The Landscape of Sodium Tellurite: Past, Present, and What Lies Ahead
Tracing The Steps: Historical Development
Looking back, sodium tellurite emerged during an age when chemists pushed past familiar acids and metals, digging deep into the periodic table’s mysteries. Tellurium itself got its scientific debut in the late 1700s, but solid advances on its compounds didn’t take off until heavy industrialization and the rise of lab analysis. Chemists discovered that mixing tellurium with sodium hydroxide left a white crystalline solid, which went on to earn a spot in labs and later, industry. Early researchers used sodium tellurite to study bacterial respiration, spotting its value as a selective agent way before antibiotics became everyday tools. With the steel and alloys sector booming in the 20th century, scientists added sodium tellurite as a finicky but effective oxidizer. Each discovery built on the steps before, carving a scientific and practical path for this once-obscure chemical salt.
Getting To Know Sodium Tellurite: Product Overview
Sodium tellurite stands out as a white or colorless crystalline powder that dissolves quickly in water. Its chemical formula, Na2TeO3, hints at its two sodium atoms clinging to a tellurite ion. Most suppliers market it in sealed plastic drums or glass jars, aware that trace water can set off slow changes in the salt. In the lab, technicians favor it for its purity and reliability; a batch that sits exposed can darken or even turn yellow if left out, hinting that the elements within stay restless. While less discussed at dinner tables, sodium tellurite holds real value in microbiology, metallurgy, and even specialty electronics. Blindly tossing it into a process rarely ends well — only those who respect its chemistry and quirks get consistent results.
Diving Into Details: Physical & Chemical Properties
Sodium tellurite comes as a fine, white powder with a faint salty-metallic smell, clocking a molecular weight near 221.6 g/mol. It melts just over 500°C, making it tough enough for most thermal work below those temperatures. Water welcomes it easily, dissolving the powder within seconds or a few minutes if the mix runs cold. Acids knock sodium ions away, leaving behind tellurium dioxide, a chalky precipitate that’s stubborn to dissolve. Chemists watch for its oxidizing punch, especially around organic matter, since careless mixing can cause rapid color changes or even toxic fumes. Tossing it next to reducing agents like sulfites or certain sugars brings out tellurium metal or black powders — proof the compound changes its stripes in reactive company. At room temperature, sodium tellurite stays stable in sealed vessels, but air and light chip away at its structure over weeks or months.
Technical Specs & Labeling In Real-World Use
Most bottles and drums list a purity between 98–99.5%, marked with batch numbers and country of origin. Labels flag the CAS number (10102-20-2), remind users to store it away from acids, and caution against direct contact with skin or eyes. More recently, regulatory rules demanded extra pictograms, so today’s containers often show hazard diamonds that make the warnings clear even in a crowded storeroom. Industry specs now require minimal heavy metal contamination and controlled sodium carbonate content, so end-users in electronics or pharmaceuticals get predictable results. Purchasing teams double-check certificates of analysis to weed out suppliers cutting corners or ignoring best practices in production and packaging. Storage rooms built on good ventilation and cool temps handle sodium tellurite just fine, but skipped inspections lead to spoiled, off-color batches and burdensome waste problems.
Walking Through The Steps: Preparation Method
Factories turn out sodium tellurite by mixing tellurium dioxide with sodium carbonate or sodium hydroxide under heated water. The process needs patience and good temperature control, since boiling too hard or skimping on stirring causes low yields and murky solutions. Once the solid fully dissolves, technicians cool the liquid until white crystals fall out, then use filtration and drying ovens to get a clean, usable powder. Scale-up means bigger tanks, better filters, and more automated controls, but even small mistakes — like letting a tank cool too slowly or missing a batch’s end-point — slash the quality. The method hasn’t changed much since last century, but process control, purity, and real-time monitoring reached new heights thanks to modern analytics. Some labs mix up sodium tellurite fresh for each experiment, claiming the product works better off the line than after weeks of shelf time.
