Exploring Sodium Phenoxide: More Than Just a Laboratory Reagent
Historical Footprint That Changed Chemistry
Sodium phenoxide has carried weight in the world of organic chemistry for over a century. Its roots trace back to the growing days of coal tar chemistry, a period when synthetic dyes and analgesics first began shaping modern medicine and industry. Ever since Friedrich Kekulé unlocked benzene’s structure in the 19th century, phenolic compounds received new attention for their role in both industrial development and scientific discovery. Sodium phenoxide emerged as a key player in several foundational reactions, especially as chemists noticed its behavior as a nucleophile. It did not take long for this substance to slide into applications, including dye synthesis and pharmaceutical intermediates. Over time, its uses extended into everything from fragrance manufacture to resin modification—demonstrating the way a fairly simple compound can leave a lasting mark across sectors.
Understanding the Product
If you pick up a bottle of sodium phenoxide in a lab, you see a light-colored solid, usually in the form of fine powder or small crystals. Its smell often hints at phenol, thanks to trace hydrolysis and its heritage as a phenol salt. Sodium phenoxide dissolves readily in water, sparking a basic solution that testifies to its place among alkali metal phenolates. You’ll often find it labeled under other names, such as sodium phenate or phenol sodium salt, but the core story remains unchanged. Its simple molecular formula, C6H5ONa, masks a world of subtle reactivity that brings new possibilities to the workbench of chemists and product engineers alike.
Physical and Chemical Properties That Matter
Sodium phenoxide stands out for its swift solubility in polar solvents and its capacity to deprotonate phenolic compounds, making it a versatile partner in organic synthesis. At room temperature, it solidifies out of solution, but once dissolved, it shows strong alkaline behavior—a natural outcome of the phenol to phenoxide conversion. This property gives it credibility in saponification, nucleophilic substitution, and other classic transformations in the chemical repertoire. As with many alkaline materials, exposure to carbon dioxide or moisture eventually alters its structure, leading to gradual conversion to sodium carbonate and regeneration of phenol. This reactivity demands proper storage and handling, especially in settings aiming for high-purity outcomes.
Technical Specs & Labeling Evolution
Over the years, chemists have debated the best packaging for sodium phenoxide to preserve its active nature and prevent unwanted moisture absorption. Although glass containers offer stability, advances in plastics and multilayered sacks have introduced cost-effective, safer solutions for bulk transport. Labels stress the importance of dry, cool storage and clear identification, often including hazard pictograms and handling instructions, especially since sodium phenoxide reacts strongly with acids and oxidizing agents. Whether bought in reagent bottles or larger commercial sacks, sodium phenoxide demands respect, and proper labeling is an essential piece of the safety puzzle, borne out of hard-earned experience on the factory and laboratory floor.
Preparation: Crafting Sodium Phenoxide
Commercial synthesis usually sticks close to the basics: sodium hydroxide and phenol join forces in an exothermic reaction, typically with water as the solvent or even under molten conditions for higher throughput. In small labs, the classic recipe involves slow addition of sodium hydroxide solution to cooled phenol under stirring—a method hammered into generations of aspiring chemists. Scale-up requires careful control of heat and mixing, as too much vigor can lead to splashing and localized overheating. The finished product often sees filtered solids washed and dried thoroughly to prevent caking and impurities. In settings where absolute purity drives outcomes, methods such as recrystallization and inert-atmosphere handling edge out the risk of decomposition and contamination by sodium carbonate or water. Over time, improvements in reactor design and process monitoring have cut costs and reduced waste, yet the fundamental essentials of this transformation stand as solid as ever.
