Succinyl Chloride: A Ground-Level Look at Its Legacy and Potential
Historical Development
Chemistry’s modern toolkit owes a lot to the methodical effort of generations, and succinyl chloride didn’t just pop out of thin air. Early research drew from the foundational studies of chlorinated carboxylic acids, routes that saw experimentation and improvements throughout the 20th century. Back in college, I remember reading about scientists wrestling with acylation reactions. Succinyl chloride’s story links with broader pushes to develop practical acid chlorides for organic synthesis, essential for pharmaceuticals and dyes in post-war industry. These efforts came from chemists seeking higher reactivity and cleaner transformations, a constant theme in both academia and manufacturing. While trends have changed, the focus on precision and reproducibility remains. Most labs, including those I’ve worked in, came to rely on versatile building blocks like succinyl chloride, not just because they're useful, but because their history has shown reliability and efficiency in real-world applications.
Product Overview
Succinyl chloride stands as more than just another reagent on the shelf. For people knee-deep in lab work, it feels familiar: a colorless to pale yellow liquid with a characteristic biting odor. This chemical helps structure-building in organic synthesis, letting chemists efficiently swap out functional groups. It usually draws attention for its ability to activate acids for acylation, especially in pharma or specialty materials. I’ve seen it bridge gaps that other reagents couldn’t when constructing heterocycles or peptide analogs. While it rarely takes center stage like some blockbuster catalysts, those who work with complex organics know its value as an accessible workhorse.
Physical & Chemical Properties
Few reagents prompt the same wariness in the lab. Succinyl chloride’s pungency, considerable reactivity with water, and ability to emit hydrogen chloride gas turn each use into a test of skill and caution. Its boiling point hovers in the mid-range compared to related compounds, and volatility remains manageable with standard glassware. Handling it reminds me of why some lab stories include cautionary legends—direct contact causes burns, and even the vapors bring trouble if inhaled without proper ventilation. These attributes mean that even the most seasoned chemists triple-check their protective gear when the time comes to measure or transfer it.
Technical Specifications & Labeling
Manufacturers agree on one thing: clarity in labeling reduces risk and confusion. Bottles of succinyl chloride, whether arriving on a loading dock or pulled from a cold-room shelf, come with explicit hazard warnings, batch data, and precise purity ratings. Genuine trust develops when specifications match what’s stated—something I’ve come to expect from trusted catalog suppliers. In day-to-day lab work, I’ve learned to double-check the lot and concentration before starting a run, since even small contaminants skew outcomes in sensitive synthetic routes. A well-labeled bottle, with details about composition and stability, reassures lab techs and researchers who rely on consistency for reproducible results.
Preparation Method
Traditional production uses reactions of succinic acid or its anhydride with chlorinating agents, such as phosphorus trichloride or thionyl chloride. Some labs pursue greener options, but efficiency and cost keep old methods in play across the globe. I recall early attempts to minimize hazardous byproducts, and while innovation continues, commercial-scale chemistry still leans on these tried-and-true approaches. The outcome, unsurprisingly, is a product ready for direct use in further synthesis—no fancy purification steps that add risk or waste.
Chemical Reactions & Modifications
Anyone who’s synthesized amides or heterocycles will recognize succinyl chloride’s knack for rapid acylation. Its two acyl chloride groups allow for tailored substitutions; alkyl or aryl amines react almost explosively in the right conditions, giving rise to diverse products from straightforward to highly specialized compounds. There’s a reason it turns up over and over in patent literature and academic journals: it delivers clean, purposeful transformations with impressive speed and reliability. I’ve used it to build both simple diacid derivatives and more sophisticated entities by leveraging its twin reactivity. Its predictability keeps process chemists coming back, especially when eliminating side products ranks high on the priority list.
Synonyms & Product Names
Reading label variations always gives new insight into chemistry’s multi-lingual world. Succinyl chloride hides behind a few aliases, like butanedioyl dichloride and succinic acid dichloride. Each synonym reveals a bit about how different traditions in chemistry name their building blocks. For the most part, the official name signals its core structure—succinate backbone, swapped hydrogens for chlorines. Catalog codes might vary, but the underlying molecule remains a constant, quietly making modern organic synthesis tick.
