Sodium Persulfate: Looking Beyond the Label
Tracing the Origins: A Century of Electrochemistry and Industrial Application
Sodium persulfate didn’t just emerge out of a laboratory overnight. Its story reflects the progress of industrial chemistry since the late 19th century. As an electrochemical creation, it came to prominence when innovative minds realized the power in splitting water and other simple molecules with electric current. Chemists understood that reacting sodium sulfate in the presence of an electric spark allowed them to pull out a strong oxidizer, which has since been called sodium persulfate. Over time, industries picked up on its use because it solved real problems— from helping textile makers bleach fabrics more efficiently to supporting etching processes in electronics. Its development moved in step with the rise of highly controlled industrial synthesis, where reliability, scale, and purity matter more each year.
What Does Sodium Persulfate Mean on the Shelf?
Most folks will never have to measure sodium persulfate or know it by its alternate names—Sodium peroxydisulfate, sodium salt of peroxydisulfuric acid, or “oxone” in a more casual manufacturing context. Labs and factories usually see it as a crystalline white powder, with a faintly sharp edge to its appearance and zero scent. It’s stable until moisture or heat start to meddle, at which point it quickly unleashes its strong oxidizing potential. This property sets it apart from household oxidizers or bleaches. Farmers, textile workers, and circuit-board producers find it useful because it lets them work at room temperature, often reducing the need to introduce extra chemicals. Yet, the same stability that makes it safe to store turns into a challenge once the container is open and the product is exposed to air.
Understanding Chemistry: What Makes Sodium Persulfate Tick
Chemically, sodium persulfate consists of a pair of sulfur atoms bound together, each carrying extra oxygen atoms—essentially stuffed with stored energy from the production process. Its formula, Na2S2O8, captures this. Each molecule holds the potential to donate oxygen. In a lab, this doesn’t just clean things— it can completely transform them. Mix it with water and a touch of heat or an activator, and you get a chemical ready to rip electrons away from almost anything it touches. It’s this characteristic that makes it prized in creating polymers or breaking down contaminants others can’t touch. Physical handling brings up real-world concerns, since that energy can turn hazardous if dust accumulates or users take shortcuts wrangling bulk powders.
Getting the Details Right: Preparation, Tech Specs, and Labeling
Production of sodium persulfate starts with passing electric current through a strong solution of sodium sulfate and sulfuric acid, often requiring specialized cells lined with premium materials to resist corrosion. As the mixture churns, sodium persulfate crystals appear and get filtered, dried, and packaged fast to avoid accidental decomposition. Lot by lot, companies check for hints of contaminants like chlorides, which affect both purity and reactivity. The labeling needs to communicate more than just the chemical name; if a drum crosses an ocean, everyone touching it deserves to know its risks, basic storage needs, and any relevant toxicity findings. Proper storage instructions make all the difference—especially in factories with high humidity, where a misplaced drum could spark a hazardous release.
Tapping Into the Full Range: Where Chemistry Meets Real Life
In the real world, sodium persulfate lifts stains from old cotton, etches intricate copper traces into circuit boards, and helps treat groundwater pollution that defies even advanced scrubbers. Its oxidative muscle also finds a place in hair bleach, where stylists appreciate how it strips pigment effectively while keeping application times short. In the manufacturing of polymers, the chemical starts chain reactions that let engineers fine-tune plastics and rubbers for everything from tires to medical devices. More recently, researchers have turned to sodium persulfate to help break down toxic organic pollutants in soil and groundwater, finding it speeds up the break without as much collateral risk as some alternatives. In all these uses, the value goes well beyond the simple notion of “bleach” or “cleaner”—it becomes essential for progress in diverse fields.
Digging Deeper: Chemical Reactions and Adaptability
Sodium persulfate is not a one-job chemical. In my own teaching lab days, students would watch it trigger a “redox” dance with dyes, metals, and organic matter. Its reactions let researchers turn dial after dial on chemical synthesis. It acts alone or partners up with iron or cobalt salts to ramp up its speed, clearing out wastes or making specialized coatings. Sometimes, a slight tweak in temperature or pH gives a completely new set of products. This adaptability keeps chemists coming back, inventing new approaches in environmental cleanup and polymer science.
