Styrene Oxide: A Comprehensive Commentary
Historical Development
Styrene oxide’s story stretches back to the deeper days of industrial chemistry. Early makers and users saw value in turning styrene—a substance already known for plastics—into something more chemically dynamic. In the mid-20th century, researchers took notice of this epoxide because it could act as a reactive building block. Factories started making it by oxidizing styrene, leading to wider availability and a surge in industrial applications. Since those first steps, advances in catalysts and engineering have sharpened the way industries approach each stage, improving both yield and purity. My own reading of industry records shows how batches grew larger yet more precise, echoing the growth of demand for strong intermediates in pharmaceuticals and polymers. Concerns about worker exposure and tight regulatory controls also began taking root as more became known about the risks associated with handling this substance.
Product Overview
Styrene oxide stands out in the market as a lively intermediate, connecting petrochemicals to finished materials that people touch every day. The industry mostly produces it as a clear, colorless liquid, packaged in steel drums or specialty containers to prevent leaks and contamination. I have seen it used in small research blends and industrial-scale batches with equal seriousness about labeling and traceability. Because buyers include everything from universities to multinational chemical plants, product quality and trust in suppliers shape the business as much as innovations in production. The best suppliers run tight ships with their purity promises, as even minor contamination can throw off a sensitive synthesis or trigger unwanted side effects in downstream products.
Physical & Chemical Properties
Styrene oxide’s appearance won’t catch the eye—just another clear liquid. What sets it apart is its molecular structure: a three-membered epoxide ring joined to a vinyl benzene group. That strained ring stores chemical energy, making it jumpy and ready for reactions that open new molecular doors. This compound boils near 194 degrees Celsius, has low water solubility, and gives a faint, sometimes sweet odor. One drop will float atop water, holding together in a layer that reveals next to nothing about what’s lurking beneath. It burns, reacts with acids and bases, and breaks down when faced with strong light or heat. My memories of working with it include a strong respect for its volatility—the smallest spill brings a sharp scent and brisk cleanup.
Technical Specifications & Labeling
Standards matter. Bottles and drums of styrene oxide carry clear hazard labels, hazard numbers, and lot specifics for traceability. Most grades meet strict requirements for purity, water content, and absence of byproducts such as benzaldehyde or acetophenone. Producers usually ship it at 98% or greater purity. Customers ask for certificates matching each container so they can trace any problem back to its source. Regulations force every packager to mark clear handling and storage instructions—this is not something an untrained worker can handle safely. I’ve seen laboratories demand sample reports and safety data sheets at every step, especially for applications tied to food, pharma, or sensitive polymerization processes.
Preparation Method
Styrene oxide typically comes from the oxidation of styrene. Factories use a range of catalysts—from peracids for small batches to silver oxide for larger setups. In smaller runs, m-chloroperoxybenzoic acid attacks the vinyl ring, folding in oxygen. Industrial plants run air or oxygen over styrene at controlled temperatures, collecting the epoxide as it forms. Some setups use benzoyl peroxide or other free radical starters to push the reaction along. Tight control over temperature and timing keeps unwanted side products at bay; just a small slip leads to ring-opened byproducts or polymerization. Good production lines recycle leftover reactants, both to keep costs down and to limit waste.
Chemical Reactions & Modifications
Chemists love styrene oxide because it reacts quickly with many chemical partners. The strained epoxide ring begs for nucleophilic attack, letting chemists open it in either acidic or basic environments. Water splits the ring to yield phenylethane-1,2-diol, an important starting material for fragrances and pharmaceuticals. Alcohols react to form ethers, amines create amino alcohols, and thiols generate mercapto-derivatives. The presence of the benzene ring adds flexibility—it encourages further reactions or modifications after ring opening. In research, I’ve witnessed how one reaction with an amine or thiol can create entirely new molecules, ready for further development. Manufacturers exploit its versatility to branch into specialty polymers, surfactants, and pharmaceutical intermediates.
Synonyms & Product Names
Chemistry’s naming habits ensure any one compound will surface under several names. Styrene oxide sometimes appears as phenyl oxirane, styrenepoxide, or vinylbenzene oxide. Trade names may add proprietary flare, but most research and shipping paperwork settles on these international names. Recognizing alternate names keeps supply chains and literature searches on track. In the past, confusion over product names has led to missed shipments or regulatory tangles, highlighting why standardized naming sticks in professional practice.