Safety & Operational Standards
Working with succinyl chloride feels like a live demonstration of everything safety instructors warn about: corrosiveness, toxic fume generation, and potential for violent hydrolysis. Personal experience has shown, more than once, that shortcuts with PPE or fume hoods can lead to disaster. Reputable labs stick to strict storage, solvent compatibility, and waste management protocols. Legal controls help, but peer-driven culture—where colleagues watch for lapses and double up on glove checks—keeps accidents at bay more often than not. Training remains critical; nothing teaches respect for reactive chemicals like a splash-burn scare or an unscheduled evacuation. Real progress in safety has come from improved engineering controls, thorough training, and open discussion of near-misses.
Application Area
Chemists prize this compound for direct, reliable pathways to other valuable chemicals. Pharmaceutical labs use succinyl chloride for building peptide drugs, functionalized biodegradable polymers, and specialty catalysts. Industrial settings rely on its efficiency in acylation steps, with end products stretching from dyes to protective coatings. Its reach extends further than most would guess. I’ve seen it appear in processes for agricultural chemicals, polymer modification, even experimental battery materials. The unromantic truth remains that many modern materials and drugs owe at least a sliver of their success to these backbone reagents, bridging starting materials with more complex targets.
Research & Development
Research communities explore new uses for succinyl chloride almost as a rite of passage. Adjusting its reactivity, pairing it with novel nucleophiles, or tuning reaction conditions for greater selectivity continues to drive papers and patents. Academic curiosity pushes for more sustainable routes, and in my own work, greener chlorinating methods receive plenty of attention. Younger chemists bring fresh energy to the search for methods that use less hazardous reagents, while still producing the flawless product quality industry expects. Startups and established suppliers alike stay alert for hints of breakthroughs, because improvements at this level ripple outward to every downstream product.
Toxicity Research
Toxicologists and occupational health experts have learned the hard way about succinyl chloride’s risks. Exposure inflicts acute irritation, especially for skin, eyes, and respiratory tract. Inhalation of vapors means trouble almost immediately, regardless of age or tolerance. Early animal studies raised enough alarms for regulatory bodies to insist on the safety standards labs and plants now take for granted. People working long-term with acid chlorides rarely forget the training or the stories—burns, asthma, and chronic respiratory issues leave little to chance. Ongoing research probes chronic effects and environmental behavior, since producers and waste handlers both face risks if breakdown proves incomplete or containment fails.
Future Prospects
Demand isn’t fading, but scrutiny keeps rising. As green chemistry matures, efforts to develop alternative acylating agents draw more funding and attention. Some projects aim to dial down environmental hazards and toxic byproducts, especially compared to traditional production routes. Digitalization and automation in chemical manufacturing also promise better control, both for quality and for minimizing human exposure. Regulatory landscapes keep shifting, requiring companies and labs to rethink handling, tracking, and disposal. Based on what I see—both in published literature and conference talks—the field moves steadily toward safer analogs, more efficient processes, and deeper understanding of exposure risks. Succinyl chloride won’t disappear soon, but its story will keep evolving as technology, art, and necessity reshape what chemists demand from their reagents.
What is succinyl chloride used for?
The Real-World Workhorse in Chemistry Labs
The name "succinyl chloride" doesn’t ring a bell for most people, but this clear, pungent liquid holds weight in the world of chemical synthesis. Anyone who’s spent time in a university lab—or worked in chemical manufacturing—knows its distinct, biting smell and the tight storage protocols that come with it. My own days as a research assistant taught me rather quickly that mishandling a reagent like succinyl chloride means an emergency shower and a pile of ruined notes. Lab work with this chemical reminds you how reactive these compounds can get.
Succinyl chloride serves as a building block in organic chemistry. Researchers lean on it when crafting complex molecules, especially those for pharmaceuticals. Chemists use it to create succinyl derivatives, which get plugged into drugs and agrochemicals all over the world. These modified compounds improve how medicines behave in the body, like unlocking better drug absorption or changing how long the effect lasts. The impact shows up in some antibiotics, muscle relaxants, and treatments for epilepsy. The pharmaceutical world would face more challenges producing life-saving treatments without this chemical in their toolkit.
