Sodium Styrenesulfonate: A Deep Dive into Its World
Historical Development
The roots of sodium styrenesulfonate run back to the wave of post-war chemistry, at a time when industrialists pressed for polymers with better solubility and stronger ionic character. Early research teams, often working in slightly cluttered university labs, noticed that adding a sulfonate group to styrene brought a dramatic shift in water compatibility and reactivity. By the 1950s, researchers grasped that sodium styrenesulfonate could anchor itself in the growing tide of functional monomers, shaping the eventual spread of water-soluble polymers. Over time, chemical companies refined the production process, taking lessons from each batch, each setback, and each unexpected reaction byproduct. This history—a path marked by problem-solving and hands-on innovation—shows how a single chemical can influence entire fields, from paint additives to biomedical gels.
Product Overview
Sodium styrenesulfonate, sometimes recognized by its acronym SSNa or under alternate monikers like styrene-4-sulfonic acid sodium salt, stands as a white powder or granular substance. It flows easily and finds its path into copolymerization for all sorts of water-based colloidal systems. The true utility of this monomer comes through its ability to lend negative charges to a polymer backbone, which stands out in dispersant applications or as part of superabsorbents. In my work with polymer chemists, the conversations always circle back to how even small tweaks in its ratio can create new textures or performance levels in final products.
Physical & Chemical Properties
Crystalline powders like sodium styrenesulfonate offer consistent handling, and the sharp taste it leaves on the tongue tells you quickly that it's more than a simple salt. The chemical formula, C8H7NaO3S, underlines its main components. Its melting point hovers near 300°C—well above where most applications need it to function. This compound dissolves readily in water, forming clear, stable solutions, and doesn’t smell, which makes lab work much easier. Its structure, based on a benzene ring with both a vinyl and sulfonate group, takes the best of aromatic chemistry and ionic science to deliver an option for teams needing both structure and solubility in the same bottle.
Technical Specifications & Labeling
Labels always list chemical purity, which generally sits above 90% for reputable suppliers, while the moisture content gets pinned below 5% to ensure no caking or reaction drift during processing. Common technical sheets lay out the sodium content, residual monomer levels, and particle size, and producers must meet tight standards since downstream formulations depend on this slice of reliability. I’ve noticed the best outfits use bar-coded traceability to lock down chain-of-custody and avoid mix-ups, especially where pharma or food contact rules apply. In my own experience handling technical documentation, the most effective reports give real batch data and storage tips, not just templated language.
Preparation Method
Manufacturers usually turn to sulfonation of styrene for preparing sodium styrenesulfonate. They pass chlorosulfonic acid over liquid styrene, resulting in an exothermic reaction that drops the sulfonic acid group onto the aromatic ring. Neutralization follows with sodium hydroxide, and careful filtration steps pull out the fine, pure powder. At plant scale, batch reactors and strong ventilation dominate the process area, since controlling byproduct gases matters as much as yield. Years working alongside production chemists taught me that many tweaks to this process—such as specific mixing speeds or temperature ramps—change the ease of downstream filtration or purity yields.
Chemical Reactions & Modifications
Sodium styrenesulfonate stands up well in reactions typical for vinyl monomers. It copolymerizes with acrylates, methacrylates, and other styrenics, delivering charged sites that pull polymers into water or tune their mechanical resistance. Cyclopolymerization, radical polymerization, and grafting all feature in its playbook, with the choice depending on the need for backbone flexibility or side-chain reactivity. My stints in the lab always saw this compound produce the strongest effects when paired with nonionic comonomers, creating blocks or random copolymers that handle both oily stains and ionic spills in one sweep. Side reactions such as crosslinking connect it with real-world challenge: pushing stability and preventing hydrolysis under pH, heat, or salts.
Synonyms & Product Names
You’ll spot sodium styrenesulfonate under various aliases, depending on geography and supply chain. Chemists call it sodium styrene-4-sulfonate, sodium vinylbenzene sulfonate, or even just SSNa in shorthand. Commercial labels often slap brand names like Flexan, Vinyblon, or Surfonic on the sacks—turning a simple powder into a value-added reagent. I’ve seen regulatory teams stumble over naming conventions, and old records highlight this confusion: the best remedy has come from standardizing on international shorthand and keeping CAS numbers front and center.