Safety & Operational Standards
Handling styrene oxide takes serious attention. The liquid poses fire risks, irritates eyes and skin, and requires vapor protection in all but the tiniest quantities. Industry facilities install specialized ventilation, robust gloves, safety goggles, and face masks to keep workers from coming in contact. Labeling spells out hazards—flammable, harmful if swallowed, may cause genetic defects—because experience and research agree the dangers are real. In larger plants, electronic monitoring and employee training prevent spills and accidental exposure. I’ve come across work sites where monthly drills test readiness, and spill kits hang in every lab corner. Disposal follows hazardous waste guidelines; any leftover or byproduct must be neutralized and tracked, both to keep people safe and meet environmental law.
Application Area
Chemists and manufacturers roll out styrene oxide in a spread of industries. Epoxy resins rely on it for strength and chemical resistance; fragrances and flavors demand certain intermediates that trace their origins to this epoxide. Some pharmaceuticals depend on its reactive ring to create new drugs or modify existing ones. Water treatment chemicals, surfactants, and even specialty plastics all call for epoxides like this at some stage. As a researcher, I’ve watched demand shift over time: epoxy applications expanded, then green chemistry pulled interest toward milder reaction pathways and alternatives. Changes in plastics demand or tighter rules around workplace exposure constantly shift the playing field, but most major chemical sectors still keep a stake in this intermediate.
Research & Development
Research teams circle around styrene oxide because it sits at the crossroads of basic and applied science. Teams in universities and industry labs look for new catalysts that lower energy needs or minimize dangerous byproducts. Computational chemists model ring-opening reactions, offering predictions that let experimentalists find new routes faster. Green chemistry programs aim for bio-based styrene sources, reducing reliance on fossil fuels and cutting carbon footprints. Other research focuses on expanding the list of derivatives, often with an eye on special polymers, new molecules for medicines, or clever surfactants. Collaborations tend to cross borders, blending experiences from bench chemistry, toxicology, and industrial engineering in hopes of winning patents and safer methods.
Toxicity Research
Evidence stacks up showing styrene oxide’s hazards. Animal studies and human monitoring both point to risks of mutagenicity and possible cancer connections. Short-term exposure burns mucous membranes and skin, with headaches and nausea at higher doses. Regulators take a hard line, setting workplace limits and requiring regular air sampling in any facility handling the substance. Chronic exposure links to cellular changes, so researchers keep pushing for better alternatives and safer handling. In labs I have visited, monitoring badges and vapor detectors stand as routine gear, showing that staying below exposure thresholds means checking every batch, every shift. Slow-moving regulations can frustrate researchers and workers, but every positive change in reporting and ventilation matters for the people doing the hands-on work.
Future Prospects
The future for styrene oxide straddles challenge and opportunity. As environmental pressures rise and new chemicals elbow their way into the market, older intermediates face scrutiny over both safety and sustainability. Many teams hunt for catalysts and production methods that cut waste, energy use, and exposure to dangerous byproducts. At the same time, new markets in pharmaceuticals and specialty materials still drive demand for reliable, versatile substances like styrene oxide. Some innovators develop bio-derived styrene routes, aiming to cut fossil fuel use and shrink carbon footprints. Advances in workplace safety and monitoring should hopefully shrink the risks for next-generation workers. In my experience, every step forward stems from close collaboration—engineers, chemists, safety experts, and end users pushing for stronger outcomes across production, application, and environmental control. Those who listen to the science and the front-line experience will steer where this compound fits in the evolving toolkit of modern chemistry.
What is Styrene Oxide used for?
Beyond the Lab: Everyday Chemicals at Work
I’ve met countless everyday products that trace their beginnings back to a chemistry lab, and styrene oxide counts among them. Walk through a factory floor that produces plastics, resins, or even fragrances, and you’ll likely spot drums labeled with this hard-to-pronounce name. So what role does this liquid play, and why should anyone outside a lab coat care about it?
Crafting Plastics and Polymers
Step into the world of manufacturing and you’ll find styrene oxide doing heavy lifting in the creation of epoxy and polystyrene plastics. In my time chatting with industrial chemists, they point to this chemical as a key ingredient for making coatings tougher and plastics more versatile. It behaves as a reactive intermediate, which means it helps other ingredients snap together into larger structures—sometimes as basic as those foam coffee cups, or as rugged as the linings in metal cans.
