Sodium Trifluoromethanesulfinate: Beyond the Bench
Historical Development
Chemists working through the middle of the twentieth century kept running up against walls in the evolution of organofluorine chemistry. Synthesizing efficient sulfinic acid derivatives just didn’t come easy. Then came sodium trifluoromethanesulfinate—often called Langlois’ reagent—shaking up synthetic labs in the late 1980s and early 90s. Langlois and his collaborators documented how this salt could slip fluorinated groups onto aromatic and heterocyclic skeletons much more smoothly than past sulfinates or the gnarly Umemoto reagents before them. Their work drew new blueprints for C–S and C–F bond formation, setting the stage for medicinal chemists diving into fluorinated bioactive molecules today.
Product Overview
Sodium trifluoromethanesulfinate, better known among chemists as “CF3SO2Na,” stands as a white to off-white crystalline powder with a punchy sulfurous odor. Its CAS Registry Number is 2926-29-6, so it pops up in catalogs under varied labels—Langlois’ reagent, sodium (trifluoromethanesulfinyl)oxidanide, or the flashed label “sodium triflinate.” Suppliers like Sigma-Aldrich or TCI America keep it stocked for labs running alkylation reactions, preparing trifluoromethyl sulfones, and building modern pharmaceutical cores. CF3SO2Na often shuffles in as a strong nucleophile and offers a reliable, shelf-stable, and cost-effective source for trifluoromethyl sulfonyl groups.
Physical & Chemical Properties
With a molecular weight of 170.07 g/mol, sodium trifluoromethanesulfinate doesn’t take up much space on the scale in the lab, but it packs a punch. This salt dissolves easily in polar solvents like water and dimethyl sulfoxide, letting chemists dial in concentrations for various transformations. It holds up well at standard room temperature, but its stability fades in acidic media—hydrolysis strips it of that precious trifluoromethanesulfonyl group. Don’t expect it to char easily; decomposition usually starts above 150 °C, releasing the distinct pungency of sulfurous compounds.
Technical Specifications & Labeling
Packaged typically in airtight, light-resistant containers to dodge hydrolysis and photo-degradation, sodium trifluoromethanesulfinate ships with purity over 98%, moisture under 0.5%, and is always checked for chloride and sulfate byproducts. Labels show hazard and precautionary statements under GHS guidelines. Transporters log the UN number if large drums cross customs lines due to its potential as a reducing agent in bulk. Documentation spells out the need for chemical fume hoods, gloves, and face protection—real-life lessons chemists learn the hard way, if not shown directly.
Preparation Method
In bulk synthesis, factories hit up methyltrifluoromethanesulfonate with sodium sulfite, converting the methyl group to the sulfinic acid through nucleophilic substitution, then basifying the mixture for sodium salt precipitation. On small scales, the path often begins with trifluoromethanesulfonyl chloride and sodium dithionite; column chromatography, crystallization, and lyophilization steps follow. In my own experience, prepping it in a university organofluorine lab always centered on minimizing exposure to moisture since even a little dampness quickly ruined a batch, turning projects into a scramble for fresh reagents.
Chemical Reactions & Modifications
CF3SO2Na jumps into radical trifluoromethylation reactions, working well under photocatalytic or copper catalysis for aryl and heteroaryl trifluoromethylations. Researchers use it to swap hydrogen for trifluoromethyl groups on drug candidates, boosting metabolic stability with a simple, economic touch. The salt goes beyond direct trifluoromethylation, too—it acts in oxidative couplings, sulfonamide synthesis, and more. Not long ago, a graduate student in my group used Langlois’ reagent to prepare new sulfone-linked liquid crystals, which would’ve been impossible with pricier or less stable alternatives. Modifications often use boron- or iron-based catalysts to tune selectivity, making the chemistry both customizable and robust.
Synonyms & Product Names
Pick up any synthesis paper or catalog, and the alternate names come out: sodium triflinate, sodium trifluoromethanesulfonate, Langlois' reagent, sodium (trifluoromethanesulfinyl)oxidanide, or simply CF3SO2Na. Suppliers sometimes push the “triflinate” branding, tying it to its pharmaceutical uses, but old-school organofluorine hands still reach for Langlois’ reagent on the bench. The name itself becomes a kind of shorthand across research and manufacturing labs, making communication simple but also necessitating careful cross-referencing in paperwork.