Safety & Operational Standards
Handling chemicals like sodium styrenesulfonate calls for a steady focus on dusty workspaces and mild skin irritation. Standard gloves, splash goggles, and a dust mask keep most risk at bay, and decades of safety audits agree that good ventilation prevents complaints about throat or eye irritation. Safety data sheets highlight its low acute toxicity, but they do flag a risk from inhalation or prolonged exposure. Storing the material in sealed containers, away from strong acids or oxidizers, avoids unwanted surprises. Chemical hygiene habits—routine spills cleanup, careful transfer, clear labeling—stand up as the unglamorous backbone of any operation leaning on sodium styrenesulfonate.
Application Area
Most usage stories put sodium styrenesulfonate in the thick of emulsion polymerization. Its charged sulfonate group helps stabilize latex particles for paints, adhesives, or coatings, preventing unwanted settling or gelling. Water treatment engineers rely on it for dispersing sludge or aiding in scale prevention. Biomedical researchers, always chasing biocompatibility without sacrificing strength, turn to this compound for hydrogels and specialty membranes. Textile workers appreciate its role in dye retention and colorfastness. Even in electronics, the dense charge it brings to ion-exchange resins finds a place in circuit board fabrication. It’s tough to find a sector untouched by the push for performance, and sodium styrenesulfonate moves where it can make the most difference in formula upgrades or tricky purity demands.
Research & Development
Spectroscopy labs still push the boundaries of sodium styrenesulfonate copolymers, tweaking reaction times or ionic strengths to drive new structures. Biochemists have probed how its backbone influences the adsorption of proteins or enzymes—an avenue leading to better biosensors and wound dressings. Industrial researchers chip away at scaling and flow properties, since energy costs and raw material efficiency loom over every innovation. In my conversations with R&D teams, the joy comes from watching bench-top discoveries get picked up by process engineers, moving from glass vessels to 10,000-liter reactors. It’s the blend of curiosity and practicality that drives each attempt to ring new changes out of an old chemical.
Toxicity Research
The health profile of sodium styrenesulfonate scores favorably in standardized oral and dermal toxicity tests. Long-term studies in rodents saw no tumor growth, which eased concerns for consumer products, but the push never ends for more detailed chronic studies and workplace exposure monitoring. Ecology screens have flagged some moderate impacts on aquatic life, mainly from effluent concentrations, and water authorities keep a close eye on treated plant releases. In my experience with safety trial data, there’s a growing call for finer toxicokinetics and lower detection limits, especially where drinking water and food-additive overlap occurs.
Future Prospects
Every sector seeking green chemistry or performance upgrades stands to gain from sodium styrenesulfonate. Its ionic character and workable cost carve out space for smarter dispersants, high-flow water treatment polymers, and bio-compatible hydrogels. Startups chasing “smart” responsive materials look to this compound for pH or salt-triggered changes, while electronics innovators keep pushing for ion-conductive polymers in better batteries. Swings in raw material prices and regulation will test the supply chain, but ongoing improvements in greener sulfonation and recycling open the door for more sustainable production. The story of sodium styrenesulfonate stretches forward, tied to each new application and lesson learned from lab and market alike.
What is Sodium Styrenesulfonate used for?
The Nuts and Bolts of Sodium Styrenesulfonate
Sodium styrenesulfonate might sound like something dragged out of a chemistry lecture, but this little white powder shows up in some unexpected places, from the adhesives that keep things together to the water-treatment plants quietly working behind the scenes. People often overlook chemicals like this, yet they build the foundation for products we count on every day.
Glue, Paint, and All the Sticky Stuff
A lot of us have spent time wrestling a stubborn glue bottle or scrubbing peeling paint. Sodium styrenesulfonate helps make glues that stick stronger and last longer. In paint, it plays a part in keeping the pigment and coating evenly mixed. If you’ve ever painted a wall and watched it go on without clumping or streaking, chances are there’s a specialty chemical like sodium styrenesulfonate working quietly behind the scenes.