Chemical Reactions and Tweaking Its Core
Sodium phenoxide’s value shines brightest in its reactivity. It triggers the famous Williamson ether synthesis, opening pathways to aromatic ethers that end up in everything from perfumes to pharmaceuticals. Alkyl halides react cleanly with sodium phenoxide, with the oxygen’s negative charge promoting nucleophilic attack and displacing good leaving groups. The sodium ion may seem secondary, but it plays a critical balancing act in stabilizing the intermediate states. In dye chemistry, this compound helps introduce sulfonic acid groups or crafts azo derivatives with high chromatic strength—proving indispensable in the quest for synthetic colors. Besides etherification and dye production, sodium phenoxide serves as a building block for phenolic resins, adhesives, UV absorbers, and antioxidants. Chemists have long explored substitutes for the sodium ion, yet its reliability and cost-efficiency keep it relevant in modern processes.
Synonyms and Alternate Product Names
It’s not uncommon to see sodium phenoxide sold under several names, both on the shelves of research suppliers and in industrial quotes. Sodium phenate and phenol sodium salt both logically draw from its chemistry. Sometimes trade literature condenses this to phenoxysodium or lists registry numbers that track closely to its international coding. No matter what name appears on a drum or bottle, the presence of this white solid signals readiness for a vast network of synthetic work.
Maintaining Safety & Reliable Operations
Years of experience have taught industry and laboratory staff that sodium phenoxide is not a benign material. It irritates skin, eyes, and mucous membranes on contact, much like other strong bases. Short-term exposure can cause burning or redness; longer exposure risks deeper tissue damage or, in rare cases, systemic effects if absorbed in quantity. Spills near acids create risk of rapid phenol liberation, which releases a strong smell and potential hazard. Storage guidelines come down to fundamentals: keep containers tightly sealed, away from incompatible materials, and at low humidity. Facilities continue to stress emergency equipment—eyewash stations, chemical-resistant gloves, and proper ventilation. These aren’t just buzzwords; they stem from hard lessons and real incidents that shape protocols designed to protect people, equipment, and products. Disposal methods reflect its environmental footprint, requiring neutralization with diluted acids under controlled conditions and careful management of any resulting phenol waste.
Where It Fits: Application Areas in Industry
Sodium phenoxide plays a quiet but crucial role across a spectrum of manufacturing sectors. In the production of dyes, it helps lay the groundwork for color-fast molecules that keep shirts and plastics looking vivid even after hard use. Resin manufacturers count on its inclusion to tweak polymer backbone chemistry and ensure the right combination of flexibility and strength, especially in consumer goods and automotive components. In the world of pharmaceuticals, sodium phenoxide helps stitching together molecules for antiseptics and analgesics, touching those finished products found in offices and medicine cabinets. Although fragrance and flavor industries once relied heavily on its etherification reactions, new green chemistry initiatives have added pressure to refine existing methods and minimize waste, sidestepping some of the pitfalls of older processes. Each application underscores the value of convenience and reliability—if a reaction needs a phenol nucleophile, sodium phenoxide rarely lets anyone down.
Research, Innovation, and the Next Generation
Research institutions and companies keep probing the capabilities of sodium phenoxide, seeking tweaks that offer cost savings, reductions in byproducts, or greater selectivity in complex syntheses. In catalysis, scientists look at how alkali phenoxides nudge organic substrates toward greener routes—sometimes with recyclable solvents, sometimes using solid supports that cut back on washing stages. Investigations pivot around not just reactivity, but also environmental compatibility, spurred on by regulations enacted in the European Union, the United States, and parts of Asia. Some labs experiment with using sodium phenoxide as a base in cross-coupling reactions, eyeing ways to ease catalyst loading and improve atom economy, which matters when scaling up for fine chemicals or pharmaceuticals. Biodegradability and water treatment craft another line of interest: researchers ask how phenolic residues break down, whether runoff retains sodium salts, and what long-term effects these have on microbes and aquatic life. Each finding adds a tile to the mosaic of modern clean chemistry—an area nobody can afford to keep on the back burner any longer.
