Sodium Selenate: A Deep Dive into Its Journey, Properties, and Possibilities
Historical Development
Sodium selenate didn’t pop up overnight; its history weaves through the early days of selenium chemistry. Scientists first recognized selenium as a curious side product in copper refining in the nineteenth century. Sodium selenate, a compound made by oxidizing elemental selenium or selenides, gradually earned attention as researchers learned more about trace minerals and their biological roles. Early chemists focused on characterization and basic applications, not always aware of the element’s delicate interplay with living organisms. The trajectory changed as nutritionists and toxicologists documented selenium’s dual nature — essential in tiny doses, perilous in larger ones. Out of the crowded field of selenium salts, sodium selenate became a go-to for supplement manufacturers, laboratory chemists, and industry, carving out a place for itself in agriculture, animal science, and analytical chemistry.
Product Overview
Sodium selenate usually presents itself as a white, very soluble powder or crystalline solid. Used in micronutrient mixes, glass coloring, and research, it attracts industries that need precision and reliability in their mineral ingredients. Labs use its standardized purity and well-understood reactions, as well as its predictable behavior in solutions, to dose selenium accurately into animal feed, fertilizer, or supplements. Its powerful oxidizing ability, and ease of measurement, bring consistency to applications where small errors could tip the selenium balance from helpful to harmful. Brands and supply chains value the tight controls around its preparation so that every batch lands within safe and useful ranges.
Physical & Chemical Properties
Sodium selenate (Na2SeO4) carries a molar mass of 188.94 g/mol and looks like a white, odorless, and granular or powdered solid, easy to mistake for plain table salt at a glance. It dissolves well in water and starts to decompose when it gets too hot, above 400 °C, releasing toxic selenium oxides. The chemical structure features a selenium atom at the center surrounded by four oxygen atoms, forming a highly oxidized oxyanion that partners with two sodium ions for charge balance. The high solubility means sodium selenate disperses quickly in water-based systems, whether in a scientific beaker or sprinkled across a crop field. The compound doesn’t vaporize or emit fumes under normal conditions, but mixing it with acids or reducing it with strong agents leads to further chemical shifts, some of them risky from a safety perspective.
Technical Specifications & Labeling
Companies that handle sodium selenate list its purity levels, often above 98% for pharmaceutical or laboratory use, and spell out water content since hygroscopic materials can pick up moisture from the air. Labels always include hazard statements, usually referencing the compound’s acute toxicity and environmental dangers if spilled. Standard labeling highlights batch number, expiration date, country of manufacture, and instructions for storage — typically in tightly sealed containers away from incompatible chemicals like strong acids. Material Safety Data Sheets (MSDS) follow regulatory rules, pointing out short- and long-term health risks, proper first aid, and spill response steps. For products shipping internationally, container markings and customs documents reference sodium selenate’s CAS number (13410-01-0) and Transport UN codes. Any supplement or additive based on sodium selenate must follow relevant food or feed regulations and never stray outside strict tolerance limits.
Preparation Method
Most commercial sodium selenate comes from a controlled reaction between selenium dioxide and sodium hydroxide. Factories expose metal selenide ores to roasting, which forms selenium dioxide gas. Chemists then dissolve the selenium dioxide in water and neutralize it with sodium hydroxide or sodium carbonate, resulting in a clear solution of sodium selenate. This liquid goes through evaporation and crystallization, then filtration and drying, to harvest pure selenate for packaging. Because selenium compounds can harm workers and the environment, facilities use fume collection, wastewater treatment, and constant monitoring. Compared to raw extraction of selenium, producing selenate involves more steps, but these procedures reduce impurities to trace levels, readying the product for stringent applications.
Chemical Reactions & Modifications
Sodium selenate reacts easily with reducing agents, forming compounds like sodium selenite or elemental selenium. Add a strong acid and the selenate ion can break down, releasing selenium dioxide or even elemental selenium with proper reducing conditions. In the lab, sodium selenate can act as an oxidizing agent, helping convert sulfides to sulfates in complex mixtures or nudging reactions toward completion in organic synthesis. Its high solubility allows for fast diffusion throughout liquid cultures, so researchers often pick sodium selenate as a selenium source in cell biology and soil science. Layer on chemical modification — for example, partial reduction or doping with other metal cations — and sodium selenate’s role stretches into advanced material synthesis, battery studies, and environmental remediation projects.
