Sodium Permanganate: A Look Beyond the Lab Bench
Historical Development
Sodium permanganate has traveled quite a path since scientists in the 19th century first tinkered with permanganates as a group. The earliest attention centered on potassium permanganate, mainly because it’s easier to handle and store. But as chemical industries expanded their reach, sodium permanganate stepped out from its cousin’s shadow. Chemists began turning to sodium salts for specific needs, especially where higher solubility mattered. Over time, as water treatment and specialty oxidation became more widespread, the sodium salt found a foothold, especially in environmental remediation, quickly catching the eye of both researchers and engineers. Increased demand for cleaner processes and tighter pollution control pushed development forward, right alongside better manufacturing methods and more reliable quality.
Product Overview
You don’t bump into sodium permanganate at your neighborhood shop, but it shows up regularly in places where strong oxidizing power makes all the difference. The compound, with the formula NaMnO4, looks like a striking, deep purple solid or sometimes turns up dissolved as a vivid purple liquid. Unlike its potassium sibling, sodium permanganate dissolves even better in water and that extra solubility makes it a top choice where strong solutions of permanganate are needed quickly and with less fuss. Whether folks are looking to clean up groundwater, bleach fabrics, or run specific chemical syntheses, sodium permanganate delivers a reliable punch.
Physical & Chemical Properties
Sodium permanganate’s deep color comes from its manganese at the +7 oxidation state. Drop a bit in water and you’ll see that signature purple—no surprise to anyone who worked in a water lab. It forms stable, highly soluble solutions at ordinary temperatures, which matters quite a bit for industrial and research settings that require consistent concentrations. The solid by itself, though, isn’t something you want lying around on the back shelf. It reacts readily with organic matter and can ignite combustibles if not handled with respect. Even the solutions pack potent oxidizing properties, so working with it isn’t about guesswork—good training keeps accidents off the front page. Mixed with acids or reducing agents, sodium permanganate quickly shifts gears, unleashing oxygen and pushing redox reactions to completion. Chemists appreciate its reliability and reproducibility, which saves both time and raw materials.
Technical Specifications & Labeling
Walk into a modern chemical plant and you’ll notice that sodium permanganate comes clearly labeled for both purity and concentration. Most industrial grades hit above 40% concentration in solution, though solid forms are available if the process calls for them. Hazard symbols pop up on every barrel, usually with warnings about handling, storage, and incompatibilities—especially with strong acids and combustibles. Globally, transportation falls under strict regulations like those enforced for other strong oxidizers: think UN numbers, specific packing group designations, and lots of documentation. You’ll see labels pointing out risks to skin and eyes, along with thorough instructions for dealing with spills or exposure, all to make sure operators keep out of harm’s way.
Preparation Method
Manufacturing sodium permanganate starts with manganese dioxide and sodium hydroxide, which get mixed up and heated with a dash of oxidizer—often sodium chlorate or direct electrolysis. Large operations now turn to continuous flow reactors to crank out consistent product, cutting down impurity levels and waste. Processing always takes the environment into account, because improper venting or wastewater management can undo the very pollution-fighting benefits permanganates are meant to provide. Over the decades, greener methods have started taking hold, cutting back on secondary pollution and improving energy efficiency along the way.
Chemical Reactions & Modifications
What sets sodium permanganate apart in chemistry circles is the predictability of its oxidizing action. Add it to organic mixtures, and it’ll cleave double bonds or strip electrons as needed, helping chemists create acids, aldehydes, and other key intermediates. In water, its role as a disinfectant or a means to address “taste and odor” issues stands out—municipal utilities often dose water lines with permanganate solutions to cut down on iron, manganese, and pesky microorganisms. In electrochemical setups, sodium permanganate finds use in specialized oxidation reactions, where fine-tuning voltage and current delivers products you can’t get with simple batch processing.
Synonyms & Product Names
In the trade, sodium permanganate sometimes gets listed as sodium manganate(VII). On the label it can show up as “permanganic acid, sodium salt” or simply as NaMnO4. If you step into a research supply catalog, you’ll see variations like “sodium permanganate monohydrate” for the hydrated solid, or “aqueous sodium permanganate solution” for the more popular liquid form. The synonyms might switch depending on local conventions or historical habits, but they all point to the same base compound.
