Sodium Thiosulfate: Looking Beyond the Lab
Historical Development
Curiosity often drives discovery, and in the nineteenth century, scientists in Europe began playing with compounds like sodium thiosulfate. The earliest records date back to the 1800s, when Jean-Baptiste Boussingault isolated it from the soda used in textile bleaching. Word spread fast. Photographers picked it up thanks to its knack for dissolving silver salts, and tanners saw value in its ability to process animal hides. These first steps set the tone for a chemical that would keep showing up in new places as research moved forward. It’s fascinating to watch a substance move from curiosity on a lab bench to something touching countless industries — proof that observation and tenacity keep the wheels of progress turning.
Product Overview
Sodium thiosulfate, commonly called hypo or sodium hyposulfite in some trades, shows up as a colorless crystal or granular solid. What strikes me is its versatility: it plays a role in photography, medical treatments, water purification, and dechlorination processes. In the world of chemicals, few can shift hats so easily. Vendors now offer several grades, from technical and photographic to analytical and pharmaceutical. Grades differ in purity and elemental contaminants, so a batch destined for clinical use faces stricter scrutiny than one for an industrial tank. Consumers and operators often care about more than purity—they look at things like shelf life, solubility, and compatibility with common additives, too.
Physical & Chemical Properties
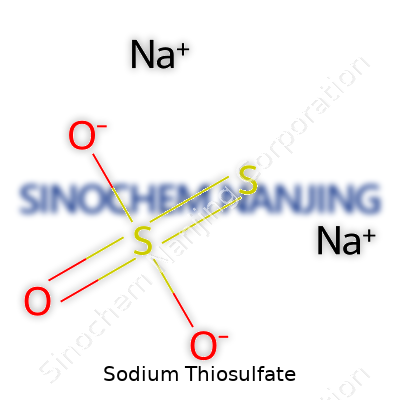
At room temperature, sodium thiosulfate looks unremarkable: transparent, odorless crystals that dissolve readily in water, stubborn in alcohol. Solubility in water sits above 60g per 100ml at 20°C, which helps with bulk handling in industry. Its chemical formula, Na2S2O3·5H2O, looks simple, but clever chemistry lurks here. This pentahydrate form means it stores a surprising amount of water, and that affects stability and storage. The ions released give it properties prized by so many sectors—the thiosulfate works as a reducing agent and a ligand, snatching up free radicals and metals. On heating, it breaks down to sulfite and sulfur dioxide, which explains why some industries avoid exposing it to high temperatures.
Technical Specifications & Labeling
Technical data forms the backbone of choosing the right product for any job. Typical assay for sodium thiosulfate pentahydrate lands around 99%, with chloride content tightly controlled at less than 0.01%. Lead, arsenic, and heavy metals must fall well below regulatory thresholds—end users expect suppliers to run precise quality checks. Routine labels list batch number, grade, purity, molecular weight (248.18 g/mol for the common pentahydrate), storage instructions, date of manufacture, and hazard statements, as required by GHS standards. Exact wording helps keep mistakes at bay during use and handling. Hospitals, labs, and plants rely on these details to stay compliant and reduce incident risk.
Preparation Method
Getting sodium thiosulfate usually starts with sodium sulfite and sulfur. Industrial production often involves reacting sodium sulfite with sulfur at warm temperatures in water, then cooling and crystallizing the pentahydrate. Some plants recycle byproducts from sulfur refining, turning waste into usable material—a win for resource-conscious operations. Experience tells me that the choice of raw materials and the purity of inputs shape the reliability of the final product. Even small contamination at this step can ripple through to downstream applications, so strict attention at every stage pays off. Filtration and washing remove lingering impurities, before evaporation produces the patient, sparkling crystals shipped worldwide.
Chemical Reactions & Modifications
Sodium thiosulfate likes to get involved in redox reactions. In iodometry, it reacts predictably with iodine, reducing it to iodide and forming tetrathionate. This lets chemists use it as a volumetric reagent for measuring oxidizers. It’s this simple exchange that guarantees its spot in schools and big labs alike. Thiosulfate also forms stable complexes with silver, which explains its starring role in developing photographic film. Modifications sometimes add co-solvents or stabilizers for niche processes, but the core chemistry stays robust. Compatibility with other sulfates and halides opens paths for custom blends, and its reactivity with acids (producing sulfur and sulfur dioxide) shapes storage rules and day-to-day handling in pharmaceuticals and water treatment.
