Sodium Salicylate: A Commentary on Its Journey and Importance
Historical Development
Sodium salicylate traces its roots back to a time before aspirin took over the medicine cabinets. It has long served as a key pain and fever remedy, especially once chemists figured out how to extract the compound from willow bark and then synthesize it reliably in labs in the late 1800s. This little white powder opened doors for everyday people who suffered from rheumatic pain and headaches, showing just how important chemical innovations can be for human health. Long before tablets came in neat foil packs, sodium salicylate was a practical, affordable answer for ordinary people. Its prominence shaped the course of drug development, as pharmacists and doctors watched it relieve discomfort that previously seemed inescapable. Knowing the roots of sodium salicylate reminds us how scientific discovery transforms daily life, especially when the benefits reach far beyond research journals and classrooms.
Product Overview
Chemists describe sodium salicylate as the sodium salt form of salicylic acid, packing analgesic, anti-inflammatory, and antipyretic properties. In practice, it’s a solid white, almost sugar-like powder with a slightly bitter taste. It dissolves well in water, which made it useful not only for oral medicines but for topical preparations and even as a base in dyes and preservatives. Decades ago, manufacturers appreciated its balance of effectiveness, safety, and low cost, and that shaped the habits of countless doctors and patients looking for help treating pain and swelling.
Physical & Chemical Properties
Pure sodium salicylate appears as a white crystalline powder. It melts at around 200 degrees Celsius before it breaks down. In water, it doesn’t resist, making preparations for ingestion and mixing fairly straightforward. The chemical formula—C7H5NaO3—gives it a molecular weight of about 160.1 g/mol. It feels smooth to the touch and has a faint but distinct odor. It’s stable under dry conditions, but exposure to moisture can clump the powder or alter its appearance. In the lab or production line, these physical traits help producers store, ship, and use sodium salicylate without dealing with complicated temperature and humidity controls. This convenience gives it a clear advantage over some pain relievers and chemical agents that break down or react at room temperature.
Technical Specifications & Labeling
Pharmaceutical-grade sodium salicylate must display high purity, usually above 99%. The labels on each batch list out not only the percentage of active compound but also water content, trace solvents, and carriers. Proper labeling details storage rules: keep sealed, dry, and away from direct sunlight. No shortcuts exist when safety and consistency matter to hospitals, researchers, or mass-market drug producers. Anyone who’s filled a prescription knows that clear, concise, and accurate labeling spells the difference between safe healing and accidental misuse. The best practices here come from years spent in the field, where missing a detail in labeling led to headaches for pharmacy staff and negative outcomes for patients.
Preparation Method
Manufacturers prepare sodium salicylate through a neutralization reaction between sodium hydroxide and salicylic acid. Technicians add the two ingredients in water, adjusting temperatures to encourage optimal reaction rates. Once the reaction finishes, they cool the solution and let sodium salicylate crystals form. Separation, purification, and drying steps follow. The yield depends on purity both of starting materials and how precisely operators control pH and temperature. In an industry where waste means lost profit and environmental risk, fine-tuning methods has always mattered. Those with experience on production floors or research benches recognize that simple chemistry still requires patience, quality checks, and skilled hands.
Chemical Reactions & Modifications
Sodium salicylate participates in a variety of chemical reactions, most notably forming esters and undergoing nitration. The carboxyl group (COOH) can react with alcohols, producing salicylate esters used in cosmetics or flavoring agents. Nitration leads to compounds used in chemical synthesis and laboratory research. The phenolic OH group allows coupling with other active pharmaceutical ingredients, expanding the range of drugs built on salicylate backbones. Labs around the world have tested tweaks to this molecule, hoping to reduce toxicity or open up new uses. Chemical flexibility means scientists rely on sodium salicylate as a stepping stone to push their discoveries into uncharted territory, whether they’re searching for better medicines or stronger preservatives.
