Sodium Peroxydicarbonate: A Closer Look at Its Evolution, Uses, and Future
Historical Development
The quest for powerful oxidizing agents has shaped the history of industrial chemistry, and sodium peroxydicarbonate found its spot in that relentless search. This compound’s roots reach back to the early twentieth century, carved out as chemists experimented with ways to stabilize peroxy groups in solid form. Early work focused on percarbonates and perborates for laundry uses, and through a web of trials, sodium peroxydicarbonate emerged as an answer for situations where a stable, easily handled solid oxidizer beat the mess of liquids or the risks of handling pure peroxides. Labs tinkered with different sodium carbonates and hydrogen peroxide combinations, trying to balance efficiency, storage safety, and price. By the middle of the last century, researchers understood the compound well enough to start considering its place outside academic circles, especially as industries began asking for better bleaching and cleaning agents that didn’t dump as many harsh byproducts into water systems. This demand marched the molecule out of the lab notebook and into serious consideration for cleaning, environmental remediation, and even some specialized pharmaceutical work.
Product Overview
Sodium peroxydicarbonate lands in a unique niche among peroxy compounds. Unlike its relatives such as sodium percarbonate, it contains a peroxydicarbonate ion that packs more oxidizing punch per sodium atom. It usually appears as a white, odorless powder or granule, easy to scoop and dissolve. People often lump it in with “oxygen-releasing” products, and it shares this reputation thanks to the way it throws off active oxygen during use. Chemists and product developers value its ability to drive oxidation reactions in benign ways, sidestepping many of the environmental headaches tied to chlorine-based chemicals. Its longer shelf-life and lower risk of volatile decomposition—compared with pure peroxides—make it attractive for both industrial and laboratory applications. It keeps for months if shielded from moisture and direct sunlight, which is important for users out in the field or dealing with hot, variable climates.
Physical & Chemical Properties
This compound stands out for its dense, crystalline form and moderate solubility in water. Sodium peroxydicarbonate doesn’t clump much under regular storage conditions, mainly because the peroxy bridges in its anion keep the structure stable against moderate humidity. Drop it in water, and the solution sparkles with active oxygen species, ready to break down stains, odors, or organic wastes. Its pH leans alkaline when dissolved, much like other sodium-based peroxides or carbonates, and this can help boost the activity of certain detergents or cleaning solutions. At room temperature, the powder stays stable for a long time; raise the heat too high or expose it to acids, though, and it starts to break down, releasing carbon dioxide, oxygen, and sodium carbonate. Its chemical stubbornness against premature decomposition helps prevent accidents and waste, and that’s valuable for anyone handling large quantities or long supply chains.
Technical Specifications & Labeling
The technical grade comes with a focus on available oxygen content as the most critical spec, usually measured as a weight percentage. Purity standards follow national or international benchmarks for peroxy compounds. Labels highlight the dangers of speedy decomposition with acids and warn about keeping it dry. While it doesn’t reach the volatility of pure hydrogen peroxide, the labeling still prominently cautions about contact with skin and eyes. The standardized packaging code usually points to UN numbers and hazard designations, marking it as an oxidizer. Most regulatory authorities require detailed listing of active oxygen content, storage guidelines, and emergency procedures for accidental exposure or spill. The biggest concern for end-users revolves around accurate measurement—since even small deviations in purity can throw off technical processes, especially in water treatment or pharmaceutical synthesis where consistency is king.
Preparation Method
Getting sodium peroxydicarbonate starts with a controlled reaction between sodium carbonate and hydrogen peroxide, often carried out in chilled, alkaline water. Some labs tweak the process by adding stabilizers to catch stray decomposition, ensuring more of the hydrogen peroxide reacts as intended. Crystal growth must be coaxed slowly, letting the compound settle out and avoiding the formation of dust or clumpy byproducts. Industrial settings rely on efficient filtration and drying systems to remove excess water while trapping as much of the active compound as possible. The chemistry seems simple at first glance, but it draws on years of troubleshooting—controlling reaction temperature, agitation speed, and concentrations so the product consistently meets oxygen-releasing targets. Inefficiencies in drying cut shelf life and affect bulk transport, which pushes manufacturers to invest in precise environmental controls across the entire production line.
