Sodium Selenite: Insight and Commentary on a Complex Compound
Historical Development
Sodium selenite stepped onto the scientific stage in the mid-19th century. Chemists were eager to isolate new elements, and selenium—a rare companion of sulfur—caught their attention. Early experimenters realized selenium’s unique properties could pack a punch, but methods for producing sodium selenite in pure form took years to finesse. Commercial motivation picked up steam during the 20th century, particularly with research into micronutrients for animal and human health. Researchers saw that small doses of sodium selenite had beneficial effects, and its value in nutritional supplements became clear as agricultural science matured. Decades of development have cemented sodium selenite’s place in laboratories, factories, food fortification plants, and fields worldwide.
Product Overview
Sodium selenite stands as an anhydrous or pentahydrate powder, recognized by its pale appearance and salty, slightly metallic taste. Industrial production caters mainly to the food, agriculture, and chemical industries. Food additive manufacturers measure out precise quantities to ensure safe supplementation, while glass factories exploit its properties to decolorize green glass. Big agriculture uses sodium selenite in livestock feed to stave off selenium deficiency, a major concern for grazing animals raised in selenium-poor soils. In pharmaceuticals, it turns up in established trace element solutions for clinical use.
Physical & Chemical Properties
At room temperature, sodium selenite usually appears as a colorless or white crystalline solid. Its chemical formula, Na2SeO3, hints at its makeup: two sodium ions and one selenite anion. This compound dissolves easily in water but hardly reacts with organic solvents, a fact that shapes its application in water-based formulations but keeps it out of oil-based products. It melts at high temperatures, near 700°C, and has a notable reactivity to strong acids, which liberate toxic selenium compounds. Handling sodium selenite requires care, as inhalation or ingestion outside controlled doses poses health dangers.
Technical Specifications & Labeling
Manufacturers usually supply sodium selenite with clear labels detailing purity (often above 98%), moisture content, water solubility, and heavy metal limits. Food and feed grades stick to regulations that define these parameters tightly. Packaging calls for air-tight containers because sodium selenite absorbs moisture and reacts with carbon dioxide from the air over time, potentially altering its quality and shelf life. Regulatory authorities, such as the US Food and Drug Administration and European Food Safety Authority, set maximum allowable limits for selenium content in dietary sources and require clear hazard warnings.
Preparation Method
Industrially, sodium selenite forms best via oxidation of elemental selenium. Producers first treat selenium metal with concentrated nitric acid, yielding selenious acid. Mixing this acid with a solution of sodium carbonate or sodium hydroxide leads to a controlled neutralization reaction, leaving sodium selenite in solution. Careful evaporation isolates the crystalline end product. Throughout, temperature, reaction time, and concentration need strict regulation, both to avoid unwanted byproducts and to cap the risks posed by selenium toxicity. Impurities—especially those containing arsenic or heavy metals—are a major concern during synthesis, given the trace amounts used in health products.
Chemical Reactions & Modifications
Sodium selenite displays keen reactivity toward reducing agents, swiftly converting to elemental selenium or selenides under the right conditions. This trait leaves it useful in redox chemistry and specialized manufacturing of selenium metal. Adding strong acids releases noxious fumes of selenium dioxide, so facilities design their processes to ventilate these hazards. The compound’s chemistry also allows for easy formation of other selenium compounds, from simple salts to organic selenium complexes, which have opening roles in further syntheses and pharmaceutical research.
Synonyms & Product Names
On shipment documents and material safety data sheets, sodium selenite might appear under several names: Disodium selenite, Selenious acid, disodium salt, and E221 in the European food additive system. Some suppliers abbreviate it as Na2SeO3, which can trip up buyers unfamiliar with chemical symbols. These synonyms help connect global markets but also increase the risk of accidental mix-ups, a problem for importers sourcing feed or food additives across borders.