Synonyms & Product Names
Over the years, sodium salicylate earned a number of alternative names, such as “Sodium o-hydroxybenzoate,” “Salicylate of soda,” and “Monosodium salicylate.” Drug labels, industrial suppliers, and academic journals might use any of these, though product purity and intended use often clarify which one gets used. Familiarity with these names matters most for those handling international shipments, imports, or research samples. A miscommunication here disrupts workflow and can even threaten patient safety, showing once more that language and tradition matter as much as modern science.
Safety & Operational Standards
Every workplace retaining sodium salicylate follows careful safety protocols. Direct skin contact can cause mild irritation, while ingestion or inhalation in powder form irritates mucous membranes and can potentially lead to harmful side effects with enough exposure. Personal protective equipment—gloves, masks, goggles—remains non-negotiable. Storage standards require dry, airtight containers away from acids or strong oxidizers. In my years working around lab chemicals, the best safety records came from teams that prioritized training and respected Material Safety Data Sheet guidelines without cutting corners. When users pay close attention to these details, workplace injuries decrease and confidence in finished products rises.
Application Area
Sodium salicylate finds a home in more than just medicine. Aspirin took center stage for oral use, but topical analgesics, toothache creams, and even veterinary solutions still call for sodium salicylate. Its chemical stability and compatibility also attract dye and pigment manufacturers, where it acts as a preservative or catalyst. Historians and chemists alike recognize that its reach goes beyond one or two industries, making it a quiet workhorse in textile workshops, photographic labs, and agriculture. Watching these shifts makes one appreciate how a chemical’s story doesn’t end with its first intended purpose. As regulations change and new needs arise, sodium salicylate adapts without fanfare.
Research & Development
Research into sodium salicylate hasn’t slowed, even with newer drugs on the market. The molecule’s ability to disrupt inflammation signals has drawn interest from teams examining autoimmune diseases and cancer. Modifications to its chemical structure have led to derivatives with altered activity, safer toxicity profiles, or extended drug release times. At scientific conferences, experts still debate the best delivery methods or explore new fields like biodegradable packaging or antimicrobial coatings. From my own time attending these events, it becomes obvious that progress stems from balancing tradition with bold leaps forward. No technology remains static; sodium salicylate proves that by evolving with each wave of research.
Toxicity Research
Toxicologists study sodium salicylate’s effects closely, as overuse or cumulative exposure leads to a condition known as salicylism—marked by ringing ears, dizziness, nausea, and more severe outcomes if left unchecked. Scientists use animal studies, cell cultures, and long-term tracking data to fine-tune safe dosage recommendations. Monitoring these side effects isn’t just for regulatory approval; it allows doctors to tailor treatments for children, the elderly, and sensitive patients. Some formulations combine sodium salicylate with buffers or other agents to further reduce risk. No chemical used in healthcare escapes scrutiny, and consumer safety groups continue to push for lower exposure limits and clear warnings. My own reading of case reports underscores how every tool in the medicine chest must balance help with harm.
Future Prospects
Sodium salicylate stands poised for further roles, despite being little changed at its core. Scientists are testing its effects in metabolic syndrome, neurodegenerative diseases, and even as an agent for water purification. Manufacturing upgrades will aim for greener, more sustainable processes, as regulatory bodies push for lower environmental impact. If history serves as a lesson, the right mix of academic vigor and industrial know-how will carry sodium salicylate forward. That journey continues, not as a relic of the past, but as a launchpad for the next generation of inventions that address pain, infection, and more. Having seen the power of collaboration between science and industry, I think sodium salicylate’s story remains one to watch.
What is Sodium Salicylate used for?
A Closer Look at a Familiar Compound
Sodium salicylate sits on the shelf in many labs and factories, but most people outside chemistry circles don’t know much about it. In my own work with pharmaceuticals and food products, this compound pops up more often than folks might guess. Chemically, it comes from the salicylate family, the same group where aspirin gets its punch. That shared heritage means you’ll find sodium salicylate in a stack of different uses.
A Reliable Ally in Pain Relief
Sodium salicylate pulls its weight as an analgesic. I’ve seen it used in medicines for aches, fevers, and inflammation. It works by blocking certain enzymes that trigger pain and swelling in the body. Some people can’t tolerate aspirin because of stomach issues or allergies. For them, sodium salicylate can serve as a gentler alternative. This kind of substitution matters, since the World Health Organization lists salicylate-based medicines among the essential drugs globally.