Synonyms & Product Names
The name sodium thiosulfate shows up in official documents, but “hypo,” “sodium hyposulfite,” and even quirky names like “fixer” in photography circles turn up in trade or conversation. Some markets list it under “EC No. 231-867-5” or “CAS No. 7772-98-7.” Off-label uses in water treatment might call it “chlorine neutralizer.” This variety can lead to confusion, especially for newcomers in procurement. Keeping an eye on synonyms across regions and suppliers helps keep ordering and regulatory paperwork tidy.
Safety & Operational Standards
Handling sodium thiosulfate doesn’t demand the same care as more reactive chemicals, but unintended mix-ups can trigger accidents. Long exposure to dust can irritate eyes and skin. Training on proper storage—dry, away from acids—forms part of every chemical handler’s standard induction. Material safety data sheets (MSDS) explain these dangers clearly. In emergencies, eating or inhaling large amounts needs medical attention, though severe effects remain rare. Facilities working with large volumes follow OSHA, EU REACH, and CLP guidelines, including emergency eye washes, respirators for dusty jobs, and clear spill management. At smaller scales, I’ve seen operators underestimate volume, so safety culture beats written rules.
Application Area
Few chemicals bridge so many professions. Water utilities use sodium thiosulfate for chlorine neutralization, safeguarding aquatic life in wastewater discharges. Film and printing experts rely on it for fixing black-and-white photographs. In labs, it carries out redox titrations and measures active chlorine. Emergency rooms stock it for cyanide poisoning, as it accelerates the body’s natural detox mechanism, improving survival odds. This seemingly unremarkable crystal even shows up in the gold extraction process, offering an alternative to more toxic options. Every industry finds a new clever use for its reactivity, supporting safety, accuracy, and efficiency across the board.
Research & Development
Research on sodium thiosulfate covers everything from fine-tuning yields in synthesis to exploring new medical uses. In oncology, teams test it as a side-effect reducer during chemotherapy, banking on its knack for neutralizing free radicals. Water technologists evaluate how it interacts with modern purification blends, looking to optimize cost and performance. Environmental chemists run trials on its behavior in ecosystems, making sure large-scale field application doesn’t accidentally upset balance. The thirst for safer, greener processes means labs stay busy investigating reuse of byproducts and possible biodegradability enhancements. No matter how much is known, more careful trials, peer review, and publications keep flowing. This ensures that each claim builds a little trust, step by step.
Toxicity Research
Acute toxicity of sodium thiosulfate ranks low compared to many industrial chemicals. Still, chronic exposure and ecological impact attract careful scrutiny. Researchers track breakdown products in soil and water, ensuring none linger longer than intended. Clinical trials look closely at dosing in medical use, aiming to avoid unintended effects during cyanide poisoning therapy. Independent studies test the risks of long-term ingestion, especially in water treatments where residual amounts may persist. Animal testing, epidemiological surveys, and regulatory assessments together help draw a full map. These efforts make its benefits safer to share with a bigger community.
Future Prospects
Demand for multi-purpose, lower-toxicity chemicals grows yearly, and sodium thiosulfate stands well-placed to take a bigger role. The push toward greener mining and pollution control favors its adoption over older, harsher reagents. Researchers put energy into expanding its use as an antidote in medical emergencies, and environmental audits promise wider regulatory acceptance. If better formulations or blending techniques emerge, even more sectors could benefit from reduced chemical footprints and simpler regulatory compliance. What comes next likely depends on success in scaling up sustainable, safe production and expanding clinical trials for new therapies. This is shaping up to be a chemical not just with a past, but a future worth paying attention to.
What is Sodium Thiosulfate used for?
A Pharmacy Staple With Unexpected Uses
Sodium thiosulfate doesn’t get a lot of time in the spotlight, but it keeps showing up in places where it matters most. I first came across it during college labs, always in a plain bottle, sitting near the chemicals for titration. Every pharmacy shelf that stocks antidotes for rare emergencies will include it. But its job goes way beyond dusty storerooms and emergency kits.