Safety & Operational Standards
Handling sodium trifluoromethanesulfinate means suiting up with gloves, goggles, and lab coats. Splashes sting, dusts irritate, and inhalation can bring about sore throats and headaches, so bench researchers work with it beneath certified hoods. Waste streams head straight to hazardous disposal, not down the sink. Ventilation matters—nobody wants a room full of sulfur dioxide byproducts if decomposition gets out of hand. Large producers check for sodium dust and maintain spill kits on hand to prevent reactive blazes. The paperwork often references ISO and OSHA standards, but real safety comes from deep familiarity with the salt’s reactivity, especially during scale-ups or modifications.
Application Area
Pharmaceutical labs lean on sodium trifluoromethanesulfinate to prepare fluorinated analogs of marketed drugs, leveraging the trifluoromethyl group to increase metabolic resistance and tweak pharmacokinetic profiles. Agrochemicals feature it in pesticide and herbicide pipelines for much the same reasons—trifluoromethylation lifts bioactive amide and heterocycle stability in harsh environments. Outside molecular design, this salt sees use in materials science, especially developing new polymers and electrical insulators with enhanced chemical durability. In my time consulting for a mid-sized specialty chemical maker, interest surged after a major client switched to CF3SO2Na in their pipeline, shaving time and cost throughout synthesis. The salt’s shelf-stability and ease of handling push it ahead of more sensitive fluorinating agents.
Research & Development
Academic labs push sodium trifluoromethanesulfinate into unexplored couplings—nickel or copper catalysis, photoredox-driven reactions, and even green chemistry adaptations. Journals fill with papers on site-selective functionalizations, or new ligand complexes that unlock reactivity against stubborn aromatic bonds. The drive to streamline late-stage trifluoromethylation has led to high-throughput screening experiments, live cell transformations, and even continuous flow processing trials for scale-up. In my own former department, projects linked CF3SO2Na to DNA-encoded library screening, propelling new ways to tweak structure-activity relationships in lead optimization. It comes down to balance—finding mild, selective chemistry that also respects environmental goals and cost ceilings.
Toxicity Research
Animal toxicity studies show that sodium trifluoromethanesulfinate carries limited acute toxicity, though the trifluoromethyl group can bioaccumulate and bring about chronic environmental load. Eye and skin irritation risks persist, and tests confirm possible mutagenicity at high exposures, making lab protocols doubly important. Acute ingestion triggers vomiting, but no long-term organ damage appears at environmentally relevant doses in most mammalian models. Waste management guidelines keep spent reagent out of water systems, recognizing the potential for aquatic toxicity tied to fluorinated breakdown products. Responsible use calls for continual review of emerging toxicological data and updating handling documents as regulatory standards grow stricter.
Future Prospects
Sodium trifluoromethanesulfinate’s future feels secure as new medicines and crop protection chemicals demand more fluorination and fewer environmental drawbacks. Catalysis research will keep opening gentler or more selective pathways, letting labs slide this salt into transformations that once needed much harsher fluorinating agents. Scale-up will challenge both chemical manufacturers and supply chains, especially as regulatory scrutiny tightens and calls for greener alternatives rise. If chemists keep reporting ways to recover or recycle fluorinated reagents, and pilot project managers lean into continuous flow processes, CF3SO2Na stands ready to stay relevant as chemistry’s appetite for trifluoromethyl builds. I learned in client-focused tech transfer years that those who link research directly to manufacturing practicality—asking, “Does this make sense at the ton scale?”—end up guiding what next-generation sulfinates and analogs will look like in both lab and industry.
What is Sodium Trifluoromethanesulfinate used for?
What’s Actually Going on With This Compound
Take a walk through any modern chemistry lab, and eventually you'll spot a bottle labeled “sodium trifluoromethanesulfinate.” The label rarely stirs excitement, but the real story starts inside the flask. Most people outside of chemistry circles have never heard of this stuff, but behind the scenes, it shapes drug synthesis, helps improve materials, and keeps research moving.
It's not as dramatic as life-or-death emergency medicine, but sodium trifluoromethanesulfinate plays a quiet but steady role in the progress of science. I once worked in a research group cobbling together building blocks for a potential cancer drug. The boss tossed me a small white jar and told me, “This is your friend today.” That jar happened to be sodium trifluoromethanesulfinate—also known as Langlois’ reagent—which lets chemists tack fluoroalkyl groups onto all sorts of molecules. Turns out, pharmaceuticals love these groups. Adding trifluoromethyl groups can toughen up molecules, help drugs travel through the body, and even stretch a molecule's patent life, which means a lot in drug development.