The Quiet Workhorse in Industrial Processes
This compound helps create dyes, flavors, and preservatives. In the work I’ve done with food labs, I’ve seen sodium salicylate act as a chemical stabilizer and preservative for a longer shelf life. Lab technicians sometimes use it to test for trace metals like iron in water samples because of its clear reaction and color change. Factories appreciate how well sodium salicylate dissolves in water, making it easy to blend into all sorts of mixtures, from antipyretic tablets to food flavorings.
Importance in Testing and Research
In hospital laboratories, sodium salicylate aids in diagnostic tests. It binds with proteins, so it’s useful in identifying conditions where protein levels drop or spike. For chemists or students learning about organic reactions, this compound helps demonstrate protein-chemical interactions in a dependable, low-cost way. In research environments, costs add up quickly— so anything that’s versatile, affordable, and safe to handle gains trust.
Pitfalls of Overuse and Safety Concerns
No conversation about chemicals in medicine and food skips safety. High doses or chronic exposure to sodium salicylate can lead to side effects such as ringing in the ears, stomach upset, or even kidney issues. In my time in pharmaceutical quality review, we tracked reports of accidental overdoses or misuse with extreme care. The FDA tightly regulates how much goes into over-the-counter drugs, limiting abuse or poisoning. Anyone with kidney trouble or a history of allergies should ask a doctor before taking products containing this compound.
Looking Toward Better Solutions
Years of experience in both product development and safety oversight tell me we need more research on long-term effects, especially with kids or vulnerable populations. Manufacturers should consider clear labeling and lower-dose formulations wherever possible. Educational outreach helps keep the public from self-medicating carelessly. As more food makers and pharmaceutical companies explore sodium salicylate’s benefits, responsibility must come first.
Grounded Use and Forward Thinking
Sodium salicylate does more than most folks guess. It eases pain, boosts industrial production, and advances science. Like with so many things, balance and careful oversight make the difference between a helpful tool and a risk. Trust in the product comes from solid evidence, open communication, and ongoing refinement of safety measures.
Is Sodium Salicylate safe for human consumption?
What Sodium Salicylate Actually Is
Sodium salicylate often shows up in the world of over-the-counter medicines. It's a salt form of salicylic acid, a chemical cousin to aspirin. For a long time, people have reached for it to ease pain and calm inflammation. You might spot it in painkillers, fever reducers, and some older cold remedies. Lab scientists sometimes use it for other things, but regular folks run into it most often with medical products or even in some food preservatives.
Why Safety Matters Right Now
Safety comes up because, like many chemicals, sodium salicylate can land on either side of the risk scale based on how and why somebody uses it. I remember my grandmother always having a tiny bottle of a salicylic remedy on her kitchen table for headaches. She’d take it without much thought. Back then, people just trusted what was on pharmacy shelves, but information and access have changed.
Modern research and medical guidelines have turned a sharper eye toward what happens inside the body. Sodium salicylate works because it reduces the chemicals behind inflammation and pain. But the line between helpful and harmful gets thin if people ignore dosages or if certain folks swallow it without guidance.
Proven Risks and Problems
Health authorities such as the FDA pay close attention to products containing sodium salicylate. Studies show that even though the compound shares the benefits of aspirin, it also shares the downsides, especially for those sensitive to salicylates or at risk for stomach trouble. High doses can irritate the stomach lining, raise the risk of ulcers, bleeding, or even kidney stress. Kids and teens recovering from viral infections should avoid salicylates due to the rare but scary risk of Reye’s syndrome—a fact hard to ignore after hearing some tragic stories from families affected in the past.
Allergies or asthma sometimes get worse after exposure. I’ve seen more than one friend reach for a pain reliever without knowing that “salicylate” in the ingredients can mean a rapid trip to the ER. Even for people without these problems, taking it alongside other blood-thinners or certain meds can stack up the dangers.
Who Watches the Store?