Safety & Operational Standards
Handling sodium permanganate is not a job for the careless. Every year, reports come in about fires, spills, and injuries tied to oxidizer mishandling. It stains hands, fabrics, and equipment with a vengeance. Inhalation or skin contact carries real risks, as the compound acts as a strong irritant and can damage tissues. Industry guidelines dictate gloves, face shields, and heavy ventilation for even short exposure periods. Training isn’t negotiable. Keeping it away from organic material or fuel sources sometimes trips up new technicians, and stories of accidental fires reinforce why operational standards have to stay sharp. Storage by itself requires sealed containers, far from incompatible chemicals, and periodic inspections to spot leaks or corrosion that could spark problems down the road.
Application Area
The biggest chunk of sodium permanganate leaves the factory loaded on trucks heading for environmental cleanup. Injecting this compound into contaminated groundwater helps tackle organic contaminants—ranging from solvents like trichloroethylene to pesticide residues—by breaking them down into less toxic substances. I’ve seen municipal water providers deploy permanganate ahead of their filtration systems: a move that keeps equipment cleaner and water safer for everyone downstream. Industrial laundries, food processing plants, and electronics manufacturers often reach for sodium permanganate when selective oxidation or deep cleaning is needed. Even in mining, it turns up in ore processing routines, helping extract useful metals or scrub out traces of unwanted elements that could foul up the end product.
Research & Development
Research teams at universities and in industry labs keep pushing the boundaries for sodium permanganate. Many studies focus on how to tolerate higher impurity levels in groundwater while still nailing remediation targets, or how to recycle permanganate more effectively after a clean-up run. I’ve seen promising experiments using permanganate in tandem with zero-valent iron or specialized resins, producing reaction chains tailored to specific site conditions. Advances in sensor technology now let operators fine-tune dosing in real time, cutting back on waste and environmental impact. Academic groups continue exploring uses in organic synthesis, sometimes adapting old textbook reactions for greener or cheaper conditions with sodium permanganate playing a key role.
Toxicity Research
Understanding the toxicology of sodium permanganate remains a priority, especially as its use deepens in fields like water treatment. Most animal studies point to moderate acute toxicity, with higher doses causing oxidative damage to tissues. Chronic exposure data is less complete, but ongoing monitoring of workers and nearby populations shapes regulatory advice. Environmental scientists track how permanganate behaves once released: it can break down into manganese oxides, which have their own environmental concerns if not properly controlled. Regulatory agencies evaluate product data to shape guidelines around permissible exposure limits, clean-up thresholds, and remediation best practices. Those involved in deploying permanganate solutions must weigh its potent benefits against the need to minimize accidental releases and protect both human and ecological health.
Future Prospects
Anybody scanning the horizon sees steady growth ahead for sodium permanganate. Demand for groundwater remediation—especially at former industrial or military sites—drives much of the market. Tighter drinking water standards put more pressure on utilities to deliver consistent, contaminant-free supply lines, where permanganate solutions keep stepping up. The push for sustainable chemistry could open new doors, as innovations in catalyst design and water recycling line up with sodium permanganate’s unique menu of chemical reactions. Research into better containment, lower-impact disposal, and safer transport will keep evolving. So will application technologies, from in situ treatment rigs that deliver permanganate straight into the earth, to smart dosing systems that shave costs and protect workers. The balancing act will always involve maximizing its environmental and economic payoff, while managing its risks with rigor and common sense drawn from both experience and evidence.
What is sodium permanganate used for?
What Sets Sodium Permanganate Apart
Sodium permanganate doesn’t show up on grocery lists or in household cleaning aisles, but it plays a key role in some of the industries most people rely on. Formed as a close cousin to potassium permanganate, sodium permanganate stands out because it dissolves more easily in cold water. That simple difference makes it a preferred choice for jobs where speed and solubility bring big benefits.
Water Treatment: More Than Just Clean Water
One of the biggest areas where sodium permanganate shows its value is in water treatment plants. Communities depend on clean water, but beneath the surface, a lot happens before a glass of tap water reaches your kitchen. Manganese and iron often sneak into water supplies and stain fixtures or cause taste issues. Sodium permanganate handles those metals by converting them into particles that water filters can grab. If you’ve ever wondered how cities deal with stubborn “rotten egg” odors caused by hydrogen sulfide, sodium permanganate is one of the solutions. Its strong oxidizing power neutralizes these smells and keeps water tasting right.