Synonyms & Product Names
People working with sodium selenate may see it listed under several names, like disodium selenate, selenic acid disodium salt, or sodium salt of selenic acid, depending on the catalog or supplier. Shortened names like “Selenate” usually mean the sodium salt in industrial and agricultural settings, though precision matters to avoid confusion with the related but less oxidized sodium selenite. Trademarks and proprietary blends for agriculture sometimes disguise sodium selenate as part of a larger mixture, so end users should always check ingredient lists, not just sales names.
Safety & Operational Standards
With sodium selenate, safety means more than wearing gloves and goggles. Even brief exposure to dust or droplets of the compound can cause acute symptoms, like nausea, metallic taste, or breathing trouble. Chronic low-level exposure increases risks for liver and kidney problems, and animal studies warn of developmental impacts. Automated dispensing, fume hoods, robust ventilation, and tight storage routines help keep workplace exposures low. Companies train staff on quick spill cleanup, medical response, and never eating or drinking near active use zones. Waste and washwater go through treatment, usually involving reduction and precipitation, before landfill or discharge. Transport regulations classify sodium selenate as hazardous, triggering extra paperwork, labeling, and route planning. Modern workplaces push for substitution with safer alternatives whenever possible, but in selenium-deficient regions, sodium selenate provides a lifeline for animal and plant health under controlled, science-backed conditions.
Application Area
Farms in selenium-poor soils lean on sodium selenate fertilizer to raise healthy cattle, sheep, and crops, since a minuscule dose corrects devastating deficiencies. Feed and supplement manufacturers use it to guarantee trace selenium content, supporting animal immunity, reproductive health, and growth. Water treatment operators sometimes draft sodium selenate into specialist roles, handling certain contaminants by targeted oxidation or selective removal. In scientific research, sodium selenate serves as a stable selenium donor, delivering accurate doses in cell culture, microbial tests, or genetic studies. Glassmakers exploit its ability to decolorize or tint, depending on how the selenate ions interact with other trace metals and furnace conditions. Since public health agencies monitor selenium exposure closely, only trained experts add sodium selenate to food or feed, following local law and scientific advice.
Research & Development
Academic labs and company R&D teams still turn to sodium selenate as a building block in trace element research. The focus often falls on comparing its biological impacts to those of other selenium compounds, tracking how different forms move through soil, water, and food webs, or designing new sensors to pick up sub-ppm contamination. Scientists explore formulations where sodium selenate works in slow-release pellets, nano-encapsulated powders, or combined with other minerals to match regional deficiencies. Medical researchers take interest in its ability to influence certain cancer cell lines or counteract oxidative damage, though such uses face extra hurdles due to safety and dosage concerns. Some inventors experiment with biofortification, mixing sodium selenate into foliar sprays for crops that end up richer in dietary selenium, investigating whether these practices can boost long-term public health without building up toxic residues.
Toxicity Research
Toxicology studies underline sodium selenate’s razor-thin margin between required and risky. Ingesting just a gram or two sometimes proves fatal for adults, and much less for children or pets. Animal and cell studies find that excess selenate disrupts key metabolic enzymes, causes oxidative stress, and builds up in soft tissues instead of flushing out harmlessly. Regulatory limits land in the range of a few micrograms per kilogram of diet or body weight, based on decades of adverse effects data. Chronic exposure links to hair loss, nail changes, neurological effects, and, in severe poisoning, fatal organ failure. Environmental scientists record quick transfers of selenate from soil to plants to grazing animals, which adds urgency to monitoring and safe application practices. Product makers invest in stable formulations and clear dosing instructions to keep benefits front and center and risks at bay.
Future Prospects
Outlook for sodium selenate rides on responsible use and judicious regulation. Precision farming, personalized animal nutrition, and demand for functional foods may prompt more sophisticated delivery systems that minimize environmental runoff. Researchers test whether silica coatings or biopolymer wraps slow down the mineral’s release and cut the danger of excess exposure. Data science and remote sensing could fine-tune how, when, and where to apply mineral supplements like sodium selenate, balancing plant and animal health with ecosystem safety. In clinical research, a few teams dig into selenate’s possible role in countering neurodegenerative diseases, tracing effects on protein folding and inflammation, but clinical progress relies on proving both benefits and air-tight safety controls. As industry and science seek better ways to detect, measure, and apply selenium, sodium selenate occupies a fragile but potentially powerful place in the toolkit — never to be used lightly, always with skill and diligence.
What is Sodium Selenate used for?
