Strontium Chlorate: A Commentary on a Storied Compound
Tracing the Past: The Historical Arc of Strontium Chlorate
Most chemical compounds have histories that trace the evolution of science, and strontium chlorate sits among the more fascinating examples. Chemists in Europe started tinkering with strontium salts in the 18th and 19th centuries, spurred by discoveries in flame colorations and eager to push pyrotechnics into dramatic territory. Strontium compounds gave fireworks that signature bold red, and chlorates in general brought a powerful oxygen-release punch. Even as folks obsessed over strontium nitrate or carbonate, a few driven scientists noticed that strontium chlorate’s oxidizing properties could play nicely in pyrotechnic mixes. My own introduction to this compound came in a cramped college storeroom, where a battered wooden box labeled “Explosives – Handle with Caution” hid small, chalky crystals that never made it into undergraduate labs. Reading up later, I learned how strontium chlorate trailed behind potassium chlorate in broader use, not because it lacked punch, but because making it consistently, and safely, wasn’t always straightforward. The world of chemistry grew more careful, and regulations tightened, shifting wider industrial appetites away from highly reactive chlorates. Yet through pyrotechnic traditions, and persistent tinkering, strontium chlorate’s value kept flickering.
Product and Properties: What Makes Strontium Chlorate Stand Out
Every time those fine, white needle-shaped crystals pour out, it’s hard to mistake the look of strontium chlorate. Its solubility in water beats many of its strontium cousins, which speaks volumes for folks who need solutions rather than just dry powders. The compound dissolves clear, hinting at hidden intensity; inside, chlorate ions wait to release oxygen in chemical reactions that need heft and speed. That powerful oxidizing property defines how this material acts. Bring a small flame even close, and strontium chlorate does its work: providing oxygen, firing up color, and supporting rapid combustion. Its melting point might not be remarkable, but that fierce reactivity calls for respect long before temperatures climb. If you’ve handled oxidizers before, you know not to underestimate the risks packed inside such an innocuous package.
Labeling and Technical Know-How
Safe handling and labeling require more than printed warnings or regulatory compliance. In practice, the risk isn’t just about following rules—it’s about deeply understanding what a compound might do outside of a chemistry manual. Strontium chlorate’s toxic, reactive nature must stay top-of-mind for educators, researchers, or anyone storing it near organic materials or fuels. Explosives regulations push strict limits for transport and storage, shaping how chemical suppliers even talk about strontium chlorate on their containers. Labels need to ring alarm bells, yes, but they also must spark real vigilance among those who open those containers—because the decisions you make in that moment, to segregate or shield from moisture, can mean the difference between safe success and a dangerous accident.
How Folks Make It—and How Chemistry Shapes the Outcome
Those who learned laboratory work the traditional way know the step-by-step patience demanded by chemicals like strontium chlorate. Standard preparation involves passing chlorine gas through a cold strontium hydroxide solution—a process that feels both old-school and risky, demanding glassware, fume hoods, and calculated nerve. Each step brings its own small hazards: venting chlorine, preventing splashes, and ensuring only purified water contacts the strontium hydroxide to reduce impurities. Chemists keen to modify reactions tweak temperatures, concentrations, and purity levels to squeeze the best results out of their starting materials. Judging success comes down to yield, purity, and physical appearance—a cloudy residue or off-color batch signals trouble somewhere along the way. I’ve seen hands tremble slightly as someone gently filters and washes these delicate crystals, knowing all too well what might happen if they dry next to anything even faintly combustible.
Chemical Reactions and Variants
Strontium chlorate responds with unmistakable vigor wherever oxidation shifts the chemistry. Watch it ignite sugar—almost instant color and light. Let it near ammonium salts and you risk more than surprise. Many chemists toy with modification by adding metal salts or even switching out chlorate for perchlorate groups in the molecular structure, seeking just the right balance between color output and stability. The practice underlines a long-standing motivation: get the brightest flame, avoid runaway reactions, keep people safe. Experiments in the lab mirror those on the manufacturing floor, where additives influence not just performance but also regulatory status and risk assessment.
The Names and Faces of the Same Compound
Walk into labs or read through international research, and the same chemical may wear a whole wardrobe of names. Strontium chlorate turns up as “strontium(II) chlorate”, or as its formula Sr(ClO3)2. Some references, especially older texts, mention it as “chloric acid strontium salt.” It’s always worth double-checking chemical synonyms in global contexts, as mistakes in translation or confusion between similar compounds spell disaster—especially where strongly oxidizing materials land in the wrong mixes.