Facing Safety and Operations Head-On
Handling sodium persulfate demands respect. Having once experienced a minor spill on unprotected skin, I can say that even brief contact can turn uncomfortable, fast. The powder irritates skin, eyes, and the respiratory tract, and factory floors know that airborne dust is not something to overlook. Consistent training, ventilation, and reliable personal protective equipment (PPE) aren’t optional—they’re hard requirements to keep incidents rare. Strict segregation from flammable or organic materials protects against accidental fires, while clear labeling and tight-lid containers buy time if moisture enters the scene. Regulatory guidance from agencies like OSHA and the European Chemicals Agency sets a baseline, but responsible handlers always go beyond minimal compliance for the sake of their teams.
Gazing Forward: The Role of Research, Ongoing Challenges, and Future Possibilities
From where the field stands, sodium persulfate still has new roles to play. While classic applications in textiles and microelectronics are consistent, the real growth comes from environmental remediation and green chemistry. Teams around the world explore how controlled oxidation, powered by this compound, might break open the toughest pollutants—think persistent pharmaceuticals or “forever chemicals” in groundwater. Research labs keep untangling its mechanism, searching for reaction setups that work under mild conditions, reducing the risk of runaway reactions or costly equipment corrosion. There’s also ongoing work on safer formulation—embedding sodium persulfate into tablets or coatings to limit dust, which could cut down on workplace exposure. Some paint strippers and chemical cleaners now use it in modified forms for precise, limited action on metals and resins, balancing performance and worker safety.
Toxicity Research: Understanding the Risks and the Path Forward
No industrial chemical earns its keep without a close look at toxicity. Animal tests and workplace exposure records have highlighted both acute and chronic risks. The respiratory risk stands out—those fine powders can lead to asthma and long-term sensitivity for anyone left unprotected. Recent research looks at breakdown products, ensuring that sodium persulfate’s use in pollution cleanup doesn’t simply trade one problem for another. Some regulators now call for regular health monitoring in facilities using the material day in and day out, and the science backs this up. Clear protocols on safe use and strict exposure limits protect the bulk of workers. Knowledge is the main shield, and ongoing education makes all the difference, especially as new uses emerge.
What Lies Ahead: Progress, Precautions, and Community Impact
In the decades ahead, industries seem destined to demand more out of every raw material, and sodium persulfate will stay part of that story. From supporting the shift toward biodegradable plastics to helping remediate old industrial sites, its utility matches the broader needs of environmental stewardship and sustainable chemistry. There’s a lesson here for chemists, regulators, and users: progress comes from using old tools in new ways, but only when everyone involved understands the balance between performance and safety. For sodium persulfate, transparency from manufacturers, ongoing research, and outspoken communities will help chart a smarter course, so its strengths serve people rather than becoming hidden hazards.
What is Sodium Persulfate used for?
What Is Sodium Persulfate Actually Used For?
Sodium persulfate shows up in places that land far from most kitchen cupboards, but it influences plenty of products people use every single day. Factories lean on it for its strong oxidizing kick, which really means it helps break things down or push chemical reactions forward. In the print shop, sodium persulfate keeps copper etched neatly from printed circuit boards. Without it, electronics would look very different. Egg beaters, televisions, or phones owe more to chemistry than most people think.
Hair salons keep big tubs of sodium persulfate on hand too, especially when someone walks in determined to go platinum blonde. By itself, hydrogen peroxide can lift a bit of color, but paired with sodium persulfate, it removes stubborn pigment. A quick chat with a colorist reveals the risks: strong stuff like this works fast but can fray hair if not handled carefully.
Hidden in the Everyday: Cleaners, Fabrics, Water Works
Sodium persulfate gets into more corners of daily routines through applications that clean or refresh. Take tired old clothes – textile mills use this powder to treat fabrics, helping dyes stick or fading colors when styles shift. That same property also makes it useful for cleaning municipal water supplies. Water treatment plants use sodium persulfate to break down pollutants that other methods miss. In polluted soil, environmental engineers pump it underground to scramble the chemical makeup of nasty spills. This can turn toxic mishaps into something safer and less likely to reach water sources people rely on.
