Sulfurous Acid: A Closer Look at Its Role and Future in Chemistry
Historical Development of Sulfurous Acid
Sulfurous acid started turning heads not in high-tech labs but as a byproduct in industries running on sulfur. Folks noticed that burning sulfur produced a pungent smell, and this led to the fume-filled experimentation that uncovered sulfur dioxide. Chemists in the eighteenth century found that dissolving this pungent gas in water gave a sour liquid, one that changed colors when mixed with other substances and hinted at something special. The notion of isolating "sulfurous acid" stuck, even though nobody managed to bottle a pure sample. Most often, it was discussed as fleeting—lost in a rush of bubbles—but it played its part in broader reactions, from bleaching to early food preservation. By the time industrial chemistry kicked into gear, sulfurous acid and its relatives handled serious business in dye-making, textiles, winemaking, and water purification.
Product Overview
Sulfurous acid doesn’t show up on store shelves as a bottle with a neat label. Instead, it exists mostly in solution, created the moment sulfur dioxide touches water. Chemists deal with it as a temporary player—useful, reactive, but always ready to disappear back into gases. Its presence gets confirmed by a taste of sour and its knack for reducing colors and killing microbes. Production plants keep it handy as a locally made solution, fresh from injected sulfur dioxide, rather than risking storage. Sulfurous acid’s value lives in these moments of use: cleaning, preserving, or shuttling electrons in the right chemical setting.
Physical & Chemical Properties
Anyone working with sulfurous acid gets right down to its most stubborn fact—it doesn’t last. Mixed in water, this weak acid works at lower pH values, usually between 1 and 2 for concentrated solutions. Clear, sometimes with a tinge, its biting odor gives away the underlying sulfur dioxide. As a reducing agent, it breaks double bonds, knocks the color out of stains, and prevents browning in fruits and fabrics alike by halting oxidation. The challenge comes from how quickly it decomposes, giving off sulfur dioxide and leaving water behind. Pure, isolated sulfurous acid never appears; only watery solutions do the job for any practical use.
Technical Specifications & Labeling
Commercial bottles or containers identify sulfurous acid chiefly by sulfur dioxide concentration—often marked as “sulfurous acid solution” or “SO2 in water”—with details about acidity, free sulfur dioxide content, and heavy metals. Labels warn of choking fumes, skin contact hazards, and offer specific density or concentration ranges, sometimes measured in ppm for food-grade applications or higher percentage for industrial cleaning. Regulations call for childproofing and venting protocols, not just for safety but to ensure nobody gets shocked by a sulfurous punch if they unwittingly open a sealed drum.
Preparation Methods
Sulfurous acid doesn’t lend itself to mass production as a stable material. Instead, plants inject clean sulfur dioxide gas under controlled pressure straight into water, chilled and stirred for maximum absorption. Sometimes chemists add a bit of alkali before the gas, which helps buffer or stabilize the solution for temporary storage, but purity and timing always matter—freshly made solutions work best. Critical to the method is off-gassing; if left open or exposed to heat, much of the acid escapes as vapor within minutes or hours, so timing application means everything.
C hemical Reactions & Modifications
The charm and frustration of sulfurous acid spring from its chemical temperament. It loves to reduce—the acid strips oxygen away from reactive compounds, which drives its bleaching action in pulps, textiles, and even wine barrels. For food preservation, it knocks out bacteria and stops browning before fruit shows a trace of rot. Its reactions branch into forming sulfites and bisulfites if mixed with bases or metal compounds, creating stable salts that can last longer and do extra jobs. Chemists depend on this reactivity to launch chains of synthesis, designer molecules, and cleanup reactions—neutralizers can finish off over-application, and careful ventilation keeps everything in check.
Synonyms & Product Names
Even with a reputation for being elusive, sulfurous acid gets several names. Bottles might mention “sulfurous acid solution,” “aqueous sulfur dioxide,” or call attention to “sulphurous acid” in older British or Commonwealth sources. Any mention of “E220” on food preservatives links back to its salt forms, sodium or potassium sulfite, both springing from sulfurous acid base chemistry. These names help sort out supply chain details, letting technicians match up what’s needed for lab-scale reactions or big-league food processing plants.