Toxicity and Environmental Caution
Toxicity studies carved out several lessons for those working with sodium phenoxide. Inhaling dust or vapor can irritate the lungs, and direct contact with solutions burns or sensitizes tissues. Accidental ingestion—an unlikely scenario in responsible workplaces—brings risk of systemic toxicity, considering phenol’s metabolic fate and its known effects on internal organs. Chronic exposure links up with concerns about ozone layer degradation, although direct evidence from sodium phenoxide stays limited compared to other halogenated pollutants. Environmental audits tend to focus on phenol liberation during disposal, monitoring wastewater for persistent organics and sodium load. Companies that handle large volumes must file safety assessments under local and international guidelines, making real investments in containment and treatment technology. With regulators in China, Europe, and North America tracking industrial emissions and chemical safety more tightly, having transparent records has become a market necessity and a moral imperative in running chemical operations.
Looking Forward: Future Prospects and Challenges
The future of sodium phenoxide is tied to the future of green chemistry and heightened industrial responsibility. Researchers look for catalysts and processes that use less energy or hazardous reagents, while factories push for closed-loop systems that keep emissions low and recovery high. As automation spreads across batch and continuous synthesis, the possibility arises to better control exothermic stages and enhance yield reproducibility—side benefits that cut down on waste and risk. Upcoming regulations and sustainability pledges by chemical giants will nudge more R&D into finding sodium phenoxide derivatives with built-in safety or degradability, especially for consumer products. At the same time, consumer scrutiny, government oversight, and global supply chain shifts will keep driving demand for products that leave a smaller environmental footprint, forcing every industry that relies on sodium phenoxide to innovate, adapt, and prove their E-E-A-T credentials.
What is Sodium Phenoxide used for?
Understanding Its Place in Daily Chemistry
Sodium phenoxide doesn’t show up on store shelves. Still, its presence trickles through many things we use or encounter. I’ve spent years following chemical ingredients from their factory start to their everyday forms. A compound like sodium phenoxide often hides behind big industry doors, yet people trust the end results every day. It shows up in dyes, medicines, and even cleaning products, playing its part quietly and reliably.
How Sodium Phenoxide Fuels Dye and Pigment Production
I’ve sat across from professionals in textile plants, watching piles of fabrics burst into new shades thanks to compounds their wearers never hear about. Chemists tune sodium phenoxide’s chemical nature to build colors with real sticking power. Manufacturers choose it for its knack at building azo-dyes, used for brightening fabrics or coloring leather. For the chemistry-minded, this comes down to sodium phenoxide’s reactivity with certain hydrocarbons. In the hands of a skilled professional, it works as a builder block, sprouting new molecules with strong color. Nobody expects their shirt or sofa to owe its color to this chemical, yet that subtle chemistry is always behind the scenes.
Pharmaceuticals: It’s All About Precision
Outside the dye world, the medicine makers rely on sharp chemical reactions. The pharma world demands accuracy, purity, and fast reaction times. Sodium phenoxide acts in the background for pain-relieving drugs and fever reducers, helping shape molecules like paracetamol from scratch. During my research into how over-the-counter drugs come to life, the consistency of sodium phenoxide’s effects kept coming up. It helps speed up reactions and build those crucial molecular pieces. Without it, everyday medicines might not meet the standards patients expect.
Everyday Products and Cleanliness
I've always found it fascinating how reliable chemistry has to be in products like disinfectants, antiseptics, or cleaning agents. Sodium phenoxide acts as an intermediate when companies build up disinfectant chemicals and certain synthetic fragrances. Think of the crisp, clean feeling after scrubbing a countertop. Often, those results owe something to the chemical chain that starts with sodium phenoxide.
Responsible Handling and Environmental Impact
Years of looking at supply chain safety protocols taught me that chemical reliability matters, but so does environmental care. Sodium phenoxide, like many reactive agents, needs careful handling. If spills reach water, trouble follows for fish and local ecology. In my work with safety educators, teams stressed personal protection and good ventilation. For waste streams, environmental engineers have pushed for neutralization steps and contained systems. It’s basic respect for chemistry’s power—something everyone from researchers to factory workers comes to appreciate quickly.