Potential Risks We Can’t Ignore
Any powerful tool can cause harm out of context. Sodium persulfate carries a health warning if handled without proper protection. Skin rashes, breathing trouble, and eye irritation come up often in safety sheets read by anyone working with it in bulk. Salon workers sometimes develop allergies after years of exposure. If dust escapes in a closed space, those sensitive to chemicals can have an especially tough time. In factories, regular handling calls for training and good ventilation, but small operations might not hear about those risks until too late.
Why Should We Pay Attention?
People like to believe danger shows up with bright warning labels or a strong smell. Sodium persulfate has little scent and hides its strength until it reacts. At home, it's rare to run across directly, but the objects treated with it touch hands every day. Knowing what’s in products and how those chemicals are made builds trust. Factories choosing safer packaging or manufacturers adding clear labeling makes a difference, especially for workers.
Retail buyers can help by asking where and how goods are processed. In cities, supporting local water cleanup plans pushes leaders to keep the most effective, least harmful chemicals in use. Schools and tech training programs should prep future workers for jobs around strong chemicals by bringing the risks and the right safety know-how into focus early, not after mistakes pile up. Life never turns out risk-free, but it gets a lot safer when people know what they’re handling and demand the companies behind those goods do better at every step.
Moving Toward Smarter Use
Safer workspaces, cleaner environments, and good hair color shouldn’t require a trade-off. Wearing gloves, setting up better ventilation, and swapping out strong chemicals when possible lighten the load on people and the planet. Sodium persulfate’s value stays obvious in high-tech fields, but it makes sense to ask if there’s a substitute where safety is a closer call. Chemistry didn’t stop changing after sodium persulfate showed up, and future generations deserve careful choices about what ends up on hands, skin, and land.
Is Sodium Persulfate hazardous to health?
What Sodium Persulfate Is and Where People Find It
Sodium persulfate turns up in all kinds of places you’d least expect. You might run across it in a hair bleaching kit, in certain cleaning products, or in the labs of high school chemistry classes. Factories rely on it, especially those making printed circuit boards, textiles, and pool chemicals. Despite its usefulness, problems start when safety takes a back seat. Stories of skin irritation and asthma in hair stylists reminded me that even common chemicals can pack a punch when handled carelessly.
What It Can Do to Health
The EPA and OSHA both write plenty about sodium persulfate’s risks. Breathing in its dust or touching it without protection triggers some strong reactions. A few years ago, there was a big study linking frequent sodium persulfate exposure to higher asthma rates among hair salon workers. Red, itchy skin, trouble breathing, and eye pain come up over and over. Even folks just cleaning their pool filters have ended up in the emergency room.
I remember helping a friend bleach his hair back in college. His hands broke out in a rash before we’d even finished. We rushed out to buy gloves, but the point stuck with me—small mistakes add up, especially with strong chemicals.
Why People Often Underestimate the Risk
The dusty white powder doesn’t look like trouble. That tricks people into thinking it’s less harsh than it is. Manufacturers often bury the warnings in fine print, and homemade “life hacks” on the internet rarely highlight the danger. News stories usually come out after something goes wrong—never before. That gap in common sense safety catches up to people in bathrooms, salons, and factories.
Science backs this up. Sodium persulfate can sensitize the skin and lungs. Once someone gets sensitized, even a little exposure later on may spark asthma attacks or rashes. Personal experience paints the same picture: Gloveless hands, rooms without enough fresh air, and equipment that spreads dust around become real problems pretty quickly.
What Can Actually Make a Difference
Protective gear makes the biggest impact. Gloves, goggles, and masks save skin and lungs, especially for folks who use persulfate day-in and day-out. Good ventilation knocks down airborne dust, keeping breathing zones a lot safer. Product instructions aren’t just legal talk—they’re based on studies and real accidents.