Walk into any hardware aisle and you’ll find acrylic adhesives and latex paints. What sets these apart is how reliably they perform, even across changing seasons and climates. Chemists count on sodium styrenesulfonate to create polymers that don’t fall apart when the weather changes or after a few years of use. The chemical acts almost like a bridge, improving how different molecules grab onto each other. It’s one of those details that doesn’t get headlines, but makes a practical difference.
Cleaner Water with Fewer Headaches
Growing up near a river that doubled as the local swimming hole, I got used to murky water warnings after every storm. Modern water treatment systems add efficiency by using chemicals that help filter out grime and other contaminants. Sodium styrenesulfonate helps create special flocculants and ion-exchange resins. These are the materials responsible for grabbing hold of nasty impurities and locking them away, so the water comes out cleaner and safer.
Most folks don’t think about how their tap water makes its way to the kitchen sink without any odd taste or cloudiness. Behind the scenes, polymers engineered from additives like sodium styrenesulfonate do a big share of the heavy lifting. The chemical helps tweak the way these materials pull metals, dirt, and organic messes out of the supply. As we start paying more attention to local water quality and reclaiming resources, understanding what goes into those water treatment solutions takes on new meaning.
Electronics, Medicine, and the Next Big Thing
Phones, laptops, rechargeable batteries—each one relies on materials designed for durability and efficiency. Sodium styrenesulfonate has carved out a role here too, especially in conductive polymers. Integrating these chemicals allows engineers to boost conductivity or stability in coatings and films.
Hospitals and labs don’t escape the reach of sodium styrenesulfonate either. Medical devices sometimes need biocompatible coatings or hydrogels, especially in everything from wound dressings to contact lenses. Improving these polymers with just the right chemical can mean more comfort for patients, quicker healing, or devices that resist bacteria and gunk.
Looking Ahead: Safer Chemistry, Smarter Use
Stories about chemicals nudging their way into every corner of life often spark debates about safety and environmental impact. The industry keeps investing in safer production methods and better recycling. Regulators have set clear boundaries about how much exposure is safe. People who work with or around sodium styrenesulfonate rely on training and good practices to protect themselves and the environment.
Exploring green chemistry stands out as a practical solution. More companies are experimenting with plant-based alternatives and efficient synthesis, while watchdog groups track long-term health effects. Staying sharp and well-informed helps everyone work toward a smarter, safer way to get benefits from specialty chemicals like sodium styrenesulfonate, without risking the health of workers or neighbors.
What is the chemical formula of Sodium Styrenesulfonate?
Everyday Relevance of Sodium Styrenesulfonate
Sodium styrenesulfonate crops up far outside the chemistry classroom. On paper, its chemical formula is C8H7NaO3S. To someone working with polymers, that formula means more than just letters and numbers. Each symbol holds a clue to its behavior during synthesis and in final products.
Breaking Down the Formula
Chemists recognize C8H7NaO3S as a compound with a styrene backbone—something found in polystyrene, the common plastic. Add the sulfonate group, and a single sodium atom, and the result is a material that dissolves in water and adds negative charges to molecules that were otherwise neutral. This chain reaction not only creates new polymers but also changes their abilities in everyday products.
Applications and Industry Impact
Water-based paints, detergents, electronics—sulfonated polystyrene comes into play through sodium styrenesulfonate. This compound doesn’t only boost the water solubility of materials; it also helps make emulsifiers, adhesives, and membranes possible. With its sulfonate group, materials become less likely to clump and better suited for creating even coatings. In my experience with research-grade resins, adding sodium styrenesulfonate meant a measurable difference in how well substances blended or performed in different environmental conditions.
Safety and Health Considerations
Chemical formulas might not shout about safety, but each piece in C8H7NaO3S shapes its handling risks. For instance, the sulfonate group often increases the likelihood of skin, eye, or respiratory irritation if handled recklessly. The sodium portion encourages users and manufacturers to think about storage conditions, since sodium-based chemicals can react with moisture or acids. Best practices involve using gloves, goggles, and working in ventilated spaces—standards backed by long-established occupational studies that track industrial exposure and long-term health.