Working Behind the Scenes in Industry and Research
The story doesn’t end in hospitals. Succinyl chloride pops up in the making of polyamides and polyesters—two large families of plastics and fibers. These materials show up in everyday objects, from car parts and clothing to packaging. The chemical helps string molecules together, shaping how tough or flexible a final product gets. That means manufacturers can design items that don’t just look good but also hold up to daily use. My old hiking boots probably owe some of their durability to the chemistry that started in a plant making succinyl chloride.
I’ve seen the headaches caused by mismanaged chemicals, and succinyl chloride demands respect. It reacts quickly with water or even moisture in the air, releasing hydrogen chloride gas—a real hazard to lungs and eyes. Strict handling and storage routines keep people safe. Every time a spill happens or a container breaks, emergency crews jump into gear. Big plants have invested in sealed transfer systems and specialized training just to lower these risks. Truth is, the chemical’s usefulness only outweighs its hazards because people stick to best practices.
Balancing Progress with Safety and Sustainability
Worries about health don’t end at the factory door. The byproducts from succinyl chloride can pollute water and harm local ecosystems if not managed right. Over the years, tougher rules have forced companies to cut leaks and invest in technologies that capture or neutralize dangerous waste. This isn’t just about fines; communities keep a close eye on what gets released near their homes. Being a good neighbor now means treating waste before it leaves the site and finding tricks to recycle leftover chemicals.
Green chemistry has started to chip away at the reliance on aggressive reagents like succinyl chloride. Researchers now hunt for milder alternatives and redesign processes to lower risk and waste. As these options grow, they promise safer workplaces and cleaner communities. Still, in many reactions, there’s no easy substitute yet. For now, succinyl chloride sticks around as a key tool, shaping materials and medicines people use every day.
What is the chemical formula of succinyl chloride?
Unpacking Succinyl Chloride's Chemical Identity
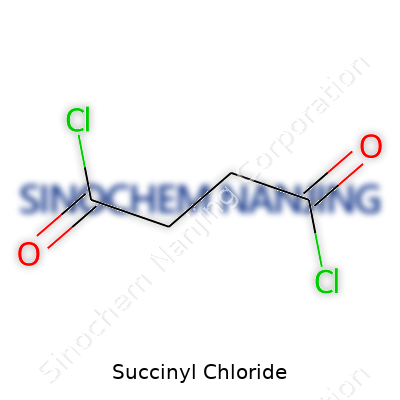
Succinyl chloride’s formula is C4H4Cl2O2. Just looking at those numbers tells a story. It’s a molecule built from four carbon atoms, four hydrogens, two chlorines, and two oxygens. If you break it down, succinyl chloride starts as succinic acid, which turns into its diacid chloride form by swapping the acid group’s OH parts for chlorine. You swap out one element for another and you often get something that acts very differently in a bottle or in a reaction flask.
Real-World Chemistry: Why We Care About Succinyl Chloride
People who work in labs like myself come across this chemical often. Succinyl chloride is a top pick as a building block. It reacts with lots of things—amines, alcohols, water—sometimes with eye-watering results. One time, a careless drop on the lab bench had the room smelling like hydrochloric acid in no time. That’s no surprise given how it's made. Take succinic acid, add a chlorinating agent (usually thionyl chloride or phosphorus pentachloride), and the reaction spits out our friend succinyl chloride along with a blast of byproducts like sulfur dioxide or hydrogen chloride gas.
Chemists use this transformation to get to other products—medicines, agricultural chemicals, dyes. I once spent a month converting amino acids into fancy peptides, and the only thing that worked consistently as a linker was succinyl chloride. Its formula gives it two outstretched arms for grabbing hold of other molecules. That makes it extremely useful in making things stick together, whether you’re designing a polymer or connecting pieces in some complex medicine.
Risks: Handling Isn’t Optional
There’s no room for amateur hour here. Succinyl chloride is nasty if handled poorly. Its reaction with water sends off clouds of hydrogen chloride, a gas that burns eyes and lungs. In our lab, working with it meant hood fans running, gloves on, goggles tight. Data suggests that exposure over time can bring lasting respiratory problems, and spills mean destroyed workspace until the area gets scrubbed clean with neutralizing agents.