Many factories consider it a bridge chemical. Picture it as a handshake between raw styrene and the finished material that lands on store shelves. Without it, certain high-performance resins used in electronics or construction would cost more, feel different, and might not stick around as long under stress.
Fragrance and Flavors: The Unseen Ingredient
Not all uses focus on building hard plastics. The chemical also finds its way into the handful of compounds that help fragrance makers create sophisticated scents and flavors. Though you won’t see “styrene oxide” on perfume labels, its presence as a building block in aromatic formulas matters. As someone who has visited flavor labs, I’ve watched chemists combine it in tiny amounts with other chemicals to mimic the sweet, spicy, or fruity notes in packaged foods and personal care products.
Research and Medical Use
Researchers value this compound as a yardstick in toxicology and genetics studies. Lab animals exposed to it help teams understand how the body breaks down foreign chemicals, and scientists use this data to spot links between workplace exposure and health risks. Talk to a toxicologist and you’ll hear how necessary such “model” chemicals can be—especially for making workplaces in manufacturing safer.
Health and Safety: Looking in the Mirror
Concern about styrene oxide springs up because it doesn’t just break down and drift away. The US Environmental Protection Agency and International Agency for Research on Cancer both label it as potentially carcinogenic. I’ve spoken with plant safety managers who track air levels religiously and suit up in heavy gloves and respirators any time they handle it. Stories surface now and then about spills or chronic small-scale exposure. These raise fair questions about oversight and regular health checks for folks who mix industrial batches all shift long.
Keeping Workplaces Safer
Solutions exist. Factories switched to closed-system handling and use smart ventilation to limit accidental contact. Monitoring equipment now gives real-time alerts if levels get risky. On visits, I’ve watched operators get training on emergency cleanups, and most wear protective gear as standard practice. Encouraging companies to replace styrene oxide with safer alternatives where possible also shows promise, especially as green chemistry grows in influence. Regulatory bodies continue to push for tighter thresholds and stricter reporting to keep things in check.
Takeaway: Styrene oxide touches many corners of industry, often out of sight. Demand for strong plastics and resilient coatings keeps it in rotation, while growing focus on safety means its story continues to evolve.
Is Styrene Oxide hazardous to health?
What Is Styrene Oxide and Where Do We Find It?
Styrene oxide shows up in industry, not something folks bump into at the grocery store. Manufacturers use it to make resins, plastics, and even pesticides. When plastic coffee cup lids get hot, some byproducts like styrene can sneak into drinks. Workers in certain factories see it more often—think coatings, insulation, and rubber lines. The rest of us rarely touch the stuff directly, but it lingers in air near industrial sites.
The Science on Health Hazards
Researchers have flagged styrene oxide for some nasty business. Back in college, I remember reading environmental health journals—animal studies raised concerns long before regulations caught up. Rats exposed to high concentrations ended up with liver and lung problems. The chemical earns its bad reputation mainly from how it affects cells. Cells react almost like they're fighting a chemical flame. DNA gets dinged up; those genetic stumbles can add up to cancer later. In humans, health authorities have focused on links to leukemia and lymphoma. Evidence points toward risks but hasn't drawn a solid, unavoidable line yet. That uncertainty always bothered me, because people aren’t rats in cages—life is much messier.
Folks working with resins and polystyrene might breathe in styrene oxide vapors. Short-term exposure feels rough—things like throat irritation, headaches, and trouble concentrating. I heard from a family friend years ago who worked outside Houston in a plastics plant. He talked about nosebleeds and migraines on days when vent fans didn’t work well. Long-term risks include nerve damage and, in some reports, impacts on memory and mood. The International Agency for Research on Cancer put styrene oxide into the “possible human carcinogen” category. In plain speak, there’s enough uncertainty that caution makes sense, especially at higher exposures.
Why This Matters Today
Not everyone will ever come across styrene oxide at work, but the choices made in production ripple out. Poor ventilation in small shops means those chemicals hang around. If rules aren’t followed, folks risk far bigger problems. I trust the stories from older generation manufacturing workers who saw safety standards get ignored. No one wants a paycheck tied to bad health down the road.
