Strontium Nitrate: An In-Depth Look
Historical Development
On the timeline of chemical discoveries, strontium nitrate stands out thanks to its association with the vivid reds lighting up festival skies. Discovered in the late 18th century, its early years were marked by the broader isolation and research into strontium compounds following the identification of the mineral strontianite in 1790. Chemists realized quickly that strontium nitrate packed a punch in pyrotechnics, outperforming previous red colorants. Over time, the production process learned from the early, messy methods, shifting toward cleaner, higher-yield reactions, guaranteeing more reliability from batch to batch. The industry also started paying real attention to purity, especially as new fields, like medicine, began exploring strontium nitrate’s potential.
Product Overview
Market shelves stock strontium nitrate chiefly as a colorant and oxidizer. Its formula, Sr(NO3)2, looks simple but delivers complexity in effect, from the rocket’s red glare to glass compositions. The product usually appears as a white, crystalline solid, easy to handle and measure. Many labs and manufacturers still prize strontium nitrate for its reliable, predictable performance in fireworks and laboratory work. People in glassmaking and ceramics also value its ability to tweak optical properties without clouding or contaminating colors.
Physical & Chemical Properties
Strontium nitrate sits as a dense, colorless, crystalline substance, with a molecular weight of about 211.63 g/mol. The solid dissolves readily in water, sparking fast chemical reactions in aqueous solutions. It does not take much to set it off thermally: at about 570°C, it breaks down, pumping out nitrogen dioxide, oxygen, and strontium oxide. Its solubility and decomposition behaviors connect directly to its main uses, offering a mix of stability at room temperature and energetic redox properties under heat. Unlike some alkali metal nitrates, strontium nitrate doesn’t absorb as much water from the air, making storage and handling more manageable.
Technical Specifications & Labeling
Industrial-grade strontium nitrate comes labeled with clear technical specs: purity level (often 99% or higher for pyrotechnics), heavy metal content, moisture level, and bulk density. Packaging gets tightly regulated to avoid contamination and accidental exposure, with containers flagged as oxidizers under GHS and DOT standards. The labeling outlines batch numbers, production dates, and safety data, lining up with regulations aimed at health and environmental safety. Employees must pay attention to the transport codes due to its oxidizing potential, which makes it unwelcome in standard shipping with flammable or organic materials.
Preparation Method
Manufacturers produce strontium nitrate through reaction of strontium carbonate with nitric acid, yielding strontium nitrate and carbon dioxide gas. This pathway gives high yields and starts with commonly available raw materials. In labs, the reaction is straightforward: dissolve strontium carbonate in diluted nitric acid, filter, concentrate, and crystallize to recover pure strontium nitrate. This process eliminates many impurities that otherwise dull the red color or introduce safety risks, making it the dominant commercial method since the industrial age. Quality checks follow crystallization to screen for contaminants like barium, lead, or sulfate, as even trace levels dent the product’s performance.
Chemical Reactions & Modifications
Once in hand, strontium nitrate acts as both an oxidizer and a steady source of strontium ions. Mix it with dextrose or elemental metals and it becomes the fire in flames, oxidizing the fuel and turning combustion into a performance. In laboratory synthesis, strontium nitrate transforms into other strontium salts through double displacement reactions, or drops out as a precipitate in the presence of sulfate, making it useful for qualitative ion tests. Chemists sometimes tweak its properties by adding stabilizers to boost shelf life or safety, but the basic reactivity—powerful oxygen release and vivid luminescence—remains the feature, not a bug.
Synonyms & Product Names
Across datasheets or supplier lists, strontium nitrate appears under names like nitric acid, strontium salt; Metstron; UN 1507; EINECS 233-131-9. Any label with those codes or the classic Sr(NO3)2 formula points to the same core product, though brand differences can hint at small purity tweaks or handling features for industrial chains versus school chemistry labs.
Safety & Operational Standards
Working with strontium nitrate means taking seriously its oxidizing power. It turns common combustibles like paper or sawdust into real hazards, so proper storage—far from fuels, in dry, cool conditions—matters. Exposure guidelines set threshold limits to avoid skin or eye irritation and stricter controls in workplaces with lots of dust. Personal experiences remind me that people sometimes skip protective equipment because it looks benign, but after witnessing fast accelerations of small fires, I always use gloves and goggles. Emergency plans emphasize neutralizing spills with lots of water, not absorbent organic material, to prevent unintended reactions. Regulators keep close tabs on its use, especially in fireworks plants and chemical warehouses, demanding regular audits and safety drills.