Chemical Behavior, Reactions & Modifications
Anyone blending sodium tellurite with acids will spot bubbling and a sharp solidification as tellurium dioxide pops out of solution. Reducing environments, such as those with hydroquinone or sulfite, coax the compound into producing elemental tellurium, forming dark or gray flakes. Sodium tellurite doesn’t stand well next to strong oxidizers; mixing with peroxides or chlorates risks vigorous reactions that turn safe handling hazardous. In specially-designed syntheses, scientists convert sodium tellurite to organotellurium compounds, prized for their use in electronics or as reagents in medical research. Every shift in bond structure hands new reactivity and risk, so chemists develop tight protocols and triple-check chemical compatibility. Some advanced labs modify sodium tellurite by doping with select metals, hoping to boost selectivity or reduce waste during catalytic processes.
What’s In A Name?: Synonyms & Alternate Products
Across documents and catalogs, sodium tellurite may appear as sodium tellurium trioxide, disodium tellurite, or just plain “tellurite.” European suppliers sometimes call it natrium telluricum, while older literature prefers the less specific “tellurous sodium salt.” Workers in the steel industry lump it with oxidizing additives, missing the full subtlety chemists assign it. Some suppliers stock potassium tellurite as an alternate, but the sodium form leads in value and range of uses due to its cost and solubility. Accurate labeling cuts confusion and keeps hazardous mix-ups off the table, especially around similar-looking sodium selenite.
Standards For Safety & Operations
Sodium tellurite doesn’t take shortcuts on safety. Regulations in the U.S. and EU describe it as a hazardous substance needing careful glove, mask, and eye protection. Direct contact leads to skin rashes or eye irritation, while inhaling dust may rattle the lungs. Most companies mandate enclosed weighing stations for dry powders and splash shields for solution prep. Spill kits designed for oxidizing agents line the walls of labs and factories wherever this compound is at play. Waste sodium tellurite can’t hit the drain; disposal contractors and neutralization steps help keep tellurium out of rivers and landfills. Experienced teams run frequent drills, knowing that cutting corners on personal protective gear or ignoring fume hoods spells trouble fast.
Where Sodium Tellurite Lands: Application Area
Microbiologists put sodium tellurite to work as a selective agent in culture media, since certain bacteria handle tellurite toxicity better than others — an edge in identifying dangerous pathogens like Corynebacterium diphtheriae. Metal processing plants toss in sodium tellurite to adjust oxidation states or tweak the properties of specialty alloys. In the past twenty years, its value crept into electronics and photovoltaics, supporting lead-free solder, semiconductors, and sensor tech. Only a handful of compounds match its oddball balance of reactivity and selectivity in these arenas. Niche users in pigment and glass manufacturing claim sodium tellurite helps fix color and refractive properties where old-school chemistry falls flat. Its place grows with each technical leap, but costs and safety hold its spread in check.
Fueling Progress: Research & Development
Research teams keep sodium tellurite under their microscopes, exploring new oxide materials, battery tech, and antimicrobial agents. Labs run fresh trials in organic synthesis, chasing molecules that bristle with tellurium atoms for improved electronics or potential cancer treatments. Collaborative projects link universities and specialty manufacturers, working to turn sodium tellurite’s risk profile into an asset through safer forms or new delivery systems. Some innovations target green chemistry, seeking milder synthesis conditions and less toxic byproducts. Open-access journals highlight steady progress on the toxicology, environmental fate, and break-down mechanisms for sodium tellurite, filling decades-old gaps and shaping regulatory guidance.