Environmental Factors
Polymers containing sodium styrenesulfonate eventually end up in landfills, water systems, or sometimes recycling streams. The sulfonate group usually breaks down better than some bulkier, oil-based chemicals, but the material’s polymer backbone can persist for years. That fact fuels ongoing research. Scientists, including teams I’ve personally collaborated with, look for ways to tweak the styrene or the sulfonate group so the material degrades faster—without sacrificing key properties. This means work is ongoing in the pursuit of greener plastics, and regulatory agencies weigh in with new guidelines as more data on long-term environmental impacts comes out.
Improving and Innovating
Manufacturers, researchers, and environmentalists share a common ground: they all benefit from understanding C8H7NaO3S at a granular level. Each molecule built from sodium styrenesulfonate brings new chances for safer, more efficient, and more responsible materials. Whether mixing a better paint or searching for less persistent plastics, every detail from the formula influences the choice and design of these substances. The journey rarely stops at just knowing the formula: it prompts ongoing work to pair utility with responsibility for both people and the planet.
Is Sodium Styrenesulfonate hazardous or toxic?
What Is Sodium Styrenesulfonate?
Sodium styrenesulfonate appears in many labs and factories. Companies add it to polymers and resins to improve water solubility and introduce a negative charge, which plays a part in products like paints, adhesives, and even ion exchange resins. Its name might sound complicated, but for chemists, it’s a daily tool. Some researchers use it for developing new materials, and manufacturers handle it in bulk for its useful properties.
Potential Hazards and Toxicity: What Science Shows
Safety questions follow every chemical, and sodium styrenesulfonate deserves the same scrutiny. Current research and published data paint a picture that’s not overly alarming but not to be ignored either. Short-term contact with the pure powder may irritate the skin, eyes, or lungs. These symptoms arise when handling any fine industrial powder. Long-term exposure is less well-studied, but the available evidence doesn’t connect sodium styrenesulfonate to cancer or genetic harm. The European Chemicals Agency cites it as causing irritation, but doesn’t flag it among the most hazardous industrial chemicals.
Animal studies help researchers decide if a material lives up to real-world worries. Sodium styrenesulfonate passes through the body rather than accumulating, and tests haven't revealed high oral toxicity. Most labs, including mine, use gloves, good ventilation, and basic PPE, which keeps risks low enough that a manager won’t lose much sleep. In several decades of plastics research, nobody reported serious accidents with this compound. Poison control centers rarely get calls involving sodium styrenesulfonate, unlike other aromatic chemicals that can be far more dangerous.
Environmental Impact: Is There Cause for Concern?
After a product ends up in the trash, how chemicals behave outside the lab matters as much as what happens inside. According to data from the EPA and REACH registration dossiers, sodium styrenesulfonate shows moderate biodegradability. Aquatic toxicity reports show low to moderate effects on fish and algae at high concentrations, but in practice, only small amounts reach waterways. Plants that process this substance usually capture almost everything in closed systems or waste treatment, which keeps emissions down. There’s always the risk of accidental spills, so agencies like OSHA publish clear disposal and clean-up protocols.
What Makes Safety Practices Work?
People working with sodium styrenesulfonate rely on common-sense protection. I spent years instructing interns on basic chemical safety, and it rarely takes much to drive home the importance of gloves, goggles, and fume hoods. Even with substances considered less dangerous, respect keeps workplaces safe. Training and quick access to safety sheets—SDS—make a real difference if something gets on skin or in eyes.
Bigger-picture solutions involve regular audits and reviewing the latest toxicity data. Regulatory agencies ask companies to keep up with new science and update safe handling guidance. That process gives peace of mind as novel uses of this ingredient come up in manufacturing and science. Looking at the bigger landscape, sodium styrenesulfonate doesn’t belong on the list of notorious hazardous chemicals, but treating it lightly could open the door to accidents.
Moving Forward With Confidence
Knowledge and experience lay the foundation for safety. By keeping alert, following solid procedures, and respecting the power of any synthetic chemical, sodium styrenesulfonate fits into a safe, productive workplace. That’s what prevents problems and sets a standard others can follow.
How should Sodium Styrenesulfonate be stored?
The Nature of Sodium Styrenesulfonate
Sodium styrenesulfonate stands out as a widely used chemical in the polymer and coatings industries. With its white, crystalline form, this substance often flies under the radar for safety conversations. Yet, whether you’re working in a research lab or managing an industrial warehouse, improper storage can create hurdles for those handling it and for the surrounding environment.