Safety & Operational Standards
Every pallet of sulfurous acid solution carries strict handling codes. The fumes can sting eyes and lungs in seconds, so storage requires tight seals, clear venting, and robust respiratory protection. Regulatory agencies demand labeling that covers accidental spillage, chemical burns on skin, and even fire risk—sulfur dioxide fumes can overpower an unprepared operator. I’ve watched warehouse teams work in pairs with air meters, keeping tabs on their gear and each other. Chemical showers sit waiting in cleaning bays; eye washes stand ready nearby, not just decoration. Regular workplace inspections check both gear and training, since a moment’s lapse can spell big trouble.
Application Areas
Sulfurous acid finds work in areas where fighting oxidation and microbial growth brings real economic and health gains. Winemakers rely on it to keep barrels crisp, stop fermentations from running wild, and protect the delicate balance of flavors against spoilage. Fruit packers keep bushels bright and fresh using gently misted acid solutions—nobody wants a crate of brown apples. Pulp mills deploy it for softening wood and producing ultra-white fibers bound for the highest grade books and magazines. In water treatment, it neutralizes chlorine leftover from disinfection, making public water safer and less irritating to skin. Laboratories lean on sulfurous acid for delicate redox work, and cleaning crews use it in diluted form to wipe away stains that resist regular scrubbing.
Research & Development
Modern chemistry still pokes at old problems—can we stabilize sulfurous acid better or harness its power with less toxicity? Biochemists search for safer, longer-lasting derivatives that keep food fresh without stirring up concern about allergies or asthma. Analytical chemists push for more precise instruments, able to measure sulfurous acid’s fleeting presence in environmental samples, wine cellars, or municipal water tanks. Green chemistry labs hunt for ways to recycle sulfur dioxide, to lessen environmental impact from big emission sources and turn waste into something usable. Advances in membrane technology hint that finer separation and storage might make sulfurous acid more controllable, opening doors to new applications, from veterinary medicine to niche microelectronics.
Toxicity Research
Studies dating back decades spell out the hazards that come from careless exposure. Inhaling strong vapors can burn throats and lungs, sometimes triggering asthma attacks or, with heavier doses, acute lung injury. Swallowing or skin contact causes burns, nausea, and sometimes even circulatory shock in high doses. Regulatory limits aim to protect factory workers and consumers alike by capping exposure in air, water, or food. Ongoing medical research digs into less obvious health effects: could long-term, low-level exposure cause chronic issues? Investigators keep revisiting these questions with better tools, searching for links to respiratory problems, allergies, or impacts in sensitive groups. The message from toxicologists stays simple: handle with care; respect boundaries.
Future Prospects
Interest in sulfurous acid isn’t fading, just evolving. Increased demand for chemical recycling, safer food preservation, and minimal-residue cleaning methods sparks new funding and university projects. Automation and online monitoring promise to make handling much safer, with remote sensors taking the guesswork out of concentration and exposure. Substitutes may appear for some applications, but thanks to its unique blend of properties, sulfurous acid sits in a fairly secure position, especially where cost, fast action, or chemical specificity demand something more than just vinegar, chlorine, or peroxide. Further down the road, biotechnologists and environmental engineers are keen on capturing sulfur dioxide streams, upgrading them onsite into precisely dosed acid for farmers, winemakers, and water treatment pros.
What is sulfurous acid and what are its main uses?
Getting to Know Sulfurous Acid
Sulfurous acid tends to pop up in chemistry lessons, often carrying a whiff of old textbooks and lab notes. Most people never see a bottle of it on any shelf, because it comes and goes in water before you get a real look. Sulfurous acid forms when sulfur dioxide mixes with water, making a weak and unstable acid that quietly dissolves before it can stand on its own. For me, my first run-in with the stuff happened in a high school class, bubbling sulfur dioxide through water only to be told, “This isn’t going to last long.”
People sometimes confuse sulfurous acid with its cousin, sulfuric acid. They’re not the same. Sulfurous acid is much weaker and tends to fade, while sulfuric acid sticks around and eats through almost anything. Sulfurous acid quietly handles jobs that need a delicate touch, rather than a strong punch.
Sulfurous Acid at Work
In the paper industry, sulfurous acid helps bleach and soften wood pulp. Fresh paper looks brighter and feels nicer because this acid can wash out unwanted colors without shredding the pulp. Cleaner paper with less yellowing doesn’t cut corners; it shows how a bit of chemistry makes everyday products better.