Staying Safe and Working Smart
In my own work, the checklist for strontium chlorate goes well beyond basic goggles and gloves. The need for isolation, temperature control, moisture management, and disposal planning defines a truly thoughtful operation. It’s easy to understand why modern regulations place hefty restrictions on both laboratory and industrial handling. Safety standards cover handling, transport, and storage, but culture matters just as much as paperwork. Even a brief lapse—mixing with sulfur, forgetting to ground containers, allowing dust to settle on open surfaces—risks catastrophe. The call for education and oversight echoes across industries, reminding newcomers and veterans alike that no shortcut stands up against the lessons of chemical history.
Uses and Applications: Lighting Up the World
Pyrotechnics absorb most of the compound’s practical demand. Whether it’s professional fireworks or stage effects, the power and color reliability of strontium chlorate tempt those seeking the purest red. Rescue signal flares, especially offshore, rely on this intense coloration for visibility in varied weather. Artisans in specialty industries might reach for strontium chlorate in niche settings—analytical chemistry, oxidizing reactions, or as a stepping stone for further strontium salt synthesis—though the risks tend to keep most commercial interests away from large-scale use. The compound’s unique blend of properties keeps it relevant despite the rise of safer, less reactive alternatives.
Research and What We Still Learn
Each new study into strontium chlorate nudges old knowledge forward. Chemists have pushed into green chemistry routes, hoping to cut down on toxic byproducts or substitute weaker oxidizers without sacrificing performance in fireworks and signaling. Journal articles detail new safety testing, contaminant analysis, and the hunt for methods that reduce hazards during rinsing and waste treatment. The drive for better processes often grows out of lived experience—one bad day in the laboratory can transform a chemist into a safety advocate. Researchers test alternative color generators and experiment with encapsulation technologies that contain the reactivity until just the right moment.
Toxicity and What It Means for People and Places
It’s impossible to discuss strontium chlorate without looking its toxicity square in the face. Even small amounts spell trouble if ingested, inhaled, or allowed long contact with skin. The chlorate ion itself tends to be the primary offender, as it can interfere with hemoglobin’s ability to carry oxygen—a risk that’s no academic abstraction. There are documented poisoning cases, some accidental, some the result of mishaps in industrial settings. The environmental side cuts just as deep: chlorate ions leaching into water systems threaten plants and aquatic life, and managing waste streams from any operation using this compound requires a hands-on, zero-tolerance mindset. Those realities shift many organizations toward researching and adopting replacements but underscore the ongoing duty to protect workers, users, and the ecosystem in every setting strontium chlorate touches.
Looking Forward: The Road Ahead for Strontium Chlorate
Even in a world obsessed with minimizing risks, the pursuit of vivid color, efficient oxidizing power, and the rich traditions of chemical artistry means strontium chlorate isn’t vanishing anytime soon. Researchers keep digging for safer modulators, process enhancements, and ways to capture the full potential of strontium’s red without triggering the hazardous legacy of reactive chlorates. Perhaps new encapsulation strategies, or alternative flame-producing salts, will carve out a path that lets fireworks light up the night sky with less worry. Until then, the compound stands as a reminder: great chemistry often walks hand-in-hand with great care, and the lessons from both past successes and failures push the field to keep innovating toward safer, smarter outcomes.
What is Strontium Chlorate used for?
On the Trail of Strontium Chlorate
Strontium chlorate doesn’t pop up much in regular conversation, yet those who pay attention to the nuts and bolts of science know it finds its way into some interesting corners of life. In chemistry labs, it puts on a show — and there's a reason for it. This chemical stands out thanks to its oxidizing power. Anyone who tried to light chemicals on fire in a high school demo probably remembers the vivid colors certain elements produce. Even now, seeing a crimson burst during a fireworks display takes me straight back to those lessons. Strontium compounds give that intense red, and strontium chlorate works as both a color source and an oxidizer for those classic shows in the sky.
Why the Fireworks Rely on It
Firework makers hunt for chemicals that burn brightly and reliably. Strontium chlorate fits the bill, releasing oxygen as it breaks down and feeding those fiery reactions. It also supplies that unmistakable red, which is hard to match with other substances. Pyrotechnics companies depend on this chemical, even though safer alternatives exist. The real challenge is the push-and-pull between color quality and risk — strontium chlorate delivers brilliant performance but packs an increased explosion hazard if mishandled.