Safety & Operational Standards
Facility standards around sodium selenite focus on worker training and robust ventilation. Direct contact spells trouble: skin, eye, and respiratory irritation lead to strict requirements for gloves, goggles, and dust masks on the factory floor. Regulatory agencies limit airborne selenium dust to very low numbers, typically below 0.2 mg/m3. Spills need prompt cleanup, with specialized disposal of contaminated materials. In storage, products keep best in cool, dry, dark places, away from acids or reducing agents. Site audits, safety data sheets, and proper labeling stand as non-negotiable in every major country for producers and high-volume users.
Application Area
Sodium selenite turns up far beyond chemistry sets. In animal nutrition, precise addition prevents white muscle disease and fertility issues in cattle and sheep, especially in selenium-starved regions. Egg and dairy producers rely on it to boost selenium content within safe dietary limits. In human health, nutritionists turn to sodium selenite for fortifying baby formula and multivitamins, as well as intravenous solutions for patients unable to eat. Glass makers also value its power to neutralize unwanted green tint caused by iron, giving bottles and windows a cleaner finish. In small doses, researchers test its effects as an antioxidant and in cancer treatment trials, probing the fine line between healthful trace mineral and toxicity.
Research & Development
Research on sodium selenite embraces both basic and applied science. Academic labs experiment with selenite’s antioxidant mechanisms at the cellular level, mapping how it interacts with enzymes like glutathione peroxidase. Teams looking for cancer prevention examine how low-level supplementation could protect DNA from oxidative damage, while toxicologists study its risks at doses only slightly above recommended dietary intakes. Feed technology experts push innovation by designing slow-release feed blocks, ensuring animals get just enough selenium over a longer period. In the energy world, sodium selenite features in studies on solar panel materials and as a potential redox mediator for batteries, a nod to selenium’s growing industrial importance.
Toxicity Research
My time in food science drove home the thin line dividing selenium’s nutritional value from its risks. Studies show selenium acts as an essential trace mineral, but its safety range is tight. At recommended levels, sodium selenite supports thyroid function, protects from antioxidant stress, and fuels animal growth. Even slightly more tips the scales toward toxicity: nausea, hair loss, nerve damage, and, in rare but catastrophic cases, lethal poisoning. Regulators use this research to lock down maximum allowable concentrations in animal feeds, baby foods, and drinking water. Newer studies check for chronic exposure risks and metabolites in various species, both safeguarding health and building the evidence base for future standards.
Future Prospects
Sodium selenite’s future looks dynamic. Population growth and food security concerns crank up demand for reliable micronutrient supplements, driving investments in production and quality control. Technological advances will likely tune release profiles in feed rations, aiming for greater safety and efficiency. Environmental toxicologists watch the rebound of artificial selenium in soils and water, spurring tighter controls and eco-friendly cleanup technologies. In medicine, the compound earns attention for its possible impacts on immune modulation and chronic disease risk. Pharmaceutical companies explore targeted delivery systems to maximize benefit and cut toxicity, a bet that new formulations could widen the safe window for using sodium selenite in therapy. Beyond health sciences, materials scientists keep experimenting with sodium selenite’s redox characteristics for use in green energy and electronics, making it a compound to watch on more than just the health front.
What is Sodium Selenite used for?
Essential Trace Mineral for Health
Sodium selenite often gets mentioned in conversations about essential nutrients. This white, odorless powder delivers selenium, a trace mineral the body uses in many processes. I remember reading about its importance the first time I set up a balanced diet plan for my family. Selenium plays a direct role in protecting cells from damage, supporting metabolism, and aiding thyroid health. Dietary shortfalls sometimes lead to muscle weakness or immune problems, especially for people who live in regions with seleniferous soils running low.
Adding Selenium to Animal Feed
Livestock require a fine balance of minerals to grow healthy and strong. Farmers often mix sodium selenite into feed rations because forage crops don’t always supply enough selenium. Healthy calves, lambs, and chicks usually start with feed that’s fortified with this mineral compound. It gets absorbed and converted by the animal’s system into active selenoproteins, keeping metabolism and growth on track. Low selenium in feed sometimes leads to serious health issues, including white muscle disease or reduced fertility in dairy cattle.
