Strontium Perchlorate: Looking Beyond The Formula
From Curiosity to Chemical Mainstay: A Brief Look Back
Strontium perchlorate, with its roots deep in the development of pyrotechnics and colorimetric chemistry, deserves a closer examination. It first grabbed attention during the heyday of elemental discovery, when researchers searched for strong oxidizers and vivid flame colorants. In my early chemistry days, I remember leafing through dusty literature noting how strontium’s deep red emission found a natural partner in perchlorate’s willingness to donate oxygen. That combination sparked interest in researchers hunting safer, more stable alternatives to chlorates or nitrates which, at times, offered unpredictability in energetic mixtures. Labs across Europe, spurred by the twin needs of analytical accuracy and better fireworks, pressed forward with studies. Before long, the compound transitioned from lab curiosity to a reliable player in fields reaching from analytical chemistry to military signaling.
The Basic Rundown: What Strontium Perchlorate Really Offers
Many chemical salts make promises on paper, but strontium perchlorate works in a very straightforward way. As a white, crystalline solid, it stands out for its extreme solubility in water, a property that opens up practical uses ranging from solution-based analytical methods to industrial oxidizers. Unlike strontium chloride or nitrate, its stability under normal storage appeals to manufacturers concerned about shelf life and ease of handling. In the workplace, the material’s high purity levels turn out essential for anyone demanding repeatable chemical yields without annoying batch-to-batch variation. I’ve seen chemists who appreciate that, just as much as the flashy uses in colored flares or tracer rounds. They need chemicals that work every time, with little fuss, and strontium perchlorate often delivers, as long as people respect its reactive nature.
Digging Into the Details: Physical and Chemical Properties
Strontium perchlorate brings interesting quirks that might not leap out from its formula. It's highly water-soluble, far more than most strontium salts, meaning that one can prepare concentrated solutions for analytical work or for use in various manufacturing processes. The crystalline solid melts at moderate heat but won’t give off noxious fumes until pushed well past that point, which makes it safer than some of its relatives. Like other perchlorates, it poses oxidative hazards, so tossing it next to combustibles has led to more than a few memorable lab mishaps. I lost count of the stories describing how a stray granule near the wrong solvent resulted in spectacular, if unintended, pyrotechnics. Chemically, it refuses to give up strontium ions unless given a strong competitor – which means it stays stable in dilute acid conditions, offering a predictable performance in many wet-chemistry routines.
The Labels Tell a Story: How Industry Measures Up
Dealing with chemicals means labels grab a lot of attention. With strontium perchlorate, technical language about percent purity, hydration state, and trace metal content crops up on commercial bottles. In grad school, I spent too many hours sweating over contamination from sodium or potassium. Minute differences mattered, and single-digit ppm could tip off the results in flame tests. The chemical industry focuses on specifications not just for lab use, but also for pyrotechnics and battery applications, where unexpected contaminants bring consequences. Even outside highly regulated environments, the trend leans hard toward transparent labeling. Anything less means you can’t trust what’s coming out of the bottle – a lesson reinforced by mishaps from quick-and-dirty suppliers that failed to deliver the goods.
The Hard Work: Preparation and Where It Takes a Turn
Getting strontium perchlorate from raw materials involves chemistry that calls for both patience and respect for safety. Strontium carbonate or strontium hydroxide is usually treated with perchloric acid, a substance that itself deserves caution. In my own attempts at small-scale synthesis, the process took several days: slow addition, careful temperature control to avoid runaway exotherms, and ongoing filtration to separate out impurities. Multiple recrystallizations can purify the product, but that also means more opportunities for labs to cut corners, or, worse, to slip up with an oxidizer that won’t forgive a careless mistake. Each batch needs close scrutiny for hydrate content—excess water can change the crystal form and cause problems later down the line in finished products or analytical runs. With production scaled up, engineering controls and ventilation systems guard against vapor buildup and dust formation. This is not the sort of material one handles without the right training and a respect for chemical unpredictability.
More Than a Formula: Chemical Reactions and Creative Modifications
You can't talk about strontium perchlorate without acknowledging its role as a reactive oxidizer. In the right hands, it's a tool for clean, high-heat reactions, often found in specialty labs or energetic material research. Through double displacement, it easily trades partners with halides, phosphates, or sulfates, producing new salts. Many researchers look for new derivatives with dopants or complex agents to modify flame colors or tweak reactivity. One of the more impressive experiments I’ve seen involved using it as a source of strontium ions in the synthesis of luminescent materials, changing photophysical properties for high-efficiency displays. Pyrotechnic chemists quickly realized that small atom tweaks could open up new application paths, though not every attempted modification proved stable enough for real-world use.