Regulators such as Health Canada and the European Food Safety Authority check the limits for sodium salicylate as an additive and drug. Foods and drinks rarely have it these days, since alternatives have replaced it as a preservative. Mainstream medicines include strong warnings and dosing info. Doctors learn to spot risks—thanks to researchers who dig into side-effect reports and population data.
Many consumer watchdogs stress that reading labels and asking a pharmacist can clear up doubts before mistakes happen. Parents, in particular, take extra steps to shield younger children from any product with salicylates.
Steps Toward Safer Choices
Facing confusion in the drug aisle, people could benefit from easier access to plain-language guides. Packaging updates help, but health care teams play a big role too. My own experience with pharmacists saved me from mixing up medications more than once—especially for someone juggling prescriptions.
Anyone concerned about side effects or compatibility with other meds deserves solid information before taking sodium salicylate. Relying on professional advice reduces risk far better than sifting through rumors online. Doctors and pharmacists stay the best line of defense when there’s any doubt, and checking in regularly about all meds used keeps things safer for everyone.
What are the side effects of Sodium Salicylate?
The Basics of Sodium Salicylate
Sodium salicylate lands in a lot of medicine cabinets as a pain reliever and anti-inflammatory. People have used it for decades to manage aches, headaches, arthritis, and even fever. Its use goes back to the roots of aspirin, and it shares some traits with that familiar tablet. Over-the-counter doesn’t always mean risk-free, so it makes sense to look closely at what this drug can do to the body — both the good and the bad.
Gastrointestinal Effects
The stomach notices sodium salicylate right off the bat. For many, nausea, stomach pain, or sometimes even vomiting show up after a dose. Some folks end up with ulcers or bleeding in the digestive tract, especially after long-term use or higher doses. The FDA and Mayo Clinic both flag stomach problems as a top concern. Even though antacids help with the burn, they won’t always stop deep ulcers or bleeding. Those who already struggle with gastritis or history of ulcers find themselves at higher risk. Watch out for dark stools or blood — those signs mean a doctor’s visit isn’t optional anymore.
Bleeding Tendency
Sodium salicylate messes with platelets, the cells that help blood clot. After years of taking care of scraped knees, I learned firsthand: these drugs can turn minor cuts into something that needs a bandage for hours. If someone’s on a blood thinner or facing surgery, this matters even more. It’s wise to talk over plans with a healthcare provider, especially for those with bleeding disorders or a family history of such conditions.
Allergic Reactions and Asthma
A small portion of people react badly, with rashes, hives, or swelling. My cousin, who has asthma, found out the hard way that some forms of salicylate trigger attacks — her breathing turned tight, and it landed her in urgent care. Those with allergies to aspirin or NSAIDs should steer clear. Swelling of the tongue, face, or a drop in blood pressure means a trip to the ER, no questions asked.
Hearing and Ringing in the Ears
High doses or long exposure can lead to ringing in the ears, a problem known as tinnitus. I once took more than suggested for my persistent headaches in college and woke up to a strange buzzing. For some, this symptom fades when the drug leaves the body. For others, it hangs around. Hearing loss can tag along if the ears don’t get a break from the medication.
Risks for Kids and Specific Groups
Children with viral illnesses should stay away from salicylates. There’s a real link to Reye’s syndrome — a rare but deadly problem that swells the liver and brain. The CDC and the American Academy of Pediatrics stand clear on this: other pains and fevers in kids call for safer options like acetaminophen or ibuprofen.
Kidneys and Liver
People with kidney or liver disease face higher stakes. Sodium salicylate runs through these organs, and any weakness in those systems slows down the removal process. Build-up can turn mild side effects into serious complications.
Solutions and Safe Use
Anyone considering sodium salicylate should talk openly with a pharmacist or doctor. Dosing matters; following the label lowers risk, and taking it with food does help the stomach. Anyone with a long list of pills or chronic conditions already in play needs extra planning. Reporting new symptoms, especially anything that feels out of the ordinary, keeps small problems from growing into big ones. Watch out for warning labels and listen to the body along the way. Better trust old-fashioned communication with a professional than guesswork from the internet.