Tackling Cyanide Poisoning
Nobody wants to imagine a cyanide exposure. Industrial accidents, smoke inhalation during house fires, or even certain old pest control methods can lead to this scary scenario. Medical teams reach for sodium thiosulfate—not because it’s flashy, but because it saves lives. It helps neutralize cyanide so the body can flush it out. EMTs and doctors worldwide have depended on it for decades. The U.S. National Library of Medicine states sodium thiosulfate works especially well combined with sodium nitrite for treating cyanide poisoning.
Purifying Water
From chemistry sets to aquariums, sodium thiosulfate makes a difference. I’ve seen aquarium hobbyists grab it off pet store shelves to remove chlorine and chloramine from tap water. Both chlorine and chloramine can cause fish stress or even kill them. Sodium thiosulfate makes the water safe, right before new fish get introduced to their tank homes. It's a favorite among folks who want safe, healthy fish, and aquarists swear by it.
Medical Treatments Beyond Poisoning
Doctors also prescribe sodium thiosulfate for calciphylaxis, a painful condition that sometimes hits people with chronic kidney disease. This condition causes calcium to build up in small blood vessels, cutting off blood supply to skin or fat tissue and causing severe wounds. The drug helps dissolve those calcium deposits, according to studies published in the Clinical Journal of the American Society of Nephrology. People dealing with calciphylaxis face long and difficult treatments, but sodium thiosulfate gives them an option when others fail.
Stopping Damage From Chemotherapy
Children treated with chemotherapy sometimes suffer from hearing loss caused by a drug called cisplatin. Hospitals use sodium thiosulfate to protect young patients’ hearing. Research funded by the National Cancer Institute reports a clear reduction in hearing loss risk. Giving these kids the chance to recover from cancer and still enjoy music and conversations afterwards matters just as much as the cancer treatment itself.
In the Lab and Industry
Chemistry classrooms lean on sodium thiosulfate for all sorts of projects: titration experiments, photographic processing, even removing iodine stains from hands. Silver halide photography almost disappeared with smartphones, but black-and-white photographers respect its history. Industrial workers use it in paper bleaching and gold mining, but the biggest impacts start with health and water safety, not profit margins.
Looking Forward
Sodium thiosulfate proves its worth every time an emergency team stops cyanide poisoning, or a young cancer survivor walks out of a hospital with intact hearing. Clean water at home, healthy aquariums, and even advanced kidney treatments—all trace back to this simple salt. If more medical centers and communities understood its ready availability and broad uses, more lives and livelihoods would see a real benefit.
Is Sodium Thiosulfate safe for human use?
Looking Closer at Sodium Thiosulfate
Sodium thiosulfate doesn’t grab headlines like new medications or miracle supplements, yet it’s popped up in conversations about everything from medicine to water treatment. Many people only hear about it in high school chemistry or during talk of heavy metal poisoning. Still, it’s slipped into clinician debates and internet health forums because some tout its ability to “detox” the body or treat specific medical problems. So, is it really safe? And what do people need to know before they jump on new health trends?
How Doctors Actually Use It
Sodium thiosulfate has a history in medicine that goes back over a century. In hospitals, doctors give it to patients after cyanide poisoning or for rare conditions like calciphylaxis, which can strike folks with kidney failure. The FDA recognizes its use in these situations, and hospitals keep vials on hand. Safety data comes from research on these controlled uses — most patients get the drug through IV, under a doctor’s direct care. At these doses, side effects like nausea or headache show up, but serious problems are rare. The American Society of Health-System Pharmacists lists sodium thiosulfate as relatively safe for approved uses, especially for short-term treatment.
In cancer care, sodium thiosulfate reduces damage to hearing that sometimes follows high-dose chemotherapy in children. The National Cancer Institute sponsored studies for this, so safety and effectiveness got careful attention. That’s more like the gold standard for medication research than internet word-of-mouth.
Safety Outside the Hospital Walls
As more people seek ways to “cleanse” their bodies or try alternative health routines, some companies market sodium thiosulfate as a supplement. This brings fresh risks. The U.S. Food and Drug Administration has not approved it for general detox use or any over-the-counter claims. Taking it by mouth, outside medical guidance, opens the door to overdose, allergic reactions, or stomach issues — all without medical benefits backed by research.