Different Names, Same Challenges: Alternative Names and Aliases
Chemistry routines love to trip people up with naming conventions. In my own work, strontium perchlorate lurked under a handful of not-quite-synonymous titles: strontium(II) perchlorate, Sr(ClO4)2, or, less often, by its hydrate forms. In literature searches, missing a synonym turned up frustrating dead ends in old patents, even though the properties stayed identical. Younger researchers often overlook these quirks, only to realize that what seems like a novel preparation or discovery had surfaced generations earlier under an alternate label.
Keeping It Safe: Handling Practices That Actually Work
The safety side of any strong oxidizer deserves more attention than it usually gets. Strontium perchlorate won't start fires on its own, but mix it with the usual organic suspects and risks follow close behind. In my early days teaching undergraduate labs, I walked students through the ways dust and static electricity can trigger unexpected reactions. Dry conditions, antistatic mats, and careful segregation from acids, flammable solvents, and reducing agents turned into the rule, not just nagging advice. Storage in tightly closed containers away from incompatible substances ranks high. For larger-scale operations, advanced dust collection and localized fume exhausts show up in well-run facilities. There’s still too little real-world research on chronic exposure risks, but the perchlorate ion’s reputation for disrupting thyroid function means anyone handling solutions, even dilute ones, needs to respect protective recommendations.
Where It Matters: Industries and Everyday Uses
Outside the lab, strontium perchlorate commands more respect. Most people encounter it through fireworks, where its reliable red flame beats almost any other coloring agent. It’s used in tracer rounds and signal flares, where performance can literally spell the difference between a successful rescue or a missed target. Analytical chemistry has relied on its high solubility and predictable behavior for certain precipitation and separation protocols. Even energy research has started exploring it for specialized batteries or propellants, although broader commercial uptake lags behind more established lithium or sodium technologies. Each application brings its own technical challenges, from purity requirements to shelf-life concerns. Any break from strict handling protocols can spark expensive or even catastrophic outcomes.
Pushing the Envelope: New Studies And Technical Hurdles
Research into strontium perchlorate hasn’t slowed down, especially when scientists look for greener, safer energetic materials. Academic and corporate labs keep probing for ways to dial back the shortcomings of perchlorates—chief among them, environmental persistence and bioaccumulation in water systems. Recent reports detail efforts to reclaim and recycle perchlorate wastes, which could reduce regulatory barriers that hamper its industrial use. I’ve read about work aiming for stabilized microencapsulated forms to reduce dust risks, as well as hybrid compounds to give more control over reaction rates for pyrotechnics. Analytical chemists want even higher purities, especially as trace contaminants can mess with sensitive detections, especially in environmental samples where strontium perchlorate sometimes functions as a reference or spike.
A Closer Look at Health: What We Know About Toxicity
Toxicity research shows mixed pictures. Acute exposure to strontium compounds can irritate skin and eyes, and inhalation of dust increases risks of respiratory distress. The real enemy comes from the perchlorate ion. Persistent studies point to how it competes with iodide in the thyroid, possibly leading to hormone disruptions and metabolic issues over long-term, low-level exposures. Occupational limits have grown stricter over the years for this reason. Most of my colleagues insist on good personal protective gear—nitrile gloves, safety glasses, and dedicated lab coats—whenever handling the dry solid or concentrated solutions. Environmental scientists keep uncovering trace amounts of perchlorate in ground and drinking water, reinforcing the need for tighter disposal regulations. While catastrophic accidents remain rare in regulated workplaces, long-term, environmental risks remain under close scrutiny.
Looking Ahead: Unpacking the Future of Strontium Perchlorate
Innovation rarely stands still. Strontium perchlorate holds future promise, especially if environmental challenges can be brought to heel. If better recovery and remediation techniques materialize, it could move beyond its current niche roles. Battery researchers, for example, want to leverage strontium’s ionic traits for new, high-performance devices. Pyrotechnicians continue chasing after ever-purer, more controllable forms for theatrical displays and military applications. With each advance, though, comes heightened responsibility. Regulation will keep growing alongside technical advances, particularly as concerns around perchlorate contamination refuse to fade away. Like so many powerful tools, the challenge shifts to using it wisely, pushing for balance between technical advantage and public health responsibility.