Communities living close to plants making styrene-based goods need transparency. I remember volunteering at health clinics near industrial corridors; some parents worried about rare cancers popping up on their street. Experts can debate numbers, but worry feels real when health is on the line.
How to Limit the Danger
Basic steps can lower risks. Strong ventilation, personal protective equipment, and regular air checks can save headaches and worse. Companies showing their air test results or switching to safer alternatives earn respect and trust. Workers should know what they’re handling and how to stay safe without drama or buried facts. Community health studies can flag problems early if funded and followed through, not just started and forgotten when headlines fade.
Styrene oxide won’t show up in daily life for most people. Still, the folks who cross its path—on purpose or by accident—deserve clear warnings and honest effort to make things safer. Health should never end up as a trade-off for quick profits or skipped precautions.
What are the storage requirements for Styrene Oxide?
Why Styrene Oxide Deserves Careful Storage
Styrene oxide can easily slip off the radar for those who don’t work with chemicals every day, but a closer look reveals plenty of reasons for caution. It takes one whiff or spill to understand that this chemical packs both power and hazard. With a structure similar to solvents and plenty of volatility, it reacts to careless handling faster than some expect. The effects reach beyond the workers on site—fumy leaks and contaminated water don’t respect fences.
Environmental and Safety Concerns
Over the years, I’ve spent time on production floors and chemical depots. One lesson stands out: basic attention often prevents big trouble. Styrene oxide shares this truth. Vapors can build up and ignite even in modest heat. Inhalation brings headaches or worse. I’ve seen delays in labeling or half-closed containers turn a routine day into an emergency rush. That’s why simple steps carry so much weight.
Storage in a cool, well-ventilated space makes a real difference. I’ve opened sheds on a hot day and been hit with the sharp tang of escaping solvents. That’s your first sign something has gone wrong. For styrene oxide, room temperature doesn’t cut it in most climates. Cold indoor storage slows down evaporation and keeps the lid on risk. Corrosion and rust from high humidity eat through drums and cause more leaks. Dry, solid ground reduces those threats.
Containers and Compatibility
Not every barrel works for every chemical. Styrene oxide gets along best with steel lined with glass or PTFE, or with high-grade plastic drums. Don’t trust basic metal barrels or reused jugs. Once, a colleague short on space stored a drum beside acids—despite the warning labels—with small leaks popping up later. Chemicals like acids and even strong bases chew through containers and spark ugly reactions.
Drums and carboys should stay tightly sealed, avoiding frequent opening or rough handling. Always check for pressure build-up before opening, as heating makes vapors expand. Labels must stay clear because fading or peeling causes confusion later. A clear marker beats hasty guesswork every time, especially after a few months on the shelf.
Preventing Accidents and Spills
Real-world slips happen where instructions get ignored. I’ve seen rushed employees skip gloves or ignore eyewash stations. Pick storage locations that keep styrene oxide away from busy traffic, heat, direct sunlight, and sources of ignition—including electrical panels or open flames. Spills demand quick cleanup with absorbent pads meant for organics, not the kitchen mop. Companies put spill kits within easy reach, but staff must get regular refreshers—once or twice a year, minimum. I’ve seen folks freeze up at the worst moment simply because their last drill was ages ago.
Keeping an updated inventory, logging old stock, and moving expired or leaky containers out brings down the risk of forgotten hazards. Training and honest communication shape habits more than rigid checklists. In all the places I’ve worked, the safest warehouses had managers who talked through every close call and stored samples for inspection.
Storing for the Long Haul
Styrene oxide doesn’t care about the profit margin or how rushed the crew feels. It just reacts—chemically and physically—according to conditions. Respect on the front end pays off later, preventing fines, health scares, and lost business from a single avoidable leak. Resilience in a storage program grows through real oversight, practical training, regular checks, and the humility to ask questions. That’s the lesson repeated experience brings to chemical stewardship.
What is the chemical formula of Styrene Oxide?