Application Area
Pyrotechnics remains the core user of strontium nitrate, for the simple reason that nothing else matches its deep, clear reds in flame tests. Beyond fireworks, its presence turns up in emergency flares, tracer ammunition, and signal lights. Glassmakers add it to lenses or color systems, trading off purity and refractive tweaks. Catalyst developers look into strontium nitrate for cleaner industrial oxidations, and some battery designers explore its use in experimental cathode blends. In medicine, researchers try out strontium salts in bone repair and imaging, with nitrate acting as the precursor for more exotic compounds. Every industry that needs clean, compact oxygen or a red indicator owes something to this salt.
Research & Development
Recent years bring a shift in focus: seeking alternatives to toxic heavy metals in colorants, strontium nitrate gets new attention. Research labs chase two main lines—improving yield and safety in large-scale production, and leveraging strontium’s properties for scientific instrumentation. One ongoing project I saw explored using strontium nitrate as a seed crystal in novel ceramics, aiming for better temperature resistance in electronics. Environmental chemistry groups test it for denitrification roles, while material scientists try new composite blends with other alkali metals to enhance properties. Each advance needs real attention to the supply chain, purity, and implications for unintended environmental release.
Toxicity Research
Toxicologists dig into strontium nitrate’s impact across routes: skin, inhalation, and ingestion. The salt travels through the body much like calcium but does not build up quickly; short intense exposures cause more harm than long, low-level contact. Acute doses trigger nausea, vomiting, and, at high concentrations, muscle cramps or heart rhythm impacts—mirroring effects seen in strontium chloride. Chronic studies suggest it slips out of the body through urine, so acute exposures get more concern than long-term buildup. Even so, handling rules push for minimized contact and testing in workplaces to head off accidental dust inhalation, especially in children or pregnant workers. Agricultural researchers keep an eye on the potential for water contamination, particularly in areas near pyrotechnic or ceramic facilities.
Future Prospects
As nations crack down on toxic emissions and seek greener flame colorants, strontium nitrate stays in demand by virtue of its low toxicity compared to barium and lead-based salts. The challenge lies in reducing its own environmental footprint, as nitrate leaching from fireworks and flares can impact water sources. Some forward-looking chemists aim for recyclable strontium compounds or biodegradable oxidizer blends. In energy tech, innovation teams are sizing up its electrochemical properties for new cell chemistries, while medical technology could unlock new imaging agents or bone treatments starting from strontium nitrate. With digital process monitoring, producers scale up efficiently, while tighter environmental rules keep waste streams cleaner. If anything, research is set to shape how strontium nitrate gets used—balancing tradition with next-gen needs in safety, sustainability, and creative science.
What is Strontium Nitrate used for?
The Fireworks Factor
Strontium nitrate does its most eye-catching work on summer nights. Ask anyone standing under a big Fourth of July show — those deep crimson fireworks almost always get their color from strontium compounds. Strontium nitrate burns with a bright red flame, perfect for putting intense color into pyrotechnics. Through college, I worked one July loading shell after shell, helping wire fuses, and got a front-row view of how strontium nitrate anchors nearly every “red” effect. Shows rely on it because other reds just blur into orange or pink. People want that clear, classic red ball bursting above the park.
Everyday Chemistry in the Classroom
High school and college chemistry labs crack open strontium nitrate for flame tests. Teachers like the reliable crimson streak it gives when the powder hits a burner’s flame — it’s much easier to identify than most other compounds. I lost track of how many times classmates guessed “strontium?” because the color stood out so boldly. Using obvious pigments keeps students alert, and the simple science sticks better than drab, forgettable demonstrations. Strontium nitrate still lands on lists of best materials for basic flame labs thanks to that strong reaction, which helps sharpen both lesson and memory.
Pyrotechnics and Safety
Mixing chemicals for big events means staying sharp about hazards. Strontium nitrate supports not only color, but also the oxidizing punch fireworks need to burst and spread fully. The danger here isn’t only with the bang. The compound by itself isn’t toxic in small amounts, but as with most oxidizers, keep it dry and away from fuels until you’re ready to use it — this isn’t something to treat lightly or leave unattended. In the wrong hands or stored carelessly, it raises the risk for fires or accidental reactions.