What is Strontium Perchlorate used for?
Fireworks and the Color Red
Anyone who has ever watched a summer fireworks show probably owes strontium perchlorate a nod, even if they’ve never heard its name before. This chemical finds its way into pyrotechnics because of its knack for coloring flames a bright, bold red. Years ago, I volunteered at a small-town Independence Day event and got a peek into the storage shed where the fireworks techs kept their compounds. There were labels I recognized—like potassium nitrate—and more exotic ones, including strontium perchlorate. Its job? Acting as an oxidizer and pumping up the show’s scarlet bursts. The vibrant red matters for flag-waving displays and competitions alike, since strontium’s hue stands out in skies full of noise, smoke, and light.
Lab Use and Water Science
In university chemistry, labs often reach for strontium perchlorate to run ion exchange tests, precipitation experiments, and occasionally for research into rare earth metal separation. The substance dissolves easily in water, leaving a clean solution where strontium ions can play their part. Some groundwater researchers even use it as a tracer. I remember one project that used diluted strontium perchlorate to check how fast water seeped through different soils—a kind of dye test but with chemistry instead of color.
Oxidizer in Industry
Manufacturers look to strontium perchlorate as a strong oxidizer for different reactions, especially when oxygen needs to be released steadily. Many oxidizers fall under safety regulations for explosive risk, and this one is no exception. Its power in giving up oxygen makes life easier for chemical engineers who want something dependable but also risky. The perchlorate group itself has a history in rocket fuel, match heads, and even safety flares. Knowing where these chemicals end up helps keep both students and factory floor workers alert to storage and handling hazards. In my own chemistry teaching days, the label’s warning kept us double-checking storage cabinets, far away from anything flammable.
Environmental Impact and Safety Questions
Urban rivers and wells sometimes show signals of perchlorate contamination, sparking plenty of regulatory and health debates. The issue? Perchlorates can disrupt thyroid function, especially in children and pregnant women. The Environmental Protection Agency keeps a close eye on perchlorate spills, which can leak from industrial operations or improper disposal. If you look beyond the chemical’s uses, stories of water contamination and emergency cleanups show the stakes. Town halls and community meetings often feature residents who just want to know the numbers and see the action plans. A little knowledge, some political will, and routine testing make a real difference. Strontium perchlorate gets wrapped up in these debates by virtue of sharing the same anion, even if it’s not the main culprit in every spill.
Finding a Safer Path Forward
For scientists searching for safer alternatives, green chemistry points to materials that break down faster and cause less trouble for the thyroid or environment. Replacing perchlorates in some cases might mean swapping a compound or tweaking the process to need less oxidizer in the first place. On the education side, science teachers spend time with students learning what red means on a chemical label—a useful habit. People in manufacturing and research benefit from exact handling, proper disposal, and regular health screenings. A big-picture fix always includes listening to workers—those who use, store, or clean up after these chemicals know firsthand which steps matter most.
Is Strontium Perchlorate hazardous or toxic?
What Makes Strontium Perchlorate Draw Concern?
Many folks outside of labs haven’t heard much about strontium perchlorate. But anyone working in chemistry or around pyrotechnics can recognize its name. This compound doesn’t just sit quietly on a shelf. Strontium perchlorate comes packed with oxidizing power. In pyrotechnics, that property helps produce the beautiful red color in fireworks. Chemically, it reacts fast and can easily trigger fires if handled without respect. Pair it with flammable materials and sparks can fly—sometimes literally.
Health Risks from Handling or Exposure
No one should take the dust or fumes from strontium perchlorate lightly. Inhaling the stuff, or getting it on your skin, can irritate the nose, throat, lungs, or skin. If you’ve ever breathed in dust from an oxidizer, you’ll know the sharp, metallic tang that hangs in your mouth and catches in your chest. In labs, gloves and goggles don’t just collect dust. They grow into habits, because compounds like this kick back hard if you treat them sloppily.
The perchlorate part of the compound can trip up the human thyroid, leading to issues with hormone production. Animal studies and research in people who work near perchlorates show that long-term exposure throws off iodine uptake, which can mean thyroid problems down the line. That’s more than just a number on a report. Fatigue, weight gain, and trouble concentrating all come with thyroid imbalance. For kids or those already struggling with thyroid issues, risk ramps up.