Human Supplements and Fortified Foods
Some communities don’t get enough selenium in their normal meals. Food companies and supplement makers step in by using sodium selenite to fortify products as a reliable, standardized source. I saw it listed as an ingredient in prenatal vitamins at the pharmacy and in breakfast cereals in supermarket aisles. For many people at risk of deficiency—older adults, vegetarians, or those with absorption problems—these extra sources matter a lot. Nutrition science has shown that selenium supports the body’s natural system to fight free radicals and helps certain hormones keep the body in balance.
Applications Beyond Nutrition
This compound also finds a place in other industries. In glass production, sodium selenite helps remove unwanted color from glass batches, producing bottles and panes that meet a higher purity standard. Water treatment plants and chemical labs use it in small amounts for specialized reactions and tests. These uses don’t usually touch most people’s lives, but they help maintain certain quality standards in products we handle every day.
Concerns and Responsible Use
Like any mineral supplement, the dose matters. Eating or drinking too much selenium over time puts people at risk for selenosis, which starts with hair loss, skin problems, nail brittleness, and sometimes much worse symptoms. Supplements and fortified products get strict regulation so overexposure doesn’t happen. Food scientists and health officials recommend sticking to recommended amounts and testing animal feeds to avoid accidental overdosing.
Looking Ahead: Safer Fortification and Sustainable Sources
New research encourages producers to examine how sodium selenite gets absorbed in both animals and humans, so this nutrient can be better targeted without creating environmental runoff. Sustainable agriculture groups now encourage tighter monitoring in farmland, along with regular soil and plant analysis, to keep the mineral cycle balanced. Safer sourcing and responsible use keep sodium selenite’s benefits available to people and animals—without putting health or the environment at risk.
Is Sodium Selenite safe for human consumption?
Understanding Where Sodium Selenite Shows Up
Sodium selenite pops up in multivitamins, animal feed, and some food products. People see it on ingredients lists and wonder if it's something to worry about. The answer isn’t black and white. I’ve spent years reading nutrition research and talking to healthcare providers about trace minerals. Selenite interests me because selenium, its core mineral, is both vital and risky.
Why Selenium Matters
Selenium keeps many enzymes running smoothly, especially the ones that protect our cells from damage. The thyroid uses it to make hormones. The immune system needs it to keep up its guard. People who don’t get enough risk problems like weakened immune function and thyroid issues. I’ve seen older adults drawn to selenium supplements, hoping to dodge illnesses or boost energy. The trick is getting enough, not too much.
How Much Is Safe?
The U.S. National Institutes of Health lists 55 micrograms a day as enough for most adults. Over 400 micrograms might cause trouble: stomach pain, hair falling out, brittle nails, even nerve problems. The window between “not enough” and “too much” stays very narrow. Most multivitamins use less than 70 micrograms, staying below the danger zone. Food sources like Brazil nuts, eggs, fish, and grains add up quickly. I learned the hard way that a bottle of extra-strong selenium tablets can tip a person over the limit faster than you’d think.
Sodium Selenite in Industry and Supplements
Companies use sodium selenite because it dissolves well in water and costs less than some other forms of selenium. Some sources call it less “bioavailable” than organic selenium found in foods, but research shows people still absorb and use it well enough. The real concern comes from over-supplementation, not the form itself.
Real Risks
I pull information from toxicology studies and poison control data: cases of serious harm always trace back to taking way too much, either by accident or believing more must be better. Sodium selenite turns dangerous at high levels because it can build up in tissues and disrupt basic cell processes. People taking large, frequent doses outside a doctor’s advice wind up with headaches, stomach upset, garlic breath, and eventually worse.
Food Fortification Practices
Food producers sometimes use sodium selenite to fortify bread or baby formula. Health authorities check the math to keep dietary intake within healthy limits. I’ve heard parents worry about every ingredient, but so far, levels in fortified foods stay far below toxic thresholds. The broader issue pops up when people mix fortified foods with high-dose supplements. Few folks read the fine print on labels until symptoms show up.