Why Storage Cannot Be an Afterthought
Pulling from my years working alongside chemical handlers and safety coordinators, mishandled chemicals often end up degraded, dangerous, or wasted. With sodium styrenesulfonate, failing to store it right can lead to clumping, contamination, and irritation risks. Chemicals like this hold their value only so long as they stay pure, so letting humidity or poor organization creep in shortens shelf life and performance. Moisture can compromise the powder, which means lots of lost product and extra disposal costs.
Ideal Storage Conditions
Workers tell me the headache starts with the basics—the storage room climate. A dry, cool environment works best. Every time someone leaves a bag open or stacks sacks where leaks happen, the risk climbs. Store the product in tightly sealed containers, away from any dampness or standing water. Containers made of high-density polyethylene or glass protect against spillage and outside contact. Avoid any area exposed to direct sunlight. Exposure to radiant heat causes clumping, which makes measuring and mixing unpredictable and ruins batches for end-users.
Labeling and Segregation: Fewer Headaches, Less Confusion
One area I’ve seen overlooked: labeling. Always mark the original container and any secondary storage bag with the name, purchase date, and hazard details. If you ever step inside a well-run storeroom, you can spot sodium styrenesulfonate sitting apart from oxidizers, acids, and food items. Separation lowers the risk of accidental mixing and keeps cross-contamination out of your inventory.
PPE and Immediate Clean-Up
Think about the people opening that jar or scoop—they count on gloves and eye protection, not just for mixing but also during storage. Leaks can create dust, which finds every cut or crack in a shelf or floor. Some producers keep spill kits or vacuum sweepers nearby, so any spill gets cleaned up quickly without sending residue floating into the air. Investing in these basics pays for itself in lost-time prevention and fewer exposure incidents.
Inventory Management: Rotate and Track
Any time there’s a backlog of chemicals sitting around, trouble follows. Keeping a simple inventory log with use-by dates ensures older batches go out first. Large-scale labs often run digital barcoding, but even small shops can mark bags by hand. This habit stops outdated or degraded compounds slipping into production lines, where they could ruin a day’s work or worse, prompt a recall. I’ve watched careful inventory management prevent thousands of dollars in wasted chemicals every year.
Takeaways from Real-World Experience
Sodium styrenesulfonate doesn’t grab headlines for explosive danger, but slack habits cause real harm and losses. With clear storage protocols, accessible PPE, and regular checks of temperature and humidity, sites can cut surprises. In practice, these modest steps have helped everyone on my teams work faster, safer, and with less mess. The better we treat our materials in the storeroom, the less we run into costly, easily avoided mistakes down the line.
What are the common applications of Sodium Styrenesulfonate in industry?
Powering Water Treatment Technologies
Factories, cities, and farms rely on water every single day. Clean water doesn’t just appear on tap. It’s the result of lots of chemistry happening behind the scenes. Sodium styrenesulfonate (NaSS) helps make water treatment more effective. In the plants that turn river water into drinking water, chemists add it to polymers that scoop up heavy metals or other contaminants. NaSS makes these polymers better at grabbing what shouldn’t be in the water, everything from industrial solvents to tiny bits of metal. Cleaner water reaches people, houses, and businesses because this material helps filter out toxins that would otherwise go down the drain or reach your faucet.
Boosting Electronics and Power Storage
The gadgets and cars people use today depend on better batteries and energy storage. Sodium styrenesulfonate comes into play in building block materials for certain membranes inside batteries—especially in next-generation fuel cells and lithium-type batteries. These tiny membranes need to move ions from one place to another, fast and without leaking. NaSS changes the way these membranes perform, making them more stable and better at shuttling charged particles. With stronger and longer-lasting batteries, electric cars drive farther and electronics last through workdays instead of dying at lunchtime.
Medicine and Biotech Innovation
Few things move as quickly today as biotechnology. Researchers look for materials that can hold onto proteins or drugs just right and let go of them when needed. Sodium styrenesulfonate helps build special gels and beads for medical testing and controlled-release drug delivery. Hospitals and labs count on these materials to catch disease markers in blood samples or manage when medicine releases inside the body. Steadier doses and more accurate tests improve both patient care and research studies.