Understanding Where Sodium Selenate Matters
People often overlook minerals like selenium, only noticing them once something goes wrong. Sodium selenate isn’t usually talked about over breakfast. Yet, this compound crops up in fields, laboratories, and even in the food chain. My own background in environmental science brought it to my attention early—after seeing the impact of trace minerals on both crops and public health.
Essential for Agriculture
In farming, sodium selenate often acts as a lifeline for soils that lack selenium. Cows or sheep grazing on selenium-poor land end up with health issues, and that trickles back to the farmer. Sodium selenate helps by being water-soluble, letting it absorb into the soil so plants can take it up. This matters, since food crops grown on those soils also move selenium up the food chain. Without some intervention, selenium deficiency can threaten both livestock health and crop safety. One fact stuck with me: Researchers at Oregon State University found that selenium deficiency can slash lamb survival rates and slow down cattle growth. Land managers don’t consider this a minor issue.
Selenium in Food Fortification
People sometimes don’t get enough selenium in their diet. That's more common in places with low-selenium soils. Sodium selenate steps in here too. Some governments require the fortification of items like table salt or flour with selenium. The additive form usually gets blended in tiny quantities, but it makes a world of difference for population health. I remember Australian news covering how wheat fortified with selenium helped improve outcomes in regions where rare, selenium-responsive heart disease cropped up. The solution turned out practical and cheap.
Scientific and Medical Uses
Researchers reach for sodium selenate in the lab. One wave of studies in Australia and Canada looked at its effect on proteins that form tangles in Alzheimer’s disease. There’s still a long road ahead before those findings become medicine, but the research keeps scientists interested. Sodium selenate also plays a part in studies on enzyme activity and cell health. Sodium selenate’s reliable chemistry lets scientists measure things with precision, which makes it more than just a basic ingredient—it lets them ask bigger questions about disease and nutrition.
Gaps and Risks
Sodium selenate sits in a tricky place. Too little selenium in food or livestock diets causes problems, but too much brings toxicity. People living near rivers or drainage flows from treated fields have seen issues pop up in fish and birds—especially where runoff isn’t managed well. One study from California’s San Joaquin Valley pointed straight at selenate runoff as a culprit in bird deformities. This highlights a reality: simple use in agriculture or food isn’t risk-free. Authorities and farmers need systems to measure and tweak selenium applications often. I wish more farm advisors stressed testing before treatment.
Looking Ahead
Bridging the gap between need and risk with sodium selenate calls for smarter management. Farms can use soil tests and slow-release fertilizers to match actual need instead of just dosing broad acres. On the public health side, tracking selenium intake through food surveys helps avoid accidental excess. Investment in research will help tune dosing, and fresh regulations can step in when trouble shows up in the environment. Sodium selenate, handled with respect, works as a vital tool for both food production and health. Its careful use hinges on science and vigilance, not just routine.
Is Sodium Selenate safe for human consumption?
Looking at What Sodium Selenate Really Is
Sodium selenate pops up in discussions about trace minerals. It belongs in the same family as selenium, an essential micronutrient your body actually needs to function. You’ll find selenium in seafood, Brazil nuts, eggs, and meat. Sodium selenate is a man-made compound, often used in fortifying cereals or animal feed, or even considered for treating certain medical conditions under strict supervision. Since the line between nutrient and poison in trace minerals stays razor thin, how safe is this chemical form?
The Line Between Necessary and Harmful
Everyone needs selenium. Your thyroid relies on it, and it helps your body clean up free radicals. Trouble comes with the margin of safety. The recommended daily amount for an adult is about 55 micrograms. Jump much above 400 micrograms and the risks start to climb, with hair loss, stomach upsets, shaky hands, and even nerve damage. Cases of selenium poisoning, like those linked to contaminated supplements in the United States in 2008, caused severe problems when daily intake shot above safe thresholds.
Facts and Regulatory Standards
Agencies like the U.S. Food and Drug Administration and European Food Safety Authority keep a close eye on how much selenium people should get. They take into account forms like sodium selenate, setting limits to prevent toxicity. Food fortification and supplements must follow legal thresholds. In practice, most folks eating a balanced diet in North America or Europe won’t see selenium deficiency or excess—unless they lean hard into supplements or work with industrial sources.
My Direct Observations from the Field
As someone who has spoken with dietitians over the years and tracked public health advice, I’ve seen both sides. On one hand, those who miss balanced meals for too long sometimes end up with low selenium, especially in regions where soil runs depleted. On the flipside, health trends push some people to take unnecessary supplements, often believing “more is better.” That’s when incidental toxicity can sneak up—a risk not just with sodium selenate, but many single-mineral supplements.