Inside the Bottle: How Chemists Use It
Not many chemicals show up in so many research projects at once. Langlois’ reagent delivers trifluoromethyl groups—the CF3 pieces that pharmaceutical companies and material scientists both chase. Toss it into a beaker with the right partners, and out comes a new molecule with properties nobody could reach before.
Researchers seeking to tweak the solubility or metabolic stability of a drug candidate turn to sodium trifluoromethanesulfinate. In one memorable project, our team used this reagent to help a candidate drug resist breakdown in the liver, getting it into the bloodstream at higher concentrations. This little boost sometimes marks the difference between a failed experiment and a promising new treatment.
Beyond medicine, the demand for rugged plastics and electronic components keeps sodium trifluoromethanesulfinate relevant. The introduction of CF3 units can turn floppy organic chains into sturdy materials. The result? Plastics resistant to flames, or coatings that shun chemicals and dirt, and even substrates for high-tech electronics.
Risks and Roadblocks
Anybody handling chemicals owes the public straight talk about risks. Sodium trifluoromethanesulfinate isn’t the most hazardous compound in a lab, but it still demands respect. Flammable dust, skin irritation, and unpredictable reactions remind chemists not to cut corners. I’ve seen a few rookie mistakes: spills that left a sharp, stinging smell, or careless disposal that troubled the building’s air systems. Most labs fix this with solid training, routine glove use, and proper ventilation, but these reminders never get old.
Room to Grow
With so much riding on the introduction of fluoroalkyl groups, access and cost remain challenges. The sheer demand pushes supply chains, especially as more chemists try to scale up their work. Some have pushed for greener synthesis—finding new ways to make or recycle trifluoromethanesulfinate from less toxic raw materials. Industry collaboration between synthetic chemists and environmental safety experts could turn these experiments into reliable, affordable options.
It’s easy to overlook humble reagents like sodium trifluoromethanesulfinate. Still, without its contributions to pharmaceuticals and advanced materials, a lot of those glossy product brochures and promising clinical studies would never reach the light of day.
What is the chemical formula of Sodium Trifluoromethanesulfinate?
What the Letters and Numbers Mean
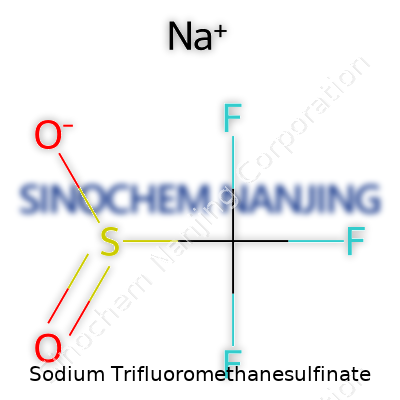
Sodium trifluoromethanesulfinate is a mouthful, and in most chemistry labs you’ll hear people call it “Langlois reagent.” Put simply, its chemical formula is CF3SO2Na. The “CF3” shows off the trifluoromethyl group, built around carbon and three fluorine atoms. “SO2” represents the sulfonyl group, a piece you’ll spot in a raft of industrial and pharmaceutical chemicals. The “Na” is sodium—a workhorse in the periodic table, always eager to form salts like this one.
How This Compound Makes a Difference
This sodium salt packs a lot of punch, especially for people working with organic molecules. It’s not as famous as sodium chloride, but chemists using it see a toolkit that opens doors to unique molecules. The Langlois reagent serves as a trifluoromethylation agent, meaning it delivers that CF3 group onto targeted molecules. The pharmaceutical industry depends on this. The trifluoromethyl group boosts drug activity, often making drugs more stable and effective in the human body.
Fluorine is rare in living things, but adding it to drugs can make a big difference: think longer-lasting medicine with fewer side effects. Drug makers reach for CF3SO2Na when the molecular architecture calls for this upgrade. I’ve spoken with colleagues in drug research who value reliable reagents; for them, getting clean yields from sodium trifluoromethanesulfinate saves time and money.
Access and Sustainability
Ease of access to this chemical has helped speed up medical research. Twenty years ago, chemists struggled with expensive or finicky reagents. The Langlois reagent, discovered in the 1980s and now widely available, sidesteps a lot of those hurdles. It’s handled as a stable white powder—a relief compared to many trifluoromethyl sources. I remember a professor once saying, “An easy-to-handle CF3 source makes my workday better.” That attitude still holds.