Medical Applications and Surprising Uses
Doctors look at strontium nitrate for how it numbs sensitivity in toothpaste. Certain toothpaste brands add small amounts, aiming to calm nerves in teeth that react badly to cold or sweet foods. There’s also interest in exploring strontium compounds for bone health. Strontium nitrate doesn’t go into supplements, but related chemicals sometimes show up in research around bone repair. No home remedies here — any potential use goes through careful clinical review first. This keeps the ingredient where it belongs: either strictly controlled in the factory, or measured properly in clinical labs.
Environmental and Health Notes
The topic of environmental impact always comes up around fireworks. Air quality takes a hit after big displays, with strontium compounds settling on nearby land and water. Studies show that most city soils can handle a few summer events, but areas downwind of frequent shows see a slight uptick in metal content. Regulatory agencies track this and set limits, and pyrotechnic companies work to use just enough strontium nitrate to get the color without going overboard. Health agencies say average hobbyists probably won’t get exposed to dangerous levels just as spectators, though anyone working with the raw powder should always wear gloves and a mask.
Smarter, Safer Use Moving Forward
Pyrotechnics enthusiasts and teachers already go through safety protocols, but broader awareness doesn’t hurt. Keeping strontium nitrate away from kids and locking up supplies can prevent accidents in homes and school labs. Fireworks insiders recognize the need for greener displays, which puts added pressure on manufacturers to find alternatives or use compounds with the lowest environmental impact. Research keeps inching forward, testing how to hold onto that signature red glow while taking the edge off of lingering fallout. Better labeling and smarter packaging make mistakes less likely, reducing risk for everyone from summer workers to curious teenagers in science class.
Is Strontium Nitrate hazardous or toxic?
What Strontium Nitrate Does in The World
Strontium nitrate gives fireworks that deep, bold red, and brings color to emergency flares. In the lab, I’ve seen it pop up in pyrotechnics and a couple of chemical tests. Its role seems small, but the bright flashes it creates come with real safety questions. I’ve watched technicians handle it like flour on a kitchen table—bare hands, quick pours—forgetting it’s more than just powder.
The Risks in Everyday Use
Breathing in strontium nitrate dust leaves the nose scratchy and the throat sore. The chemical doesn’t just stop at an annoying cough. Doctors point to real signs in workers exposed to nitrates—skin redness, itchy eyes, and, over time, trouble breathing. Young kids could mistake its white crystals for sugar, which turns a small spill into a big deal. I’ve read incident reports involving backyard fireworks makers rushed to clinics with burns or unusual heartbeats after handling sparkler ingredients bare-handed.
Fire and Health Hazards
Light a fire near strontium nitrate, and things can escalate fast. It decomposes, feeding oxygen to flames and boosting fire risk, especially in dry spaces where old paper or sawdust hide. Firefighters I’ve talked with say nitrate fires burn hotter and resist water—so fire safety plans need to be serious in places where storage happens.
Ingesting even small amounts can shock the digestive system. The nitrate part drives up blood’s ability to carry less oxygen, making some people lightheaded or short of breath. It shares this risk with other chemicals in the nitrate family, which have earned a reputation in medical journals for long-term effects on the liver and kidneys if exposure stacks up over years.
Environmental Impact
Spills move fast through soil and water. I’ve heard stories from lake-side towns worried about nitrate runoff. Algae blooms feed on leftover chemicals, leading to fish kills and thick, green water. This isn’t just theory—monitoring from state agencies keeps turning up nitrate spikes near fireworks show sites or old storage yards. Plants and insects get wiped out, breaking links in the local food chain.
What Makes Handling Safer
Most injuries come from ignoring common sense—no gloves, crowded storage, loose lids, and working without proper ventilation. Wearing gloves, keeping containers sealed, and using dust masks became routine in labs where people cared about safety. School chemistry sets now carry warnings for good reason.
Toward Responsible Use
Safer storage needs straightforward changes: lock chemicals up, separate them from fuels and paper, and make sure no random sparks or open flames get too close. Training matters. I’ve walked into facilities where folks have laminated checklists and yearly refreshers; accident rates drop.