Environmental Hazards
Anyone dumping chemicals like strontium perchlorate into the water table is courting trouble. Perchlorates don’t tend to break down quickly in soil or water. A contaminated site can leech perchlorates for years, causing ripple effects in plants and wildlife. In southern California, for example, groundwater tainted with perchlorate has led to millions in cleanup bills after the chemical entered sources for drinking water.
Plants and animals face their own problems. Fish exposed to perchlorate experience thyroid disorders, impacting growth and reproductive health. Communities depending on well water near industrial sites have had to bring in bottled water until cleanup can remove the contamination.
Managing Risk and Moving Toward Safer Practices
Safety doesn’t happen by accident. Facilities using strontium perchlorate now rely on proper ventilation, moisture control, and rigorous training. Handling and storage guidelines have grown stricter for good reason. Wrapping containers tightly, securing them away from organic material, and locking everything down limits those disastrous chain reactions chemists fear. It may seem like overkill, but nobody questions the value of extra steps after an incident.
Management can’t rest on rules alone. Regular air and water monitoring, health checks for workers, and emergency plans that staff actually practice make all the difference. In my own time working in industrial labs, spill drills and surprise audits annoyed everyone until we caught a small leak—which could have turned much worse if ignored.
Research, Awareness, and Substitutes
Science offers hope for breaking the cycle of hazardous exposure. Green chemistry efforts focus on finding alternatives that don’t upend the thyroid or pose persistent danger to water supplies. Some pyrotechnics manufacturers have swapped out traditional oxidizers for less dangerous formulas, steering clear of perchlorates. The movement grows at the same pace as public knowledge. Awareness among workers, supervisors, and even local communities shrinks risks far more effectively than paperwork.
Bottom Line
Strontium perchlorate demands attention and caution at every stage, from storage to disposal. The risks touch both people and their environment, especially where safety checks get treated like paperwork instead of protection. Keeping this compound in check requires more than good intentions—it takes real knowledge, clear procedures, and a willingness to put health and safety first.
How should Strontium Perchlorate be stored?
Understanding the Material
Strontium perchlorate isn’t what most people keep around the house. You’ll find it mainly in labs, fireworks, and sometimes in pyrotechnic displays where that sharp red flame lights up the sky. It brings together strontium and perchlorate, both elements that don’t play nice with water or anything flammable. Handling it safely isn’t just a matter of reading labels; it’s about knowing what could go wrong and preparing for that. My time as a lab tech hammered home that no amount of labeling replaces real respect for reactive chemicals.
Challenges with Storing Strontium Perchlorate
Moisture loves to find its way into just about everything, and strontium perchlorate takes up water from the air like a sponge. I’ve seen containers clump up into sticky, useless cake just because of a cracked lid. This isn’t just inconvenient; the chemical also holds on to that water, which can mess up measurements and, worse, increase the risk of an unwanted reaction. Keeping humidity low means more than comfort—it directly affects safety here. Labs often use desiccators and silica gel; a dry space isn’t optional.
It's not only water causing trouble. Any time perchlorates get in contact with something that can burn, that mix can become dangerous, sometimes explosive. Strontium perchlorate may start out looking calm, but it will quickly fuel a fire if it escapes its container and meets organic material, reducing agents, or accidental sparks. I’ve witnessed what happens when a nearby chemical leaks—cross-contamination can turn a benign storage shelf into an emergency. Never trust shared containers or pantry-style storage. Each incompatible material needs its own dedicated spot.
Proper Conditions Matter
Experience has taught me to never trust makeshift packaging. Screw-top glass bottles with robust seals, labeled clearly, away from sunlight—these steps seem basic, but skipping one invites disaster. Secure shelving well above ground, never stacked with acids, fuels, or any powdery organic material, keeps risks in check. If a spill does occur, quick cleanup with completely dry tools is crucial. Even small leaks or powder trails can build up over time into a real hazard. Safety goggles and gloves join the routine for a reason. My colleagues and I always made a habit of checking expiration and seal integrity before and after each use.
Supporting Safety with Facts
Research puts perchlorates, including strontium perchlorate, right up there with dangerous oxidizers. Reports from OSHA and the National Fire Protection Association point to dozens of lab fires caused by mixing oxidizers with combustibles by mistake or through accidents. The CDC reminds that even inhaling perchlorate powder isn’t safe, as it can disrupt thyroid function over time. This isn’t just theory; annual incidents keep fire departments and hospital emergency rooms busy. People often think of science as precise and controlled, but safe storage often relies on choosing simple routines—and following them without exception.