Facing The Risks: Toxicity Research
Studies continue to show sodium tellurite’s power to harm at modest doses. Lab animals exposed to high concentrations develop liver and kidney trouble, while fish and plant cells suffer at much lower levels. Human exposures, whether by inhalation or skin contact, trigger nausea and acute pain, with long-term buildup leading to metallic taste, fatigue, and nerve symptoms. Early workers saw “tellurium breath,” a garlic-like odor, after accidental dosing. Modern toxicology efforts center on mapping safe limits and recovery times, using real-world exposure scenarios rather than only animal tests. Regulators use these data points to shape permissible air and water levels, always lagging slightly behind new findings. Risk remains high enough that regular monitoring, technical controls, and professional training aren’t optional anywhere responsible work with sodium tellurite takes place.
The Road Ahead: Future Prospects
Looking forward, advanced electronics and green tech could give sodium tellurite new relevance. As researchers chase better, lighter batteries and more transparent conductors, tellurium-based salts like sodium tellurite could claim new ground in flexible devices, solar panels, and photoactive coatings. On the other side, stricter environmental controls might shrink its role, especially as alternatives or recycling methods reduce net tellurium use. Investment in closed-loop manufacturing and improved disposal could ease health and environmental burdens, letting responsible users tap sodium tellurite’s chemistry without facing regulatory backlash. Ongoing R&D aims to stretch its benefits while taming its downsides, keeping this old compound in the mix for another round of industrial and scientific breakthroughs.
What is Sodium Tellurite used for?
What Sets Sodium Tellurite Apart?
Not every laboratory chemical boasts the kind of specialized role sodium tellurite fills. In science classes growing up, the names of heavy metals rarely came with exciting baggage—most sat in storerooms, next to dusty bottles of things like barium or selenium. But sodium tellurite marks its own space thanks to what it brings to microbiology and chemical engineering.
This white, crystalline powder grabs attention in microbiology labs. Anyone who’s tried to identify a troublesome bacterial contaminant knows how subtle some bacteria can be. Enter sodium tellurite. In culture media, sodium tellurite acts as a selective agent. Certain bacteria get knocked out quickly when this substance enters the mix, while a handful manage to adapt and grow, revealing themselves in the process. Microbiologists working in public health depend on this ability, especially when testing food samples or monitoring water safety. Foodborne pathogens like Listeria monocytogenes tend to survive where others falter, making detection possible without second-guessing colony identities.
Why Does Selectivity Matter So Much?
Public health lives and dies on accuracy. Think about the stakes in a hospital kitchen or a dairy plant. Contaminants need to be tracked and stopped before outbreaks start. Sodium tellurite provides that edge, pulling specific threats out from the crowd fast. This saves money on unnecessary treatments, cuts back on lost product, and—most importantly—saves lives. According to the Centers for Disease Control and Prevention (CDC), listeria infections kill hundreds each year in the United States. Fast, reliable detection through tellurite-based media puts a real dent in those numbers.
People outside the biology world might see sodium tellurite as esoteric, but in research and environmental monitoring, it’s a staple. I remember volunteering at a local water treatment testing center. The techs routinely reached for media containing sodium tellurite any time a water sample arrived. Testing would show high selectivity—few false positives. In hands-on scenarios like this, compounds don’t stay just science terms—they decide how communities face critical safety issues.
Other Uses Worth Mentioning
Sodium tellurite plays a part in some specialty industrial applications too. In metallurgy labs, technicians use it as a reducing agent. They’re tasked with extracting and purifying metals, a process filled with technical pitfalls. Sodium tellurite steps in to help reduce toxic tellurium compounds and assist with refining, an important move when producing high-purity metals for electronics or solar technology.
Historically, some companies worked sodium tellurite into glass and ceramics manufacturing—the aim being to improve optical qualities for scientific glassware or colored filters. As industries chase better materials for electronics, batteries, and renewable energy, demand for traditional compounds to adapt keeps growing.
The Safety Side of the Coin
Of course, any story on chemical use needs honesty about risk. Sodium tellurite isn’t something to use lightly. Inhalation and skin contact cause real harm, so strict lab protocols matter. Regulatory bodies like OSHA and NIOSH recommend gloves, eyewear, and fume hoods. Proper disposal also demands care, and responsible manufacturers share detailed safety data and encourage user training. Ignoring these basics puts both laboratory staff and downstream environments in harm’s way.