Hidden in Lab Drawers
In research, strontium chlorate does more than just light up celebrations. Chemists use it as an oxidizing agent for controlled reactions. Here, labs trade off convenience for risk — this chemical accelerates some processes that might crawl with weaker oxidizers. It’s not the most famous tool, but users value its reliability. Still, every chemistry teacher I’ve known swears by a strict set of precautions around it. Even outside of commercial production, students and researchers must understand the hazards and treat every step with care.
Environmental and Health Realities
Chemicals with strong oxidizing punch often cause trouble in the wrong hands or wrong places. Strontium chlorate ranks high on lists of reactive substances thanks to its knack for setting off unwanted fires or even explosions in the presence of fuel or friction. The environmental side of things can't go unnoticed either. Unlike sodium and potassium chlorates, which got banned or restricted in many places, strontium chlorate hangs around in niche uses. Direct exposure can irritate skin and eyes, and ingestion poses risks to organs. Waste management and careful handling stand as the only real option for limiting its impact. Folks managing old fireworks or chemical stockpiles often need practical training and solid safety gear.
Room for Better Alternatives
There’s growing interest in pushing safer substitutes. Potassium nitrate and strontium carbonate don’t burn quite as hot or as red, but they’ve carved out space in some pyrotechnic recipes. Groups like the American Pyrotechnics Association support research into greener chemistry. From my own experience watching the slow shift in chemistry teaching, regulators and fire marshals remind us every season that even old-school spectacle must meet new standards of safety. That means every time regulations tighten or new tech emerges, experts debate the future of chemicals like strontium chlorate.
Smart Handling and a Focus on Safety
Working around chemicals that boast such pure energy requires real respect and skill. Manufacturers, educators, and researchers find value in strontium chlorate but won’t stop teaching safe storage, proper labeling, and emergency cleanup. Anyone who has ever heard about an accidental fire in a school lab knows these rules aren’t mere formalities. Safety data sheets and up-to-date protocols help manage the risks. As long as people chase brighter displays or faster reactions, learning to balance spectacle, science, and safety will shape how chemicals like strontium chlorate earn their place in the toolkit.
Is Strontium Chlorate hazardous or toxic?
A Look at the Risks Behind Strontium Chlorate
Strontium chlorate doesn't sound like a household name, but its dangers are real, even in small quantities. The first thing anyone should know—this compound shows up in the world of pyrotechnics. Many fireworks flickering across summer skies owe their red glow to strontium compounds. Strontium chlorate, with its strong oxidizing nature, plays a part in these vibrant displays.
All those bright colors come with a hidden cost if not handled carefully. Strontium chlorate ranks among hazardous materials thanks to its chemical structure. It reacts fast with organic material. Toss in a bit of friction, heat, or an unfortunate spark and you’ve got a real risk of fire or even explosion. I’ve watched a demo in a chemistry class where a tiny amount nearly caused a panic when a student got careless—a fizz of sparks, a burst of heat, and a cloud of panic as everyone moved back. In the wrong setting, things could’ve gone worse.
Health And Environmental Concerns
The fuss doesn't end with flammability. Exposure brings toxicological concerns. Breathing in dust from strontium chlorate may irritate the nose and throat. Swallowing a mouthful, even by accident, brings bigger problems. It can irritate the stomach, mess with blood cells, and, in high-enough doses, threaten life as it interferes with the blood's ability to transport oxygen. Workers in labs, factories, and even unlicensed fireworks shops face risk if safety standards slip or if don’t work with proper ventilation and gear.
Skin contact rarely causes trouble unless exposure happens again and again. Eye exposure needs quick attention. I once helped a colleague rinse out his eye at an outdoor event when wind blew colored pyrotechnic dust his way—luckily, we kept saline on hand and acted fast, but pain and redness lasted for hours. That’s a lesson no one forgets.
Beyond immediate harm, there’s an environmental story as well. Water-soluble strontium chlorate means runoff doesn’t stay put. Spilled or improperly stored chemical finds its way into soil or streams, hitting aquatic life and soil health. We all know too many pollution stories already—from pesticides to industrial accidents—so the need for responsible handling can’t be overstated.