Keeping Industrial Pipes in Shape
Corrosion and scale inside industrial pipes can slow production, spike energy costs, and lead to expensive repairs. Factories and municipal facilities don't have the luxury to ignore buildup. Sodium permanganate acts as a cleaning agent, breaking down organic material and removing built-up deposits. Its use helps companies sidestep unexpected shutdowns and keeps water flowing smoothly, reducing repair bills and keeping employees safe from leaks or waterborne hazards.
Soil and Groundwater Remediation: Battling Pollution
Many old industrial sites leave a legacy of pollution buried deep below the surface. Chlorinated solvents like trichloroethylene and vinyl chloride used in past decades can seep into groundwater, posing health risks for nearby residents. Sodium permanganate gets used to treat these chemicals directly in the ground. The process, known as in-situ chemical oxidation, neutralizes toxic pollutants. I’ve spoken with environmental engineers who explain that without oxidizers like sodium permanganate, cleaning up legacy pollution might drag out for generations, leaving entire neighborhoods stuck with unsafe water and property values in freefall.
Fine-Tuning the Use: Challenges and Ideas
Using a chemical this strong brings some headaches. Dose it too high, and treatment systems can get clogged by manganese dioxide solidifying out, leading to maintenance headaches. Worker training and close monitoring help manage these risks. Then there’s the issue of storage and transportation — sodium permanganate is corrosive and reacts if mishandled. Good storage practices, staff education, and careful transport dramatically cut down the dangers.
From my time working with municipal utilities, I’ve seen teams innovate by automating dosing or by mixing sodium permanganate with other oxidizers to increase effectiveness and keep costs down. Cutting-edge research looks at greener alternatives, but right now, very few products match sodium permanganate’s ability to solve tough environmental problems.
Weighing Impact on Health and Environment
A pound of prevention still beats a ton of cure. By tackling pollution, odor, and metal problems where they start, sodium permanganate protects drinking water, public health, and the long-term value of infrastructure. Its track record stretches back decades, yet researchers and utilities keep fine-tuning how it’s used to shrink risk and squeeze out even more benefit. Whatever the future holds, understanding and respecting this chemical lets industries keep delivering safe water and a cleaner environment.
Is sodium permanganate safe to handle?
Practical Risks Behind the Scientific Name
Sodium permanganate stands out as a powerful chemical in water treatment and various cleaning industries. Its reputation for cleaning up groundwater and removing iron or hydrogen sulfide from water comes from its strong oxidizing nature. The same reason it’s so effective also creates the main challenge: handling it opens the door to irritation and real risks with careless use.
Understanding How It Behaves
With a vivid purple color and the ability to stain on contact, sodium permanganate leaves a mark. That’s not just on your skin — splash it, and you’ll deal with stains on clothing, floors, and even metal equipment. Short exposure might seem harmless at first, but the chemical lingers. Direct skin contact doesn’t hurt right away, but over time it can dry out the skin, cause redness, or trigger allergic reactions for people with sensitive skin. Touch it to your eyes, and you’re looking at everything from mild discomfort to serious injury.
Breathing It In or Swallowing
Most of us would shudder at the idea of inhaling any type of chemical dust. Sodium permanganate powder isn’t an exception. Dust can irritate the nose and throat, possibly even the lungs, especially after repeated exposure in poorly ventilated workspaces. Ingesting it could mean nausea, burning in the mouth or stomach — a clear medical emergency. Emergency rooms sometimes see industrial workers who didn’t take proper precautions and learned the hard way.
Fire Hazards and Chemical Reactions
Sodium permanganate reacts strongly with many other substances. It doesn’t burn itself, but it feeds flames if it comes in contact with flammable material. Spilling some on paper or oil can set off a fire or explosion. Forgetting about that in a busy industrial setting only increases risk for everyone nearby. Handling this chemical with wet hands or mixing it poorly can also end up creating dangerous byproducts like permanganic acid or chlorine gas.
My Experience and Best Practices
Years spent working with water treatment systems taught me two lessons: always wear gloves and never skip the goggles. Even the most experienced operators sometimes get in a hurry, thinking nothing will happen just this once. One slip-up and purple stains don’t wash away for days. Worse still, if it touched my eye, an injury like that could last far longer. Extended use without proper skin protection led a coworker to develop chronic skin issues — nothing taught us faster than seeing that firsthand.