How should Sodium Salicylate be stored?
What Happens with Poor Storage
Sodium salicylate rarely makes headlines, but its storage reveals a lot about responsibility and safety with chemicals. I’ve seen what happens when labels fade, caps come loose, or powder picks up moisture from the air. Things start small—lumps in the jar, a bit of stickiness—but those details can snowball. In laboratories and storerooms, a small failure in routine can mean wasted stock or a much messier hazard if someone doesn’t spot it early.
How the Environment Changes Everything
Temperature and humidity will always win if you ignore them. In my own experience, summer heat finds its way into storerooms. Leave sodium salicylate in a warm, damp corner, and soon, you’re dealing with degraded material—sometimes changing its appearance or even picking up a faint, odd smell. Anyone who’s ever dug into a clumpy jar knows the frustration—plus the suspicion that something dangerous could follow if the compound starts reacting with moisture or dust in the air.
The Case for a Tighter Lid and Good Labeling
A tight seal isn’t an optional step—it makes or breaks the shelf life of this chemical. Any experienced technician can spot badly stored sodium salicylate. Good jars use thick glass or high-quality plastic. The best ones carry clear labels with the date and batch number. Flip up the lid, and you find a dry, free-flowing powder every time. I’ve run into more than a few old pill bottles repurposed in home labs. That temptation often backfires: poor lids, weak seals, and unclear labeling spell trouble.
Why Sunlight Means Trouble
Strong sunlight kicks off slow changes in many chemicals, especially over several months. Sodium salicylate left on a sunny windowsill can break down, become discolored, or cake up. At that point, you’re hoping the active ingredient hasn’t lost potency. This is why pharmacy storerooms and serious lab shelves block out UV rays. Light-proof containers or even a cupboard can improve outcomes far more than fancy technology ever will.
Real Consequences for Small Mistakes
It’s easy to dismiss storage as someone else’s job, but mistakes create bigger problems. I once worked in a teaching lab where a jar had soaked up enough water to trigger worries about toxicity and shelf life. We had to track every person who’d used that batch for weeks. Students learned more from that slip-up than any safety handout. Clear protocols exist for a reason: keep containers well-sealed, dry, and out of direct sun. Keep temperatures steady, ideally around room temperature. Store away from acid fumes and reactive compounds. Safety data sheets say this clearly, but people forget and then scramble when mistakes surface.
Better Practices Make for Safer Work
Tighter routines could stop most storage issues before they start. Regular checks, clear logs, and educating new lab members make the difference. Simple steps—fresh labels, weekly inspections—add up. Even at home, storing chemicals isn’t about paranoia, just respect for what’s in the jar. A little care keeps work safe and reliable and protects health in the long run.
What is the recommended dosage of Sodium Salicylate?
Looking Past the Label: Dosage Knows No Shortcuts
If you’ve ever opened the medicine cabinet and noticed a bottle marked “Sodium Salicylate,” chances are it came with a healthy dose of confusion. This compound, a cousin to aspirin, gets plenty of attention in textbooks and chemistry labs. Out in the real world, the question that always surfaces goes straight to the heart of safety: how much is too much?
Trust the Numbers, Not Urban Legends
Doctors and pharmacists have long agreed on one thing—there isn’t a one-size-fits-all answer. Age, health status, and even body weight change the conversation. For most adults, the standard guideline points to a dose of 325 mg to 650 mg every four hours, but never exceeding 4 grams within twenty-four hours. Kids live by a different rulebook altogether, so only health professionals should set their limits. My own aunt once tried “just one extra” for her arthritis, and landed in the ER with stomach pain that took days to settle. That memory makes me check—twice—before pouring out any pills.
Risks Stick Around Longer Than the Pain
Sodium Salicylate drops inflammation and soothes aches. On paper, it looks like a breeze. In practice, misuse pins people with side effects that blindside families every year. Bleeding, ringing in the ears, even dangerous metabolic problems—these don’t show up on the warning label in big red letters. The U.S. National Library of Medicine lists nausea, heartburn, and kidney strain among common complaints. People with asthma, ulcers, or existing kidney issues risk far worse. There’s a reason careful voices—like Mayo Clinic—urge routines and doses tailored to each body, not just the numbers on a bottle.