Managing Confusion and Clear Solutions
I tell friends: Know the safe range. Skip “megadose” trends. Check labels, and count everything from multivitamins to the handful of Brazil nuts you might snack on. Doctors and nutritionists should help patients tally up what they really consume before recommending any selenium supplement. Manufacturers ought to keep doses small and warning labels clear. Simple blood tests can spot those rare cases of low or high selenium and take the guesswork out of it. Being mindful beats fear or carelessness, especially with a nutrient where the margin of error stays so tight.
What are the potential side effects of Sodium Selenite?
Sodium Selenite: A Double-Edged Sword
Sodium selenite shows up everywhere in the health world, especially in supplements, animal feeds, and sometimes even in baby formula. As someone who’s spent years reviewing supplement trends and researching minerals, I’ve seen both the hype and the hidden dangers behind this chemical. Yes, it gives the body a trace of selenium, something everyone needs for thyroid function and immune defense. But sodium selenite is no magic powder—dose matters, and so do the risks.
The Risk of Getting Too Much Selenium
The recommended dietary allowance for selenium lands at about 55 micrograms daily for most adults. In small amounts, sodium selenite helps prevent deficiency. Yet the line between helpful and harmful isn’t all that thick.
I’ve seen people think more is better. That idea quickly runs into trouble with selenium. The tolerable upper limit for adults is only 400 micrograms per day. Taking extra, even by mistake, leads to the risk of selenium toxicity.
What Overexposure Really Looks Like
One of the first signs: stomach upset. Nausea, diarrhea, and abdominal pain hit some people quickly. With more exposure, the symptoms can grow worse. I remember a case reported in the New England Journal of Medicine where a batch of selenium supplements contained selenium levels far above what the label promised. The fallout involved hair loss, brittle nails that literally fall apart, a strong metallic taste in the mouth, skin rashes, and fatigue. All of this falls under a condition called selenosis.
For folks working in manufacturing or animal care, breathing in or touching sodium selenite powder gets risky, too. There are known cases of respiratory irritation and eye problems from industrial exposure. OSHA actually regulates selenium at the workplace because of these risks.
More than Physical Symptoms: The Ripple Effect
Toxicity doesn’t just attack the obvious places. High selenium can lead to neurological issues: dizziness, nervousness, and sometimes even trouble remembering things. Chronic exposure, repeatedly getting too much, can hurt the kidneys or liver, both of which work to flush metals and minerals from the body. It’s not only the short-term upset stomach or nail changes—over time, the body’s organs can struggle to keep up.
Real Solutions for Real People
Most people reading a supplement label don’t have lab equipment at home. So the single best step is to stick to reputable brands that can verify dose and ingredient quality. Doctors can run blood tests for those at risk, like people on total parenteral nutrition, or those with malabsorption concerns. Another smart move: check if you already get enough selenium from food. Brazil nuts pack plenty, and a normal diet with grains, poultry, and eggs supplies enough for most.
This trace mineral helps keep cells healthy and immune systems moving. But too much sodium selenite tips the scales in the wrong direction. One tablet can make a difference for some, but more isn’t always better. Always weigh the label and the science together, and ask the tough questions before adding anything new to your routine.
How should Sodium Selenite be stored?
Plain Truths About Sodium Selenite Risks
Sodium selenite has uses in supplements, animal nutrition, and even some scientific research. I’ve handled more than a few chemical bottles in my days working in labs, so keeping supplies safe has become second nature. It’s worth remembering: despite the benefits, this is a compound with real health hazards. The Centers for Disease Control and Prevention (CDC) identifies sodium selenite as toxic if swallowed or inhaled and as an irritant upon contact with skin or eyes. Product labels typically warn of health risks, but those often gather dust on the shelf like the chemical they’re supposed to protect you from. So, what should we actually do to keep sodium selenite safe?