Anyone who thinks these risks aren't real only needs to check the statistics. Each year, chemical injuries in labs still happen, often from folks thinking they can cut corners on safety when working with compounds like this. Fortunately, solid policies and real hands-on training stop most accidents. Teaching new lab members proper protocols sticks out as one of the best things I've done—not just for their own health but for everyone who shares space or equipment.
Solutions and Next Steps
Better labeling and improved education help reduce mistakes. Digital tracking systems sound fancy but have saved plenty of panic and confusion. If you don’t know exactly where chemicals are and how much you have, problems compound quickly. Beyond this, companies and universities have started requiring refresher training for returning workers. I’ve seen the impact—a few hours a year can mean the difference between a clean record and a costly accident.
Getting the formula for succinyl chloride right isn’t just an exercise in memorization. It’s about understanding how one compound fits into dozens of essential processes, knowing what makes it useful, recognizing the risks, and making choices to keep people safe without shutting down progress. The right knowledge always pulls its weight far beyond the lab.
What safety precautions should be taken when handling succinyl chloride?
Why Succinyl Chloride Demands Respect
Most chemists eventually cross paths with succinyl chloride. I remember the moment I opened my first bottle. An invisible fog seemed to seep from it, followed by the distinct scent that tells you trouble could be lurking. Succinyl chloride reacts quickly with water and produces corrosive fumes, particularly hydrogen chloride. Skin, lungs, and eyes all face real risk. There’s no room for shortcuts when handling this substance; past mistakes from others have written every line of lab protocol for a reason.
PPE: Suit Up for Safety
Lab coats and safety goggles offer a good baseline, but succinyl chloride asks for more. I never open a bottle without splash-resistant goggles, and every bit of skin needs coverage — including chemical-resistant gloves. Nitrile or neoprene gloves outperform latex here. Face shields step in for large transfers, since accidents often catch you looking the other way. Long sleeves, buttoned cuffs, and even a disposable apron keep exposure to a minimum.
Ventilation: Keep the Air Moving
I always work with succinyl chloride under a well-maintained chemical fume hood. Hydrogen chloride fumes irritate mucous membranes and can set off asthma attacks within minutes. Good airflow pulls fumes away, while keeping concentrations below dangerous levels. Poor ventilation or lack of a functioning hood ramps up risk faster than most people realize.
Bottle Handling, Storage, and Preparation
Succinyl chloride bottles demand respect during every transfer. I check the integrity of containers and avoid glass whenever possible if any pressure could build up. Only the smallest amount leaves the stock bottle at a time. Each transfer stays slow and close to the bench — spills and splashes happen fast, especially when gloves grow slick. Secondary containment trays help catch every drip and drool.
I find dry, well-marked cabinets away from water sources keep containers safe until they’re needed. Humidity, even from a loose sink pipe nearby, spells disaster. A bottle left open or without a tight seal eventually sucks in water and turns to a sticky, fuming mess.
Reactivity and Incompatibles
Succinyl chloride picks fights with water, alcohols, amines, and strong bases, generating heat and toxic gas. Even a trace of moisture lights the fuse. I learned quickly to check all glassware for dryness. Any cleaning solvent must dry fully before use. Mixing with amines makes things heat up — sometimes much faster than you’d expect. These reactions can go runaway if left unsupervised or measured carelessly.
Spills and Emergency Response
Accidents happen, no matter how careful the plan. I keep neutralizing agents like sodium bicarbonate or calcium hydroxide powder nearby, but never pour water on a spill. Proper chemical spill kits sit within arm’s reach. Bigger spills mean pulling the fire alarm and getting everyone out, not heroics. For contact with skin, flushing with water is the only move, yet with serious exposure, medical help becomes urgent.
Education and Training
Nobody should handle succinyl chloride without understanding its hazards in detail. Labels, current safety data sheets, and clear instructions belong front and center before any experiment starts. I still review up-to-date resources from trusted institutional safety programs and industry standards. Staying up-to-date with real case studies and fresh protocols helps avoid mistakes others already paid for.
How should succinyl chloride be stored?