Water treatment plants turn to sulfurous acid for another job—reducing chlorine or cleaning up traces of chemicals. Most of the tap water folks drink started off a lot rougher, and sulfurous acid helps remove excess chlorine or stubborn pollutants. This means cities can deliver water people want to drink instead of something that smells like a swimming pool.
The wine world also knows a thing or two about sulfurous acid, though not always by name. Winemakers need to control bacteria and yeast that can spoil a barrel. They rely on sulfur dioxide, which, when dissolved in water, creates sulfurous acid. So, those delicate flavors in white wines or unspoiled batches come partly from chemistry at work in the background.
Why Good Handling Counts
Anytime a chemical gets used to clean up drinking water or improve food safety, there’s a double duty. Quality control matters more than ever. Poor handling means leftover chemicals in water or tainted food supply chains. There’s also the issue of air pollution—burning fossil fuels or smelting ore releases sulfur dioxide, the parent gas of sulfurous acid, and that means acid rain. People may think of acid rain as a thing of the past, but the damage still surfaces, stripping paint, killing plants, or messing with soil balance. Smart handling keeps sulfurous acid in check and puts it to work where it helps, not harms.
Better Paths Forward
Cleaner energy can cut down on sulfur dioxide in the air in the first place, making it less of an industrial burden. Simple sensors and better training in labs and factories lower the odds of accidental spills or misuse. Urban wastewater plants might phase in newer technology for sulfur and chlorine removal, dialing in just enough sulfurous acid—no more, no less.
It helps to remember that sulfurous acid rarely stands in the spotlight, always behind the scenes. Its main uses keep products clean, water safer, and even preserve flavors, showing there’s real value in understanding even the fleeting, nearly invisible members of the chemical world.
Is sulfurous acid safe to handle and what precautions should be taken?
What Sulfurous Acid Brings to the Table
Sulfurous acid appears in labs as a weak, unstable acid. It pops up during efforts to dissolve sulfur dioxide gas in water. You won’t see a bottle marked “sulfurous acid” sitting on a shelf; the solution forms as you bubble the gas through water. A lot of people, drawn by the chemical’s bleaching and reducing properties, turn to it for tasks ranging from lab work to water treatment trials.
Understanding the Risks
This acid does not last long before breaking down to its original ingredients. Still, it brings a bite. Direct contact causes skin irritation. Breathing in vapors hits the throat and lungs with a sharp sting — shortness of breath, coughing, and chest discomfort can follow. Sulfurous acid solution gives off sulfur dioxide fumes, and those with asthma or other breathing problems know just how rough that can get. In larger doses, this can send a person straight to urgent care.
Learning from Experience
Years back, my chemistry instructor tried a classroom demo with sulfur dioxide and water in a poorly vented room. Within minutes, people started coughing. Luckily, everyone got fresh air. Looking back, this memory taught me respect for even mild-sounding chemicals. Not every dangerous substance comes flagged with a skull and crossbones — sulfurous acid proves the point.
Steps for Safe Handling
Think gloves, goggles, lab coats — that’s the minimum. Chemical splash goggles keep the solution away from sensitive eyes. Replace gloves if you feel any tingling or see a tear. If you get this acid on your skin, rinse it off right away with water for several minutes.
Fresh air matters. Work in a fume hood or near an open window to keep fumes from building up. One whiff of sulfur dioxide is sometimes enough to trigger headaches. If your workspace doesn’t run fans or vents properly, postpone the experiment.
Keep sulfurous acid well labeled and sealed. It breaks down quickly, but fumes seep from open bottles. Storing it with a tight-fitting cap and a clear label helps everyone know what they’re working with. Avoid mixing it with oxidizers or strong bases; unpredictable reactions sometimes lead to pressure buildup or dangerous gases.
Antidotes and Emergency Steps
If you get splashed in the eye, rinse with water for at least ten minutes and head straight to medical help. For inhalation, get out to fresh air. Anyone who can’t breathe easily afterwards deserves medical attention. Never try to neutralize spills using household chemicals, since the wrong move generates more toxic fumes. Use an absorbent, plastic or ceramic scoop, and ventilate the area before cleaning up.