Chemical Reactions & Modifications
Sodium peroxydicarbonate shows its true value in the way it interacts with both organic and inorganic materials. In water, the peroxydicarbonate ion breaks apart, shooting off a stream of reactive oxygen that attacks stains, microbes, and certain inorganic pollutants like sulfides. Chemists often use it as a mild but persistent oxidizer, able to steer reactions toward certain end-products without the unpredictability of harsher reagents. They experiment with tweaks—adding catalysts or adjusting temperature and pH—to push reactions further or slow them down. It can pair with transition metals to speed up bleaching or cleaning, or team up with surfactants for stronger stain removal. Organic chemists dig into its selective tendencies, especially in synthesizing fine chemicals where side reactions from chlorine or ozone would mess up purity. The product also proves useful in creating modified forms, such as surface-coated particles that slow the release of active oxygen, which has implications for water treatment and even controlled agricultural applications.
Synonyms & Product Names
People might call this compound sodium percarbonate dihydrate, sodium persalt, or give it another localized name depending on the market. It appears in literature under chemical labels like sodium carbonate peroxyhydrate, though this sometimes causes confusion with classic sodium percarbonate. Some regions refer to it simply as “oxygen bleach,” folding it into the broader category despite its deeper chemical distinctions. Trade names vary, and while branded forms can add stabilizers or package it alongside supporting agents, the backbone chemistry stays the same—an oxidizing agent that offers a cleaner alternative to chlorine and other harsh industrial staples.
Safety & Operational Standards
Working safely with sodium peroxydicarbonate means treating it with the same respect as other strong oxidizers. Even though it’s less reactive than pure hydrogen peroxide, moisture will start the decomposition, so airtight containers and dry rooms have become the norm. Eye and skin protection stops accidental burns, and local exhaust ventilation helps in bulk operations where airborne dust could cause respiratory irritation. Industries must prepare spill kits with ample water, but also be prepared to contain runoff, since it still counts as a hazardous substance at scale. Compliance with standards comes straight from the playbook of chemical risk management: proper labeling, clear storage procedures, and thorough worker training. Fire departments consider it a moderate oxidizer—capable of intensifying fire in the presence of flammable material—so storage often keeps it isolated from paper goods, fuels, and organic bulk cargo. Wastewater rules sometimes require pre-treatment to remove residual oxidant before it enters city systems, addressing both environmental safety and infrastructure protection.
Application Area
Bleaching and cleaning drive its largest market segments, because active oxygen handles stains and microbial contamination without the toxic residue of chlorine bleach. Laundry powders and machine cleaners use it to lift stubborn dirt and reduce odor. Water treatment plants dose it to break down unwanted chemicals, slashing the need for hazardous byproducts or heavily acidified water streams. Environmental engineers turn to it for remediating spills in soil and groundwater, making the most of its ability to oxidize organic contaminants without leaving behind problematic salts. In pharmaceutical synthesis, it serves as an intermediate reagent, helping produce compounds where purity and non-toxic residue make all the difference. Research communities also keep it handy for green chemistry pilot studies and educational labs, as it lets students run oxidation demonstrations with much lower risk than liquid peroxides. Farmers experiment with it as a pre-treatment for seeds or bulbs, and some horticulturalists recommend controlled doses for combating fungal disease in bulbs and tubers. The key lies in its versatility—strong enough to drive professional results, yet gentle enough not to poison soil or waterways when properly managed.
Research & Development
R&D teams keep seeking ways to make sodium peroxydicarbonate more stable, cheaper to produce, and less environmentally burdensome. Journal articles dig into process engineering—lowering the energy footprint of production, swapping out stabilizers for biodegradable versions, or boosting yield to compete with percarbonate. Work at universities and corporate labs examines catalytic methods to improve selectivity and speed of oxidation, hoping to expand the list of treatable pollutants or supports for pharmaceutical synthesis. The most creative research launches into controlled-release formulations: encapsulation technology that delivers the oxidant to target areas, cutting waste and improving outcomes in agriculture or closed-system wastewater treatment. Others look at hybrid chemistries, mixing peroxydicarbonate with enzymes or transition metals for deeper cleaning or better breakdown of biofilms. These advances hinge on direct partnerships between chemical engineers, suppliers, and end-users, looping field data back into the design cycle and speeding up innovation. Open publication of methods and results helps keep the risks transparent, encouraging responsible scaling and wider adoption.