Future Challenges and Smarter Production
As demand grows for safer, greener products, the whole industry faces calls for reducing hazardous byproducts. My conversations with sustainability officers circle back to innovation: changing manufacturing setups, recycling solvents, or shifting toward less harmful production paths. Some new processes aim to squeeze every bit of value out of each molecule, cutting down on waste. It’s the ongoing challenge: keep products reliable, keep people safe, and keep one eye on the river downstream.
Is Sodium Phenoxide soluble in water?
What is Sodium Phenoxide?
Sodium phenoxide steps out from the chemistry textbook and into real life as the sodium salt of phenol. It shows up in dyes, drugs, and as a building block for more complex molecules. If you’ve heard of phenol from old-timey antiseptics or coal tar, sodium phenoxide plays a key role on the synthetic side. In the lab, tossing sodium metal into phenol can make it, but commercial setups often use sodium hydroxide instead.
Does Sodium Phenoxide Mix with Water?
Yes, sodium phenoxide dissolves in water. No fancy trick needed; just give it a stir. The sodium part splits off, and phenoxide ions scatter throughout the liquid. This happens because sodium phenoxide holds ionic bonds that break up easily when water surrounds them. Chemists call this “hydration”— something those sodium ions lap up readily.
Solubility isn’t just a desk fact. If you’ve ever tried to dissolve table salt in water, you already get the rough idea: the polar ends of water molecules tug at the charged sodium and phenoxide, prying them apart. Room temperature water handles the task with ease.
Why It Matters
Solubility shakes up how chemicals move and react. Take pharmaceuticals — a compound that lifts out of water quickly can reach the bloodstream faster. For sodium phenoxide, its solubility turns it into a useful carrier in making painkillers and antiseptics. Even in dye manufacturing, it lets color stick to fabric more evenly without clumping or settling out.
In my college lab, I ran into sodium phenoxide during an experiment synthesizing dyes. Back then, if the substance didn’t dissolve, all bets were off. You ended up with patchy color floating on top instead of the even pigment the professor wanted. In industrial chemistry, that difference costs money or ruins products.
Comparing to Phenol
Some people imagine sodium phenoxide will behave like its close relative, plain phenol. That doesn’t hold. Phenol dissolves a bit in water, but sodium phenoxide sweeps right through because its ionic structure hits the right note with water’s polarity. Try mixing, and you’ll spot the change: sodium phenoxide becomes clear, not cloudy, every time.
Risks and What to Watch For
Solubility doesn’t always signal safety. Sodium phenoxide packs enough punch as a strong base to burn skin and eyes. Water helps dilute it, but hands-off is the best rule. Accidentally getting some on you can feel like a spritz of oven cleaner—chemical burns follow. Labs working with it push safety with gloves, splash goggles, and nearby running water in case of spills.
Looking Forward: Better Handling and Waste
Soluble chemicals like sodium phenoxide don’t stay put. Rinsing a beaker or tossing waste down the drain carries that chemical far from the lab. Water treatment plants may not break it down fully. Scientists explore better capture or neutralization steps to keep trace chemicals like these away from drinking water or wildlife. One promising fix: using activated carbon filters or tweaking pH in holding tanks to help trap or neutralize the chemical before it leaves the plant.
Simple as it sounds, the journey of sodium phenoxide through water ties back into big-picture questions: human health, environmental protection, and the nitty-gritty nature of chemical handling. It’s not just a lesson in solubility — it’s a reminder that every lab decision ripples far beyond the bench.
What is the chemical formula of Sodium Phenoxide?