Why Purity, Dose, and Source Matter
Not all selenium sources act the same in your body. Organic types, like selenomethionine from plants, process differently compared to the inorganic sodium selenate. The latter absorbs well but doesn’t linger in tissues as long. In medicine, high doses only get used with close monitoring. Self-dosing is where risks show up: loose regulation in certain supplement markets, unclear labeling, and lack of awareness may lead to people getting far more than their bodies can handle. A 2014 study in Clinical Toxicology chronicled an outbreak of selenium poisoning from a misformulated supplement that used sodium selenate—demonstrating how having the right form at the right dose prevents harm.
Smart Approaches and Possible Solutions
Healthcare providers should encourage people to look for food sources before reaching for extra pills. If a supplement becomes necessary, transparency, third-party quality assurance, and clear labeling limit confusion. Policy makers can reinforce this with stricter labeling for selenium content, and public education campaigns explaining why ‘more’ isn’t always wise. In agriculture or animal feed, monitoring residue levels ensures the food supply stays within safe windows.
Final Thoughts on Safety and Use
Sodium selenate in controlled doses supports health. Once exposure drifts above the safe range or comes from unknown sources, safety vanishes fast. People should ask their doctor before taking selenium supplements and remember that food delivers not just selenium, but the complex web of nutrition that keeps bodies running right.
What are the side effects of Sodium Selenate?
Getting to Know Sodium Selenate
Anyone who's handled micronutrients in labs, farming, or health care has probably come across sodium selenate. This compound delivers selenium, an essential trace element, but in the wrong dose or setting, sodium selenate flips from helpful to harmful. Despite coming with some benefits in small amounts, its side effects deserve just as much attention, especially among people considering supplements without proper guidance.
Common Side Effects and Why They Happen
Nausea comes up a lot for anyone who’s taken higher doses. The body sees an overload of selenium, and the gut reacts almost immediately. I’ve seen this first hand working with folks at a community pharmacy. Some tried boosting their intake after reading about selenium’s role in thyroid health or cancer prevention—and the sick feeling kicked in faster than any results they were hoping for. Stomach pain, vomiting, and diarrhea can follow.
Skin reactions can catch people off guard. A garlic odor on the breath and skin doesn’t sound dangerous, but it signals selenium building up in the body—a warning that’s easy to miss unless you know the pattern. Hair loss can sneak up over weeks or months, sometimes paired with brittle fingernails riddled with white spots. These are not everyday supplement side effects; they’re signals your intake is overshooting safe boundaries.
Neurological symptoms can worry anyone. Studies, including research published in the Journal of Clinical Toxicology, describe dizziness, irritability, and sometimes confusion. I remember a middle-aged client reporting feeling “foggy” and moody after what he thought was a routine supplement dose. Blood tests told the story: selenium levels far above the recommended range.
Who Faces the Biggest Risks?
People with kidney problems can’t clear extra selenium efficiently. This wasn’t obvious to many people I’ve helped until they mentioned existing medical conditions. Toxicity risk rises as the mineral stays stuck in the system, amplifying symptoms more quickly.
Kids and older adults get a stronger punch too. Their bodies process minerals less efficiently, so what looks like a minor dose for an adult in good health may become a real problem for them.
Supporting Evidence: What the Data Shows
The U.S. National Institutes of Health pegs the tolerable upper intake level for selenium at 400 micrograms per day. Going above that even for a few weeks can raise the odds of side effects. Documented cases report hair loss, nerve damage, and digestive issues—all tied to dosages over this mark.
Long-term exposure deserves a hard look. In rural areas where groundwater contains too much selenium, researchers have tracked communities with brittle nails, skin rashes, and even chronic fatigue. These aren’t short-term problems; they point to the lasting cost of ignoring dose limits.
What Helps: Managing the Downsides
Talking to a doctor or dietitian about supplements isn’t wasted time. Medical professionals look at your full health picture—medications, organ function, diet—before suggesting selenium intake. Lab testing supports safer use.
Reading labels and understanding dosing instructions matter more than most folks expect. Supplements vary in strength and serving size. The “more is better” approach never works here. Balance requires accurate measurements and trusted sources.
If you do notice symptoms like stomach upset or odd smells, stopping the supplement and seeking advice makes a world of difference. Managed early, selenium toxicity often reverses over time. Never ignore symptoms in children, pregnant women, or those with chronic illnesses; their risk just runs higher.