Toxicity Research
Most evidence suggests sodium peroxydicarbonate presents much lower toxicity than chlorinated oxidizers, both for users and the environment. Once it decomposes, the main byproducts—oxygen, water, and sodium carbonate—don’t introduce new toxicants. Still, toxicity studies warn about the dangers of concentrated exposure, particularly from accidental ingestion or inhalation of dust. Eye and skin irritation comes up often in occupational case reports, especially when personal protective gear lapses. Animal studies show low acute toxicity at realistic workplace concentrations, but repeated, long-term exposure data remain scarce. Environmental toxicologists keep tabs on possible side-reactions if heavy metals are present, as some byproducts could drive mobility of harmful ions in soil or water. Regulatory reviews lean on these toxicity profiles to justify looser restrictions compared to halogenated cleaners, but they keep a wary eye on emerging data, especially as new modifications or formulations move from pilot to market.
Future Prospects
Sodium peroxydicarbonate sits at a crossroads as businesses and governments demand chemicals that pack a punch without leaving scars on people and ecosystems. The drive toward sustainable chemistry should open up wider uses, especially as efforts to ban or phase down persistent, chlorine-based compounds pick up steam. The appetite for waterless or cold-water cleaning pushes product developers to harness its activity under gentler conditions. Agricultural trials hint at value for gentle seed and soil treatments, as food safety rules crack down on residues from harsher agents. Startups and established players see opportunities in “green” formulations for household and institutional markets, backed by consumer desire for effective products with smaller health and environmental risks. If industry and research manage to smooth out production wrinkles and control costs, sodium peroxydicarbonate could form the backbone of next-generation oxidizers for decades to come. Researchers, customers, and advocates will need to keep pushing for transparency and real-world testing to make sure its promise translates into widespread benefit, ensuring cleaner processes and safer work spaces wherever it finds a home.
What is Sodium Peroxydicarbonate used for?
The Role Sodium Peroxydicarbonate Plays
Sodium peroxydicarbonate sits high on the list of modern cleaning and bleaching agents. In my experience working in facilities management and talking with colleagues in the laundry business, I get asked about these kinds of chemicals all the time. Sodium peroxydicarbonate stands out for its oxygen-based cleaning power and eco-friendly footprint.
How Sodium Peroxydicarbonate Works
Picture handling stained white uniforms or scuffed-up children’s sports gear. Mix sodium peroxydicarbonate into the wash and you see a bubbling action as it hits the water. This isn’t just a visual fizz—oxygen gets released and lifts dirt, brightens colors, and works on smells that can hang around even after regular soap. I’ve seen it used in homes as a serious stain remover, but it also has a place in large-scale laundry operations, from hospitals to hotels.
Beyond Laundry: Disinfecting and Cleaning Applications
Hospitals and clinics often aim for surface disinfectants that do more than push dirt around. Sodium peroxydicarbonate gets picked because its oxygen breaks down grime and acts against bacteria. Kitchens and food factories go for this cleaner since residues don’t cause issues the way chlorine or ammonia would. Surfaces get rinsed off easily, without a chemical odor or taste hanging around.
Safer Alternative for the Environment
Households worry about chemical impact on pets and kids. In my family, I look for cleaning solutions that work and leave the fewest toxic leftovers. Sodium peroxydicarbonate fits this bill, breaking down into oxygen, water, and soda ash. According to the European Chemicals Agency, this compound doesn't hang around long enough in soil or drains to cause long-term harm, unlike some older cleaning agents that show up in our rivers and lakes.
Pool and Spa Maintenance
Anyone whose summer job involved cleaning pools knows that strong oxidizers can turn a cloudy pool clear. Sodium peroxydicarbonate brings oxygen to water treatment and doesn’t create the sharp smell or skin irritation that people often blame on chlorine. Pool companies offer it as a gentle “shock” against algae growth and bacteria. It works quickly, and you don’t need to shut down pools for days while chemical levels return to normal.