Breaking Down Sodium Phenoxide
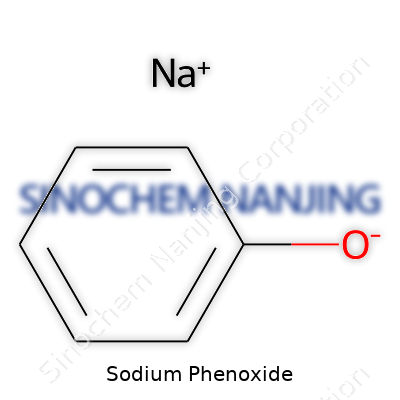
A lot of chemistry students and lab techs run across sodium phenoxide early on. It pops up during synthesis work, sometimes in organic chemistry labs when benzene rings get involved. Its chemical formula is C6H5ONa. That’s nothing more than a sodium ion hooked onto the oxygen where a hydrogen atom used to sit on a phenol molecule. You could call it a simple swap—remove the hydrogen from phenol’s hydroxyl group and slot in the sodium. That single change has real impact on its reactivity.
Why This Formula Matters
During a summer job at a research lab, the protocol actually listed sodium phenoxide by its chemical makeup, not its trade name. If someone didn’t have those basics down, it created confusion and sometimes mistakes during reagent prep. Simple errors here waste time, contaminate reactions, and drain budget. Clear, correct chemical formulas are the backbone for good research—even more so as regulations demand stricter safety data and traceability.
Sodium phenoxide acts as both a strong base and a nucleophile in organic synthesis. Chemists draw on this when making things like dyes, drugs, or specialty plastics. C6H5ONa kicks in as an intermediate for Williamson ether synthesis and a bunch of other coupling reactions. Its structure lets the aromatic ring stay stable while the oxygen with sodium attached goes after electrophiles. This ability stems straight from its formula—a phenyl group (C6H5), one oxygen, and a sodium atom. Each part shapes its behavior in the flask or reactor.
Safety, Handling, and Real-World Impact
I’ve seen what happens when people don’t understand the nature of the chemicals they’re handling. Sodium phenoxide doesn’t just float harmlessly in water. Add it carelessly to an acid, or even just water with a slightly acidic pH, and it can decompose to phenol and sodium ions, creating toxic vapors if things heat up. There’s risk of burns and irritation; nothing abstract about that if you get it on your skin. The clear identification—C6H5ONa—helps safety officers recognize what neutralizing agents to use and what PPE is required.
Many labs skip over the basics, thinking everyone gets the formula after their first semester of chemistry. Yet confusion shows up outside the classroom. Even experienced researchers from other disciplines can mislabel bottles, or grab similar looking compounds—mistaking sodium phenoxide for sodium benzoate, for example. That small error can sabotage weeks of work.
Transparency and Trust in Sharing Information
People expect and deserve accurate, straightforward answers to basic chemistry questions. In my experience, clear language and verification always wins over jargon or abbreviations, especially when dealing with mixed teams from engineering, biology, and materials science. Google helps more people find chemical suppliers, registration databases, and open-access safety data, but clear naming—like spelling out C6H5ONa instead of vague descriptions—improves global communication and lab safety.
Miscommunication about formulas isn’t just a teaching issue. Regulations require pinpoint accuracy in inventory control, shipping, and storage. A misprint could end with someone ordering the wrong material, causing supply chain headaches or safety incidents down the line. For big companies, that can run into significant losses; for students, it can cause experiments to flop until someone spots the error.
Solutions and Good Habits
Cementing the formula of sodium phenoxide, C6H5ONa, in every chemist’s mind sets a foundation for safe, productive work. That means keeping chemical labels clear and current, double-checking reference materials, and encouraging people to ask questions if they hit even basic uncertainties. Supporting this with everyday vigilance saves time, protects people, and keeps research moving forward.
How should Sodium Phenoxide be stored safely?
Understanding the Substance
Sodium phenoxide doesn’t get as much spotlight as strong acids or industrial solvents, but storing it wrong can trigger big headaches. Unlike plenty of household chemicals, this solid poses some persistent risks that call for a solid safety routine in labs and factories. Even in small operations, a careless moment can end up with health issues, equipment damage, or regulatory trouble.