Breaking Down the Basics
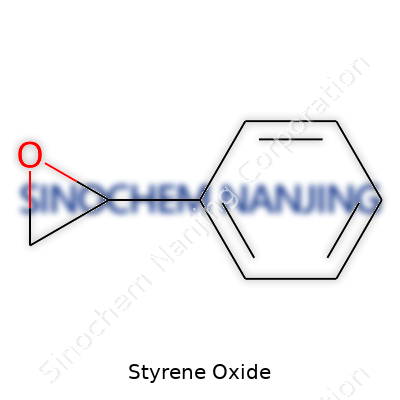
Styrene oxide sits in the corner of organic chemistry with the formula C8H8O. One oxygen atom, eight carbons, eight hydrogens. Sometimes it doesn’t get the attention other compounds receive, but its structure carries a punch. The presence of an epoxide ring makes it far more reactive than basic styrene. This detail matters, especially for anyone working hands-on with chemicals in labs or factories.
Why Knowing the Formula Matters
During my years studying and working in a lab, I ran into styrene oxide while prepping experiments for polymer research. The chemical's properties took me by surprise the first time. With an extra oxygen packed on a three-membered ring, reactivity shoots up. You can’t approach it like you would other aromatic compounds. If you spill this stuff, you need to pay full attention to ventilation and protective gear. Even a small error could lead to skin irritation or worse.
The C8H8O formula isn’t something just for chemistry exams. It’s there for safety data sheets, for figuring out toxicity, for making sure that you understand how much to measure in a reaction. Forgetting a single atom changes results, changes risk, and can affect everyone involved.
Health Hazards and Industry Use
Styrene oxide pops up in the production of specialty polymers, pharmaceuticals, and sometimes even in research settings. Its reactivity helps make new molecules, but that same strength can turn on you in poorly ventilated labs or tight manufacturing spaces. Studies from NIOSH and the EPA show that styrene oxide can damage DNA, and animal tests reveal clear links to liver toxicity and cancer risk. Out there in manufacturing, where exposure can last all shift long, health safeguards need to keep up.
Facts are facts: according to the International Agency for Research on Cancer, styrene oxide counts as a possible human carcinogen. Consistent low-level exposure increases the odds for mutations in cells, and years of monitoring workers in resin plants show elevated cancer rates. Outdated factories and poorly maintained safety protocols only worsen potential hazards. Paint thinner smells, headaches at the end of the day, and a rash that took weeks to fade — I’ve seen all these result from ignoring the chemical’s structure and toxicity.
Tackling Safety and Solutions
It’s tempting to power through a shift and overlook that odd smell or tingling in your hands, but health lasts a lifetime. For those who work directly with styrene oxide, updated personal protective equipment and regular air monitoring make a world of difference. I remember the first time a plant I visited upgraded to fume hoods with proper filtration — absenteeism from illness dropped within a month. Building a system where every worker gets practical training, not just a quick sign-off, pays off over time.
On an industry level, switching to less hazardous epoxides or engineering safer closed-loop systems shows promise. Some companies now use substitutes in manufacturing where possible, saving money and improving worker health. Substitution and airtight automation don’t just lower risk; they draw a map for the rest of the industry. Watching old colleagues make it to retirement without serious health problems tells you everything you need to know.
Small Formula, Big Impact
C8H8O isn’t just a label for a lab bottle. It stands for real-world decisions about safety and sustainability, tied up in the practical work of chemistry, manufacturing, and health. Taking its risks seriously means better choices on the floor and safer results all around.
How should Styrene Oxide be handled safely?
Why We Should Pay Attention
A lot of us rarely think about the chemicals that turn up inside industrial settings, labs, or even the background of everyday goods. Yet, take something like styrene oxide—a clear, colorless liquid with an almond-like odor. People handle this stuff in places from plastics manufacturing to research labs. It’s not about being afraid. It’s about knowing what’s on the line for you and folks you work with.
Facts about the Health Risks
Styrene oxide doesn’t just smell; it packs a punch on contact. Touching it can cause skin burns and irritation. Breathing it in can hit your eyes, nose, throat, and may even reach the lungs, sparking coughing and shortness of breath. Deeper exposure brings deeper problems—long-term research connects styrene oxide to mutagenic effects, hinting at cancer risk. No need to tally off medical journal citations to see this calls for respect.
It enters the body through the skin, breathing, or swallowing. If left unchecked, it works its way in and messes with cells. Short-term dizziness, headache, or even fainting are warning signs screaming for better safety. So, the issue grows bigger than just following a rulebook.