Industrial Applications and Worker Safety
Factories that handle textiles, pulp, or paper rely on bleaching agents. Sodium peroxydicarbonate is less harsh to handle than some peroxides or hypochlorite compounds, reducing the chances of chemical burns. In textile dyeing, workers benefit from predictable results and a safer workspace. Companies report reduced chemical waste, as this compound does its job, then tapers off without leaving a trail.
What Could Improve?
Cost stays on people’s minds. Sodium peroxydicarbonate takes more energy to produce compared to basic cleaning powders. That shows up in the price. Some buyers still opt for cheaper or stronger chemicals and ignore the risks. Substituting sodium peroxydicarbonate can cut those risks but sometimes doesn’t get the nod in tight budgets. I’ve seen schools and businesses stretch their cleaning supplies by combining this product with other simple cleaners, and it still outperforms harsher options.
Final Thoughts on Practical Uses
What stands out is the balance: strong cleaning, gentle impact. Cleaner factories, safer homes, places where kids and workers aren’t exposed to long-term harm. As access grows and manufacturers expand supply, more people will swap old habits for smarter choices, and sodium peroxydicarbonate plays a part in that shift.
Is Sodium Peroxydicarbonate safe to handle?
Understanding the Chemical
Sodium peroxydicarbonate doesn’t show up in everyday conversation. This white, powdery substance plays a role in some niche cleaning and bleaching products. The name hints at its punch: it unleashes a steady stream of oxygen when it touches water, delivering stain-fighting power. That property may sound familiar to anyone who’s used an oxygen bleach for laundry, but sodium peroxydicarbonate isn’t the same as sodium percarbonate or sodium perborate. Small tweaks in chemistry lead to big changes in how a compound behaves.
Real Risks, Not Science Fiction
Most folks don’t have sodium peroxydicarbonate in the kitchen pantry. If you ever have to handle it, pay attention to the warnings. This powder doesn’t belong on bare skin. Contact causes irritation. Breathing the dust burns the nose and throat, sparking coughing fits, and can make it hard to catch your breath. Eyes take the biggest hit; a small amount can sting badly and may cause lasting damage. These effects aren’t rare horror stories—they happen because of the strong oxidizing action that makes the chemical useful in the first place.
Mixing with combustible materials raises the risk. Storing sodium peroxydicarbonate next to paper, sawdust, or anything organic sets up a recipe for fire. If the chemical spills and gets wet, the slow release of oxygen can push up the risk even further. Once, working in a research lab, I saw a bottle start fizzing in a storage room when water vapor leaked in. The fizz itself looks harmless until you remember what’s happening: oxygen builds up, and a tiny spark can turn things dangerous fast.
Staying Smart: PPE and Ventilation
Working with sodium peroxydicarbonate means gearing up. Gloves that keep chemicals out and tight goggles are the base kit. Forgetting to slip on a dust mask spells trouble. A direct puff of fine powder can turn a regular afternoon into an emergency room visit.
Labs and manufacturing sites nail this point with training. Clear safety sheets back up the message: don’t eat, drink, or smoke nearby. Wash up after each use. If it hits the skin, don’t wait—flush the area right away. Dumping any leftover powder down the sink also leads to issues since the chemical’s oxidative punch can trigger problems for plumbing and local waterways.
Safety: More Than Label Warnings
The record on sodium peroxydicarbonate stays clear—handled carelessly, it bites back. The U.S. Occupational Safety and Health Administration (OSHA) recommends keeping exposure as low as possible. The European Chemicals Agency (ECHA) flags similar risks, highlighting the strong oxidation potential.
Not every risky chemical deserves a ban, but proper respect lays out a path to safety. Spill kits, eye-wash stations, and strict storage rules create a defensive wall. Limiting access ensures only trained staff get near open containers. Revisiting handling procedures helps everyone stay sharp. In my years around chemicals, complacency causes accidents, not the powder itself.
Room for Better Protections
Some workplaces move faster than others to update their safety rules. I’ve seen smaller outfits ignore old stock tucked away in forgotten cabinets. Regular safety audits catch that sort of oversight. Creating a culture where anyone can speak up about hazards (and get listened to) makes a difference. Safety thrives where embarrassment takes a backseat to open discussion and honest fixes.