Using substitutes gives a way out. Some companies have switched to colorants and oxidizers that burn less hot and don’t carry the same health concerns, especially for large public events. These options cut down on the environmental and health risks, even if the color looks a little different.
Weighing Benefit Against Risk
Strontium nitrate won’t vanish overnight, but putting health and safety higher on the list changes how we make and use it. Awareness makes each step—from school labs to large fireworks—less about excitement and more about keeping people and the environment out of harm’s way. People have to respect the power of these compounds to keep their families and neighbors safe.
How should Strontium Nitrate be stored?
Why Taking Storage Seriously Matters
Strontium nitrate looks innocent at first glance—white, powdery, easy to mistake for basic chemicals used in classrooms. But it brings more risk than most realize. Some people know it by its brilliant red flames in fireworks. Underneath the spectacle, it carries real hazards for fire, health, and the environment. I’ve handled this chemical in small batches for pyrotechnics, and the lessons stick with me: storing it the right way keeps everyone safer.
Recognizing the Hazards
Strontium nitrate belongs on a shelf with oxidizers. Left near fuels, organic material, or anything even a little bit oily, it speeds up burning and can trigger fires most folks struggle to control. It also carries health risks if inhaled or ingested, irritating the respiratory system and causing long-term health issues with repeated exposure. Once I helped clean up a spill where improper packaging led to a minor reaction—eye-watering smoke and a sharp smell nobody wants at their workplace. Forgetting proper storage brings these incidents far too close for comfort.
Building a Safe Storage Plan
So, how do you store strontium nitrate safely? I’ve learned that the basics done right beat complicated fixes every time. This chemical calls for containers that protect against moisture—sealed good and tight with sturdy lids. Skip anything flimsy or easily broken. Keep it in a dry spot, far away from sinks or areas likely to get wet. Even a bit of humidity can cause clumping, ruin the chemical, and possibly set off unwanted reactions.
Strontium nitrate should not mix with combustibles, acids, sulfur, or anything reactive. In the pyrotechnics shop, our supply sits on shelves far removed from wood shavings, paper, and solvents of any kind. I prefer steel or thick plastic bins with labels in bold letters so there’s no guessing or mix-ups, especially with similar-looking compounds.
Choosing the Right Environment
Temperatures matter too. Store strontium nitrate in cool, stable conditions—not next to a heater or in spots that bake in the afternoon sun. Direct sunlight and heat speed decomposition or reactions with other materials. If the building has temperature fluctuations, I’d pick the steadiest corner, away from windows and ventilation systems blowing hot air.
I’ve seen some try to keep large amounts in standard storerooms, but anyone working with quantities beyond a kilogram should use a dedicated storage cabinet rated for oxidizers. Many states or countries also require this by law, and with good reason. It limits the risk if fire breaks out somewhere else in the building.
Training and Emergency Preparedness
Even the best container can’t fix careless handling. Every person with access should know the risks and proper routines: never return unused powder to the storage drum, always clean up spills right away, and never eat or drink in the same space. Trust in checklists and clear labeling rather than memory.
Spill kits, fire extinguishers (not just any kind—preferably those for chemical fires), and proper ventilation help turn accidents from disasters into minor setbacks. In my experience, running emergency drills makes these steps feel automatic, reducing panic when something actually happens.
Staying Grounded in Experience
A safe storage plan for strontium nitrate doesn’t rely on theory. It draws from lab incidents, fire marshal reports, and the hard lessons of those who work with chemicals every week. Stick with sealed containers, dry conditions, and separation from any flammable or reactive substance. Combine that with smart labeling, regular training, and a little common sense, and you won’t just avoid fines—you’ll give everyone peace of mind. Proper storage doesn’t add cost; it saves you from problems that are too expensive to fix after the fact.
What is the chemical formula of Strontium Nitrate?
Getting to Know Strontium Nitrate
People sometimes breeze right past the details in chemistry, but a single line formula often determines everything about a material. Strontium nitrate, for instance, comes with the chemical formula Sr(NO3)2. This string of letters and numbers provides real insight into what the compound can do. If you’ve ever been at a fireworks display and seen stunning red bursts against the night sky, you can thank strontium nitrate. It's the go-to source manufacturers rely on for that bold color. That real-life impact is the sort of thing students remember long after chemistry class.