Responsible Use and Safer Alternatives
Manufacturers can’t cut corners. Regulatory bodies like OSHA and EPA treat strontium chlorate as a chemical needing strict control. The hazmat signs aren’t there for show. Clear labeling, information sheets, and staff training protect workers and the public. As someone who grew up near a fireworks plant, memories of drills and frequent inspections were as common as hearing distant pops during festival season.
There’s some hope for safer alternatives. Chemists continue searching for colorants with less toxicity and lower reactivity. In some settings, strontium carbonate steps in as it’s less troubling from a safety point of view. Firework displays should amaze, not endanger. Regular audits, better storage practices, and advances in chemical substitutes bring us a step closer to making that a reality.
People deserve celebration and safety together. Respect for dangerous materials like strontium chlorate, strict safety rules, reliable data, and a willingness to innovate can stop headlines about accidents before they ever happen.
How should Strontium Chlorate be stored?
Everyday Importance for Labs and Warehouses
Storing chemicals always calls for sharp attention, but some compounds raise the stakes. Strontium chlorate, for example, holds a spot on the list of strong oxidizers. My time in university labs showed me the quiet risks that come with routine handling—one absent-minded move can turn a storage room into a headline.
Strontium chlorate doesn’t wait for mistakes. Its oxidizing power means contact with organic material, powders, or reducing agents can trigger fires or even explosions. A forgotten beaker of acetone, a wooden shelf, or simple paper packaging can serve as the spark. Countless incident reports trace their origins to storage mix-ups that most people figure could “never happen here.” The EPA and OSHA both highlight the same point: these chemicals prove less forgiving than their dusty plastic jars suggest.
Storage Practices that Prevent Trouble
The safest spot for strontium chlorate is a cool, dry room, locked against visitors and marked with clear hazard signage. Anybody without direct business shouldn’t walk in. Metal cabinets, especially those lined with corrosion-resistant coatings, make for reliable storage. Stainless steel buckets leave fewer places for leaks or chemical buildup, and they won’t catch fire if something sits too close.
Rowdy humidity or summer heat does more than spoil food. Damp spaces speed up decomposition, which can generate oxygen and create pressure inside sealed containers. That’s how cabinets become bombs instead of barriers. Good ventilation sucks out vapors and prevents dust buildup, which matters since inhaling even a small amount of the powder can damage lungs and gut lining.
Separation proves just as crucial as container choice. No flammable liquids or organic solids belong anywhere nearby. I saw a storeroom get shut down once after someone left a jug of ethanol next to an oxidizer shelf “just for a weekend.” Rules made in the name of safety seem like overkill, all the way up until the moment a small reaction goes large.
Labeling and Inventory Save Lives
Labels turn blind guesswork into informed caution. Every bottle or drum deserves a date of arrival, and every batch must tell you when it’s time to get rid of it. Unused strontium chlorate ages poorly—it clumps, it reacts with air, and it’s nobody’s friend. A simple laminated sheet on the inside of every cabinet door keeps tally. If that sounds tedious, think about the effort law enforcement and fire crews put in after an accident. I’ve watched a whole day go by as crews sorted unlabeled jars after a fire alarm in my old building—nobody knew what they were handling, so crews had to assume the worst.
Training and Access Controls Build a Culture of Safety
No safety precaution works unless people know why it matters. Every employee or student who handles strontium chlorate should get face-to-face training before touching a container. Training covers not just the chemical’s dangers, but also accident drills and real-world case studies. Sharing stories—the near misses, the lessons after close calls—keeps attention sharp. Restricting who can even open the storage space cuts down “routine accidents” to almost zero. One responsible custodian keeps better track than a dozen half-trained hands ever could.
Simple Steps Outweigh Fancy Gear
Most accidents start with a shortcut. Keep strontium chlorate in tight-lidded metal cans, separated from anything flammable. Factor in temperature, moisture, and airflow. Use physical locks and logbooks. Regularly talk through what could go wrong, so everyone keeps respect for a chemical that shows little mercy for slips. A pile of regulations won’t keep people safe if safe practices don’t become habits—habits carry weight long after the training pamphlets disappear.
What safety precautions should be taken when handling Strontium Chlorate?
Understanding the Risks
Strontium chlorate stands out as a powerful oxidizer. This means even a little carelessness can invite danger. A person who’s moved boxes of lab chemicals learns quickly which ones demand full attention. Strontium chlorate ranks with substances that make you pause before opening the lid. Add a little heat, a stray spark, or contact with organic matter—suddenly there’s a dramatic reaction. Accidents don’t politely wait for warnings; they just happen. Stories from old labs and accident reports show combustions can erupt with little provocation. If you’ve read one too many case files, you treat this white powder with real respect. The dust alone can make eyes sting and lungs burn.