Sodium peroxydicarbonate delivers real value in industrial and lab settings. No tool comes risk-free. Knowledge, training, and teamwork keep the harm at bay. Respect turns a strong oxidizer from a threat into a productive partner.
How should Sodium Peroxydicarbonate be stored?
Not Your Everyday Powder
Sodium peroxydicarbonate sounds like the stuff of labs, but people in water treatment, detergent production, and specialty cleaning have handled it for years. Some folks call it sodium carbonate peroxyhydrate. Its strong oxidizing properties can make a mess if not treated with respect. I’ve watched operators misjudge its stability and pay the price: jars swelling up, leaks popping through weak seals, or surprise fizzing that turned a tidy bench into a foamy disaster.
The Risk You Don’t See
The stability of sodium peroxydicarbonate can fool newcomers. Dust floats up when poured carelessly, irritating lungs and eyes. Humidity kicks off slow reactions, making the powder clump or worse, release oxygen. Heat can quietly push the compound toward dangerous self-decomposition. The EPA lists oxidizers like this among chemicals demanding special handling because they boost fire risk if stored near flammable materials. Even a forgotten beaker with some leftover product has triggered unexpected reactions in warm or damp warehouses.
Storing It Right: The Basics
The best place for sodium peroxydicarbonate? Dry, cool, well-ventilated, and clearly labeled. I’ve always pushed colleagues to keep containers tightly sealed with robust threading and gaskets. Even the pros sometimes use ill-fitting lids only to find their chemical crusting up and losing strength. Polyethylene and other chemical-resistant plastics work better than metals which could corrode or promote dangerous decomposition.
Don’t shelf it near acids, fuels, papers, or anything prone to ignite. The smallest spill in the wrong corner can build tension in the air, especially if the room never quite cools down in the summer. I remember a well-meaning intern who stored a drum near some cleaning solvents. Lab supervisors had to clear the whole wing after a suspicious smell signaled a reaction.
Lessons in Labeling and Segregation
Risk doesn’t just come from chemical properties. Human error often stacks up over months. Outdated or missing hazard signs have led to unprotected hands, contaminated spatulas, or half-hearted inventory checks. GHS-compliant labeling and written procedures really make a difference, especially as team members change over time.
Don’t mix sodium peroxydicarbonate with organic materials. This can't be stressed enough. My experience says complacency slips in fastest in busy, shared storage rooms. Segregating oxidizers away from everything else and using locked cabinets brings peace of mind. Accidents from mixed chemicals may seem unlikely, but every producer and large-volume storage facility has their stories.
Solutions the Industry Trusts
Routine audits catch bad habits. Locking in a regimented training schedule helps both newcomers and old hands. I’ve seen a lot of labs get comfortable and skip regular checks. They only realize their mistake after an unexpected reaction or near-miss incident wakes them up.
Monitoring humidity and temperature with simple, inexpensive sensors pays dividends. Even big manufacturers use these, because nobody trusts HVAC systems to stay perfect year-round. If the area feels muggy or hot, chemical life drops, and accidents grow likely.
Disposing of old or spilled sodium peroxydicarbonate by professional channels protects not only the staff but also the public and environment. EPA and OSHA guidelines lay out clear steps, and following them strictly keeps everyone on the right side of the law and out of harm.
Storing It Safely Pays Off
Sodium peroxydicarbonate won’t forgive shortcuts. Tidy, segregated storage under dry, cool conditions in fully sealed, chemically compatible containers prevents waste and protects people. Regular checks and a healthy respect for oxidizers keep the workplace safe and the product usable.
What are the main applications of Sodium Peroxydicarbonate?
Finding the Role in Cleaning and Disinfection
Sodium peroxydicarbonate draws most of its attention as a source of hydrogen peroxide. Imagine the job of keeping public spaces–think gyms, schools, hospitals–free of tough germs. Tablets and powders based on this chemical release hydrogen peroxide the moment water touches them. This reaction produces oxygen, breaking down stains and unwanted microbes. From a user’s standpoint, quick action matters. The fizzing shows the product is working. Surfaces get cleaner, but people also avoid the heavy chlorine smells that linger with old-school bleaches.