Direct Experience with Chemical Surprises
Anyone who’s ever cracked open a container from the back of a storage shelf knows surprises happen. Sodium phenoxide reacts with air and moisture. Leave its jar exposed just once, and the next person could face lumps and degradation. Years ago during a student project, someone forgot to close a lid tightly. The next day, we found a solid turned clumpy and slightly colored, likely from reacting with the air. Cleaning that spill was tough, and reminders about “just seal the lid” stuck with me ever since.
Why Sealed Containers Matter
There’s more than neatness at stake. Sodium phenoxide absorbs moisture from the air and produces sodium hydroxide as it reacts. Here’s the catch — sodium hydroxide can corrode metal and burn skin. Fewer open containers mean fewer chances for the powder to break down, and fewer breakdowns mean less harmful byproduct floating around.
Keeping chemicals in airtight, proper containers saves more time and money than cleaning up after problems. Glass or high-density polyethylene keep air and humidity at bay. Metal containers struggle because this powder can slowly corrode them, so using them brings unnecessary risk.
The Importance of Labeling
Sodium phenoxide may look like a simple powder, but mislabeling turns it into a guessing game. Improper labeling means the next shift or new team member can’t tell what they’re handling, raising exposure risks. I remember seeing a storeroom where different white crystalline powders sat inches apart. With one faded label, emergency response would have stalled, wasting minutes in an incident.
Date everything and use clear, chemical-resistant markers or pre-printed stickers. Standardizing labels across a lab or storage area isn’t just about organization. It’s lifesaving. If an accident occurs, responders need to know what spilled before grabbing the wrong neutralizer or burning through gloves.
Keep Water and Ignition Sources Away
Sodium phenoxide reacts strongly with water and can ignite combustibles. Store it away from any sinks, eyewash stations, or emergency showers to avoid accidental splashes. Avoid shelving near heat lamps, open flames, or heating elements because some dust in the air will catch fire more easily than expected.
People and PPE
PPE isn’t about ticking boxes — it’s real protection. Gloves, goggles, and long sleeves shield skin from the corrosive mix after exposure. Even experienced workers get complacent, so regular check-ins and real-world drills boost habits. Emergency kits should stay within arm’s reach, and training sessions have a bigger impact when they use real-life scenarios, not just lectures.
Safer Facilities, Fewer Accidents
Smart storage design solves problems before they ever reach personnel. Secure shelves, warning signs, ventilation, and routine checks cut risk. Regulations like OSHA’s hazardous material standards aren’t paperwork exercises — they’re grounded in stories of real injuries where people learned safety the hard way. Meeting these requirements keeps both people and research moving forward without interruption.
What are the potential hazards of handling Sodium Phenoxide?
Understanding What’s at Stake
Anyone who’s spent time in a lab knows chemical safety isn’t just busywork set by overzealous regulators. Sodium phenoxide has a reputation — and it’s earned. It irritates the skin on contact, and a small splash can bring on burning sensations or welts. Once, during my early days in a university organic lab, I saw a colleague hustle straight to the eyewash station after a slipup. He had a mild case, thanks to quick action, but not everyone’s so lucky. That brief encounter hammered home why this compound gets flagged in safety protocols. Skin and eyes take the brunt, with redness, pain, or even deeper tissue damage if you wait to rinse.
Breathing In Trouble
Fumes sneak up in poorly ventilated spaces. Inhaling dust or mist from sodium phenoxide invites coughing fits, headaches, or sore throats. Over time, repeated exposure can mess with your lungs. A 2019 review in the Journal of Occupational Medicine linked improper handling of phenolic compounds, sodium phenoxide included, with increased rates of bronchial issues among workers who skipped protective masks. Nobody wants to develop chronic respiratory problems just for mixing a reaction or prepping a solution.