Solid Safety Measures
Personal protective equipment is mandatory, not just advised. Wearing gloves and long sleeves minimizes exposure. Eye protection isn’t optional. Operating in well-ventilated areas keeps dust away from your lungs and reduces accidental inhalation. Storage in a dry place far from combustible chemicals helps avoid nasty chemical interactions. Any spills need containment with lots of water, while staying clear of acids or organic material nearby.
Real-World Solutions
Routine training keeps safety rules from becoming just background noise. Labeling all containers carefully, even temporary ones, avoids confusion on site. Rapid access to emergency showers and eye-washing stations saves precious seconds if an accident happens. Encouraging honest reporting of close calls builds a stronger safety culture — one that everyone on the team trusts.
How should sodium permanganate be stored?
What Makes Sodium Permanganate a Storage Challenge?
Sodium permanganate draws attention in water treatment and chemical manufacturing for its strong oxidizing powers. Its deep purple color might look stunning in a lab jar, but the risks painted by its reactivity highlight the need for careful storage. In my years working around treatment chemicals, I’ve seen what can go wrong when someone shrugs off common-sense practices. Leaks, heat, and dodgy containers invite problems, and that goes double for sodium permanganate.
Keep It Dry, Keep It Safe
Chemicals like this don’t play well with moisture. Sodium permanganate meets humidity, and soon clumps or even leaks start. Water doesn’t just mess with the product—it can also help generate heat, and that spells danger for a strong oxidizer. Storing the chemical in a dry, well-ventilated space keeps risks lower and shelves cleaner. In facilities I’ve visited, dry rooms set away from doorways and wash-down areas help keep chemicals from ruining both product and peace of mind.
Strict Containers Only
Some substances tough out mediocre packaging, but sodium permanganate expects more. Polyethylene or stainless-steel drums with tight seals don’t just help stop leaks—they also prevent accidental mixing with incompatible materials. I remember a maintenance manager who once stored it in a rusty steel drum, thinking it didn’t matter. That mix-up led to a leaky mess and ruined product. High-quality, chemical-resistant containers might cost more, but the cost of an incident runs higher.
Separate from Flammable and Reducing Agents
Oxidizers like sodium permanganate find trouble fast when stored near fuels, solvents, or other reducers. I’ve seen fires ignited in similar cases, sparked by nothing more than poor planning and a stray spill. Physical segregation—never just a label on a shelf—keeps things calm. On a well-run site, oxidizers stand far from organics, acids, and other chemicals that could feed an accidental reaction.
Cool, Shaded Environments Stop Heat Build-Up
Exposing sodium permanganate to sunlight or high temperatures raises the odds of a fire or decomposition event. Rather than risking it next to furnace rooms or windows, a climate-controlled area works best. Warehouse managers who install temperature logs don’t do it to look fancy; they know that spikes in heat have led to chemical accidents in real life. Consistent, moderate temperatures not only protect product quality but also safeguard workers.
Proper Labeling and Emergency Prep
Clear, tough labels that stand up to spills or abrasion help workers avoid “what’s in that drum?” mistakes. I remember a close call in a small plant where masking tape doubled as a label—the wrong chemical got added to a reaction tank, jeopardizing the whole shift. Durable labels and a rule that nothing moves without a Safety Data Sheet (SDS) nearby have prevented repeats of those days.
Training Makes the Difference
Written protocols and safety videos only go so far unless people know why precautions matter. I’ve sat in trainings full of yawns, but nothing sticks like seeing the aftermath of a mishandled drum. Hands-on teaching, covering transfer procedures, spill cleanup, and protective gear, turns book learning into habit.
Solutions that Work Every Day
Facilities that invest in secure, ventilated chemical storage and tough containers see fewer incidents with sodium permanganate. Periodic safety drills, clear chemical segregation, and a badge system for those who handle the substance further reduce risk. My advice: never assume an oxidizer “can wait” for proper handling. Years in the field have proven that good storage practices save product, time, and—most importantly—lives.
What is the difference between sodium permanganate and potassium permanganate?
What Happens in Real-World Use?
Walk into a water treatment plant or a chemistry storeroom, and you’ll spot the deep-purple sparkle of permanganate compounds. People often ask about the difference between sodium permanganate and potassium permanganate. Anyone who’s used these in a lab or on a job site can tell you: the choice between them isn’t just about which is easier to spell.