For businesses, routine training and regular safety reminders go further than warning signs on a wall. Keeping emergency wash stations and clear labeling on hand actually changes behavior. For homes, buying single-use packets and tightly resealing chemicals avoids spills and accidental contact, making a world of difference, especially where kids and pets live.
Laws push companies to test for persulfate allergies in workers, especially in Europe. This matter didn’t get serious attention in the United States until injury rates got too high to ignore. If you ask any seasoned stylist or pool maintenance worker, plenty will have a story about ignoring safety and paying for it.
Understanding chemicals like sodium persulfate is less about alarm and more about respect. Smart habits, good gear, and honest information protect people, long before regulators step in.
How should Sodium Persulfate be stored?
A Chemical With a Serious Reputation
Sodium persulfate shows up all over modern industry. Bleaching, etching, polymerizing—this chemical’s role is wide-ranging. I’ve spent time around labs and workshops, and the minute someone brings out a drum of sodium persulfate, the mood shifts. Stories of burnt skin and coughing fits travel fast, so people treat the stuff with real caution.
Hazards Demand Respect
Direct experience makes the safety warnings hit home. Sodium persulfate behaves as a strong oxidizer. I’ve seen tiny pinholes form in metal tools left lying near a spill—something the manufacturer’s warnings back up. Contact with organic material, reducing agents, even dust or shavings, can start a reaction that’s not just a little smoke and smell. That’s why I don’t cut corners.
The CDC, NIOSH, and the chemical’s own safety data sheets point out acute irritation to eyes, lungs, and skin. Long-term exposure can bring on asthma-like effects. Once, after a cracked container leaked into a storage closet, it took days and a professional team just to make the air safe again. So you won’t find me hauling an open bag around or storing it under a bench in the old supply room.
Storage Tips Backed by Experience and Evidence
I keep sodium persulfate in a cool, dry spot—more than a few feet from heat sources or windows. Humidity is trouble, so airtight containers with gasketed lids become my go-to choice. The original packaging might seem tough, but a double layer, or a sealed drum inside a vented cabinet, adds an extra layer of peace of mind.
Chemical compatibility deserves real attention. In one job, a storage cabinet mixed oxidizers with flammable solvents. That set off alarms and led to an afternoon of re-labeling and moving containers. Since then, I always separate sodium persulfate from acids, bases, metal powders, and anything flammable, just as the American Chemical Society advises.
Eye-level labeling—big, bold, chemical-resistant—prevents mistakes. Accidents can happen in a rush, but a bright label and clear hazard symbols make a difference, especially when teams rotate or work under pressure.
Ventilation, Spills, and Access: Layers of Protection
The best storage sites offer good ventilation. If the material starts to break down, small traces of sulfur oxides and other gases can drift into the workspace. I always prefer ventilated storage cabinets with sturdy shelving and a sump or tray underneath, just in case a container cracks. That spill a few years back proved that reacting quickly matters.
Restricted access is another practice that comes from hard experience. Only trained staff open the doors to the chemical storage area. Logs and check-out sheets help track who last handled the compound. That oversight not only prevents theft or prank misuse—it also stops a careless mistake that could end in a hospital visit.
Personal Attention Makes Storage Work
Sometimes guidelines can feel abstract until you’ve seen a small mishap turn big. Sodium persulfate isn’t just a chemical on a shelf—it’s a substance that commands respect. Reading the safety data, checking government agency guidance, and using airtight, compatible storage containers aren’t extra steps; they’re ways to keep people out of harm’s way. Small details prevent large disasters, and those details start the moment the drums arrive and don’t end until the last bit leaves the site.
What is the shelf life of Sodium Persulfate?
Understanding Sodium Persulfate’s Longevity
Sodium persulfate acts as a strong oxidizer in industries from electronics to water treatment. Unlike flour or rice, you won’t see mold or sourness if it’s past its prime. Instead, shelf life with chemicals boils down to a steady battle: moisture in the air, heat from light, and the chance of stray contaminants all chip away at stability. Most suppliers stamp a shelf life between one to three years, usually erring on the side of caution, but the real story digs a little deeper.
