Strontium Arsenite: An In-Depth Commentary
Historical Development
Looking back at the origin of strontium arsenite, research shows that early chemists often crossed paths with obscure compounds long before anyone fully understood their nature or risks. Strontium was uncovered in Scotland at the end of the 18th century, its name coming from the town of Strontian. Arsenic compounds date even farther back, known and feared for their toxic potential since ancient times. Those mixing strontium and arsenic in a lab a century ago weren’t running sophisticated control systems or thinking about environmental impact. They eyed new dyes, glass additives, and pigments, while often downplaying dangers. Today things look different. Guidelines and safety expectations changed, especially with what we know now about arsenic exposure. Yet, each modern study adds to the stack of knowledge built by these early experiments, proving there’s always more to learn from the past in handling materials that come with such baggage.
Product Overview
Strontium arsenite shows up as a heavy, crystalline material, sporting colors from pale white to yellow, depending on purity and source. Factories often produce it for its pigment qualities, especially where resistance to heat matters. The formula sticks to Sr3(AsO3)2. It’s not a compound you find around the kitchen or local hardware shelf. Industry leans on it as a specialty item for ceramics, glassmaking, and sometimes for limited chemical syntheses. Because arsenic sits at its core, producers mark strontium arsenite for professionals with suitable training—not your average general-use chemical.
Physical & Chemical Properties
You handle strontium arsenite as a dense powder, grainy and tough to dissolve in water. The stuff resists melting at high temperatures, making it handy for processes facing serious heat. It breaks down in strong acids, giving off arsenic trioxide fumes, which demand heavy ventilation and expert handling. No one should overlook the weight of this powder or its stubborn resistance to most ordinary solvents. So, labs with the right credentials keep it under lock and key, stressed by the dual threat—chemical reactivity and potent toxicity.
Technical Specifications & Labeling
Product datasheets for strontium arsenite highlight the importance of batch consistency. The strontium to arsenite molar ratio stays fixed, keeping up with chemical standards. Impurities—especially lead, barium, and unreacted arsenic—get measured down to ppm levels. Labels on drums or bags always feature international hazard symbols for acute toxicity and environmental risk. Producers stamp proper United Nations numbers and safety codes on every package. Accuracy here means everything, since one slip could spell a breach of health regulations or introduce risks in downstream applications.
Preparation Method
Makers prepare strontium arsenite by mixing strontium salts and arsenite sources under tightly regulated lab conditions. The usual process starts with strontium chloride or strontium nitrate dissolved in water, meeting sodium arsenite under controlled temperature and pH. Precipitate forms, workers filter and rinse the solid, then dry and grind it. Labs treat waste streams as hazardous, collecting everything for specialized disposal. Clean production means tailoring steps to minimize dust and keep emissions to a minimum, stepping up with scrubbing filters and chemical neutralization for waste gases.
Chemical Reactions & Modifications
Drop strontium arsenite into acid, and you get a fast reaction releasing arsenic trioxide and strontium salts. Act with heat, and decomposition shows up as gaseous arsenic compounds, pushing for top-tier ventilation and gas capture technology. Try pairing with oxidants, and the compound reacts quickly—giving off both strontium oxide and various arsenate byproducts. There’s not much flexibility for benign changes. Every modification opens doors to dangerous forms of arsenic, which health experts keep a firm leash on. Workers wear full gear because even small mishaps can cause huge problems for people and the surrounding environment.
Synonyms & Product Names
In chemical catalogs, this compound stands under names like tris(strontium) arsenite, strontium(II) arsenite, and sometimes by internal numbering in research lab inventories. Synonyms carry across languages, but the hazard stays the same. Buyers and handlers must check root chemical names and cross-check with safety data to avoid mix-ups, since similar-looking labels can lead to life-threatening confusion.
Safety & Operational Standards
Nobody downplays the hazards here. The regulation lines stretch further for strontium arsenite than most specialty chemicals. Strict rules govern storage—only sealed, labeled containers, locked storage, and spill kits ready for action. Workers need respirators certified for arsenic dust, heavy-duty gloves, and sturdy eye protection. Contract labs adhere to local, national, and international codes, including OSHA, REACH, and GHS regulations. Emergency protocols see regular drills, and all waste finds the hands of licensed hazardous material handlers. Even lab ventilation must reach benchmarks set by industrial hygiene experts.