Store It Like It Matters
Forget about just sticking it anywhere. Many old storerooms end up with chemicals shelved alongside paints, cleaning products, and even food supplies. The National Institutes of Health recommends a dedicated, labeled storage area for any toxic compounds, including sodium selenite. Mixing it up with less hazardous products invites accidents. From my own experience, adopting a “one shelf, one purpose” philosophy prevents a lot of confusion. If you catch a whiff of rotten garlic in an area with poor ventilation, that’s the time to audit your chemical storage.
Keep Things Cool and Dry
Proper temperature and humidity safeguards the stability of sodium selenite. Many compounds break down in the heat and moisture, and sodium selenite is no different. Leaving it where the temperature flares during summer or in a humid basement makes problems likely. Shield containers from sunlight, since some decomposition products could form otherwise. The U.S. Pharmacopeia recommends a dry, cool area—typically below 25°C (77°F). Dryness matters as moisture can create clumpy, unstable powders. Airtight containers keep water in the air at bay. Storing it this way has helped me avoid both ruined stock and unplanned reactions.
Chemical Hygiene Starts with a Good Label
Even after years handling chemicals, reading what’s in the jar saves mistakes. Clear, legible labels matter more than anybody likes to admit. Scratched, faded, or handwritten notes on tape can lead to costly mix-ups. Printing the full name, hazard statements, concentration, and date received creates a record that works when memory fails. The U.S. Occupational Safety and Health Administration (OSHA) backs this up with standard label requirements. This habit kept one coworker from mixing up two white powders in our lab—one handy label saved hours of paperwork and a potential ER visit.
Prepare for Accidents Before They Start
Every chemical presents some risk. Spills, broken containers, or accidental contact can happen to anyone, no matter how careful. Spilling sodium selenite calls for good gloves and quick cleanup—never leave it for tomorrow. Keeping cleanup kits close means anyone can handle a minor disaster without hunting through drawers. Having a practiced process, including safe disposal containers and knowing where emergency eyewash and showers are, lowers the stakes if something does go wrong. It’s not about paranoia; it’s about respect for what these substances can do.
Training and Oversight Make a Difference
Relying on written protocols without hands-on training misses the point. Every person who might handle sodium selenite deserves real guidance and a chance to practice safe storage and handling. Group walkthroughs and asking for feedback make the system better. My best lessons came from seasoned chemists sharing stories of things gone wrong—and how to avoid repeat mistakes. Honesty about the risks opens the door for smarter decisions and a safer workplace for everyone.
What is the recommended dosage of Sodium Selenite?
Why People Care About Sodium Selenite Intake
Sodium selenite has gained attention because it provides selenium, a mineral that the body uses for everything from immune support to thyroid function. Most people pick up their selenium from food, but in some situations, a supplement gets the nod—especially for those with special dietary restrictions, certain health conditions, or when soil in their area simply doesn’t hold enough selenium for local crops. Getting too little or too much can both lead to health problems.
Recommended Dosage for Adults
Research and public health guidance usually point to a recommended dietary allowance (RDA) for selenium, not sodium selenite specifically, since selenium can come from various forms. The RDA for adults sits at 55 micrograms of selenium daily. Pregnant people need a bit more, at 60 micrograms, and breastfeeding people look at about 70 micrograms. Most supplements, including sodium selenite, list the amount of elemental selenium they provide, not the weight of sodium selenite itself.
Doctors sometimes suggest higher doses for short periods, such as for someone with a proven selenium deficiency or for certain medical protocols under strict supervision. Here, the dosage can go higher, but always under the watchful eye of a health professional, because crossing into the territory of too much selenium risks selenosis—a toxic effect with symptoms like hair loss, stomach upset, and nerve issues.
Why Precision Matters
My own push to understand supplement safety grew after seeing friends self-medicate with minerals from the internet, based on loose advice. Too much selenium over time can sneak up with subtle symptoms. The U.S. sets the tolerable upper intake level for selenium at 400 micrograms per day for adults. The body doesn't need megadoses. Some people imagine that more equals better, but selenium works best in a narrow range—not too little, not too much.
People who work on farms or in environments with industrial exposure also have to keep an eye on total selenium intake, since they might face higher accidental exposure. Children need even stricter limits, as their developing bodies can’t handle high levels.