What Breaks It Down
From my experience handling oxidizers in a university chemistry stockroom, the biggest enemy is moisture. Sodium persulfate grabs onto water from the air fast, which then encourages it to decompose. Leave a jar open in a humid room, and you’ll notice the powder start to clump and lose potency long before the stamped date. If contamination creeps in—from scooping with a not-quite-clean spatula or from dust floating in—you’re looking at less reliable results in your next reaction.
Heat doesn’t do the powder any favors either. Direct sunlight—not just a hot room—pushes decomposition, and you end up with less active ingredient. Every proper chemical safety course drills this in early. Sodium persulfate isn’t a set-it-and-forget-it material, and I’ve seen more than one research project go sideways after someone pulled an old, partially used bottle off the back shelf.
How to Store It Right
Storing sodium persulfate in sealed containers, away from damp and heat, stretches life closer to three years. Glass jars with tight lids and desiccant packs can work well, and a dark cabinet or cool chemical fridge truly adds extra protection. Some laboratories even track open dates and run occasional tests on older stock, saving big headaches. This small step can stop a batch of etching solution—or a research grant project—from falling apart due to weak reagent.
Fact-Checking the Expert Guidance
Publicly available safety data and material standards, like those from Sigma-Aldrich and Fisher Scientific, consistently note that sodium persulfate keeps its best qualities for about 24 months if kept dry and cool. The breakdown rate can jump fast if open containers sit out. Laboratories working under Good Manufacturing Practice rarely trust anything near expiration. Most scientists—myself included—prefer to replace old stock after two years, even if it looks unchanged. The cost of wasted experiments far outweighs the price of a fresh jar.
Expanding on Solutions
Routine inspection and proper labeling carve out extra life for sodium persulfate. Newer digital tracking tools let teams set reminders about expiration, and some labs use color-changing desiccants inside stocked jars to flag high humidity instantly. For industry, suppliers can offer smaller packaging so that entire contents get used before the risk of aging creeps in. Keeping quality high means less chemical waste and better results—which matters in every lab and production line.
Effective shelf life isn't a one-size-fits-all promise. Paying attention to storage conditions and handling methods puts more control back in researchers’ and manufacturers’ hands, saving both money and time. Sodium persulfate rewards care and vigilance, making routine updates and check-ins just part of any smart operation.
Is Sodium Persulfate soluble in water?
Everyday Chemistry: Mixing Science Class with Real-World Uses
Bags of sodium persulfate tend to show up in labs, industry workshops, and even tattoo parlors. Once you’ve worked around this chemical, its behavior becomes familiar, almost routine. One thing stands out right away: sodium persulfate dissolves in water. Pour it into a beaker, watch those white crystals vanish, and get a clear solution. That’s not just a science demo—it's how this chemical shows up for work across so many areas, from etching circuit boards to cleaning stubborn stains.
Why Solubility Matters in Real Life
It’s easy to take for granted why a powder dissolves. In practice, solubility drives the usefulness of a lot of materials, and sodium persulfate is no exception. In textile shops, workers rely on this feature when bleaching fabrics or prepping them for dye. The fact it disperses evenly in water means results actually look bright and even, not blotchy or faded.
I’ve watched copper etchers in electronics apply sodium persulfate solutions for printed circuit boards. If it clumped or refused to dissolve, the jobs would stall—or worse, parts get ruined. Solubility keeps the process smooth and cuts down on both messes and waste. A solution you can trust also makes workplace safety easier, letting workers know what to expect every time they measure and mix.
What Science Says: Solubility Stats
The data backs up what folks see in person. Sodium persulfate breaks up in water at up to about 55 grams per 100 milliliters when that water sits at room temperature. That’s serious solubility. Scientists call this an ionic compound, which means the charged parts pull apart when they hit water and stay put in solution. This is why DIY chemistry kits and professionals both choose sodium persulfate when looking to get quick, reliable dissolving action.
Discussions around water temperature come up naturally. Most people notice: as water heats up, more sodium persulfate fits in before it hits its limit. Anyone who’s steeped tea or mixed up sugar knows how temperature changes the game.