Handling and Dissolving the Compounds
Potassium permanganate forms solid crystals and doesn’t dissolve as easily as sodium permanganate. Anyone who has prepared solutions with it knows it takes some patience and a fair bit of mixing to get a uniform one. Sodium permanganate, on the other hand, dissolves quickly and completely in water, which means no waiting for stubborn grains to break down. For large-scale dosing—like in groundwater remediation—this saves real time and avoids uneven concentration. Fast, predictable mixing helps avoid mistakes that cost money and have real consequences if you’re dealing with dangerous pollutants or tricky chemical reactions.
Cost and Availability
Cost sometimes decides the winner. Potassium permanganate is less expensive, and supplies reach a broader customer base. Sodium permanganate costs more to produce and often comes in liquid form, which adds shipping concerns. Still, spend any time cleaning up environmental spills or treating municipal water, and you may discover that the initial cost doesn’t always tell the whole story. The easier handling and higher solubility of sodium permanganate might mean lower total chemical costs, fewer shutdowns, and safer storage for teams who use these chemicals daily.
Safety and Storage Concerns
Ask anyone who has handled these chemicals—permanganates act as powerful oxidizers, so respect is key. Both compounds stain skin and equipment, and they react fiercely with organic material. Liquid sodium permanganate arrives ready to go, without dust, which means less inhalation risk than with granular potassium permanganate. Spills of sodium permanganate also clean up more quickly, since undissolved chunks aren’t left behind. Every worker knows which means fewer stained hands and less time scrubbing tanks or filters.
Uses and Situational Suitability
In the field, sodium permanganate shows clear advantages for groundwater clean-up projects or continuous dosing jobs, thanks to its faster mixing and liquid form. Potassium permanganate stands out for water treatment plants, aquaculture, and disinfection, especially in places where cost pressures run high. Homeowners and aquarium hobbyists usually stick with the popular purple granules for simplicity and affordability.
Environmental Impact and Sustainability
Both chemicals break down into manganese dioxide after use, but the high solubility of sodium permanganate ensures a more controlled and complete reaction, leaving fewer residues behind. Potassium permanganate, with its solid form, sometimes causes over-treated “hot spots” if mixing falls short. A chemist or project manager seeking best outcomes for both people and the environment often weighs the long-term impact of residues and the reliability of dosage.
Potential Solutions for Better Decision-Making
Many organizations benefit from straightforward chemical audits. Take stock of project goals and risks before picking a compound. Brief safety training or mixing demonstrations cut accidents and save money. Partnering with trusted chemical suppliers can give teams early heads-up on supply chain hiccups or price movements. If costs or safety worries keep popping up, switching from one permanganate to the other sometimes pays off in ways that don’t show up on a spreadsheet—cleanup time, peace of mind, even morale in the maintenance crew.
What precautions should be taken when using sodium permanganate?
Understanding the Risks
Sodium permanganate stands out as a heavy-duty oxidizer. In water treatment or chemical processing, folks trust it to clean up organic muck, control odors, and tackle contaminants. But its strong nature brings risk into the workplace. Small mistakes can lead to fires, toxic fumes, or nasty burns. Respecting the properties of this chemical helps prevent emergencies and protects those who work with it every day.
Personal Protection Comes First
Staying safe doesn’t take complex gear, but nobody should skimp on the basics. Goggles shield your eyes from splashes. Gloves protect hands from irritation or burns. Aprons or long sleeves spare your skin from accidental spills. Even with ventilation, a good mask keeps out fine dust. In my experience around hazmat sites, the difference between an ordinary day and a painful rush to the ER often lies in remembering to gear up before touching the bag or bottle.
Safe Storage Prevents Trouble
Many accidents begin at the storage shelf. Sodium permanganate reacts aggressively if it meets the wrong substance — for instance, glycerin, strong acids, or even some solvents. Trouble brews fast when containers sit near combustibles or ordinary cleaning agents. Dry, cool places with tightly sealed containers say “no” to moisture, ignorance, and random mix-ups. At one plant I toured, bright safety labels on each drum and a separately locked closet for oxidizers slowed down mistakes before they could start.