Pushing for Responsible Chemistry
Science and industry both owe their future to smarter, safer chemical handling. As more people look to microbiology and material science to solve society’s problems, sodium tellurite’s story keeps evolving. Its role in public health and technology rests on constant accountability—suppliers, labs, and end users all have to keep safety and accuracy at the front of every decision.
Is Sodium Tellurite hazardous or toxic?
What is Sodium Tellurite?
Sodium tellurite isn’t something most folks run across in daily life. You find it mostly in laboratories, some electronics manufacturing, or maybe in places tinkering with microbial growth mediums. At first glance, the compound doesn’t look that menacing—just another white or colorless powder. The real concern hides behind the science and the handling it demands.
How Toxic is It, Really?
Toxicity isn’t always about immediate danger. Sometimes, it’s slow, sneaky, and cumulative. Sodium tellurite falls in that camp. The compound contains tellurium, a heavy element whose chemistry isn’t as notorious as mercury or lead, yet it still packs a punch. Breathing in even small amounts of the dust can prompt nausea, vomiting, or a nasty metallic taste in the mouth. Skin contact with solutions might irritate but doesn’t usually do lasting harm unless it happens over and over again.
Reports show that ingestion does the most damage. Swallowing a small dose can trigger stomach cramps, diarrhea, and kidney trouble. Years ago, toxicologists tested its effects and saw that higher exposures might mess with nerves or even disrupt how blood cells carry oxygen. Sodium tellurite isn’t classified as a cancer-causing chemical, yet its acute effects shouldn’t be brushed off.
What About Environmental Impact?
Few people talk about where trace amounts of sodium tellurite end up after disposal. Industry doesn’t use loads of it, but improper handling sends it down drains, into soil, or sometimes even waterways. This brings trouble for tiny creatures first—bacteria, invertebrates, maybe fish. Tellurium builds up, which researchers call bioaccumulation, and though rare, can disrupt whole food webs. The element isn’t essential for life, so when it shows up, living things struggle to process it. Even though not many communities deal with tellurium pollution, the science suggests caution.
Personal Perspective on Safety
Having worked in a college lab years back, I remember the day our instructor showed us the bottle stamped “Na2TeO3.” We wore double gloves, masks—no shortcuts allowed. The safety protocols came down from people who’d seen slips turn into emergency calls. Back then, it sunk in that handling risk isn’t about fear, but respect for the chemicals.
Eye protection, solid ventilation, and proper disposal matter even for uncommon substances like sodium tellurite. Training makes sure the balancing act between scientific progress and personal safety doesn’t tip the wrong way. There’s no shame in over-preparing, especially when the risks reach beyond the user to the environment and community.
Better Solutions
A few labs and manufacturers have started shifting to less hazardous chemicals when they can. Green chemistry aims to swap risky compounds with safer options—sometimes it works, sometimes the alternatives lag behind in effectiveness or price. For those who have to use sodium tellurite, the answer lies in airtight processes, well-marked containers, protective gear, and robust education. Emergency procedures and quick access to wash stations can make a major difference if exposure happens.
Communities benefit when colleges and factories share information on chemical use and disposal, so they can respond fast to any potential leak or spill. Encouraging others to report concerns and maintaining up-to-date environmental assessments helps catch small problems before they spiral.
The Importance of Informed Use
Sodium tellurite will stick around in specialty settings, but open discussion, awareness, and the right safety measures keep its risks in check. Chemical hazards become manageable when those handling them don’t ignore the facts. Guided by real experience and good science, users can work safely—protecting themselves, their teams, and the places they call home.
What is the chemical formula of Sodium Tellurite?