How should Sodium Selenate be stored?
Experience Reminds Us: Safety Gaps Spell Trouble
Sodium selenate serves important roles in agriculture, glass manufacturing, and chemical research—but its benefits don’t erase the serious risks that come with careless handling. One lesson that gets repeated, both in high school chemistry labs and any industrial stockroom, is just how quickly a day can turn from routine to emergency. A careless container placement, a missed leak, or some heat in the wrong place—these little shortcuts can add up to health hazards and environmental guilt.
Walking through a storage area, I've seen far too many close calls. Spills, broken seals, and cross-contamination between incompatible substances still happen, especially where people rush or take shortcuts. Sodium selenate’s dangers come from both toxicity and reactivity. Getting the storage wrong isn’t just an inconvenience; it can make people sick and land businesses in regulatory hot water.
Containers Matter: Tough, Tight, and Clearly Labeled
Forget improvising with leftover bottles or questionable bags. Store sodium selenate in sturdy, tightly sealed containers—glass is best for lab use, but chemically resistant plastic works in bigger facilities. Anything less can let moisture or air sneak in, and sodium selenate doesn’t just sit quietly when that happens. Exposure to water and acids can produce toxic selenium fumes, and any residue on skins or surfaces can be dangerous over time.
Labels should jump out. Nothing’s worse than mystery dust in an unmarked jar—routine labeling mistakes can lead to someone mixing the powder with the wrong substance or not understanding what they’re handling.
Temperature and Light: Cool, Dry, Dark Wins Every Time
Some people treat temperature control like an afterthought, but keeping sodium selenate cool and dry keeps it stable. Heat speeds up chemical reactions, and even “room temperature” can be too much in a warehouse during summer. Moisture is another enemy—dry spaces with desiccants nearby help keep clumps and breakdowns at bay. Direct light only adds to instability, slowly breaking down the compound and reducing safety.
No Loose Rules on Ventilation
Good ventilation separates safe sites from unsafe ones. Open shelves in tight spaces? That’s a recipe for trouble if anything leaks. Fume hoods and ventilated cabinets give an extra layer of protection. In bigger facilities, dedicated chemical storage rooms with filtered airflow keep workers and visitors from breathing in toxins.
Keep Friends Close, Incompatible Chemicals Far Away
One of the simplest storage missteps: putting incompatible substances too close together. Store sodium selenate away from acids, strong oxidizers, and foods or animal feeds. It’s tempting to group powders for convenience, but this habit can start a chain reaction if containers get damaged.
Training and Documentation Build Lasting Habits
Most accidents happen because someone didn’t know better or took a shortcut on safety steps. Routine training helps keep old habits in check and makes sure new team members know the risks from day one. Good recordkeeping—tracking storage dates and checking expiration—helps prevent waste build-up and keeps inspections smooth.
Simple Changes Save Lives
Storing sodium selenate safely comes down to respect for the chemical and the health of everyone using the facility. Well-maintained storage, regular safety reviews, and a willingness to call out risky habits make all the difference. From science classrooms to industrial stockrooms, these steps aren’t just best practices—they’re lifelines.
What is the recommended dosage of Sodium Selenate?
Understanding Sodium Selenate
Talking about sodium selenate feels different from most daily supplements. This isn’t something you just pick up at the pharmacy and take with breakfast. Sodium selenate brings real benefits but even more real risks if misused. As someone who’s worked with nutrition professionals and read through more than a few clinical guidelines, I’ve seen how a micronutrient can turn into a double-edged sword.
Recommended Dosage and Current Guidelines
The mainstream consensus among established sources—like the National Institutes of Health—places a safe level of elemental selenium intake at 55 micrograms per day for most adults. Sodium selenate is a salt form, so a milligram of it doesn’t equal a milligram of pure selenium. About 44% of its mass is elemental selenium. If someone’s taking sodium selenate specifically, a simple calculation gives the right amount to match the 55-microgram target. Eating a Brazil nut or two might hit the recommendation, but supplements need closer attention. The World Health Organization and other bodies cap the total daily selenium limit at 400 micrograms to avoid toxicity.
Risks of Overdoing It
It only takes a small dose above the safe range to start seeing dangerous effects, everything from hair loss to nerve damage. I’ve seen research that chronic overdose—even as low as 400 micrograms a day—can cause selenosis. It doesn’t always show itself quickly, which makes it especially sneaky. The body can’t just flush out excess selenium, so it builds up over time. Some stories in the medical literature come from supplements with labeling problems or mistakes made by companies during manufacturing. One batch error is all it takes. Real people ended up in hospitals.