Personal Protection, Not Optional
You want gloves—nitrile, not latex. Chemical splash goggles. A sturdy lab coat. Some might shrug at a simple apron, but I’ll take melted plastic and a ruined pair of pants over a trip to the ER any day. I’ve watched colleagues learn the hard way what happens without a face shield when the air stinks of chemicals and the skin prickles. Some safety lapses leave lasting marks. Inhaling even a little bit of oxidizer-laced powder can kick off coughing fits or more severe reactions, since lung tissue doesn’t repair itself fast. Respirators avoid these problems, especially if strontium chlorate becomes airborne during handling.
Working Conditions Matter
It makes a big difference to do everything in a fume hood or well-ventilated spot. Poor air circulation means everyone in the room gets exposed, sometimes without realizing. I learned after moving a supposedly empty bottle and breathing in stale dust. Cold, dry, and clean surroundings keep accidents at bay. Humid rooms encourage powders to clump or react, often in ways nobody expects. Keeping the work area clutter-free is practical. Strontium chlorate doesn’t belong anywhere near combustible materials or flammable solvents. Cross-contamination isn’t just sloppy—it’s risky. Static control steps like grounding yourself or the workspace can stop tiny sparks that otherwise might go unnoticed.
Handling and Storage: No Shortcuts
Original containers, clearly labeled, with tight-fitting lids: it sounds simple, but sloppy storage crops up in too many labs and classrooms. Every new bottle deserves a careful check, especially for signs of moisture or degradation. Store the containers away from direct sunlight, heat sources, and any chemicals that produce acids. Adventures in mixing oxidizers with reducers rarely end well. I’ve spent time sorting cabinets and always find odd bottles sitting where they shouldn’t. Peace of mind comes from double-checking every shelf. Spills happen—plan for them. Spill kits with proper neutralizers, absorbent pads, and washing stations make the clean-up process less frantic.
Waste Disposal: Not a Side Thought
Pouring leftover strontium chlorate down a drain would violate safety rules and pollute the water system. Hazardous waste containers—durable, leak-proof, and correctly labeled—keep it out of the wrong places. Trained staff or specialized disposal services manage the rest. I’ve watched schools scramble to fix mistakes when chemicals reach the wrong bins. It takes one phone call to prevent a mess and save on clean-up costs. Regular training updates stick in your mind, especially after hearing warnings from those who have seen equipment melt or pipes burst.
Learning from the Past
Each safety measure around strontium chlorate comes from real injuries, lost equipment, and costly interruptions. People who handle this chemical safely never see their precautions as a burden. They see it as a way to go home in the same shape as they arrived. No flashy tech replaces solid judgment and clear procedures. Respect for the chemical—and the people sharing your workbench—makes all the difference.
Where can I purchase Strontium Chlorate?
Strontium Chlorate Draws Caution and Curiosity
Search for chemicals online, and few raise eyebrows like strontium chlorate. Chemistry students catch wind of it in pyrotechnics, and some hobbyists remember seeing old formulas that used it. These days, finding a reliable source is a tall order. In the United States and much of Europe, you won’t find strontium chlorate tucked away in art supply stores or your neighborhood hardware shop. The reason for these roadblocks boils down to safety and legal controls. Strontium chlorate is a strong oxidizer. That means it feeds fire. If it mixes with certain other materials—a spark, friction, or heat—suddenly things get unpredictable or even dangerous.
Sourcing Is More Than Just Clicking “Buy”
People who search for strontium chlorate want to do things right. Most look for lawful sources, like chemical supply companies that cater to schools, industry, or research labs. These sellers don’t just hand it over. They require business licenses, legitimate purposes, and plenty of paperwork. Some ask for a statement of intended use. Others want proof you belong to an institution with a safety officer. My own experience in university labs made these rules familiar—and sometimes frustrating—but also critical for safety. A friend in the fireworks field said his suppliers kept careful tabs on orders, filed reports, and stayed up to date with federal guidelines like those set out by the U.S. Chemical Facility Anti-Terrorism Standards.