Smarter Standards and Training
Anyone using sulfurous acid should know local workplace rules and safety procedures. Training makes a difference — seeing an experienced chemist go through step-by-step handling beats reading instructions off the web. Regular drills prepare people for spills or accidental releases. Nearby eyewash stations make the difference in an emergency.
Official guidelines from trusted bodies like OSHA or the NIOSH outline limits for exposure and handling standards, backing up real-world experience with evidence. When a problem emerges, consulting materials safety data sheets (SDS) on sulfurous acid keeps both old hands and newcomers informed.
Solutions and Smarter Practice
Risk drops sharply in well-ventilated, organized workspaces with clear labeling and fresh training. Emergencies rarely turn serious when people have protective gear ready and know how to use it. Sharing firsthand stories of what went wrong, instead of hiding mistakes, builds a culture where people stay alert and encourage careful steps every time someone reaches for a chemical bottle.
What is the chemical formula and properties of sulfurous acid?
Getting to Know Sulfurous Acid
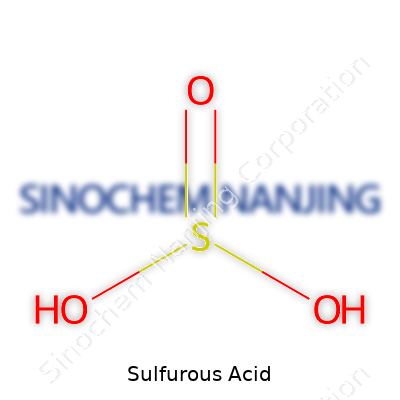
Sulfurous acid comes with the formula H2SO3. If you picture it, the molecule forms through the addition of water to sulfur dioxide gas—a process that almost feels old-fashioned in chemistry labs. The catch: the acid rarely exists on its own in a bottle. Add enough water to SO2, and you only find it dissolved, never isolated as a pure liquid. That vanishing act has sparked a fair share of questions during late-night cram sessions or practical labs.
Basic Properties and Behavior
Most folks expect acid to stick around—like the classic battery acid, sulfuric acid (H2SO4). Sulfurous acid refuses to play by those rules. You’ll only find it in solution, where it breaks down pretty easily into water and sulfur dioxide. Yanking the gas from that mix takes away the acid. Its weak, unstable nature gives it more of a backstage role in chemistry than the spotlight.
Dip a pH strip into a fresh sample and you’ll see readings beneath 7 but nothing extreme. The acid only half-commits to giving up its protons in water. Nobody’s cleaning batteries with the stuff; its use centers on its gentler, less corrosive touch. Lab work on reducing reactions often brings up sulfurous acid, thanks to its knack for acting as a reducing agent. That reducing power helps clean up or change other chemicals without throwing the balance way off.
Where You Meet Sulfurous Acid
Winemaking and food preservation sometimes dip into sulfurous acid’s territory. It’s the unseen hand behind sulfites, holding back spoilage or browning in fruits and drinks. Years working with commercial kitchens and small wineries taught me that most folks never see sulfurous acid head-on. They notice its effects when wine stays fresh and fruit doesn’t turn brown after slicing.
Its fleeting nature has another side. Folks handling fresh solutions know to watch for the strong smell of sulfur dioxide gas—pungent, stinging, impossible to ignore. In closed spaces, SO2 exposure can hurt the lungs. Failing to handle it well causes coughing, eye irritation, and headaches. Safety protocols become more than just good advice; they’re the difference between a simple job and a trip to the ER.
Challenges and Points of Care
Nobody gets by in chemical work without paying attention to instability. Sulfurous acid disappears, leaving behind water and sulfur dioxide. Storage barely works, so folks mix it right before using it. This brings up issues about waste and air quality. Many cities limit the use or release of SO2 because it harms human health and the environment. As someone with experience in environmental labs, I know air filtration and careful venting trump just about every shortcut.
Solutions start at the source. Reduce how much SO2 enters the workspace, and keep reactions contained. Simple improvements, like fume hoods or better training, help protect workers and bystanders. Innovation matters too—new alternatives for food preservation keep popping up, making reliance on sulfurous acid less central. At the same time, regulations demand close logging and reporting, so surprises stay few.
Looking Forward with Caution
Science, and even home winemaking, leans on sulfurous acid now and then. Knowing what makes it tick, respecting its limits, and following practical safety steps keep things in balance. Understanding its properties offers ways to get the job done without risking health or environment.