Getting Serious About Chemical Safety
I’ve spent years around chemical storerooms, and nothing gets my attention like succinyl chloride. If you scan a safety data sheet, the dangers jump right out. This stuff reacts fast with water, including the moisture in the air. Leave it open on a bench, and you get hydrochloric acid vapor in seconds. For anyone breathing nearby, that’s a direct ticket to respiratory trouble. Fumes smell sharp and irritable—your eyes and throat will tell you something's wrong before you read any labels.
This isn’t a chemical to shove in the same place as everyday solvents. The best approach: keep it in a tightly-sealed glass bottle, with the lid double-checked every time after use. Plastic bottles don’t cut it here, since succinyl chloride can eat through certain plastics and start leaks that no one wants to find. Glass keeps things contained and doesn’t interact with the compound, so it stands up to both time and chemical reactivity.
Humidity: The Invisible Enemy
Humidity sneaks up on you. Open a bottle for a few quick seconds in a damp storeroom, and unwanted reactions begin. The solution lies in a good-quality desiccator. I’ve learned from past mistakes: extra steps pay off. Add silica gel or phosphorus pentoxide close by to soak up stray water vapor. Experienced lab workers double-pack the bottle—first inside a sealed bag, then inside the desiccator. Every added layer cuts the odds of a surprise reaction on the shelf.
Storing Cool and Isolated
Heat speeds up reactions, so room temperature isn’t the way to go. I always look for flameproof refrigerators rated for hazardous materials. Succinyl chloride doesn’t belong in a crowded fridge with peroxides or acids. Jumping chemicals close together in storage makes a perfect storm—mix ups or leaks could set off dangerous chain reactions. Set up a specific spot, sectioned off from oils, oxidizers, and anything with alcohol groups. Even a small splatter can lead to choking fumes and a rush for the eye wash.
Respect the Label, Track the Material
Labels often fade, so I use thicker permanent ink for compound names and hazard warnings. It’s tempting to skip strict tracking on chemicals you open rarely. That’s a mistake. I learned the hard way that busy team members may forget about a half-used bottle hidden at the back. Inventory checks each month help spot old materials fast. Succinyl chloride hydrolyzes slowly, so any old bottles can build up pressure and lead to broken lids or more serious accidents.
Planning for Spills and Training
Even the tightest protocols can fail. I always remind juniors: spills aren’t rare, and hesitation compounds trouble. Keep neutralizing agents close, like calcium carbonate or sand—not water. Goggles and gloves are great, but a splash-proof lab coat is better. Spill kits belong in arms’ reach. Training drills, not just written instructions, help everyone respond quickly and cut down risks when something goes sideways.
Succinyl chloride won't give second chances. Careful storage comes from knowing the actual dangers and building habits. Ingredient labels or datasheets won’t protect you. Consistent routines and honest respect for chemical hazards do far more to keep every workday safe and productive.
What are the potential hazards and first aid measures for succinyl chloride exposure?
Real Dangers in the Lab and Workplace
Succinyl chloride shows up mostly in chemical production, research, and some industrial processes. Handling something like this brings real risk, especially if you’ve ever worked on a factory floor or in a college chemistry lab. Transparent liquid, sharp strong odor, and the label "corrosive" always slapped front and center—there’s a reason for that. Getting careless with it or skipping gloves can cost you. Even a single splash can burn your skin within seconds.
Now, most folks don’t realize succinyl chloride reacts with water, steam, or even moist air, producing hydrogen chloride gas. That means eye exposure, a lungful of fumes, or even breathing near an open container can send you running for help. Many researchers learn early after their first scare that goggles, gloves, and a fume hood aren’t suggestions—they’re bare minimum safety.
Immediate Health Hazards
Most of us can recall the stinging feeling of getting solvent or acid on our skin. Multiply that by ten with succinyl chloride. Even a small spill leads to deep burning, blisters, and terrible pain. Eyes take damage fast, putting vision in serious danger.
Inhalation is worse. Breathing its vapors causes severe coughing, choking, and sometimes swelling in your airway. Permanent lung damage isn’t just a threat in textbooks. Even indirect exposure—maybe from a neighboring bench or faulty hood—can set off headaches, nausea, and burning lungs. In my grad school days, a poorly labeled bottle led a classmate straight to the ER.