Getting Straight to the Facts
In everyday conversation, people rarely mention sodium tellurite. Most folks outside of chemistry classrooms or laboratories probably wouldn’t recognize this compound or what it does. Yet, anyone with a background in science—and especially those who care about practical chemistry—should know its formula: Na2TeO3. A simple string, but it tells a story about how sodium, tellurium, and oxygen work together. Each part of this formula holds a clue about the substance’s structure, behavior, and uses.
It’s Not Just About Memorization
Looking at the chemical formula, I see more than just elements; I see connections. Sodium tellurite contains two sodium atoms for every tellurium atom, and the tellurium is bonded to three oxygen atoms. In college, professors drilled formulas into our heads, but the importance goes beyond passing tests. Understanding this structure helps predict how the compound will react with other substances, how it dissolves in water, and whether it’s safe to handle.
Experience in the lab taught me that handling sodium tellurite isn’t the same as handling sodium chloride. Tellurium compounds have unique toxicity to bacteria, which makes sodium tellurite handy as a selective agent in microbiology. The presence of tellurium sets it apart and brings in a layer of complexity that straight sodium compounds lack.
Safety and Environmental Concerns
Every chemical brings potential risks, including sodium tellurite. Its toxicity impacts both humans and environmental systems. As someone who worked in waste disposal as a lab tech, I remember the importance of managing chemical waste for compounds like this. If small amounts slip into a water supply, fish and aquatic invertebrates face harm. It’s not paranoia; reports show that tellurium compounds, especially in ionic forms like Na2TeO3, poison organisms more easily than elemental tellurium.
The safety data on sodium tellurite also deserves attention. Too often, organizations overlook training when introducing chemical agents. Not just gloves and goggles, but clear guidance on handling, disposal, and spill response. Each bottle of sodium tellurite on a shelf needs a clear label and a corresponding safety sheet. Speaking from personal experience, one careless moment can prompt hours of emergency cleanup. Sending that message home early helped keep lab teams, janitors, and visitors safe. Real-world responsibility goes far beyond reading the barcode on a reagent bottle.
Where Sodium Tellurite Enters Our World
Sodium tellurite shows up in more places than most expect. The role it plays in biotechnology earned it a badge among researchers testing new antibiotics and screening for bacterial strains. With its selective toxicity, labs use it to single out microbes that tell other stories—like whether a hospital’s waterline carries dangerous bacteria.
Outside the lab, this compound finds work in specialty glass and ceramics. Its oxidizing properties make it valuable in niche manufacturing, and some commercial chemical syntheses depend on it. Knowing how Na2TeO3 acts, and respecting its chemical character, makes the difference between failure and a successful outcome.
Better Handling Through Education and Policy
Clear, experience-driven education paves the way for better safety with sodium tellurite. Instead of tossing formulas on chalkboards, educators and workplace managers can connect the dots: teach real stories of mishaps, demonstrate correct disposal, and push for stricter oversight where chemicals threaten vulnerable communities. This compound’s formula—Na2TeO3—offers a lesson in respect for the intertwined worlds of science, industry, and public health.
How should Sodium Tellurite be stored?
Understanding Sodium Tellurite Risks
Sodium tellurite often looks harmless, showing up as a white crystalline powder. Beneath the surface, it carries health risks that many people miss. If it touches skin or eyes, or if dust clouds drift into your lungs, it triggers irritation and may cause bigger health problems later on. Not everybody keeps chemical safety in mind, though, so clear steps need to guide safe storage at all levels—from labs to industry.
Choosing the Right Location
I’ve worked around a lot of chemical storerooms. Space matters, but so does what sits nearby. Never park sodium tellurite close to acids or other incompatible materials—add a splash of acid and you might release toxic fumes. Dedicated shelving, labeled well, makes a huge difference. If labels fade or fall off, confusion spills over, so checking storage conditions should become routine instead of an annual panic.