The Cleveland Clinic and Mayo Clinic both highlight that sodium thiosulfate’s safety isn’t assured for self-experimentation. Side effects like vomiting, low blood pressure, or even severe reactions can hit people who don’t realize they have allergies or pre-existing medical conditions. Besides, “cleansing” myths don’t match up with biology. The liver and kidneys filter natural toxins every day, no need for drastic interventions.
Staying Safe: What People Can Do
Many drugs, including sodium thiosulfate, play important roles in hospitals under doctor supervision. These medicines save lives in emergencies or help patients with rare diseases. Problems arise when people start self-medicating based on internet trends or non-medical advice. Google searches often put unfiltered content in front of desperate or curious people. Anything that claims to be a universal cure or detox tool deserves a double check. The National Institutes of Health recommends using medicines only for their approved purposes, unless in the care of an expert who can explain side effects and potential dangers.
Anyone thinking of using sodium thiosulfate for unapproved uses would do well to ask their doctor. The urge to try new supplements is strong — but science sticks to safety, and that calls for real medical knowledge, clinical research, and honest facts. Watching people I care about flirt with novel health fixes reminds me that simple questions about safety demand clear, experienced answers, not promises from untested fads.
How should Sodium Thiosulfate be stored?
Handling a Sensitive Chemical
Sodium thiosulfate isn’t a household name, but it pops up in all kinds of lab work. Hospitals use it as an antidote for cyanide poisoning. Photographers used to rely on it in darkrooms. Water treatment facilities lean on it to neutralize chlorine. So, knowing how to store this chemical really matters—not just for safety, but for making sure it actually works when called upon.
Moisture: The Biggest Enemy
Sodium thiosulfate comes as a clear crystal or powder, and moisture is its sworn enemy. Once exposed to humidity, it cakes up, forms lumps, and loses effectiveness. I’ve seen a jar stored near a sink turn solid as a rock, and it ended up in the trash. Dry, airtight containers are non-negotiable here. If there’s any doubt in the seal, throw in a pack of desiccant. A small investment saves headaches down the line.
Keep the Temperature Steady
Room temperature suits this material just fine. Extreme heat will break it down, leaving nothing but chemical dust. Cold, damp storage leads right back to the moisture problems. After a summer heatwave, a colleague once found degraded sodium thiosulfate that wouldn’t dissolve completely during titration. Reliable temperature—think 15-25°C—keeps things predictable. That’s straight from the Sigma-Aldrich datasheets and my own experience working with reagents on university campuses.
Avoid the Sunlight
Direct sunlight throws a wrench into the works. It speeds up oxidation, which can put a batch well past its prime. Laboratories usually have their storage shelved in the back, away from windows, for good reason. In one amateur experiment, a student left a container on a windowsill. The product turned yellow, signaling breakdown. Even for short-term storage, tuck it away from the sun's reach.
Keep Away From Acids and Oxidizers
Mixing sodium thiosulfate with acids kicks up sulfur dioxide gas, which stings the nose and shouldn’t exist outside a fume hood. Store it apart from acids and strong oxidizing agents. In shared spaces, clear labeling and separation go a long way. Accidental mixing, especially in a cluttered storage area, leads to dangerous surprises. I've seen mismatched shelving in badly organized spaces, and one careless moment led to a small spill and a big panic over toxic fumes. Segregated shelves and unmistakable containers prevent mix-ups.
Practical Steps For Safety
Security starts with labeling. Every bottle should spell out “Sodium Thiosulfate” with clear hazard symbols. Emergencies get a lot messier if someone grabs the wrong container. Personal protective gear—gloves, goggles, lab coat—keeps users safe during handling, just in case the unexpected happens. Small spills come up fast with water, but for bigger messes, call for help and follow your institution’s chemical protocol.
Regulations vary, but OSHA calls for “cool, dry, well-ventilated storage, away from incompatible substances.” That matches real-world practice in the labs where I’ve worked. Double-check for leaks in containers before putting anything away, and record how much is on hand. The little stuff—like jotting down the received date—helps prevent using old, unreliable product.