How to Approach Supplementation
Before picking up a bottle of sodium selenite, look at your diet. Foods like Brazil nuts, fish, meat, and grains offer plenty in most diets. Actual deficiency shows up in lab tests and with symptoms, not just guesses based on occasional tiredness. For anyone who suspects a gap in their nutrition, the smartest first step is checking in with a doctor. Blood work, not web searches, gives a clearer picture.
For people with kidney issues or on certain chemotherapy drugs, using sodium selenite supplements requires even more caution because of the risk of unwanted interactions or side effects. Supplements sold online don't always guarantee accurate labeling, so buying from trusted companies matters.
Solutions for Safer Selenium Use
Education about dietary needs should start with yearly checkups and stronger public health messaging. Food fortification offers a safety net in areas with low-selenium soil, as some countries have discovered. For individuals, using apps or tracked meal plans can help spot hidden sources of selenium and avoid accidental overdose from multiple supplements.
Reasonable use of sodium selenite supports good health. Open conversations with healthcare providers and awareness of the risks set the foundation for safer, evidence-based choices.
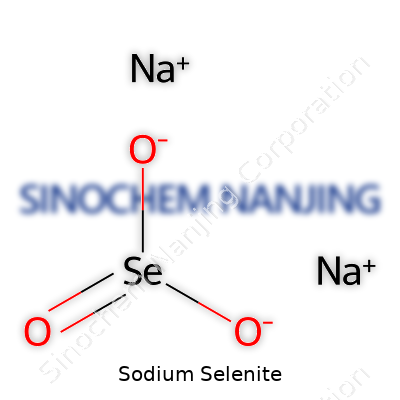
| Names | |
| Preferred IUPAC name | Sodium selenite |
| Other names |
Disodium selenite Selenious acid, disodium salt Sodium selenite pentahydrate Sodium selenite anhydrous |
| Pronunciation | /ˌsəʊdiəm səˈliːnaɪt/ |
| Identifiers | |
| CAS Number | 10102-18-8 |
| Beilstein Reference | 4039357 |
| ChEBI | CHEBI:13401 |
| ChEMBL | CHEMBL1386 |
| ChemSpider | 21162106 |
| DrugBank | DB11131 |
| ECHA InfoCard | 100.029.203 |
| EC Number | 231-892-1 |
| Gmelin Reference | Gm05800 |
| KEGG | C01758 |
| MeSH | D020564 |
| PubChem CID | 24935 |
| RTECS number | VV7400000 |
| UNII | 2F6VXO3UW0 |
| UN number | UN2630 |
| Properties | |
| Chemical formula | Na2SeO3 |
| Molar mass | 172.94 g/mol |
| Appearance | White or colorless crystalline powder |
| Odor | Odorless |
| Density | 2.62 g/cm³ |
| Solubility in water | Freely soluble |
| log P | -2.6 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 3.1 |
| Basicity (pKb) | 3.4 |
| Magnetic susceptibility (χ) | -42.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.509 |
| Viscosity | Low viscosity |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 127.1 J/mol·K |
| Std enthalpy of formation (ΔfH⦵298) | -711.4 kJ/mol |
| Pharmacology | |
| ATC code | A12CE02 |
| Hazards | |
| Main hazards | Toxic if swallowed. Causes severe skin burns and eye damage. May cause cancer. Very toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS07, GHS09, GHS06 |
| Pictograms | GHS06,GHS08,GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | P260, P264, P270, P273, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P311, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Lethal dose or concentration | LD50 oral rat 7 mg/kg |
| LD50 (median dose) | LD50 (median dose) Oral (Rat): 7 mg/kg |
| NIOSH | WH6785000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Sodium Selenite: 0.2 mg/m³ |
| REL (Recommended) | 0.03-0.05 |
| IDLH (Immediate danger) | 1 mg/m³ |
| Related compounds | |
| Related compounds |
Selenium dioxide Sodium selenate Selenous acid |