Handling with Common Sense
Pouring, scooping, or mixing sodium permanganate needs a calm hand. Pouring quickly stirs up dust, which stings lungs and lashes eyes. Slow, deliberate movements in a well-ventilated area keep particles from flying. Tools get cleaned before and after use. Containers should stay closed except for the minutes they’re needed. Workers get training so they know what to expect from the chemical’s reactions, especially if a spill happens. Strict clean-up routines become second nature after you see what an unplanned reaction can do.
Dealing with Spills or Exposures
If a spill lands on the floor, water alone makes things worse by spreading contamination. Absorbent, non-flammable materials like vermiculite come in handy, scooped up and sealed in sturdy bags. Wash minor skin contact right away, but never use bleach or acid, since it sparks more reactions. Every emergency eyewash and shower should be kept clear, checked often, and always within a short sprint from where chemicals are handled. Once I saw a forgotten eyewash station bench-blocked by boxes, and it chilled me — emergencies don’t wait for someone to move clutter.
Training and Response
No one should work with sodium permanganate without solid training. Emergency drills, clear signage, and phone numbers for poison control or the fire department stay posted at eye level. A prepared team knows how to warn the rest of the building and how to spot a medical emergency. Following OSHA guidelines means fewer guesses and safer outcomes. If you forget why the rules seem bothersome, speak to someone who has seen what happens without them.
Solutions That Work
Industry groups keep improving safety data sheets and sharing incident reports so lessons spread quickly. Developing better packaging cuts down on leaks or confusion. Some facilities install sensor-activated exhaust fans or spill alarms, adding another layer of protection. Listening to veteran workers and involving everyone in safety talks builds a stronger workplace culture and keeps complacency off the floor.
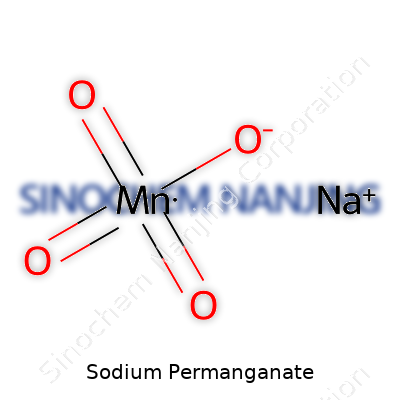
| Names | |
| Preferred IUPAC name | sodium manganate(VII) |
| Other names |
Sodium manganate(VII) Permanganic acid, sodium salt Sodium permanganate(VII) |
| Pronunciation | /ˌsoʊdiəm pərˈmæŋɡəˌneɪt/ |
| Identifiers | |
| CAS Number | 10101-50-5 |
| Beilstein Reference | 385873 |
| ChEBI | CHEBI:78035 |
| ChEMBL | CHEMBL1201567 |
| ChemSpider | 153429 |
| DrugBank | DB11304 |
| ECHA InfoCard | 040000019875 |
| EC Number | 232-232-5 |
| Gmelin Reference | 33037 |
| KEGG | C18698 |
| MeSH | D017758 |
| PubChem CID | 24414 |
| RTECS number | SD8750000 |
| UNII | KM2S90M2NO |
| UN number | UN3214 |
| Properties | |
| Chemical formula | NaMnO4 |
| Molar mass | 141.94 g/mol |
| Appearance | Dark purple to almost black crystalline solid or granular powder |
| Odor | Odorless |
| Density | 2.703 g/cm³ |
| Solubility in water | Very soluble |
| log P | -4.00 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 26.0 |
| Basicity (pKb) | pKb ≈ 6.37 |
| Magnetic susceptibility (χ) | +11800.0e-6 cm³/mol |
| Refractive index (nD) | 1.39 |
| Viscosity | 3 cP at 20 °C |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 174.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -813.4 kJ/mol |
| Pharmacology | |
| ATC code | V03AB19 |
| Hazards | |
| Main hazards | Oxidizer, causes severe skin burns and eye damage, harmful if swallowed, may intensify fire |
| GHS labelling | GHS05, GHS07, GHS09 |
| Pictograms | GHS05,GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H272, H302, H314, H410 |
| Precautionary statements | P210, P220, P221, P280, P370+P378, P305+P351+P338, P310, P501 |
| NFPA 704 (fire diamond) | 3-0-2-OX |
| Lethal dose or concentration | LD50 Oral Rat 750 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 750 mg/kg |
| NIOSH | RN9940000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 12 mg/m³ |
| Related compounds | |
| Related compounds |
Potassium permanganate Permanganic acid Sodium manganese(IV) oxide |