Application Area
Strontium arsenite isn’t a household word outside specialist circles. Glass and ceramic outfits look for it when certain color effects or chemical resistances matter—especially in making glass for science and custom fixtures. Some old textiles featured it in pigments, though modern codes largely stopped that. Limited chemical research sometimes involves strontium arsenite as a precursor to other compounds. No broad consumer uses exist because arsenic’s toxicity puts the brakes on popularity and legal access. Industrial users shoulder responsibility for keeping the supply chain bulletproof.
Research & Development
Researchers in materials science keep looking for ways to make the best use of its unique properties while keeping risks manageable. Synthetic chemists explore routes to tweak crystal structure, hoping for new glass or ceramic behaviors. Environmental scientists study how strontium arsenite interacts with common waste streams, searching for strategies to block arsenic pollution. Laboratory security teams set standards to detect even trace leaks, often inventing new monitoring devices in the process. Yet, most modern R&D focuses on reducing or replacing arsenic-based chemicals altogether, given the mounting health concerns and regulatory pressure worldwide.
Toxicity Research
Arsenic-based compounds carry a story filled with medical intrigue and tragedy. Toxicologists document dangerous effects even from low exposures to dust or vapor. Cancer risk spikes sharply for those breathing arsenite particles. Animal studies uncover damage to organs, DNA, and the nervous system. Workers dealing with strontium arsenite face added scrutiny for early symptoms—skin lesions, nerve problems, and respiratory distress. Decades of research guide medical staff in both treating accidental exposures and setting workplace thresholds. Testing goes far beyond guesswork, following international consensus on the maximum allowable exposure, but the margin for error keeps shrinking as scientists learn more about long-term effects.
Future Prospects
Moving forward, tight rules promise reduced production and limited access. Researchers keep their eyes out for safer replacements in specialty ceramics and glass, hoping for materials that keep performance while ditching the toxicity profile. Companies invest in methods to recover arsenic and strontium from industrial waste to shrink environmental impact. Digital monitoring and advanced filtration technology stay on the rise to catch even the tiniest escape from production lines. If more sustainable substances take over, strontium arsenite will fade further into a footnote, with lessons learned closely informing future chemical development. Every update to global guidelines reflects a world less willing to trade safety for short-term technical gains—pointing chemical innovation in new, safer directions.
What is Strontium Arsenite used for?
The Role Strontium Arsenite Plays
Strontium arsenite doesn’t show up much in the headlines unless you look into specialized industries or the history of pigment manufacturing. This compound carries a legacy—a legacy both in color and caution. For decades, people mixed it into paints to lay down strong, bright reds. Ceramics and glassmakers used it in glazes, drawn by its ability to maintain color intensity even after high-heat firing. When you see vintage reds in old glassware or antique signs, there’s a fair chance some of that glow comes from strontium arsenite.
Industry Choices: Beauty and the Beast
Bright pigments sell paint, ceramics, and art supplies, and some of the most stubborn reds on the palette once came from strontium arsenite. The pigment’s stability under the sun and intense heat made it a go-to for outdoor uses: road signs, warning labels, signal flares. Manufacturers often favored it because it stuck around—it didn’t fade or turn muddy under tough conditions. If you ever worked in a paint shop, you’d recognize the value in a color that doesn’t disappoint six months after going on a wall.
The Health and Safety Factor
On paper, combining strontium and arsenite gives you a substance with practical perks. In real life, arsenic flips the situation on its head. There’s a reason most households don’t stock strontium arsenite: arsenic is toxic. Long-term arsenic exposure damages organs and increases cancer risk. That brings up stories of factory workers from decades ago—anyone who spent time around arsenic compounds knows caution is not optional. I remember touring an old ceramics plant where red pigments demanded separate ventilation; safety gear covered everyone mixing powder. One careless move could haunt you for years.