Working Toward Better Storage Practices
Some places now invest in climate-controlled cabinets or automated warning systems, but even these don’t replace face-to-face training. Online safety sheets give details, yet nothing replaces hands-on experience, careful labeling, and reliable habits. I found that assigning a single point-person—someone who checks the area each week—cuts down mistakes more than a stack of reminders ever could. Regular safety reviews, peer accountability, and easy-to-understand signage keep everyone honest and sharp. Whether in academia, industry, or a small private lab, these straightforward steps save lives and property. Real diligence, not just compliance, creates real safety.
What is the chemical formula of Strontium Perchlorate?
A Closer Look at Strontium Perchlorate
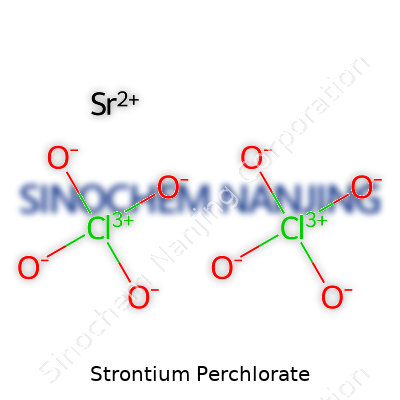
Strontium perchlorate, which goes by the formula Sr(ClO4)2, finds its way into many chemistry labs and industries. Chemistry classes have taught me plenty about how different compounds behave and why their formulas matter. In this one, strontium, a reactive metal, teams up with the powerful perchlorate ion. The combination delivers a salt with some striking features: it's highly soluble in water and delivers more than just textbook interest. Fireworks, pyrotechnics, and research applications all draw on chemicals like this for their unique properties.
Living With Risks: Why the Formula Matters
Getting this formula right isn’t just a math exercise. Chemistry is not forgiving about detail. A number in the wrong place, a misunderstanding about charges, and a chemical accident can follow faster than expected. The strontium ion (Sr2+) pairs with two perchlorate ions (ClO4-) for a perfectly balanced, neutral compound. Pulled from my experience, lab safety jumps to mind. Perchlorates don't just sit on shelves. These ions pack energy and can spark fast oxidation, so any confusion in their composition can lead to unsafe handling.
Strontium Perchlorate in the Real World
Industries rely on consistency. In fireworks, for instance, strontium compounds produce bright red flames. Purity and accuracy in the formula mean audiences get spectacular colors, not unpredictable results. Water solubility means the salt dissolves fast, an advantage in certain chemical syntheses but also a risk for the environment. Water sources exposed to perchlorates face contamination, which several studies, like the ones published in Environmental Science & Technology, have linked to thyroid problems in humans and animals.
Looking for Solutions: Safety, Regulations, and Alternatives
Responsible chemistry calls for keeping compounds like strontium perchlorate under control. Regulatory bodies, including the Environmental Protection Agency in the US, set limits and procedures to keep perchlorates away from drinking water and food supplies. Personal experience reminds me that those rules work only if everyone in the chain knows what they’re handling. Proper labeling, hazard training, and storage—to keep moisture and heat away—reduce the odds of accidents and spills. Some labs and industries explore less reactive oxidizers when possible. Chemistry professors sometimes push for greener alternatives, seeking substances with reduced toxicity for both workers and the environment. The textbook formula Sr(ClO4)2 might look straightforward, but the implications ripple through public safety, industry output, and environmental health.
Checking Your Work: Why Accuracy Protects People
Students and professionals stay sharp by turning to reliable sources—peer-reviewed studies, textbooks, and regulatory databases. That habit of checking the formula pays off. Mislabeling might not sound like much, yet a single error could turn a safe experiment into a near miss. Good science, in my view, springs from knowing the details by heart and never taking shortcuts with the facts.
Can Strontium Perchlorate be shipped internationally?
Handling Hazards and International Rules
Strontium perchlorate stands out as a powerful oxidizer. This makes it useful in science labs and even pyrotechnics, but also brings real shipping challenges. Countries have strict rules because strontium perchlorate reacts fast with organic material and can help a fire get out of control. Think back to basic chemistry class — strong oxidizers speed up combustion. Now imagine that on a cargo plane. That's why hazardous classification matters so much. According to the UN’s Model Regulations on the Transport of Dangerous Goods, strontium perchlorate falls under Division 5.1 (oxidizing substances), which triggers extra precautions.