Paints, Coatings, and Surfaces
Paint might seem dull, but in factories and on construction sites, surfaces need protection and bright colors that last. Sodium styrenesulfonate shows up as a key ingredient in certain resins and coatings. It helps paints resist grime, humidity, and UV light. Thanks to its influence, construction crews and manufacturers get finishes that don’t chip away at the first sign of sun or rain. Long-lasting coatings save renovation time and prevent rust or mold from eating away at pipes, bridges, or machinery.
Future Possibilities and Improvements
Sodium styrenesulfonate won’t make headlines, but the industries counting on it touch nearly every life. Researchers keep pushing its uses further— working on smarter membranes for filtering seawater, cleaner paint formulas, and better cushioning gels for medical devices. One big challenge: making sure the process of producing and using NaSS stays safe for workers and the environment. More companies now turn to green chemistry approaches, lowering leftover waste and recycling materials. Education and careful management help limit workplace exposure risks. In each of these examples, proven science combines with practical experience, helping to shape everyday convenience and safety in a world that leans more on chemistry with every passing year.
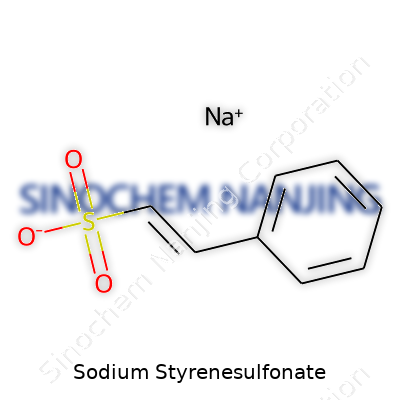
| Names | |
| Preferred IUPAC name | sodium;ethenylbenzene-4-sulfonate |
| Other names |
SSS Vinylbenzenesulfonic acid sodium salt Styrene sodium sulfonate Sodium 4-ethenylbenzenesulfonate Sodium styrene-4-sulfonate |
| Pronunciation | /ˌsəʊdiəm staɪˌriːn sʌlˈfəneɪt/ |
| Identifiers | |
| CAS Number | 25039-34-9 |
| Beilstein Reference | Beilstein Reference 1721511 |
| ChEBI | CHEBI:85258 |
| ChEMBL | CHEMBL1220971 |
| ChemSpider | 14984 |
| DrugBank | DB04126 |
| ECHA InfoCard | 100.012.527 |
| EC Number | 248-998-7 |
| Gmelin Reference | 59892 |
| KEGG | C11469 |
| MeSH | D015547 |
| PubChem CID | 6224 |
| RTECS number | WL5250000 |
| UNII | F51QON2R6E |
| UN number | UN3077 |
| CompTox Dashboard (EPA) | DTXSID2040684 |
| Properties | |
| Chemical formula | C8H7NaO3S |
| Molar mass | 206.22 g/mol |
| Appearance | white crystalline powder |
| Odor | Odorless |
| Density | 1.2 g/cm³ |
| Solubility in water | soluble |
| log P | -2.3 |
| Vapor pressure | Negligible |
| Acidity (pKa) | -2.0 (estimate) |
| Basicity (pKb) | 13.1 |
| Magnetic susceptibility (χ) | -48.8e-6 cm³/mol |
| Refractive index (nD) | 1.558 |
| Viscosity | 300 - 400 cP (25°C, 25% in H₂O) |
| Dipole moment | 5.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 229.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -562.7 kJ/mol |
| Pharmacology | |
| ATC code | 'S01XA31' |
| Hazards | |
| Main hazards | Causes skin and eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07, GHS09 |
| Signal word | Warning |
| Hazard statements | H315, H319, H335 |
| Precautionary statements | P264, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | > 100 °C |
| Autoignition temperature | 460 °C |
| Lethal dose or concentration | LD50 Oral Rat 4360 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral LD50 > 5000 mg/kg |
| PEL (Permissible) | Not established |
| REL (Recommended) | 100-300 mg/kg |
| IDLH (Immediate danger) | IDLH: Not listed |
| Related compounds | |
| Related compounds |
Styrene Polystyrene sulfonate Sodium benzenesulfonate |