There’s a flip side. The global push on sustainable chemistry asks everyone to think about fluorinated compounds. Some fluorochemicals stick around in the water, soil, and people for decades. Even small quantities in pharma research can add up over the years, especially as industries worldwide ramp up their output.
Investing in Smart Choices
Green chemistry strategies keep gaining steam. Companies now look for ways to recover or recycle fluorinated byproducts. Labs develop new reactions that cut down or avoid harsh waste. Some universities train students to scan every procedure for hidden environmental costs. I’ve helped undergrads set up water filtration; we noticed how much less landfill-bound material we used once we planned smarter. Progress depends on people with good data and practical tools—this approach gives the best odds of building a future where chemistry grows with responsibility.
Trust and Quality Assurance
Scientists, including myself, double-check data on every new batch of sodium trifluoromethanesulfinate. Trust in results doesn’t come easily—you look for certificates of analysis, and peer-reviewed studies, and keep an eye on the supply chain. That’s not just accountability; it’s survival in tough research and stricter regulations. A paper in the Journal of Fluorine Chemistry in 2020 showed how the reagent streamlines late-stage functionalization, but only when handled by chemists who know their stuff.
Any chemical, even one with a solid track record, demands respect. The future belongs to researchers and companies blending curiosity with ethics: taking the time to know their reagents inside and out, including something as specialized as sodium trifluoromethanesulfinate.
How should Sodium Trifluoromethanesulfinate be stored?
Why Storage Matters for Sodium Trifluoromethanesulfinate
Most people think about chemicals as mysterious powders or clear liquids in a bottle tucked away on a shelf. Few realize that a decent chunk of lab accidents start with sloppy storage. Sodium trifluoromethanesulfinate isn’t something you’ll find in a grocery store. This stuff goes into specialty reactions, making unique additives and pharmaceutical ingredients. A careless approach can cost labs—sometimes even lives.
Real World Risks
Ask any chemist who’s worked with reagents like this: bad storage turns a helpful chemical into an expensive headache. Moisture creeps in, purity goes south, and soon nothing reacts how it should. Exposed to air and water for long, sodium trifluoromethanesulfinate can decompose or even give off sulfurous smells. Worse, improper storage can trigger hazardous fumes or fire risks, especially if contaminants touch the solid.
Smart Storage Isn’t Optional
Glass bottles with airtight seals give real peace of mind. Forget those cheap snap-caps from dollar stores. A quality cap stays tight, and the neck remains clean, so you don’t have to mop up leaks. Clear labeling—the product name, the date you opened it, hazard symbols—saves everyone’s time and maybe their skin if things go wrong. Based on my own time in a busy teaching lab, one misfiled, unlabeled bottle brought an entire class to a halt. Time spent labeling is always well invested.
Store it in a cool, dry cabinet—ideally, a chemical storage area with climate control. Humidity in most labs varies a lot through the year. If the room’s steamy, even a sealed bottle will eventually show signs of clumping or caking. Keeping desiccants in the storage container helps prolong shelf life, and solid silica gel pouches do the trick without making a mess.
Keep Separation from Danger Companions
One lesson every chemistry student learns: not all bottles play nice together. A basic compound like sodium trifluoromethanesulfinate stays more stable away from acids and oxidizers. Mixing fumes in a closed cabinet turns a benign storage area into an unpredictable reaction chamber. I’ve seen bottles sweat inside cabinets where peroxides, amines, or simple strong acids were stored nearby. Their vapors interact, corrosion starts, and lids seize up, making safe handling almost impossible.
Storing for Stewardship, Not Just Safety
Wasting specialty chemicals, particularly ones with complex synthesis routes, adds up fast—both financially and environmentally. Flushing degraded chemicals isn’t an answer. Responsible storage protects the bottom line and avoids unnecessary waste. Fact: about one-third of chemical disposal costs in university settings tie back to reagents improperly stored and rendered useless. Proper care on day one saves months of regulatory hassle down the road.
Modeling Solutions for Any Lab
Each new purchase offers a chance to double-check setup. Stackable, ventilated cabinets with built-in spill trays make a huge difference. Rotating stock: first in, first out. Look for a dry, dark place, out of direct sunlight, and never forget secondary containment in case a jar breaks. Quick spill response kits nearby save time and stress. If unfamiliar with the material, ask a senior colleague or refer to an updated safety data sheet. Responsible storage only takes a moment, but it sets the tone for safe science, every day.