Getting Advice That Fits the Whole Story
Most people skip the fine print and ask a neighbor for advice the way someone asks about casserole recipes. That can lead to serious mistakes. Sodium Salicylate interacts with common drugs—blood thinners, diabetes medicines, and even some antidepressants. No quick search beats the voice of someone who handles your medical record. That’s what makes direct conversations with pharmacists and physicians so vital.
Why Proper Use Is Everybody’s Business
Stories about misusing over-the-counter pain relievers drive home a point: careful dosage isn’t just for health nuts or the elderly. It’s for everyone who wants relief without regret. One friend ended up missing work for a week after ignoring her doc’s advice and doubling what she thought was a harmless anti-inflammatory. She never saw the rebound headaches coming. Real solutions come from building habits—measuring out each dose, spacing them as directed, never mixing with alcohol. For people with chronic pain, talking with their provider about alternatives—physical therapy, topical options, non-salicylate choices—opens new doors.
Better Safe Than Sorry
We all want something to melt pain away without a second thought. But every shortcut carries a price. If you don’t remember the last time you checked your dosage, or aren’t sure if Sodium Salicylate is right for you, the safest move is to ask. One honest chat can spare a world of worry—and set you on a path that feels right, body and mind.
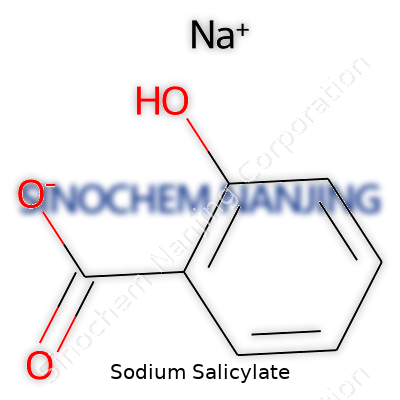
| Names | |
| Preferred IUPAC name | Sodium 2-hydroxybenzoate |
| Other names |
Salicylic acid sodium salt Sodium o-hydroxybenzoate Sodium 2-hydroxybenzoate |
| Pronunciation | /ˌsoʊdiəm səˈlɪsɪˌleɪt/ |
| Identifiers | |
| CAS Number | 54-21-7 |
| Beilstein Reference | Beilstein Reference: 96965 |
| ChEBI | CHEBI:40083 |
| ChEMBL | CHEMBL1357 |
| ChemSpider | 21568936 |
| DrugBank | DB01401 |
| ECHA InfoCard | 100.019.064 |
| EC Number | 205-749-9 |
| Gmelin Reference | 8417 |
| KEGG | C01762 |
| MeSH | D012474 |
| PubChem CID | 16760719 |
| RTECS number | VO5950000 |
| UNII | O414PZ4LPZ |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C7H5NaO3 |
| Molar mass | 160.105 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | Density: 1.44 g/cm³ |
| Solubility in water | Very soluble |
| log P | -1.3 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 13.6 |
| Basicity (pKb) | 7.63 |
| Magnetic susceptibility (χ) | -40.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.567 |
| Dipole moment | 4.24 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 110.50 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -760.5 kJ/mol |
| Pharmacology | |
| ATC code | N02BA04 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | Hazard statements: H302, H319 |
| Precautionary statements | Precautionary statements: "P261, P264, P270, P271, P272, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P332+P313, P337+P313, P362+P364, P403+P233, P405, P501 |
| Autoignition temperature | 450 °C |
| Lethal dose or concentration | LD50 oral, rat: 930 mg/kg |
| LD50 (median dose) | LD50 (median dose): 930 mg/kg (oral, rat) |
| NIOSH | WF1225000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 15 mg/kg b.w. |
| Related compounds | |
| Related compounds |
Salicylic acid Methyl salicylate Aspirin (acetylsalicylic acid) Sodium benzoate Sodium acetylsalicylate |