Environmental and Safety Thoughts
Sodium persulfate’s ability to disappear into water brings some headaches if folks don’t handle it with thought. This chemical packs a strong oxidizing punch. The same reason it can strip copper or bleach clothes means it shouldn’t end up in streams or sewers. If too much washes into water supplies, aquatic life may catch the bad end of things. Safety sheets warn about eye irritation and breathing difficulties if someone gets careless. Gloves, masks, and smart disposal all become part of the routine.
Smart Use and Better Practices
Most people I know who use sodium persulfate stick to mixing only what they’ll use and label every solution for later. Smaller batch prep helps keep old, concentrated liquids from sitting around too long. In school and small labs, teachers remind students to dilute leftovers and neutralize solutions before pouring them away—to prevent the most common accidents.
Technology for water treatment does help mop up industrial spills, but at the local level, spreading awareness and training workers prove more effective than just chasing leaks. Simple habits like accurate measuring, fast cleanup of spills, and careful labeling keep everyone safe.
Sodium persulfate’s success in so many fields owes a lot to its honest, reliable way of dissolving in water. People trust the results, but respect for safety and the environment keeps us all out of trouble.
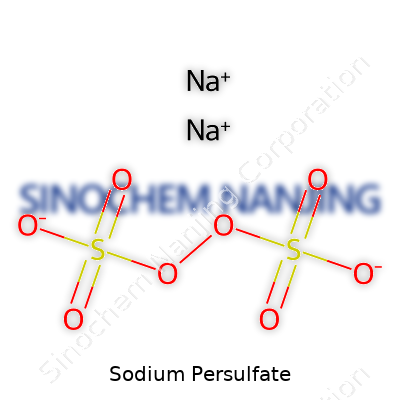
| Names | |
| Preferred IUPAC name | Sodium peroxodisulfate |
| Other names |
Peroxydisulfate sodium Sodium peroxydisulfate Sodium persulphate Disodium peroxodisulfate Disodium persulfate |
| Pronunciation | /ˈsəʊdiəm pəˈsɜːlfeɪt/ |
| Identifiers | |
| CAS Number | 7775-27-1 |
| Beilstein Reference | 1206952 |
| ChEBI | CHEBI:32144 |
| ChEMBL | CHEMBL1356 |
| ChemSpider | 12166 |
| DrugBank | DB09318 |
| ECHA InfoCard | '03a7ceeb-fff4-40a8-a4f6-1b772f50272c' |
| EC Number | 231-892-1 |
| Gmelin Reference | 101149 |
| KEGG | C18697 |
| MeSH | D013014 |
| PubChem CID | 24413 |
| RTECS number | CN7850000 |
| UNII | G8JLV8359Q |
| UN number | UN1505 |
| Properties | |
| Chemical formula | Na2S2O8 |
| Molar mass | 238.10 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 2.59 g/cm³ |
| Solubility in water | 70.6 g/100 mL (20 °C) |
| log P | -2.4 |
| Vapor pressure | Negligible |
| Acidity (pKa) | > -3.0 |
| Basicity (pKb) | pKb: 1.2 |
| Magnetic susceptibility (χ) | -36.4e-6 cm³/mol |
| Refractive index (nD) | 1.497 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 149.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -985 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -145.4 kJ/mol |
| Pharmacology | |
| ATC code | V03AB17 |
| Hazards | |
| Main hazards | Oxidizer, harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS07, GHS08 |
| Pictograms | GHS03,GHS07 |
| Signal word | Danger |
| Hazard statements | H272, H302, H318, H335 |
| Precautionary statements | P210, P220, P221, P234, P264, P273, P280, P302+P352, P305+P351+P338, P304+P340, P312, P332+P313, P337+P313, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 3-0-2-OX |
| Autoignition temperature | 100 °C (212 °F) |
| Lethal dose or concentration | LD50 Oral Rat 895 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat 1190 mg/kg |
| NIOSH | RN0189 |
| PEL (Permissible) | 15 mg/m3 |
| REL (Recommended) | 250 mg/L |
| IDLH (Immediate danger) | 'UR' |
| Related compounds | |
| Related compounds |
Ammonium persulfate Potassium persulfate |