Support for using sodium peroxydicarbonate in cleaning comes from studies documenting its ability to degrade viruses and bacteria. For community health workers, using a solid, stable product means fewer worries about storage, a big plus compared to liquid disinfectants that can break down on the shelf. In schools, tablets cut waste by dissolving completely, so there’s less residue washed down the drain.
Laundry and Household Use
Washing white fabrics or removing pet stains takes something tough on grime but gentle on fibers. That’s where sodium peroxydicarbonate steps up, releasing oxygen to help lift soils. I’ve watched the transformation of old T-shirts dulled by years of use, dulled by spills or sweat. A soak with this chemical can help restore brightness without using strong bleaches that damage the threads. Households favor products that give results and keep hands safe, especially parents watching over kids or pet owners cleaning up after a spill.
Consumer demand for less-harmful laundry aids has sparked reformulations. Most “oxygen bleach” powders on store shelves fall back on this ingredient. Its safety record aligns with advice from experts who urge less harshness in everyday cleaning, both to protect health and keep household chores from being a hassle.
Industrial, Medical, and Food-Related Fields
Industry uses sodium peroxydicarbonate for more than surface cleaning. Breweries, dairies, and food factories adopt these products to keep equipment safe. Organic residues vanish, and there’s little risk of ammonia-like smells lingering. As food safety rules get stricter, operators rely on proven results. This chemical breaks down without leaving harmful by-products that would threaten meals or drinks.
Hospitals also need hard-hitting, quick-acting sanitation. Time and again, studies show sodium peroxydicarbonate-based cleansers eliminating difficult pathogens. Tablets and powders that store well are easy to share across wards. Less chemical residue means equipment can return to service faster, so staff lose less time in between procedures.
In water treatment and pool care, this compound’s ability to generate oxygen becomes handy. Algae and contaminants get tackled while the water stays clear. Regular users–public pool managers or spa owners–like a chemical that works reliably and doesn’t leave behind sludge or strong chemical smells.
Looking Toward Sustainability
Regulators and consumer advocates call for safer chemicals and lower-impact cleaning routines. Sodium peroxydicarbonate checks many boxes: It breaks down to water, oxygen, and simple salts, reducing the risk of long-term pollution. Each use cuts back the demand for harder-to-handle legacy products. This lines up with what I’ve seen as people look for solutions that clean up after themselves, not just the mess.
What is the shelf life of Sodium Peroxydicarbonate?
A Closer Look at Sodium Peroxydicarbonate
Sodium peroxydicarbonate shows up in labs and cleaning products as a strong oxidizer, releasing active oxygen in contact with water. The white powder or granule will boost stain removal and sanitizing properties in detergents, and people turn to it for eco-friendly bleaching solutions. It gets compared to sodium percarbonate, but it gives off active oxygen at a slightly different rate, making it useful in some formulations over others.
What Determines Shelf Life in Real-World Use
With chemicals like this, degradation starts the day the drum gets sealed. The shelf life of sodium peroxydicarbonate usually falls between 12 and 24 months under cool, dry conditions. Heat, humidity, and exposure to air all speed up the breakdown, so the granules clump or fizz sooner than they should. I’ve seen cases where leaving the lid loose cuts the effective shelf life in half, even if the batch never looked much different to the eye.
Keeping the stuff in a sealed, moisture-resistant container makes all the difference. Throwing the bag onto a pallet in a hot, wet warehouse will pretty much guarantee that you lose most of your oxidizing power by the end of the year. Even a swing of a few degrees in storage temperature can shorten usable life. The European Chemicals Agency notes that oxidizers like sodium peroxydicarbonate have shelf lives listed by manufacturers based on their own storage standards. That kind of guidance gives a baseline, but my experience says local conditions always matter more.
Why Shelf Life Means More Than an Expiration Date
The real risk with expired sodium peroxydicarbonate sits in its performance. As oxygen release drops, cleaning and bleaching effects plummet. Industries relying on precise dosing—pharmaceuticals, water treatment, food processing—see measurable losses. I’ve witnessed complaints from buyers who claim a product ‘goes dead’ halfway through its expected lifespan, usually traced to a storage room that’s either too warm or sits unsealed for days.