How should sulfurous acid be stored and disposed of?
Getting Real with Sulfurous Acid
Sulfurous acid doesn’t usually become a hot-topic chemical outside a lab or industrial facility, but anyone who’s ever had to handle this stuff knows it takes respect and a plan. This isn’t just another bottle on the shelf. It reacts fast with air, and it can burn. That’s not something to brush off, especially for small labs and classrooms where training sometimes lags behind the reality of daily work.
The Right Storage Starts with Awareness
Walking into a storage room, I can always spot trouble by seeing where acids get shoved. Sulfurous acid demands someplace cool, dark, dry, and away from anything reactive. Strong bases or oxidizers nearby spell disaster. Plastic containers work, especially ones built to handle acid. Glass containers might hold up but only if stored far from vibrations or traffic; glass loves to surprise you with cracks, especially when chemicals react with corks or rubber stoppers. Tightly sealed containers stop leaks and prevent air from turning that already-fussy solution into sulfur dioxide gas.
Ventilation Matters
Opened a sulfurous acid bottle without a fume hood and you feel it in your lungs immediately—the gas is no joke. A lot of injuries come not from spills but from fumes slowly built up in cramped storerooms. A room with steady ventilation, not just a window cracked open, keeps the air safe. Even school science closets need something more than a fan stuck in the doorway.
Record Keeping and Training Save Trouble
Plenty of near-misses I’ve seen over the years started with someone forgetting what’s in the bottle. Every container deserves a label with the concentration, date mixed, and a big warning sign. Staff should run through emergency drills at least every semester. New techs and students should never puzzle their way through “How do I get rid of this?” without guidance.
Disposing Sulfurous Acid Without Cutting Corners
People sometimes treat sulfurous acid like it’s vinegar or lemon juice and pour it away. Chemistry teachers from the old school remember dumping stuff down the drain but that stopped making sense once we realized its effects on wastewater and pipes. Neutralizing with sodium carbonate or bicarbonate works, but only in small batches—dumping it all in at once creates a storm of off-gassing and heat. Slow addition, with stirring and plenty of time for each bit to react, spares everyone a chemical cloud.
Once the bubbling stops, most labs test pH until the waste sits between 6 and 8. Anything outside that means going back and adding more base or acid as needed. Then the solution joins the hazardous waste drum, labeled and scheduled for certified pickup. Waste haulers check logs, so skimping on details has bitten more than one department with surprise fines. Trust me—answering to regulators about skipped steps beats spending your weekend in safety retraining.
Better Tools, Fewer Accidents
Modern safety tools show how much easier life gets with the right gear. Splash goggles, face shields, nitrile gloves, and absorbent pads change disaster to a near-miss. Even spill kits that seem unnecessary become quick heroes the moment a container slips. An ounce of preparation in storage and disposal beats a pound of panic after a spill.
Proper storage and safe disposal aren’t complicated if we don’t cut corners. Every accident avoided tells you that respect for what sulfurous acid can do goes further than rules on a clipboard.
What are the common industrial applications of sulfurous acid?
Sulfurous Acid in Water Treatment
Clean water usually depends on a series of behind-the-scenes chemical reactions. Sulfurous acid finds a solid place here, mostly because it makes chlorine removal look easy. Anyone who's dealt with drinking water systems or wastewater treatment knows the headache of leftover chlorine. It not only affects taste but also forms unwanted byproducts. Adding sulfurous acid reduces excess chlorine effectively, making water safer for downstream use and keeping systems running smoothly.
Bleaching Agent in the Paper Industry
Working in or with paper mills, you quickly learn about the battle with impurities and stubborn colors. Bleaching wood pulp isn’t just about getting that crisp white finish—it demands a careful touch to preserve fiber quality. Sulfurous acid stands out because it’s gentler than stronger bleaches like chlorine. Mills often turn to it for a mild bleach that gets results without tearing up the delicate pulp fibers. It also helps limit equipment corrosion, so mills often save on maintenance in the long run.
Preservative Power in the Food Industry
Sulfurous acid sounds like something that shouldn’t go near food, but reality says otherwise. Both small breweries and big food producers rely on it for preservation. The acid inhibits unwanted microbes in fruit juices, wines, and dried fruit processing. Having worked with small-scale cideries, I’ve seen firsthand how it prevents spoilage and maintains flavor. Facts back it up: safe concentrations prevent bacteria and fungi growth, cutting down on food waste while keeping products shelf-stable.