Regulatory Changes: The Search for Safer Alternatives
Once experts saw the health damage from chronic arsenic exposure, regulators didn’t take long to limit its use. Paint and pigment industries moved away from strontium arsenite. Safer synthetic reds and oranges showed up in the 20th century. You can walk the aisles of a modern hardware store and pick up can after can of bright red paint without worrying about arsenite dust. Many governments now ban or strictly regulate compounds containing arsenic, cutting down occupational illness and environmental risk.
What Matters Now
Few modern businesses put strontium arsenite on their ingredient lists. The risks outweigh the rewards. Today’s pigment makers craft stable reds from alternatives—think iron oxides, organic dyes, or engineered nanoparticles—without dealing with toxic heavy metals. It’s still possible to come across strontium arsenite in museum restoration labs. Some collectors and chemists hunt down old stocks for research, but those cases are rare and regulated closely.
Moving Forward Safely
It’s easy to focus on the science, but experience shows that good safety procedures win the day. I’ve seen skilled artists and manufacturers protect themselves by asking questions about what’s in their materials, wearing the right gear, and demanding clear labeling. Whenever chemicals like arsenic get involved, trust in regulation, transparency, and personal responsibility makes all the difference. Keeping the past in mind helps keep workers healthier and the art world just as vivid—without risking lives.
Is Strontium Arsenite toxic or hazardous?
Looking at Chemical Risks Up Close
Strontium arsenite isn’t a chemical found in most homes, but people working in lab environments or certain industries might come across it. Just hearing “arsenite” can cause some alarm – arsenic has a long reputation for being poisonous, both in movies and real life. Strontium itself lands on the less-famous side, more known for its use in fireworks and some medical imaging tests. Once these two elements bond together, you get a compound with risks that anyone handling it should not ignore.
Poison in the Details
Arsenic stands out for its toxicity. The World Health Organization hasn’t minced words about arsenic compounds: exposure links clearly to cancer, skin problems, heart disease, and damage to the nervous system. Arsenite forms of arsenic pose more threat than arsenate forms, because the body absorbs them more easily. Strontium arsenite follows the same rules. Inhaling its dust, swallowing even small amounts, or taking it in through the skin can harm people. This stuff packs a punch not just for the lungs and liver, but for pretty much the whole body if exposure lasts or the dose climbs too high.
Personal Perspective: Safe Practices Make a Real Difference
Early in my career, I worked at a university lab tracing heavy metals in water. Safety gear felt bulky, goggles steamed up, and gloves slowed everything down. Still, I saw what happens when handling protocols get skipped. One grad student wearing no mask ended up with blisters after a splash from a much less nasty compound than strontium arsenite. Harsh chemicals teach tough lessons fast. Respecting proper handling pays off, especially with something carrying the one-two punch of strontium and arsenic.
Backed by Hard Data
The U.S. Environmental Protection Agency identifies arsenic compounds as hazardous air pollutants. OSHA restricts exposure tightly, with workplace air limits set at a few micrograms per cubic meter. In Europe, regulators treat these compounds with similar caution. Any site storing or using strontium arsenite falls under strict chemical inventory and disposal reporting. This regulation comes from solid research: chronic arsenic exposure links to lung, skin, and bladder cancers, among other issues. Strontium doesn’t cancel out these effects – it just rides along for the trip.
Path Forward: Reducing Risk Where It Counts
Substitutes or alternative processes should get priority if possible. Quite a few industries already look for less toxic routes before pulling arsenic off the shelf. In university and advanced manufacturing settings, education and enforcement must go hand-in-hand. Clear signs, routine training, and strong oversight help cut down sloppy mistakes. Even simple moves like using a fume hood and wearing proper gloves stack the odds toward safety. Prompt cleanup of spills, careful disposal, and good recordkeeping keep legal and health troubles far away.
Why This Matters
Few people walk through life thinking about compounds like strontium arsenite. But workers and communities living near industrial sites deserve upfront information and support. Controlling exposure doesn’t just protect employees; it shields neighborhoods, water supplies, and the environment from hazardous runoff and accidental contamination. Science makes the risks plain, and common sense calls for action. Chemical safety isn’t a luxury. It’s a necessity for everyone down the line.