OSHA and chemical safety agencies recommend keeping the product in air-tight containers away from acids, moisture, and direct heat. Small leaks or spills attract humidity, leading to clumping—once that happens, the risk for unexpected reactions rises, and chemical potency drops fast. The importance of handling and regular checks grows as the stock ages. Solutions that come from experience, rather than blind trust in the ‘best before date’, save money and headaches for every facility I’ve worked with.
Finding Practical Solutions
Training staff counts more than any warning label or standard operating procedure on paper. Not everyone in a storeroom knows the difference between a batch that’s merely dusty and one that’s gone flat. Labeling the containers with clear storage instructions and reminding employees during safety walks goes a long way. Adding a monthly log for inspection ensures material that’s started to degrade gets flagged and replaced. I’ve found humidity sensors do well enough in small storage rooms, giving an early sign of whether to step up air conditioning or dehumidification.
Trusting manufacturer timelines sets a reasonable baseline, but always double-check by opening the drum, looking for lumps, and confirming performance in practice. A simple test with water will show if the material still fizzes and reacts as needed. Staying practical, storing right, and empowering the team with information keeps sodium peroxydicarbonate working as it should, making the material safer and more reliable for everyone who handles it.
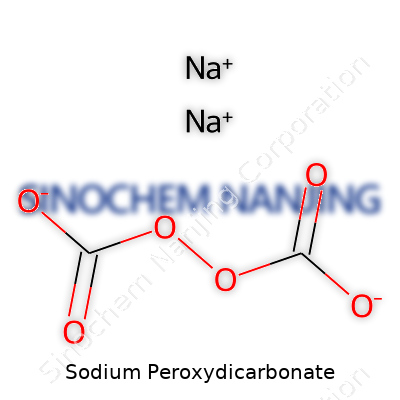
| Names | |
| Preferred IUPAC name | disodium peroxodicarbonate |
| Other names |
Disodium peroxydicarbonate Sodium carbonate peroxide Sodium peroxycarbonate Sodium percarbonate |
| Pronunciation | /ˈsəʊdiəm pəˌrɒksaɪdaɪˈkɑːbəneɪt/ |
| Identifiers | |
| CAS Number | 15630-89-4 |
| Beilstein Reference | 3551877 |
| ChEBI | CHEBI:91250 |
| ChEMBL | CHEMBL1201811 |
| ChemSpider | 21482929 |
| DrugBank | DB11530 |
| ECHA InfoCard | 03b08eaf-778e-4a3f-bca2-aa6b0d0ba7d6 |
| EC Number | 239-154-0 |
| Gmelin Reference | 319928 |
| KEGG | C18615 |
| MeSH | D017782 |
| PubChem CID | 16211238 |
| RTECS number | NJGWQ0407E |
| UNII | DS602OJK5M |
| UN number | UN3378 |
| Properties | |
| Chemical formula | Na2C2O6 |
| Molar mass | 157.99 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 2.14 g/cm³ |
| Solubility in water | soluble |
| log P | -4.37 |
| Vapor pressure | Negligible |
| Magnetic susceptibility (χ) | -45.0e-6 cm³/mol |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 216 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1160 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | −820 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V03AB52 |
| Hazards | |
| Main hazards | Oxidizing, harmful if swallowed, causes serious eye damage, may cause respiratory irritation. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS03,GHS05 |
| Signal word | Danger |
| Hazard statements | H272, H302, H318 |
| Precautionary statements | P210, P220, P221, P234, P264, P270, P280, P283, P305+P351+P338, P337+P313, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 2-0-1-OX |
| Autoignition temperature | > 130°C |
| Lethal dose or concentration | LD50 Oral Rat: > 2,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 2210 mg/kg |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Sodium Peroxydicarbonate is not specifically established by OSHA. |
| REL (Recommended) | 160 mg/m³ |
| Related compounds | |
| Related compounds |
Sodium percarbonate Sodium peroxide Sodium carbonate Potassium peroxydicarbonate |