Chemical Synthesis and Reducing Agent
Many manufacturing tasks hinge on reliable reducing agents. In the production of chemicals like sodium sulfite or as an intermediate in dye-manufacturing, sulfurous acid enters the scene. It helps convert raw materials efficiently and keeps oxidation in check. In my days shadowing a chemical engineering team, I saw entire shifts dedicated to monitoring redox balances in reactor vessels—getting the right reducing agent makes all the difference for quality and yield.
Disinfection in the Brewing Industry
Brew houses need a cleaning regime that's thorough but doesn’t leave unwanted residues. Sulfurous acid is often chosen to sanitize bottles, barrels, and equipment because its breakdown products—mainly sulfur dioxide—work fast and leave little trace. Brewers can reuse clean containers quickly, cut water consumption, and move product to market faster.
Challenges and Safer Handling
Anyone working with sulfurous acid realizes it’s not all smooth sailing. It’s unstable, quickly decomposing to sulfur dioxide and water. Safe handling requires good ventilation, protective equipment, and tight controls. There’s always a risk of sulfur dioxide gas leaks, which brings regulatory scrutiny and health concerns. Facilities often install sensors and provide routine worker training, both to protect employees and meet environmental compliance rules.
New Pathways: Sustainability and Alternatives
New research looks at how renewable sources or alternative chemicals can provide the same results as sulfurous acid—especially in food and water applications. Some companies explore microbial treatments or green chemistry, aiming to lower environmental impact. Still, it’s tough to beat sulfurous acid’s balance of effectiveness and cost in many traditional roles.
Sulfurous acid shows up in key industrial moments, where versatility and reactivity matter. Safer practices and new innovations keep it working for industry without putting health or the planet on the backburner.
| Names | |
| Preferred IUPAC name | sulfurous acid |
| Other names |
Sulphurous acid dihydroxidooxidosulfur sulfur dioxide solution E220 aqueous sulfur dioxide |
| Pronunciation | /ˈsʌlfjʊərəs ˈæsɪd/ |
| Identifiers | |
| CAS Number | 7782-99-2 |
| Beilstein Reference | 1204124 |
| ChEBI | CHEBI:26851 |
| ChEMBL | CHEMBL1233328 |
| ChemSpider | 595 |
| DrugBank | DB14534 |
| ECHA InfoCard | ECHA InfoCard: 031-007-00-2 |
| EC Number | 016-094-00-1 |
| Gmelin Reference | 1364 |
| KEGG | C01353 |
| MeSH | D013436 |
| PubChem CID | 1099 |
| RTECS number | WS4250000 |
| UNII | K1CVM13F96 |
| UN number | UN1845 |
| CompTox Dashboard (EPA) | DTXSID5027287 |
| Properties | |
| Chemical formula | H2SO3 |
| Molar mass | 82.07 g/mol |
| Appearance | Colorless solution |
| Odor | Pungent odor |
| Density | D1.64 g/cm3 |
| Solubility in water | miscible |
| log P | -0.471 |
| Vapor pressure | 14 mm Hg (20°C) |
| Acidity (pKa) | 1.9 |
| Basicity (pKb) | 6.99 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 1.332 |
| Viscosity | Water thin |
| Dipole moment | 1.17 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 111.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -611 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -771 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | V07AB52 |
| Hazards | |
| Main hazards | Corrosive, causes severe skin burns and eye damage, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H314: Causes severe skin burns and eye damage. H331: Toxic if inhaled. |
| Precautionary statements | P260, P264, P280, P301+P312, P305+P351+P338, P310, P330 |
| NFPA 704 (fire diamond) | 2-0-0-Acid |
| Lethal dose or concentration | LD50 (oral, rat): 820 mg/kg |
| LD50 (median dose) | Sulfurous acid LD50 (median dose): "Sulfurous acid LD50 (oral, rat): 280 mg/kg |
| NIOSH | RN636 (string) |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Sulfurous Acid: "2 ppm (as sulfur dioxide, SO2) |
| REL (Recommended) | 1 – 10 mg/L |
| Related compounds | |
| Related compounds |
Sulfuric acid Sulfite Sulfur dioxide Thiosulfuric acid |