Climate and Container Selection
Ventilation becomes more than a comfort feature in chemical storage. If air doesn’t move, escaping dust or gasses stay trapped, raising health risks. Sodium tellurite fares best in a cool, dry, well-ventilated area. Don’t let it absorb moisture—humid rooms speed up degradation and open the door to chemical reactions. Air meets sodium tellurite, moisture seeps in, and pretty soon you’ve got clumping or unpredictable byproducts.
Glass and HDPE (high-density polyethylene) containers hold up under usual lab conditions, resisting breakdown and keeping contents dry. Forget plastic bags or containers with a worn seal—cuts corners turn into bigger headaches once the chemical starts to react with the air. Tight-fitting lids and secondary containment (like a small bin to catch leaks) stop accidents from spreading.
Protecting People and Property
People working near sodium tellurite deserve clear rules. PPE (personal protective equipment) should never collect dust on a shelf, especially gloves and eye protection. If someone has to move the chemical, clear instructions and proper retrieval tools—scoops and antistatic brushes—should sit close by. Don’t stack tons of containers—gravity and glass never mix well. Store containers at waist or chest height so accidents lead to less damage.
Emergency Preparedness
Sometimes, things go wrong. Spills demand fast cleanup with the right tools—no dry sweeping, which tosses dust in the air. Use a HEPA vacuum or damp towels, dispose of waste in properly labeled hazardous waste containers, and ventilate the area. Every site I’ve managed, we checked the eyewash stations and showers monthly. That habit kept minor splashes from becoming medical emergencies.
Accurate record-keeping runs hand-in-hand with storage. Maintaining a log of inventory reduces forgotten or expired chemicals collecting in dark corners. Local regulations often set the baseline for storage, but going above that keeps everyone safer. Workers and managers should update procedures together, because what looks safe on paper sometimes falls apart in real life. A well-informed team can spot storage problems early and step in before risk turns into harm.
Conclusion
Sodium tellurite brings value to research and industry, but respect for its risks shows through safe storage. Careful climate control, tough containers, and rigorous handling habits form the backbone of chemical safety. The real solution combines respect for the hazards, commitment to clear procedures, and constant teamwork to protect workers and the wider environment.
What precautions should be taken when handling Sodium Tellurite?
Why Treating Sodium Tellurite with Respect Matters
Most people won’t run across sodium tellurite unless they’re working in a lab, but for those handling it, the stuff comes with real risks. Sodium tellurite can cause serious irritation to your skin, eyes, and lungs. Swallowing even small amounts kicks off nausea and more dangerous symptoms. So, the laid-back approach doesn’t work here—taking good care protects not just yourself but everyone sharing the space.
Personal Protective Equipment Is Non-Negotiable
Gloves are a must. Not just the usual latex gloves you might keep around, but chemical-resistant material like nitrile. Standard lab coats help, but long sleeves and goggles cover the bases better if an accident splashes up. Inhaling tellurite dust is where things get much trickier. Breathing this in isn’t just uncomfortable. It creates a chemical risk that can lead to coughing, headaches, or worse over extended periods.
A decent fume hood is absolutely worth the investment. The kind of lab that works with sodium tellurite regularly often deals with other toxic substances, so solid ventilation goes a long way toward protecting workers. Relying on an old fan or open window isn’t enough.
Storage: Keep It Separate and Secure
Overpacking and cluttered shelves sometimes slip through when labs get busy. Sodium tellurite demands its own space, sealed tightly and clearly labeled. Mixing it up with acids or other reducing agents can trigger dangerous reactions, so keeping it away from other chemicals means fewer surprises. Using shatter-proof storage containers cuts down on messy accidents if something slips off the shelf.
Every trained scientist has seen how a tiny bit of powder, spilled in the wrong place, can snowball into a big headache. Avoiding shared scoopulas for different chemicals can save everyone time and hassle because even trace contamination in a lab adds up.
Don’t Underestimate Training and Communication
Getting everyone in the lab on the same page beats winging it when something goes wrong. Regular safety talks make a bigger difference than most folks realize. Not everybody remembers the exact difference between sodium tellurite and other white powders sitting nearby, so reviewing labels together, checking for proper storage, and walking through spill drills can prevent panic when things go sideways.