Is Sodium Trifluoromethanesulfinate hazardous or toxic?
The Realities Behind the Chemical
Sodium trifluoromethanesulfinate has made its way into many chemistry labs, thanks to its use in organic synthesis and pharmaceuticals. Lab workers recognize this salt as the “Langlois reagent.” On paper, it looks harmless. Yet the story changes when you dig into the material safety data sheets and talk to people who’ve actually handled it.
Digging Into Toxicity Concerns
Information about its toxicity starts with the basics: substance exposure happens mostly through skin contact, inhalation, or swallowing. Some assume its sodium base makes it safer. The reality is that the trifluoromethyl group doesn’t always play nice with the body. Most manufacturers label the reagent as harmful if swallowed, irritating to the eyes and skin, and risky for the lungs if aerosols form. According to suppliers, irritation and redness follow direct contact. Inhaling the dust or vapor stings the respiratory tract and the eyes.
Scientific research hasn’t piled up mountains of chronic exposure data for sodium trifluoromethanesulfinate. Still, chemical relatives tend to disrupt cellular functions. Fluorinated compounds don’t break down in the body or the environment very easily. Researchers at the American Chemical Society have warned about the persistence of sulfinates and their downstream effects. Some fluorinated chemicals, when not handled right, stick around in the body longer than anyone hopes.
Environmental Impact and Workplace Reality
Disposal presents headaches for the environmentally-minded. Pouring sodium trifluoromethanesulfinate down the drain only spreads fluorinated chemicals further. Compounds with fluorine chains resist normal water treatment. Scientists spotlighted the risk of introducing new persistent organic pollutants this way. Responsible disposal requires incineration at specialized facilities. Not every lab or workshop has those connections, so safe storage and less frequent use remain better options.
From experience, even small spills cause trouble. White powder whips up into the air easily and coats work surfaces. Gloves and goggles do more than meet regulations. They become an everyday necessity for anyone who handles a bottle. Anyone who’s brushed off a chemical spill on bare skin remembers the stinging sensation and rushing to the sink. Fume hoods cut down the risk, but a surprising number of folks skip that step due to over-familiarity or broken equipment. That increases the chance somebody breathes in the dust or drags it home on their clothes.
Diving Into Safer Habits and Alternatives
Health and safety folks argue in favor of closed system transfers and pre-weighed reagent packets, since weighing loose powder can release small clouds. Training new chemists in good habits helps, too. Some universities now require students to log each use and follow a checklist for personal safety gear and ventilation. The move toward alternatives comes up in academic conferences, with green chemistry taking center stage. Many labs experiment with less persistent sulfonates or completely different fluoroalkyl sources. Each has its own trade-offs, but the conversation shows people care about health and long-term safety.
Everyone in chemical research knows the stress from juggling deadlines, experiments, and health concerns. Nobody wins if a split-second mistake turns into a hospital trip. Being honest about chemical risks builds actual trust in workplaces, and drives home the need for protective gear and safe handling routines. Sodium trifluoromethanesulfinate deserves respect, not panic. Safe habits, real information, and community accountability turn costly mistakes into rare events.
What is the purity and typical packaging size of Sodium Trifluoromethanesulfinate?
Real-World Labs and Industry Standards
Anyone who steps into a synthetic chemistry lab knows the scramble for reliable reagents gets real fast. Sodium trifluoromethanesulfinate shows up most often as a white, solid powder, usually positioned near reagents crucial for trifluoromethylation reactions. Researchers and chemical suppliers recognize the difference a few points of purity can make when reactions get picky or when scale increases beyond bench work.
Why Purity Matters
Purity directly shapes reaction outcomes. Chemists working with sodium trifluoromethanesulfinate usually seek grades about 98% or better. Numbers dip lower and that crisp white powder can end up tinged or speckled—sometimes even sticky with unhelpful moisture. These impurities may not sound like a big deal in theory, but the real cost shows up during workup or analysis. The smallest unknown byproduct can ruin yields or force extra hours in cleanup.
Quality control data from recognized reference labs reinforce these standards. Trace metal content, moisture, and residual solvents—the regular culprits—matter because downstream chemistry leans heavily on every reagent living up to its label. When you see 98% or above, that batch passed HPLC, NMR, and ICP-OES checks—not just an easy glance at color or granularity.