Unexpected Fires and Explosions
Sodium phenoxide isn’t an inert powder. Toss it near acids, and it reacts violently. Spills next to moisture or water set off heat and create phenol — another hazardous chemical. A single misstep can escalate into a fire or explosion. Years ago, a local chemical warehouse stored incompatible chemicals close together, and a leaking drum started a small fire that forced an evacuation. The investigation later pointed to careless storage of reactive compounds, sodium phenoxide being one of them. Firefighters had to move quickly to contain toxic smoke before it drifted through the neighborhood. The lesson sticks: chemical reactivity cuts deeper than most people realize.
Environmental Fallout
People forget that improper disposal also invites trouble. Pouring sodium phenoxide down the drain exposes wildlife and water sources to toxic waste. Aquatic organisms, in particular, are at risk. The American Chemical Society published a study showing phenolic waste can disrupt reproduction in fish, even at low concentrations. I’ve volunteered on stream cleanup crews, and any chemical spill is a nightmare. Waterways deserve respect, and dumping hazardous chemicals is lazy and shortsighted.
Smart Safety Steps
There’s no magic fix, just solid, day-to-day habits. Gloves, goggles, and lab coats create the first layer of defense. Fume hoods matter; working in the open risks inhaling more than anyone wants. Spills need quick, informed responses—never paper towels or running water; use proper neutralizing agents and a chemical spill kit.
Good training beats guesswork. Every lab session should cover emergency guidelines, not just basics. It’s worth reading beyond labels: consult safety data sheets for details on reactivity and treatment. Facilities storing sodium phenoxide must label and separate chemicals, review regulations, and provide regular refresher courses for both new folks and veterans. Too many accidents start with “I thought I knew what I was doing.”
| Names | |
| Preferred IUPAC name | sodium phenolate |
| Other names |
Phenol sodium Sodium phenolate Sodium monophenoxide Sodium phenol Phenolate sodium |
| Pronunciation | /ˈsəʊdiəm fəˈnɒksaɪd/ |
| Identifiers | |
| CAS Number | 139-02-6 |
| Beilstein Reference | 1907449 |
| ChEBI | CHEBI:59750 |
| ChEMBL | CHEMBL1356 |
| ChemSpider | 54686 |
| DrugBank | DB11174 |
| ECHA InfoCard | 100.004.417 |
| EC Number | 205-488-0 |
| Gmelin Reference | 57779 |
| KEGG | C01828 |
| MeSH | D010702 |
| PubChem CID | 9009 |
| RTECS number | SJ3325000 |
| UNII | LKY2B3Y8E8 |
| UN number | UN2811 |
| Properties | |
| Chemical formula | NaOC6H5 |
| Molar mass | NaOC6H5: 116.09 g/mol |
| Appearance | White to light yellow crystalline powder |
| Odor | characteristic phenolic odor |
| Density | 1.3 g/cm³ |
| Solubility in water | soluble |
| log P | -1.98 |
| Vapor pressure | Negligible |
| Acidity (pKa) | pKa ≈ 10 |
| Basicity (pKb) | 9.98 |
| Magnetic susceptibility (χ) | -54.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.541 |
| Viscosity | Viscous liquid |
| Dipole moment | 4.5 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 151.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -328.8 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -374.7 kJ/mol |
| Pharmacology | |
| ATC code | C01EB13 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes severe skin burns and eye damage, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | Harmful if swallowed. Causes severe skin burns and eye damage. Toxic to aquatic life with long lasting effects. |
| Precautionary statements | P210, P261, P280, P305+P351+P338, P310, P370+P378, P403+P233 |
| NFPA 704 (fire diamond) | 2-3-1 |
| Flash point | 86°C |
| Autoignition temperature | 540°C |
| Lethal dose or concentration | LD₅₀ oral rat 980 mg/kg |
| LD50 (median dose) | LD50 (median dose): 900 mg/kg (oral, rat) |
| NIOSH | RN3500000 |
| PEL (Permissible) | Not Established |
| REL (Recommended) | 100 mg/L |
| IDLH (Immediate danger) | 50 ppm |
| Related compounds | |
| Related compounds |
Phenol Sodium benzoate Sodium salicylate Sodium cresylate Potassium phenoxide |