Every new lab member should go through training that includes handling sodium tellurite in real-world terms—not just reading the safety data sheet. Sharing stories about near-misses or slip-ups keeps things real and drives home why these routines exist.
Quick Steps If Things Go Wrong
Spills happen, even in careful environments. Immediate action beats hesitation every time. Covering the spill with inactivated absorbent material, ventilating the area, and using proper disposal methods stop problems from getting worse. Reporting the incident—rather than cleaning up in silence—means the whole team can double-check for lingering risks or missed spots.
If someone comes into contact with sodium tellurite, flush the affected skin or eyes with running water. Medical follow-up is not just a safety net. Nobody wants to risk long-term complications from what feels like a minor slip.
Learning from Past Experience
Working with sodium tellurite has taught me that accidents seldom happen the same way twice. Small lapses add up, and corners cut today can spell big problems tomorrow. The best labs I’ve ever worked in weren’t the ones with the fanciest gear but the ones where everyone took safety seriously, consulted with each other, and called things out before a simple mistake turned into an emergency.
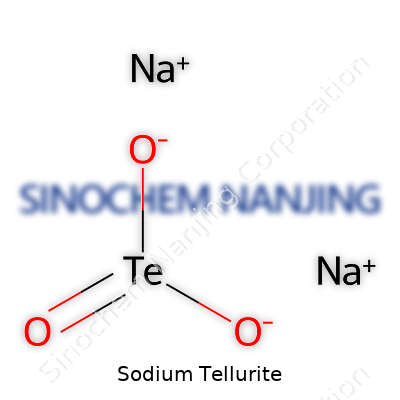
| Names | |
| Preferred IUPAC name | Sodium tellurite |
| Other names |
Sodium tellurite (Na2TeO3) Disodium tellurite |
| Pronunciation | /ˈsəʊdiəm tɛˈljʊəraɪt/ |
| Identifiers | |
| CAS Number | 10102-20-2 |
| Beilstein Reference | 356675 |
| ChEBI | CHEBI:63051 |
| ChEMBL | CHEMBL1231861 |
| ChemSpider | 18763 |
| DrugBank | DB11230 |
| ECHA InfoCard | 100.970.326 |
| EC Number | 231-145-7 |
| Gmelin Reference | 130145 |
| KEGG | C52744 |
| MeSH | D013700 |
| PubChem CID | 24816 |
| RTECS number | WS4900000 |
| UNII | WQ2U855OT1 |
| UN number | UN3288 |
| CompTox Dashboard (EPA) | DTXSID5020677 |
| Properties | |
| Chemical formula | Na2TeO3 |
| Molar mass | 165.59 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | D=4.39 g/cm³ |
| Solubility in water | Soluble |
| log P | -4.06 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 2.5 |
| Basicity (pKb) | 6.05 |
| Magnetic susceptibility (χ) | -47.2e-6 cm³/mol |
| Refractive index (nD) | 1.73 |
| Viscosity | 3 cP (20°C) |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 121.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -586.0 kJ/mol |
| Pharmacology | |
| ATC code | V03AB54 |
| Hazards | |
| Main hazards | Toxic if swallowed, harmful if inhaled, causes skin and eye irritation. |
| GHS labelling | GHS05, GHS07, GHS09 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | P264, P270, P273, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 2-1-2-OX |
| Lethal dose or concentration | LD50 oral rat 185 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 160 mg/kg |
| NIOSH | WT2575000 |
| PEL (Permissible) | PEL: 0.1 mg/m³ |
| REL (Recommended) | 200 mg/L |
| IDLH (Immediate danger) | IDLH: 25 mg/m³ |
| Related compounds | |
| Related compounds |
Sodium selenite Sodium sulfate Sodium selenate Potassium tellurite Tellurium dioxide |