Safe Handling Starts with Smart Choices
First step for anyone facing styrene oxide is a real-deal protective routine. Picture a worker in a plant pouring out styrene oxide to mix up resins. Proper gloves (think nitrile or neoprene, not the thin, cheap kind), safety goggles to block splashes, and a fitted lab coat should be the day-to-day armor. Good ventilation matters too: open windows don’t cut it. You want local exhaust right where the chemical hits the air. The reason: vapors travel and collect fast in low spots, carrying real risks for anyone nearby.
Emergency showers and eye-wash stations shouldn’t be afterthoughts hidden at the back of the building. In one factory I visited, an accidental splash meant a quick rush to rinse—nothing fancy, just fast, easy access. It made a difference. Being ready means stationing first aid gear within reach and drilling staff until it’s muscle memory.
Training Empowers, Not Just Protects
No one benefits from a thick binder of rules nobody reads. Training should go past slide shows. Live demonstrations, hands-on sessions, even watching someone else go through a spill and cleanup: these things stick. People remember what they’ve done and seen far longer than pages of bullet points. Regular refreshers keep the knowledge sharp so nobody forgets which mask to grab, or how to check for leaks in a glove.
Waste disposal counts as much as what happens during the shift. Styrene oxide waste can’t get dumped with regular trash—it belongs in sealed, correctly labeled containers, stored upright. Experienced folks double-check the seals and labels before taking things to the disposal area. Law and ethics sync up here: cutting corners can lead to fines, but it’s the people’s health that actually gets hurt most.
Building a Culture That Cares
Protecting against styrene oxide isn’t only a personal task. Peers look out for each other, catching possible mistakes or forgotten steps. Companies that reward good safety practice encourage everyone to stay alert. Leadership has a job to do here—open conversations about what worries staff and fixing trouble spots before someone pays the price.
In the end, keeping safe with styrene oxide means blending tough science with honest, day-to-day caution. When people feel respected and heard, they look out for themselves and everyone else—and that matters more than any wall poster covered with warnings.
| Names | |
| Preferred IUPAC name | phenyloxirane |
| Other names |
Phenylene oxide Epoxybenzene Oxirane, styrene Styroxide |
| Pronunciation | /ˈstaɪriːn ˈɑksaɪd/ |
| Identifiers | |
| CAS Number | 96-09-3 |
| Beilstein Reference | Beilstein Reference: 1919404 |
| ChEBI | CHEBI:17215 |
| ChEMBL | CHEMBL1234859 |
| ChemSpider | 5586 |
| DrugBank | DB02235 |
| ECHA InfoCard | ECHA InfoCard: 100.003.375 |
| EC Number | 202-476-7 |
| Gmelin Reference | 6362 |
| KEGG | C01778 |
| MeSH | D013343 |
| PubChem CID | 7515 |
| RTECS number | WL2455000 |
| UNII | 9S1U4J8U7Q |
| UN number | UN2056 |
| CompTox Dashboard (EPA) | DTXSID2020227 |
| Properties | |
| Chemical formula | C8H8O |
| Molar mass | 120.15 g/mol |
| Appearance | Colorless liquid |
| Odor | aromatic odor |
| Density | 1.045 g/mL at 25 °C |
| Solubility in water | 7.1 g/L (20 °C) |
| log P | 2.1 |
| Vapor pressure | 0.6 mmHg (20 °C) |
| Acidity (pKa) | 19.0 |
| Basicity (pKb) | 15.55 |
| Magnetic susceptibility (χ) | -54.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.495 |
| Viscosity | 2.5 mPa·s (20 °C) |
| Dipole moment | 4.11 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 322.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 82.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4320 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07, GHS08 |
| Pictograms | GHS02,GHS05,GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H332, H341, H351 |
| Precautionary statements | P201, P261, P264, P280, P301+P310, P303+P361+P353, P305+P351+P338, P308+P313, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2 |
| Flash point | 80°C |
| Autoignition temperature | 430 °C |
| Explosive limits | Upper: 6.8%, Lower: 1.1% |
| Lethal dose or concentration | LD50 oral rat 640 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 640 mg/kg |
| NIOSH | NIOSH: QO8225000 |
| PEL (Permissible) | PEL: 50 ppm |
| REL (Recommended) | 50 ppm |
| IDLH (Immediate danger) | 200 ppm |
| Related compounds | |
| Related compounds |
Phenylethanolamine Phenylethylene glycol 2-Phenylethanol Styrene Phenylacetaldehyde |

