry.
Back in the 1980s and ‘90s, not every professional measured gas residue as carefully. Accidents happened. Some involved beloved family pets who were let back inside too soon. Those stories led to tighter standards and required training for anyone handling fumigation chemicals. It doesn’t erase the risk but reduces the likelihood of accidental poisoning.
What Science Says
Peer-reviewed studies published in journals like Environmental Health Perspectives show that sulfuryl fluoride doesn't linger in houses after enough ventilation. Within 1-2 days, almost all traces are gone from indoor air if the company follows current guidelines. The EPA sets permissible limits for residues on treated surfaces to keep both people and pets safe after reentry. California’s Department of Pesticide Regulation has tracked hundreds of cases. Most serious health problems linked to sulfuryl fluoride occurred when someone broke safety protocols and went inside early.
Possible Solutions and Precautions
No one likes to trust their family’s safety to warning tape wrapped around a building. More consumer education would help. Pest control operators should give plain language instructions, not just hand over technical pamphlets. Families should pack a go-bag for pets and be ready to spend a night or two away from home, not rush back too early.
Smart technology could help. Remote sensors can check air quality without unlocking doors. Better tracking of who enters or leaves during fumigation would prevent tragedies from simple miscommunication.
Choosing a licensed, well-reviewed pest management company matters. Reviews from real neighborhood customers mean more to me than a glossy brochure.
Personal Takeaway
Sulfuryl fluoride isn’t something for amateurs to handle. With proper precautions, documented protocols, and a willing pause in daily routines, people and pets get through fumigation safely. Shortcuts and skipped steps put families at risk. The science backs up safety—if everyone follows the rules.
How does Sulfuryl Fluoride work as a fumigant?
Understanding the Battle Against Pests
Grain storage, flour mills, shipping containers—if you have ever worked in any of these spaces, you know bugs find their way in, no matter how much you sweep or patch up holes. Nobody wants to bite into bread and wonder about the journey those grains took. Sulfuryl fluoride steps in as a gas that gets released inside structures to kill pests deep within walls, equipment cracks, and stacks of goods.
How Sulfuryl Fluoride Hits Its Target
Sulfuryl fluoride, discovered back in the 1950s, doesn’t work like some older chemicals that choke bugs by smothering them. Instead, this gas gets inside the insect’s body and interrupts basic cell chemistry. It blocks an enzyme that insects need to produce energy. Without the ability to generate energy, bugs—everything from termites to beetles and bed bugs—stop moving and die off. This isn’t just guesswork; pest management pros use gas detection tools to make sure they’re hitting the right levels in every nook and cranny.
What Sets Sulfuryl Fluoride Apart From Its Predecessors?
A lot of folks talk about methyl bromide, which dominated the game before environmental and health risks led people to look for alternatives. Sulfuryl fluoride doesn’t break down the ozone layer the way methyl bromide does. On top of that, companies can clean up after a treatment and get goods back to market in hours. Methyl bromide, by contrast, required drawn-out aeration times and strict cross-contamination avoidance, which complicated schedules and budgets.
Real Risks and Safety Questions
As someone who’s seen a fumigation crew in action, the whole process feels a bit like cordoning off a crime scene. Teams seal up entire buildings with tarps and post warning signs everywhere. Staff use gas monitors to patrol the perimeter and check for leaks. Human health stands at the top of everyone’s mind, since sulfuryl fluoride is toxic if inhaled directly. Mismanaged fumigation leads to real tragedies; incidents in California and Florida show why strict training and clear protocols mean everything in this line of work.
Sustainability and the Bigger Picture
Sulfuryl fluoride skips ozone damage, but the story doesn’t end there. This gas sticks around in the atmosphere for decades and traps heat, which means it adds to climate change if overused. The scientific community flags this every time they update guidelines: balance fast pest control with the planet’s long-term health.
What Matters for the Future
Newer techniques focus on using less gas and supplementing with heat treatments, vacuum, or even controlled atmospheres with carbon dioxide. Digital monitors and airtight seals cut down on waste. Some farms turn toward integrated pest management (IPM) to reduce heavy chemical reliance altogether. Local laws shape how, and even if, sulfuryl fluoride enters the toolkit.
At its core, sulfuryl fluoride offers fast, deep-reaching results in the unglamorous world of keeping food unspoiled and buildings safe. But every practioner has a responsibility to keep each treatment contained, respect the science behind exposure limits, and look for smarter ways to stop bugs from becoming tomorrow’s problem. Food safety always matters, but thinking about the next generation matters just as much.