Responsibility Comes Standard
Chemicals don’t forgive laziness. A little care with sodium thiosulfate goes a long way—protecting people, budgets, and experiments. There’s never been a shortcut for good habits, and skipping them isn’t worth the cost.
What are the side effects of Sodium Thiosulfate?
Understanding Sodium Thiosulfate’s Role
Sodium thiosulfate has helped a lot of people battling cyanide poisoning and some certain chemotherapy side effects. Doctors sometimes use it to treat calciphylaxis, a rare and painful complication that strikes patients with kidney disease. This isn’t some supplement anyone finds at a health food store. When doctors put it in a treatment plan, it’s usually because other options don’t work or the situation’s urgent.
What People Have Experienced
Sodium thiosulfate does a number on the body in both good and not-so-great ways. Some patients get relief from tough medical issues. Many others feel the flipside: nausea, vomiting, headaches, or even low blood pressure. These are more than side notes. Feeling dizzy, weak, or nauseous can mess up a person’s day and make recovery slower.
Some folks run into problems like pain at the injection site — burning, swelling, redness. A burning feeling in the veins makes people think twice about coming back. Those who already wrestle with heart or kidney issues sometimes slide into trouble with fluid overload. This means puffy legs, sudden weight gain, and a hard time breathing. Doctors have flagged this because it tends to spark trips to the emergency room, especially for older patients or those on dialysis.
Rare and Serious Reactions
Most people do fine with careful dosing. Things turn serious quickly if sodium thiosulfate triggers an allergic reaction. Shortness of breath, rash, or a pounding heart signal the body’s not handling it. On the lab end, this medicine sometimes triggers metabolic acidosis, where blood gets too acidic. For kids or those who are fragile already, this can be a real threat.
If someone already deals with low calcium, problems get worse. Calcium drops even further with sodium thiosulfate. Seizures, muscle cramps, tingling, or spasms get much harder to control if doctors don’t spot the change early. The whole experience often leaves people drained, tired, and confused, especially when they don’t know what to expect.
Why Side Effects Get Overlooked
Hospital routines move fast, and a medicine like sodium thiosulfate can seem like just another IV bag. Sometimes, side effects get brushed off, blamed on “the illness” instead of the medicine. I’ve seen patients in clinics struggling to describe what they feel after an infusion because they weren’t told what might come up. Families start searching for answers only when their loved one’s condition changes overnight. It leaves everyone feeling frustrated, doctors included.
Better Pathways Forward
People do much better when they know what’s coming and can flag problems early. Nurses and pharmacists can walk patients through honest, practical warnings. Having clear checklists posted near patient beds helps everyone ask the right questions. Blood tests need to stay up-to-date, watching for acidosis or falling calcium long before symptoms show. Simple routines—asking about headaches, checking for swelling, tracking blood pressure—keep reactions from getting out of hand.
Doctors carry a tough job deciding who needs a powerful drug like sodium thiosulfate. No one should walk into those treatments guessing about the risks. Listening, regular follow-up, and realistic information turn a scary process into something people can handle. It’s not about scaring folks, just giving them the awareness to call for help the minute things feel off.
Can Sodium Thiosulfate be used to treat cyanide poisoning?
Every Second Counts in Cyanide Poisoning
Anyone who’s worked near industrial sites or heard stories from mining communities knows cyanide brings danger. Accidents involving this substance land people in the emergency room with little time on the clock. Cyanide starves cells by shutting down their oxygen use, leading to collapse within minutes. People living or working near fire sites or chemical plants face risk, too. For these folks, lifesaving knowledge means knowing which antidotes ER doctors reach for.
Sodium Thiosulfate: A Foundation in Cyanide Antidotes
Doctors have turned to sodium thiosulfate for decades in poisoning emergencies. It works by giving the body’s own enzymes more to work with, helping turn deadly cyanide into less harmful thiocyanate. The liver then sweeps this aside, disarming the threat. Hospitals keep sodium thiosulfate ready because of this chemical teamwork, and old-school medics have plenty of stories where it tipped the odds for people caught in desperate situations.