Why This Formula Matters
Getting hands-on with chemistry reveals just how much power a simple formula packs. Knowing the formula Sr(NO3)2 tells you the compound holds one strontium atom paired with two nitrate groups. Strontium itself is an earth element, pretty common in minerals dug up worldwide, while the nitrate part ties it to the family of strong oxidizers. In practice, this means strontium nitrate helps drive reactions where a spark needs fuel—think pyrotechnics, or even certain kinds of flares used at sea to call for help.
I’ve spoken with folks who’ve worked in pyrotechnic industries, and they don’t just think of strontium nitrate as a textbook entry. They lean on it for consistency in color and reliability in burn. They buy it in bulk, weigh it out against other components, and use that chemical formula as gospel to balance the mix. Without that repeatability, shows would be less predictable and maybe less safe.
The Risks Lurking Beyond the Formula
It’s smart not to lose sight of the risks. Strontium nitrate holds a place in the group of chemicals that can accelerate fires. Accidents in small fireworks shops or student labs remind us that careful handling is non-negotiable. Strong oxidizers, especially when combined with organic matter or metals, can turn a tiny spark into a much bigger mess. Some years back, news broke about a warehouse fire linked to improperly stored oxidizing agents. Incidents like that fuel stricter guidelines and workplace safety routines in the chemical trades.
It’s not only about fire. Mishandling can lead to environmental harm. Nitrates can drift into groundwater and kick off algae blooms that sap oxygen from lakes. The formula Sr(NO3)2 shows there are two nitrate pieces per molecule, so carelessness can double the trouble if spills go unchecked.
Steps Toward Safer Use
A practical approach helps control the risk. Regular training for those who handle strontium nitrate goes a long way. Labeling and clear storage rules limit confusion, and spill kits in the right spots let professionals act fast if things go sideways. Peer-reviewed studies back up the need for strong safety cultures wherever chemicals mix with fireworks or flares. Regulators keep pushing for updates to standards, especially for high-volume users and those near water sources.
Schools have an extra job: turning chemical curiosity toward responsibility. Teachers can show future scientists that formulas like Sr(NO3)2 aren’t just answers on a quiz—they’re the backbone of real projects and the reason for safety conversations. From lesson planning, I’ve seen engagement skyrocket when students understand how the right information leads to real-world impact.
Looking Ahead
Chemists, teachers, and safety pros all agree: respecting the formula means understanding its potential, both for creativity and for caution. In a way, every chemical formula tells a story. Strontium nitrate writes its own—starring fireworks, science labs, and safety checklists in equal measure.
What are the safety precautions for handling Strontium Nitrate?
Understanding the Risks
Strontium nitrate isn’t something most folks keep in their garages, but for those who work with fireworks, signal flares, or research labs, it’s pretty common. It gives fireworks that deep red glow and shows up in some science classrooms. Despite its usefulness, this chemical packs some risks. I remember my first lab job—there’s always a seasoned technician who warns you early about keeping your hands clean, goggles on, and chemicals contained. That lesson sticks for a reason. Strontium nitrate can irritate the skin, eyes, and especially the lungs, leaving your body worse for wear if you don’t respect it.
Personal Protective Gear Isn’t Optional
It’s tempting to skip the gloves when you’re in a hurry. I’ve seen too many people pay for that mistake with burns, rashes, and time away from work. Strontium nitrate reacts with skin moisture, so gloves are non-negotiable. Proper lab coats or aprons, long sleeves, and—crucially—chemical splash goggles should become routine. This isn’t just for your peace of mind. Protective gear stops small accidents from becoming real emergencies, and it’s hard to recall a single day in any decent lab where anyone skipped that step.
Good Ventilation Saves Health
Whenever someone opened a new jar of strontium nitrate in a stuffy storeroom, the air felt heavy. The dust lingers, and if it drifts up your nose or down your throat, irritation follows. Fume hoods or at least well-placed exhaust fans keep the workspace safe. If you lack proper ventilation, things get dicey fast. Chronic inhalation means trouble for lungs and may lead to headaches or nausea, so air movement is no detail to brush aside. Trust the old hands: clean air counts every time.
Fire Hazards Aren’t Hype
This is an oxidizer. I learned early on that mixing strontium nitrate with sawdust, sugar, or even some metals isn’t just theoretical trouble—it can blow up in your face. No hot plates right next to your chemical containers, no open flames around the workspace. Always keep this compound separated from combustible stuff. I’ve watched a small spark cause a sudden flare more than once, which really presses the point home: treat it like it wants to light up, because it does.