What precautions should be taken when using Sulfuryl Fluoride?
Why Sulfuryl Fluoride Calls for Respect
Sulfuryl fluoride plays a big role in fumigating buildings and silos. Many professionals turn to it because pests don’t stand much chance against its power. I’ve spent some time on job sites where this stuff clears out termites or beetles with ruthless efficiency. But you quickly learn respect for that pale gas. Most folks I’ve worked with grew up hearing stories about accidents. One thing gets clear — a casual attitude can send anyone to the hospital, if not worse.
Don’t Skip the Safety Gear
You cannot spot sulfuryl fluoride in the air, nor can you smell it. This isn’t the world’s way of keeping danger interesting; it’s why masks and gas detectors become lifelines. Full-face respirators designed to block this specific gas keep your lungs safe. Relying on any basic dust mask simply puts your health at risk. Workers might roll their eyes at “all the gear,” but it only takes a few minutes of exposure to regret skipping it. A gas meter should be clipped to your collar, set to alarm if levels climb. I’ve watched alarms go off because a door didn’t seal properly. That minute extra set-up paid off.
Seal off and Warn Others
Doors and windows need more than a “good enough” tape job. Sealing every crack feels tedious, but loose seals can leak poison to spaces people never expect. Every access point needs bright, unmissable warning signs. When crews set up for treatment, they put up blinking lights and pile on layers of red tape. The aim is not to impress anyone — it’s to make sure no one stumbles inside. It’s easy to forget how many service workers or neighbors can wander into a danger zone if things aren’t clearly marked off.
Only Trained Teams
No shortcut replaces real training. I watched a seasoned handler walk a first-timer through protocol once, step by step: double-checking cylinders, using proper tools, setting up monitors, locking entry points. No step rushed. I hear stories of people learning on YouTube, which gives me chills. Handling a toxic gas has no room for “making it up as you go.” Legal requirements usually demand certification for a reason. There’s too much risk for workers, customers, pets, and anyone nearby.
Ventilation and Follow-up
Once the treatment wraps up, patience wins the day. Opening up property for immediate use, just to meet a deadline, can leave folks breathing toxic air. Ventilating the building takes longer than anyone hopes, but air has to flow through every space and proper testing needs to happen before declaring the building safe. Teams now use real-time sensors to check for trace gas before handing back keys. I’ve seen jobs where the number stays stubbornly high and keeps workers outside for hours. The frustration grows, but nothing justifies skipping this check.
Think Emergency Plans and Community Safety
Things do not always go to plan. In the middle of a job, an unexpected leak or a sudden illness can mean seconds count. Everyone on site must know the nearest exits, emergency contacts, and the fix for basic exposure — that could mean rapid evacuation and calling for help. Sharing plans with local fire departments and alerting neighbors in advance builds a stronger safety net. Communities depend on transparency and quick action in the rare event something goes wrong.
Looking for Better Solutions
Many homeowners and building managers ask about alternatives to sulfuryl fluoride. Research into less toxic pest control options has picked up over the years, which makes sense given both the effectiveness and the risks of this gas. Integrated pest management, heat treatments, and traditional baiting get considered more often. Every method carries its own set of challenges, but nothing beats thinking twice before using a substance that demands so much care.
What are the environmental impacts of Sulfuryl Fluoride?
Fumigation and Its Unseen Toll
Sulfuryl fluoride gets used in pest control, mainly in agriculture and for treating wood. Farmers and businesses have leaned on it for decades because it wipes out tough insects without leaving residue on food. It’s tempting to call it a miracle chemical, especially compared to some of the nasty pesticides of the twentieth century. But anything that can clear out warehouse-sized infestations deserves a closer look.
This gas floats up into the air after fumigation wraps up. Early on, experts assumed it broke down pretty fast. That’s not the case. Research from institutions like UC Irvine found sulfuryl fluoride sticks around for decades. It doesn’t react easily in the lower atmosphere. That means even small leaks or routine use contribute to a slow but meaningful build-up over time.
Climate Change Adds Weight
Few realize how powerful sulfuryl fluoride is as a greenhouse gas. Researchers found it traps heat thousands of times more effectively than carbon dioxide. The Environmental Protection Agency estimates the gas stays in the atmosphere for nearly forty years. Add up those fumigation cycles every month, multiply by the facilities involved, and the impact starts to come into focus.
I’ve worked with communities close to agricultural hubs, and farmers usually hear a lot about crop yields. The conversation rarely brings up longer-term impacts on the climate. Yet every extra ounce of greenhouse gas draws out droughts and shifting rainfall that hit their bottom line just as hard as any beetle ever could.