Why Doctors Often Pair It With Other Treatments
Sodium thiosulfate never works alone in top emergency rooms. Fast-acting antidotes like hydroxocobalamin (a form of vitamin B12) or sodium nitrite enter the bloodstream and jumpstart rescue, attacking the poison straight away. Hydroxocobalamin grabs cyanide, traps it, and leaves the kidneys to do the rest. This combo, used together, covers different angles and boosts survival. In smaller clinics, or when supplies run short, sodium thiosulfate sometimes acts as the main player, though it's used with careful monitoring.
Safe Use Comes With Experience
Even though sodium thiosulfate has a long track record, no drug comes without warning labels. High doses can shift electrolyte levels, lead to low blood pressure, or rarely spark allergic reactions. In poison control centers, staff balance the lifesaving impact against side effects, always choosing the lesser evil. Folk wisdom and official medical training both teach that slow and steady IV drips prevent most trouble.
Access and Awareness: Biggest Challenges
Hospitals in big cities often keep full antidote kits within reach. Rural clinics sometimes rely on sodium thiosulfate because it’s more affordable or already sitting on dusty pharmacy shelves. Emergency crews need regular training on when to use antidotes and how to spot poisoning early. Sometimes, language barriers or fear of medical bills keep victims from getting help until it’s too late. Public campaigns and clear protocols make treatment smoother for all.
Looking for a Safer Future
Newer research experiments with faster-acting drugs and easier-to-carry treatments. For now, sodium thiosulfate holds its ground in many emergency rooms, thanks to history, low cost, and solid results. Some projects work on making antidotes available in places people need them most, pushing for more education around poisoning risks in schools and workplaces. These efforts matter, because when cyanide hits, every bit of preparedness translates into lives saved.
Closing the Knowledge Gap
I remember a medical conference where a trauma surgeon shared a hard story of a factory worker exposed to cyanide. Quick action, including sodium thiosulfate, turned tragedy into survival. Stories like that underscore why knowing how antidotes work goes far beyond chemistry—sometimes it’s the difference between life and death.
| Names | |
| Preferred IUPAC name | Sodium thiosulfate |
| Other names |
Sodium hyposulfite Thiosulfuric acid, disodium salt Disodium thiosulfate |
| Pronunciation | /ˌsoʊdiəm θaɪˈɒsʌlfeɪt/ |
| Identifiers | |
| CAS Number | 7772-98-7 |
| Beilstein Reference | 425332 |
| ChEBI | CHEBI:32145 |
| ChEMBL | CHEMBL1366 |
| ChemSpider | 21510 |
| DrugBank | DB00765 |
| ECHA InfoCard | 100.111.799 |
| EC Number | 231-867-5 |
| Gmelin Reference | 778 |
| KEGG | C01814 |
| MeSH | D013019 |
| PubChem CID | 24842 |
| RTECS number | WS4900000 |
| UNII | O4V940HO96 |
| UN number | UN3077 |
| Properties | |
| Chemical formula | Na2S2O3 |
| Molar mass | 158.11 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.667 g/cm³ |
| Solubility in water | Soluble |
| log P | -4.5 |
| Vapor pressure | Negligible |
| Acidity (pKa) | ~11 |
| Basicity (pKb) | 6.63 |
| Magnetic susceptibility (χ) | −76.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.333 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 241.8 J·K⁻¹·mol⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -635.1 kJ/mol |
| Pharmacology | |
| ATC code | V03AB02 |
| Hazards | |
| Main hazards | Not a hazardous substance or mixture according to Regulation (EC) No. 1272/2008. |
| GHS labelling | GHS07 |
| Pictograms | GHS07,GHS09 |
| Signal word | No signal word |
| Hazard statements | May cause eye irritation. |
| Precautionary statements | P264, P270, P301+P312, P330 |
| Lethal dose or concentration | LD50 Oral (Rat): > 5,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral, rat: 7,000 mg/kg |
| NIOSH | WI6725000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 15 to 30 g in 300 ml water as a 10% solution |
| IDLH (Immediate danger) | Not listed |
| Related compounds | |
| Related compounds |
Sodium sulfate Sodium sulfite Sodium dithionite Potassium thiosulfate Calcium thiosulfate Sodium tetrathionate Sulfur dioxide |