What are the storage requirements for Strontium Arsenite?
Understanding Strontium Arsenite
Strontium arsenite draws attention due to its toxicity. Coming across it in a lab or an industrial setting reminds anyone of its potential dangers: both strontium and arsenic compounds pose serious health risks. That means folks handling it hold a special responsibility. Some say keeping things safe is about trusting your gear and your training, and for strontium arsenite, that belief rings true more than ever.
What Makes Storage Critical
The powder is both a chemical and an environmental hazard. Any accident—spill, leak, dust, or vapor—doesn’t just affect the handlers but also the larger community. Over the years working in labs, I’ve seen seasoned technicians triple-check labels and storage spaces just out of sheer respect for substances like this. Mishandling strontium arsenite risks poisoning through inhalation or accidental touch, and the cost can last a lifetime.
Key Storage Principles
1. Cool, Dry, Isolated SpacesStore strontium arsenite in a well-ventilated, locked cabinet. Moisture plus certain chemicals equals trouble. Humidity leads to corrosion of containers, and chemicals leaching out raise the risk for everyone nearby. Any room storing it ought to stay as dry and cool as possible. Forced ventilation keeps the air in check and reduces the risk of inhaling anything by mistake.
2. Secure, Well-Labeled ContainersUse airtight glass or high-quality plastic jars. From experience, metal isn’t wise since strontium arsenite can react if moisture finds its way inside. Good labs invest in chemical-resistant labels—faded, peeling stickers spell disaster if things get shuffled around. Even folks who’ve spent decades in a lab can get tripped up by poor labeling.
3. Keep Away From Incompatible SubstancesFlammable solvents, acids, and alkalis have no place nearby. Chemical databases flag strontium arsenite as risky with oxidizers and reducing agents both. Accidental mixing can produce toxic gases or explosive compounds. Segregated shelving works well. Many companies fine-tune their storage protocols from earlier close calls, logging everything and doubling back on regular audits.
Protecting Humans and the Environment
Any chemical storage area with strontium arsenite needs spill kits on hand. These aren’t standard kits—sorbents, neutralizers, and special gloves all play a part. Personal protective equipment (PPE) saves lives. Workers should always suit up with gloves, goggles, and a fitted mask. I remember my first experience in the chemical storeroom, and the mentor there used to say, “Treat arsenic like you’d treat a loaded gun. No shortcuts.” That lesson sticks.
Disposing of strontium arsenite leftovers matters as much as storage. You can’t just toss residues in the garbage. Facilities often set up strict waste management workflow, where hazardous waste contractors cart off sealed containers for proper treatment. Reports of groundwater contaminated by improper disposal of heavy metals have surfaced before—that risk is real.
Improving Chemical Safety Standards
Continuous education changes outcomes. Everyone who handles dangerous substances should know exactly what they’re dealing with, every time. Posters, emergency instruction cards, and clear lines of communication help reinforce what’s at stake. Audits, checklists, and peer oversight bring extra security. I’ve watched safety officers catch errors the rest of us missed; sometimes it's human diligence that stands between business-as-usual and a major health scare.
More investment in modern ventilation and alarm systems goes a long way. Bigger organizations track usage electronically, logging each access and providing just-in-time training reminders. For smaller teams, pooling resources or sharing best practices pays off just as well. It all comes back to taking chemical hazards seriously, day in and day out.
Final Thoughts
Every rule about storing strontium arsenite serves a real-world purpose. Respect for these protocols keeps people healthy and stops needless headaches down the road. Years of lab work have convinced me—good habits are the difference between routine and regret.
What is the chemical formula and structure of Strontium Arsenite?
A Closer Look at Strontium Arsenite
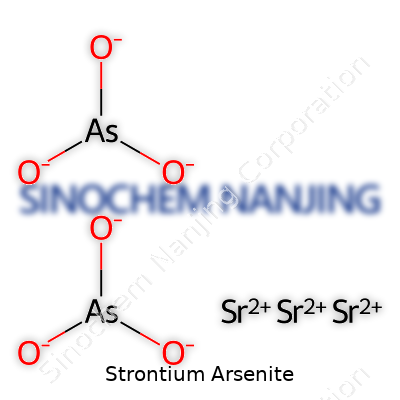
Chemical formulas carry a certain honesty that helps chemists, teachers, and students speak the same language worldwide. Strontium arsenite comes up in a few textbooks and reference materials, even if you don’t spot it lining the shelves of your local pharmacy or hardware store. The chemical formula for strontium arsenite is Sr3(AsO3)2. Each formula unit features three strontium atoms paired with two arsenite groups.