Air and Human Health
There’s something worrying about a substance that’s tough to see or smell but floats around for years. Most people have no clue when sulfuryl fluoride has been used nearby. Testing for the gas isn’t part of standard home or building checks. That means we roll the dice on exposure, especially for those living close to fumigated sites. The American Lung Association points out that the chemical can cause breathing difficulty, nausea, and sometimes more serious reactions if not handled with care.
Bigger Picture: The Link with Food Regulation
Government agencies recognize the risks but wrestle with their own tradeoffs. Banning sulfuryl fluoride outright could mean more crop loss, less food security, or a return to even harsher chemicals. The EPA tried to phase out its use on food in 2011 but ran into resistance because the alternatives carried their own problems. Navigating pesticide policy often means accepting lesser evils, but the greenhouse gas angle gives this debate new urgency.
Looking at Solutions
No single fix covers all the challenges tied to sulfuryl fluoride. Technology offers some hope. Better sealing and targeted fumigation equipment can cut leaks. Some research labs work on biodegradable options that break down safely in sunlight. Farmers know the lay of their land best—tapping into that local knowledge through education and incentives makes more sense than blanket bans handed down from above.
Consumers can play a small part by supporting growers who use integrated pest management. It’s not just about buying organic. It means taking time to know where fruit, nuts, and grains come from. Standing up for labeling and transparency pushes regulators and companies to dig deeper into the true costs behind what ends up on our shelves. Each decision nudges the balance from quick fixes toward solutions that last beyond the next harvest.
| Names | |
| Preferred IUPAC name | sulfuryl difluoride |
| Other names |
Vikane Sulfuryl difluoride SO2F2 |
| Pronunciation | /ˈsʌlfjʊr.aɪl ˈflʊəraɪd/ |
| Identifiers | |
| CAS Number | 2699-79-8 |
| 3D model (JSmol) | `JSmol.loadInline("data:text/plain;base64,CmlzbyB2ZXJzaW9uIDQuMApTdWxmdXJ5bCBGbHVvcmlkZQogUyAxLjAwMDAgMC4wMDAwIDAuMDAwMCBTIDAuMDAwMApPIDAgMC4wMDAwIDAuMDAwIE8wCk8gMC4wMDAwIDEuNzkwOCAwLjAwMDAgTzEKRgAgMC4wMDAwIC0xLjQzMzQgMC4wMDAwIEY=");` |
| Beilstein Reference | 3565952 |
| ChEBI | CHEBI:29379 |
| ChEMBL | CHEMBL1201865 |
| ChemSpider | 52548 |
| DrugBank | DB11586 |
| ECHA InfoCard | 03ee191e-6ab7-4b34-80c1-7108a53c2467 |
| EC Number | '016-029-00-8' |
| Gmelin Reference | 37830 |
| KEGG | C06325 |
| MeSH | D013471 |
| PubChem CID | 24552 |
| RTECS number | WM8400000 |
| UNII | Y3U394W9A4 |
| UN number | UN2191 |
| Properties | |
| Chemical formula | SO2F2 |
| Molar mass | 102.06 g/mol |
| Appearance | Colorless gas |
| Odor | Odorless |
| Density | 1.67 g/cm³ |
| Solubility in water | 0.75 g/L (20 °C) |
| log P | 0.29 |
| Vapor pressure | > 1950 mmHg (20°C) |
| Acidity (pKa) | ~ -2.0 |
| Basicity (pKb) | 15.0 |
| Magnetic susceptibility (χ) | -9.6×10⁻⁶ |
| Refractive index (nD) | 1.0008 |
| Viscosity | 0.415 cP |
| Dipole moment | 1.18 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 297.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1240 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1326 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V3FA000 |
| Hazards | |
| Main hazards | Toxic if inhaled, causes respiratory irritation, may cause central nervous system effects, harmful to aquatic life. |
| GHS labelling | GHS02, GHS06 |
| Pictograms | GHS04,GHS06 |
| Signal word | Danger |
| Hazard statements | Hazard statements: "H220, H280, H331, H373 |
| NFPA 704 (fire diamond) | 3-0-0-OX |
| Autoignition temperature | 400°C |
| Lethal dose or concentration | LD50 oral rat 113 mg/kg |
| LD50 (median dose) | 150 mg/kg |
| NIOSH | SFV |
| PEL (Permissible) | 5 ppm |
| REL (Recommended) | 0.03 |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Sulfuryl chloride Sulfuryl bromide Sulfuryl iodide Thionyl fluoride |