Accepted Packaging Sizes in Practice
Small-scale labs don’t want five kilos hogging shelf space. Suppliers understand that half the customers want just enough for a handful of runs. Typical packaging ranges from as small as 5 grams up to 100 grams for research needs. Packaging at these scales helps reduce waste and makes hazardous goods handling less of a headache for staff without industrial-grade storage.
For production environments, sodium trifluoromethanesulfinate often arrives in 500-gram to one-kilogram containers, sometimes even five-kilo drums for big-volume manufacturing. Each package comes with its own challenges—smaller ones mean less risk of contamination, but pricier per gram. Larger sizes cut cost but demand careful handling. I’ve seen instances where a badly sealed bag led to water clumping the entire batch, turning a few thousand dollars into landfill. So, effective packaging means sealing out moisture, often through double bagging and desiccant packs.
Safe Handling: More Than Labels
Any chemist worth their salt checks for the smallest hint of past mishandling. Moisture, visible caking, or funky odors often hint at poor storage, which cuts into actual purity—regardless of what the label claims. I always check packaging integrity before opening, and some colleagues use secondary containers during storage just to keep humidity at bay.
Building on Experience and Trust
Trust builds through supplier transparency. Certificates of analysis, clear lot numbers, and third-party test results all become routine safeguards, not extras. It’s the difference between reliable, repeatable chemistry and a project stalling over trace impurities. Labs working under strict regulatory frameworks—think pharma or regulated agrochem—never let a batch in without exhaustive review over both purity and packaging logs.
Improving Industry Practices
Moving forward, the chemical industry can help by offering more detailed data sheets, including recent moisture content and clear insight into storage conditions. More communication between suppliers and end-users protects both investments and research. Nobody wants to repeat a week of experiments due to a batch that picked up water in transit.
Conversations between buyers and suppliers raise standards across the supply chain. Better packaging seals, clear labeling, and responsive support don’t just protect product quality—they build the trust and reliability that science and manufacturing rely on every day.
| Names | |
| Preferred IUPAC name | sodium trifluoromethanesulfinate |
| Other names |
Trifluoromethanesulfinic acid sodium salt Sodium triflinate CF3SO2Na Sodium (trifluoromethylsulfinyl)ide |
| Pronunciation | /ˌsoʊdiəm traɪˌflʊəroʊˌmɛθeɪnˈsʌlfɪneɪt/ |
| Identifiers | |
| CAS Number | 2926-29-6 |
| 3D model (JSmol) | `3D Structure; JSmol; [Na+].C(F)(F)(F)S(=O)[O-]` |
| Beilstein Reference | 3582229 |
| ChEBI | CHEBI:39098 |
| ChEMBL | CHEMBL1375647 |
| ChemSpider | 61354 |
| DrugBank | DB14580 |
| ECHA InfoCard | 03b0e5f5-1be0-4b1e-90c7-8e1bc105b956 |
| EC Number | 206-201-1 |
| Gmelin Reference | 122111 |
| KEGG | C14415 |
| MeSH | D017616 |
| PubChem CID | 2734768 |
| RTECS number | YW8560000 |
| UNII | N3Q8ERO2GR |
| UN number | UN3261 |
| CompTox Dashboard (EPA) | DTXSID7036796 |
| Properties | |
| Chemical formula | CF3SO2Na |
| Molar mass | 172.06 g/mol |
| Appearance | White to off-white crystalline powder |
| Odor | Odorless |
| Density | 1.57 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -4.3 |
| Acidity (pKa) | 4.89 |
| Basicity (pKb) | 7.2 |
| Magnetic susceptibility (χ) | -33.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.377 |
| Dipole moment | 2.17 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 207.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -978.54 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1208.8 kJ mol⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H319 |
| Precautionary statements | P261, P264, P271, P273, P280, P302+P352, P305+P351+P338, P312, P337+P313, P362+P364, P501 |
| Lethal dose or concentration | LD50 oral rat 1750 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 >2000 mg/kg |
| NIOSH | TT4550000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | Not established |
| Related compounds | |
| Related compounds |
Trifluoromethanesulfonic acid Trifluoromethanesulfonic anhydride Trifluoromethanesulfonamide Sodium methanesulfinate Potassium trifluoromethanesulfinate |