Understanding Its Structure
Many people think of chemistry as this world of mysterious shapes and bonds. Take a look at the arsenite ion first—its formula (AsO3)3- feels simple but packs plenty of detail. Arsenite usually builds itself out in a sort of triangular-pyramidal shape. Arsenic settles in the middle, coordinating with three oxygen atoms that form the corners. The leftover electron pairs on arsenic nudge the geometry out of being a flat triangle, so you’re left with a three-cornered pyramid.
Strontium, sitting right beneath calcium on the periodic table, exists as a Sr2+ cation. It teams up with arsenite’s 3- charge in a crystal lattice that balances out each charge, letting the solid hold together. In a classroom or research lab, the arrangement might take the form of repeating units—clusters of strontium ions bridged by the arsenite anions. You get an extended lattice, pretty much like other ionic salts, which influences melting point, solubility, and chemical stability.
Why Strontium Arsenite Matters
I’ve noticed that strontium compounds pop up more often than most people guess. Strontium itself gives fireworks their bright red color, and arsenic compounds played a huge part in pigments and glassmaking in the past. But make no mistake—arsenic carries a hefty toxicity warning. As with all arsenites, strontium arsenite must be handled with care, following strict guidelines to reduce exposure and contamination.
These risks don’t mean scientists and teachers avoid the topic. In fact, understanding compounds like strontium arsenite builds respect for laboratory safety. The awareness carries over to water supplies and soil analysis, since arsenites often point to pollution that can threaten crops, livestock, and people. Regulators and researchers around the globe watch for these compounds, especially where industrial waste or mining activity might leave traces behind.
Developments and Responsible Handling
Health professionals and chemical safety officers spread the word about responsible use wherever arsenite compounds appear. In recent years, strict environmental laws across many countries clamp down on arsenic pollution. Strontium arsenite’s name sometimes comes up in academic circles looking at waste treatment or environmental clean-up. Scientists keep searching for effective ways to break down or lock away these kinds of hazardous compounds, turning to advanced filtration techniques and specialized bacteria capable of changing arsenic’s chemical form.
For someone coming up in the science world, paying attention to strontium arsenite’s formula and structure serves as a wake-up call about chemistry’s impact on society. Learning about chemical bonding, crystal formation, and toxicology builds more than just exam knowledge—it shapes real-world decisions in laboratories, classrooms, and policy offices.
How should Strontium Arsenite be handled safely?
Why Strontium Arsenite Demands Respect
Strontium arsenite, a compound found in some pigments and industrial products, brings serious risk straight to the workbench. It contains arsenic, a chemical people have learned to treat with respect for centuries, and strontium, active enough to pose its own set of concerns. Toxic dust and fumes stick to this material, and the stakes remain high for both people and the environment.
Personal Safety in Practice
Experiencing work in labs where hazardous compounds like this are on the inventory, personal responsibility becomes hardwired from the first training day. Forgetting to put on goggles or gloves—even once—leaves lasting memories. Gloves that fit well, goggles with anti-fog features, and a fitted lab coat form the basic armor. For airborne risk, nothing beats a well-fitted respirator. Feeling that tight seal and the slightly labored breath makes the risk real: the press of fabric, the filter’s insistence, all reminders that one careless breath can spell trouble.
The Right Space for the Job
Working with strontium arsenite happens in a fume hood or a specially ventilated area. The whir of a fan overhead signals safety. Dust control starts before any work—wetted towels or antistatic mats keep stray dust from traveling. In my time on clean-up crews, floors and benches got swabbed with solutions known to trap fine particles, never just swept or dry-wiped. A spill, even small, leads to a full halt and a focused cleanup with dedicated tools.