Many people assume all chemicals travel the same path across customs. They don’t. Customs brokers and freight agents work with different lists from the International Air Transport Association (IATA) and International Maritime Organization (IMO). Strontium perchlorate makes those lists, not just as something to flag, but as something to double-check for packaging, labeling, and paperwork. I’ve helped startups move chemicals for research, and the long forms and inspections weren’t overkill — they protected people and prevented deadly mistakes.
Pitfalls in International Transport
Anyone trying to ship strontium perchlorate overseas finds layers of policy, cost, and liability. Every package must carry safety documentation, like Material Safety Data Sheets (MSDS), and the packaging must pass tests for leaks, impact, and corrosion. Not all countries let someone import a strong oxidizer, either. For example, New Zealand and Australia run chemical import alert programs due to environmental and terrorism fears. That means a shipment stuck at the border translates to lost money, wasted time, and sometimes investigations.
Another critical spot comes with airline and shipping firm decisions. Top couriers like FedEx and DHL keep updated lists of what they carry and what they won’t touch. Chemicals with “perchlorate” in the name often get an automatic rejection, even with all paperwork lined up. I’ve watched colleagues spend weeks searching for a freight company with the proper permits and security protocols.
Responsible Shipping Practices
Chemical safety isn’t just bureaucracy; it’s about protecting people from risks they might never see coming. Misdeclared oxidizers explode in the news every few years. Solid partnerships with certified dangerous goods shippers cut through most of the red tape, as they know how to pack, declare, and notify authorities at each stop. Each country applies its own twist to the international codes, so a shipment that sails through Germany may not clear customs in Brazil. Errors, even honest ones, face big fines.
The Path Forward
Safer alternatives and digital tracking help some. But the best solution comes from investing in staff training — not just the chemistry pros, but also the admin workers booking freight. Regulators want shipper declarations, accurate hazard labels, and emergency response plans in every box or pallet. More collaboration helps too. Industry groups could push for more uniform standards and push back on knee-jerk import bans that don’t truly boost safety.
At the end of the day, moving dangerous chemistry across borders means more than clearing customs. It means accountability for every link in the chain, from manufacturer to end user. The risks aren’t abstract, they’re specific, and good practices couple caution with know-how.

| Names | |
| Preferred IUPAC name | Strontium perchlorate |
| Other names |
Perchloric acid, strontium salt Strontium(II) perchlorate |
| Pronunciation | /ˈstrɒnʃiəm pərˈklɔːreɪt/ |
| Identifiers | |
| CAS Number | 13465-18-6 |
| Beilstein Reference | 358925 |
| ChEBI | CHEBI:84941 |
| ChEMBL | CHEMBL4266271 |
| ChemSpider | 110529 |
| DrugBank | DB11451 |
| ECHA InfoCard | 100.015.659 |
| EC Number | 223-960-9 |
| Gmelin Reference | 2860 |
| KEGG | C18698 |
| MeSH | D013326 |
| PubChem CID | 16211577 |
| RTECS number | TH8225000 |
| UNII | X6K3VN0FQ2 |
| UN number | UN1502 |
| CompTox Dashboard (EPA) | urn:epa.compound:56908 |
| Properties | |
| Chemical formula | Sr(ClO4)2 |
| Molar mass | Sr(ClO₄)₂: 286.53 g/mol |
| Appearance | white crystalline solid |
| Odor | Odorless |
| Density | 3.25 g/cm³ |
| Solubility in water | Soluble |
| log P | -2.01 |
| Vapor pressure | Negligible |
| Magnetic susceptibility (χ) | +11800.0e-6 cm³/mol |
| Refractive index (nD) | 1.433 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -743.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1184.8 kJ/mol |
| Pharmacology | |
| ATC code | V07AA |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS01,GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H271, H272, H302 |
| Precautionary statements | P210, P220, P221, P280, P370+P378, P501 |
| NFPA 704 (fire diamond) | 3-0-2-OX |
| Lethal dose or concentration | LD50 oral (rat) 584 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 460 mg/kg |
| NIOSH | SN12240 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Strontium Perchlorate: Not established |
| REL (Recommended) | 400 mg/L |
| Related compounds | |
| Related compounds |
Calcium perchlorate Barium perchlorate Magnesium perchlorate Strontium chlorate Strontium nitrate Strontium sulfate |