First Aid People Remember
Experienced folks know rinsing with water, and plenty of it, proves the best first move. For accidental skin contact, pull off contaminated clothes. Stand under an emergency shower. Don’t skimp on time—fifteen solid minutes under cold running water can save skin tissue.
Facing splashes to the eyes, speed is life. Head for the eyewash station without delay and flush eyes wide open for at least fifteen full minutes. Most eye injuries recover if rinsed right away, but too many put off the wash, hoping the sting fades. It doesn’t.
Take inhalation emergencies seriously. Quickly get to fresh air. Don’t tough it out. If someone looks dizzy or has trouble breathing, call emergency medical help—don’t wait. Succinyl chloride fumes can worsen asthma or trigger swelling in folks with allergies. In riskier jobs, having a designated buddy system helps spot trouble before it spirals.
Building a Better Safety Mindset
People can’t control every accident, but preparation makes the difference between real harm and a close call. Everyone in the workspace should know what’s in each bottle, keep protective gear on hand, and actually walk through spill drills a couple of times a year. Label everything. Unmarked bottles cause as many emergencies as outright carelessness.
For supervisors and safety leads, extra training helps. Not just in chemical handling, but in spotting small lapses—unbuttoned lab coats, loose stoppers, broken fume hoods. Routine checks, open conversations about "scary near-misses," and quick access to first aid supplies matter more than fat safety manuals gathering dust.
Resources and Real World Solutions
Government agencies like OSHA and the National Institute for Occupational Safety and Health lay out clear guidelines for handling hazardous chemicals. Make use of the tools: up-to-date safety data sheets, on-site eyewash and shower stations, spill kits, and proper ventilation. If you’re ever unsure, or working with new chemicals, stop and review the materials before diving in.
No shortcut matches reading up and preparing. Lives change with a single misstep. Succinyl chloride teaches hard lessons, but solid training, real respect for the risks, and quick first aid keep people working safely—every single day.
| Names | |
| Preferred IUPAC name | Butanedioyl dichloride |
| Other names |
Butanedioyl chloride Succinic acid dichloride |
| Pronunciation | /ˈsʌk.sɪ.nɪl ˈklɔːr.aɪd/ |
| Identifiers | |
| CAS Number | 4790-18-5 |
| Beilstein Reference | 1720932 |
| ChEBI | CHEBI:131278 |
| ChEMBL | CHEMBL46306 |
| ChemSpider | 12626 |
| DrugBank | DB14181 |
| ECHA InfoCard | ECHA InfoCard 100.003.679 |
| EC Number | 208-866-2 |
| Gmelin Reference | 8377 |
| KEGG | C00956 |
| MeSH | D013429 |
| PubChem CID | 61317 |
| RTECS number | WL8225000 |
| UNII | N8KGR0476D |
| UN number | UN3261 |
| Properties | |
| Chemical formula | C4H4Cl2O2 |
| Molar mass | 183.03 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | Pungent |
| Density | 1.401 g/mL at 25 °C |
| Solubility in water | Reacts violently with water |
| log P | 0.83 |
| Vapor pressure | 0.4 mmHg (20 °C) |
| Acidity (pKa) | 1.90 |
| Basicity (pKb) | pKb: -1.3 |
| Magnetic susceptibility (χ) | -5.11 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.473 |
| Viscosity | 1.436 mPa·s (20 °C) |
| Dipole moment | 2.64 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 354.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -572.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -878.1 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS02,GHS05 |
| Signal word | Danger |
| Hazard statements | Hazard statements: "H302, H314, H335 |
| Precautionary statements | P260, P261, P264, P271, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P311, P312, P321, P330, P363, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-1-W |
| Flash point | 71 °F (22 °C) |
| Autoignition temperature | 365 °C |
| Lethal dose or concentration | LD50 oral rat 1660 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 371 mg/kg |
| NIOSH | SCB |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.5 ppm |
| IDLH (Immediate danger) | IDLH: 3 ppm |
| Related compounds | |
| Related compounds |
Succinic acid Succinic anhydride Succinimide Succinyl-CoA |