Training and Vigilance Matter More Than Labels
Labels mark the risks, but real understanding comes from repeated drills and shared stories. Handling strontium arsenite calls for chemistry teams to go beyond required checklists. Peer checks, double-bagged waste, a visible log sheet for every removal—all parts of routines that can save a person’s lungs or skin. Hearing veterans talk about long-term exposure makes the dangers stick.
What Happens to Waste?
Waste disposal shapes my own respect for chemicals like strontium arsenite. The red bags, locked barrels, and scheduled pickups are visible in any facility dealing with hazardous waste. Each gram gets recorded, weighed, and tracked so nothing slips into the regular trash. I’ve spoken with waste management specialists who never stop emphasizing traceability—they keep tabs on every transfer, because one missed step can contaminate water or land for years.
Education and Oversight Still Play Catch-Up
Too often, training programs stick to the rulebook, leaving out vivid case studies that hit home. People remember stories and real consequences. Materials like strontium arsenite push schools and workplaces to up their game: regular refreshers, anonymous reporting for unsafe acts, and cross-training so no one stands alone with dangerous stuff. Government inspections add teeth to these expectations. When inspectors walk the floor, teams pay attention and safety culture strengthens.
Room for Better Solutions
Safer substitutes and new containment tech can lower risk, but industry change lags behind scientific discovery. Some companies install air sensors that text a supervisor if dust concentration climbs, making employees part of the system’s feedback loop. Others limit access to only the tasks that truly need strontium arsenite, choosing automation where possible. Personal accountability means filling out checklists, but it also relies on the know-how picked up from mistakes and near-misses.
It’s About Real People
What sticks with me isn’t just the procedure or the gear—it’s people: the feeling of walking out of the lab at the end of the day, knowing your team looked out for each other and every ounce of strontium arsenite stayed contained. The chemical challenges workplaces to match science with action, experience with routine, and respect for health with the realities of the job.
| Names | |
| Preferred IUPAC name | Strontium trioxidoarsenate(III) |
| Other names |
Strontium(II) arsenite Strontium orthoarsenite |
| Pronunciation | /ˈstrɒn.ʃi.əm ˈɑː.səˌnaɪt/ |
| Identifiers | |
| CAS Number | 12006-54-7 |
| Beilstein Reference | 1901873 |
| ChEBI | CHEBI:88214 |
| ChEMBL | CHEMBL4262983 |
| ChemSpider | 21844392 |
| DrugBank | DB15954 |
| ECHA InfoCard | 03c41d40-694a-4bb0-b5cd-dc274d1f3dda |
| EC Number | 232-225-4 |
| Gmelin Reference | 84845 |
| KEGG | C18612 |
| MeSH | D013324 |
| PubChem CID | 101976798 |
| RTECS number | XW6475000 |
| UNII | F52XU69A3L |
| UN number | UN1557 |
| CompTox Dashboard (EPA) | DTXSID6012536 |
| Properties | |
| Chemical formula | Sr₃(AsO₃)₂ |
| Molar mass | 309.40 g/mol |
| Appearance | White solid |
| Odor | Odorless |
| Density | 4.56 g/cm³ |
| Solubility in water | Insoluble |
| log P | -2.0 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 6.36 |
| Magnetic susceptibility (χ) | Paramagnetic |
| Refractive index (nD) | 1.755 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 120.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1026 kJ/mol |
| Pharmacology | |
| ATC code | V10BX03 |
| Hazards | |
| Main hazards | Harmful if swallowed, toxic if inhaled, may cause cancer, causes damage to organs, environmental hazard |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | P261, P264, P271, P272, P273, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P311, P314, P330, P362+P364, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-2-OX |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 (oral, rat): 20 mg/kg |
| LD50 (median dose) | LD50 (median dose): 14 mg/kg (Oral-rat) |
| NIOSH | B017 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Strontium Arsenite: 0.01 mg/m³ |
| REL (Recommended) | 0.01 mg/m³ |
| IDLH (Immediate danger) | IDLH: 5 mg/m3 |
| Related compounds | |
| Related compounds |
Strontium arsenate Barium arsenite Calcium arsenite |