Online Marketplaces and the Black Market Temptations
If someone tries searching eBay or obscure online retailers for strontium chlorate, they end up disappointed. Big platforms block listings of regulated oxidizers for good reason. Sketchy websites may claim to offer it, but those present steep risks—a knock on your door from law enforcement or worse, exposure to toxins or scams. Trustworthy chemical suppliers like Sigma-Aldrich, Fisher Scientific, or Carolina don’t risk their reputations with careless sales. They have clear policies, shipment tracking, and proper documentation, and they don’t cut corners for hobbyists or unauthorized buyers. Even the most persistent backyard chemists run into walls.
Why the Drive for Access Surfaces
Sometimes, curiosity pushes people to learn more about chemical reactions, color effects, or science demos. DIY hobbyists and teachers want hands-on experiences, not just textbook diagrams. Limited access to chemicals can be a real barrier to deep learning, pushing some to skirt the rules. But I’ve watched more harm than good come from shortcuts—fires, chemical burns, and a few close calls during high school chemistry experiments gone wrong. Stories from emergency rooms back me up: rare chemicals like strontium chlorate need respect and tight handling.
Pathways for Learning, Not Just Buying
Moving forward, schools and hobby groups can work with universities and registered labs, arranging safe demonstrations or supervised sessions for advanced experiments. Some organizations offer outreach programs or open-house lab days. These events let people see chemical reactions with proper controls and trained professionals nearby. If someone’s determined to work with strontium chlorate, formal education is the path to access: gaining chemistry credentials, joining a research group, and practicing solid lab safety.
Better Safety, Clear Rules
Guidelines from groups like the American Chemical Society and regulatory agencies help keep curiosity from turning into trouble. The best solution isn’t looser restrictions but more hands-on opportunities under expert supervision. Learning comes with responsibility; the rules teach us why certain compounds stay off household shelves.
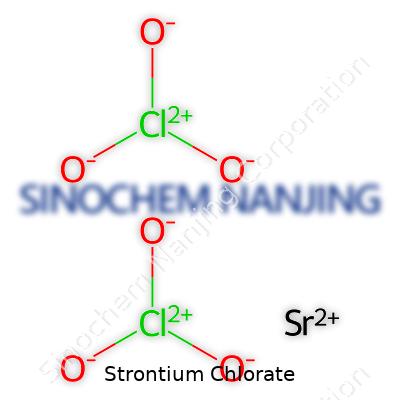
| Names | |
| Preferred IUPAC name | Strontium chlorate |
| Other names |
Chloric acid, strontium salt Strontium chlorate(V) Strontium dichlorate |
| Pronunciation | /ˈstrɒn.ti.əm ˈklɔː.reɪt/ |
| Identifiers | |
| CAS Number | 10025-70-4 |
| Beilstein Reference | 127122 |
| ChEBI | CHEBI:86458 |
| ChEMBL | CHEMBL1233067 |
| ChemSpider | 22354 |
| DrugBank | DB15640 |
| ECHA InfoCard | 100.028.859 |
| EC Number | 233-961-8 |
| Gmelin Reference | 1877 |
| KEGG | C18682 |
| MeSH | D013325 |
| PubChem CID | 24828 |
| RTECS number | TH8225000 |
| UNII | MX2J699ZV8 |
| UN number | 1507 |
| Properties | |
| Chemical formula | Sr(ClO3)2 |
| Molar mass | 261.53 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 3.52 g/cm³ |
| Solubility in water | soluble |
| log P | -3.07 |
| Vapor pressure | Negligible |
| Magnetic susceptibility (χ) | +79.0e-6 cm³/mol |
| Refractive index (nD) | 1.443 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 208.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -877.2 kJ/mol |
| Pharmacology | |
| ATC code | V09XX03 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS03,GHS07 |
| Signal word | Danger |
| Hazard statements | H271: May cause fire or explosion; strong oxidizer. |
| Precautionary statements | P264, P280, P370+P378, P501 |
| NFPA 704 (fire diamond) | 2-2-3-OX |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 oral rat 1040 mg/kg |
| LD50 (median dose) | LD50 (median dose): 600 mg/kg (oral, rat) |
| NIOSH | SN 8850000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | REL (Recommended Exposure Limit) of Strontium Chlorate is "0.5 mg/m³". |
| IDLH (Immediate danger) | IDHL: 50 mg/m3 |
| Related compounds | |
| Related compounds |
Barium chlorate Calcium chlorate Magnesium chlorate Strontium perchlorate Strontium nitrate |