Practice Good Storage
Don’t keep strontium nitrate on a shelf next to anything flammable. Use tightly closed, labeled containers, stashed in a cool, dry cabinet that stays locked. In every facility I’ve worked, someone’s been assigned to check that chemicals are stored right. It might seem boring, but a little diligence here keeps property and people safe. Moisture and heat make it break down faster, so controlling those two factors makes all the difference.
Emergency Preparedness
No one expects spills or splashes, but they always come sooner or later. Eyewash stations and safety showers can’t just be afterthoughts—they need to be easy to reach. Immediate rinsing after exposure minimizes harm, and coworkers should know where to go and what to do. If a fire starts, reach for the right type of extinguisher, not just the nearest one. Practice helps everyone stay sharp, just like regular fire drills at school or work.
Responsible Disposal
Tossing leftover chemicals down the drain doesn’t just break rules, it can harm water sources and local wildlife. At every workplace I’ve known, proper disposal meant following protocols and paperwork, sending hazardous waste for approved treatment. It takes more effort, but keeping the community safe beats cutting corners.
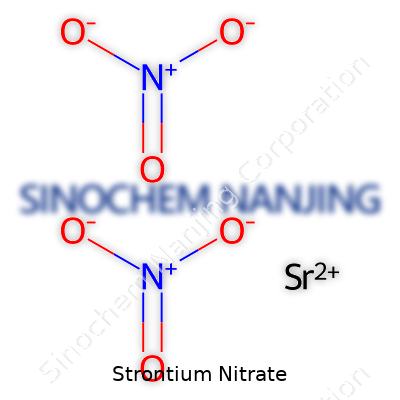
| Names | |
| Preferred IUPAC name | Strontium dinitrate |
| Other names |
Strontium dinitrate Nitrate de strontium Strontium(II) nitrate |
| Pronunciation | /ˈstrɒn.ti.əm ˈnaɪ.treɪt/ |
| Identifiers | |
| CAS Number | 10042-76-9 |
| Beilstein Reference | 1908730 |
| ChEBI | CHEBI:76284 |
| ChEMBL | CHEMBL1201567 |
| ChemSpider | 12181 |
| DrugBank | DB11198 |
| ECHA InfoCard | 100.102.379 |
| EC Number | 233-131-9 |
| Gmelin Reference | 86137 |
| KEGG | C18641 |
| MeSH | D013329 |
| PubChem CID | 24592 |
| RTECS number | WK7650000 |
| UNII | RFQ8151U48 |
| UN number | 1507 |
| CompTox Dashboard (EPA) | DTXSID0021625 |
| Properties | |
| Chemical formula | Sr(NO3)2 |
| Molar mass | 211.63 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 2.99 g/cm³ |
| Solubility in water | Moderately soluble |
| log P | -4.62 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 6.16 |
| Basicity (pKb) | 6.20 |
| Magnetic susceptibility (χ) | -48.0e-6 cm³/mol |
| Refractive index (nD) | 1.564 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 116.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -945.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -542 kJ/mol |
| Pharmacology | |
| ATC code | V07AA04 |
| Hazards | |
| Main hazards | Oxidizing solid, harmful if swallowed, causes eye and skin irritation, may intensify fire. |
| GHS labelling | **"GHS02, GHS07, GHS09"** |
| Pictograms | GHS01,GHS03,GHS07 |
| Signal word | Danger |
| Hazard statements | Hazard statements: H272, H315, H319, H335 |
| Precautionary statements | P210, P220, P221, P264, P280, P370+P378, P305+P351+P338, P337+P313, P501 |
| NFPA 704 (fire diamond) | 2-0-3-OX |
| Autoignition temperature | Does not ignite easily |
| Lethal dose or concentration | LD50 oral rat 2750 mg/kg |
| LD50 (median dose) | 1892 mg/kg (rat, oral) |
| NIOSH | SN1225000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Strontium Nitrate: "10 mg/m3 (total dust) (OSHA) |
| REL (Recommended) | 30 mg/m³ |
| IDLH (Immediate danger) | IDLH: 1300 mg/m3 |
| Related compounds | |
| Related compounds |
Strontium nitrite Strontium chlorate Barium nitrate Strontium carbonate |