Uses Outside of Nutrition
Sodium selenate has been put to the test against certain diseases, such as Alzheimer’s and cancer, in experimental clinical trials. Doses used by researchers go way above what’s considered safe for daily intake, but these situations involve rigorous supervision. No one should self-prescribe the levels used in these studies. If someone stumbles across a forum post suggesting miracle results from high-dose selenium, turn to a real evidence-based source and talk to a healthcare professional.
Quality and Source Really Matter
Supplements can vary. One bottle might contain much more or less than the label claims. Organizations like the U.S. Pharmacopeia or NSF certify some brands, but not all products get this seal. Quality control gaps still show up, and regulators have caught contaminated or misformulated products on the shelf within the last decade. Not every supplement marketer follows the rules. My advice—if you need sodium selenate, don’t order from a random online shop. Ask a doctor or pharmacist for options with proven track records. If you’re already eating a mixed diet with seafood, grains, eggs, or Brazil nuts, you probably get enough selenium without added risk.
Safe Usage Means Partnering with Experts
If sodium selenate is part of a treatment plan—as in some very specific cases involving metabolic or neurological conditions—the dosing always comes from a specialist. Self-medication just doesn’t work here; mistakes hurt people. Medical supervision includes regular blood checks for selenium and liver function. It still blows my mind how easily overlooked a “supplement” can spike up health risks.
Moving Forward
There’s no magic shortcut—find your information from clinical sources, speak with someone who understands the difference between supplement labels and elemental selenium, and remember the line between sufficient and toxic remains thin. That’s the reality for sodium selenate: useful when handled with respect, harmful when misunderstood.
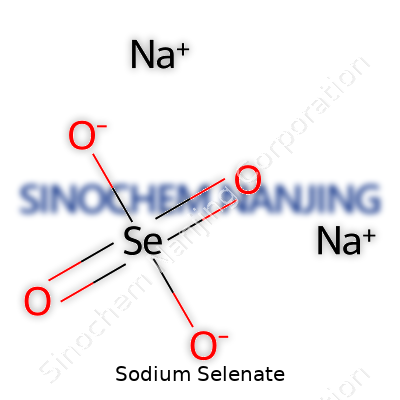
| Names | |
| Preferred IUPAC name | Sodium selenate |
| Other names |
Disodium selenate Selenic acid, disodium salt Sodium selenate(VI) Selenate de sodium |
| Pronunciation | /ˌsəʊdiəm səˈliːneɪt/ |
| Identifiers | |
| CAS Number | 13410-01-0 |
| Beilstein Reference | 1303280 |
| ChEBI | CHEBI:48841 |
| ChEMBL | CHEMBL1201811 |
| ChemSpider | 61930 |
| DrugBank | DB11131 |
| ECHA InfoCard | 100.030.036 |
| EC Number | 231-987-8 |
| Gmelin Reference | 6268 |
| KEGG | C18616 |
| MeSH | D018080 |
| PubChem CID | 25156 |
| RTECS number | VS8750000 |
| UNII | 7Q966D49RG |
| UN number | UN2630 |
| Properties | |
| Chemical formula | Na2SeO4 |
| Molar mass | 188.94 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 3.098 g/cm³ |
| Solubility in water | Soluble |
| log P | -4.3 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 2.0 |
| Basicity (pKb) | 1.92 |
| Magnetic susceptibility (χ) | -57.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.384 |
| Viscosity | Viscous liquid |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 125.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -889.7 kJ/mol |
| Pharmacology | |
| ATC code | A12CE02 |
| Hazards | |
| Main hazards | Toxic if swallowed. Causes damage to organs. Very toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS05, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | P210, P220, P221, P261, P264, P270, P271, P273, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P313, P311, P330, P361, P370+P378, P391, P403+P233, P405, P501 |
| Lethal dose or concentration | LD50 Oral Rat 1.6 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1.6 mg/kg (oral, rat) |
| NIOSH | TE7700000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Sodium Selenate: "0.2 mg/m3 (as Selenium, OSHA PEL, 8-hour TWA) |
| REL (Recommended) | 0.15 mg/kg |
| IDLH (Immediate danger) | 1 mg/m³ |
| Related compounds | |
| Related compounds |
Sodium selenite Selenium trioxide Sodium sulfate Sodium tellurate |

