A Real-World Look at Sodium Periodate
Historical Development and a Ground-Level Introduction
Sodium periodate doesn't make front-page headlines, but the folks in labs and chemical plants have counted on it for over a hundred years. In the early days of organic chemistry, the old-school researchers realized they needed a cleaner way to break open stubborn molecules without making a mess. Enter sodium periodate. Chemists like Wigand and Fenton, working in Europe at the turn of the 20th century, figured out how this salt could oxidize glycols, opening new doors in both industrial and academic realms. As scientists dug deeper, they saw how wide the ripple effect could be, from basic research to large-scale production.
Core Characteristics: More Than Meets the Eye
Sodium periodate comes in two forms that matter in the lab: the anhydrous version and the hydrated variety, usually the dihydrate. Both pack a punch in the oxidizing department. They look like simple, white, almost crystalline powders, but the real action hides in the chemical makeup. With the formula NaIO4 or Na5IO6 (in the case of metaperiodate and orthoperiodate), this stuff handles water like a pro—it dissolves cleanly, and the presence of iodine in a high oxidation state sets the tone for its reactivity. Many people are surprised at how stable it sits on the shelf if kept away from moisture and light, but on command, it becomes a workhorse for oxidative cleavage.
Technical Specs and Labeling: More Than Just a Bag of Powder
Most bottles of sodium periodate must be handled with respect. You’ll see purity levels up to 99% for reagent grade, which matters for research but also impacts cost in industrial batch processes. Labels focus on the oxidizer symbols, storage warnings, and batch numbers. Chemists appreciate how tightly specs tie into expected reactivity and ease of audit for traceability in critical applications. If you’ve ever had a batch that underperformed, you know specs are not just legal fluff; they’re the backbone for reproducibility and trust.
Preparation Method: From Theory to Practice
On paper, sodium periodate looks simple to make: it grows out of the reaction between sodium iodate and chlorine gas in an aqueous medium, or from the electrochemical oxidation of sodium iodate. Scale it up, though, and it takes real-world skill. Workers wear serious PPE, and quality matters at every step, including temperature control and precise dosing of reactants. Pilot plant engineers have tales of rushed batches or missed endpoints that cost time and money. The game changes when impurities creep in, so routine purifications follow, often with filtration, washing, and drying under vacuum.
Chemical Reactions and Modifications: How It Actually Gets Used
The bread and butter for sodium periodate lies in breaking carbon-carbon bonds in vicinal diols, a mainstay in carbohydrate chemistry. Analytical labs rely on this for structural work, while medical research uses it for surface modification of polymers, notably in the world of biomedical hydrogels and tissue scaffolds. Periodate’s ability to react cleanly, without over-oxidizing, explains why it remains a staple in oxidative cleavage even as trendier reagents pop up. Synthetic chemists appreciate its selectivity and the old-fashioned reproducibility.
Other Names on the Bottles
Walk through any storeroom, and sodium periodate will show up as sodium metaperiodate, sodium orthoperiodate, or just “periodic acid sodium salt” on older stock. The IUPAC naming can trip up students, but seasoned researchers know to cross-check CAS numbers and melt points to avoid surprises in sensitive experiments.
Working Safely: No Room for Complacency
Anyone who’s handled sodium periodate knows it earns the oxidizer symbol for a reason. Toss it near organic materials or store it next to combustibles, and you invite trouble. Ventilation matters. There’s no substitute for fume hoods and dust control, especially when weighing powders for batch synthesis. Reports of skin and respiratory irritation remind workers to suit up, check glove compatibility, and avoid open flames or sparks. Regulatory bodies like OSHA and the European Chemicals Agency treat sodium periodate as a hazardous material, so transport and disposal rules come down hard and clear. In my own lab experience, slip-ups often come from seasoned workers who get too comfortable, reinforcing the need for recurring safety drills and reviews.
Applications Outside the Textbook
Sodium periodate performs wherever selectivity and oxidative muscle outweigh the drawbacks of handling it. Analysts use it in determining carbohydrate structures, mapping out plant polysaccharides, or creating defined aldehyde groups for further linkage. Its reputation shines in blood glucose assays, where enzymes paired with periodate-generated intermediates power diagnostic kits that see action in clinics across the globe. Beyond the bench, textile and water treatment facilities turn to sodium periodate to degrade dyes and stubborn organics. Environmental chemists use it to help break down pollutants that resist conventional treatments.
New Research Directions and Real-World Hurdles
Academic and industrial researchers keep tweaking how sodium periodate gets put to work. Innovations focus on greener synthesis, both to cut emissions during manufacture and to recycle residues after use. Electrochemical approaches hold promise for reducing chlorine use during production and minimizing waste streams. Bioengineering fields count on sodium periodate for gentle modification of natural polymers like cellulose, aiming for new biomaterials and smart drug delivery gels. Yet hurdles remain. Sodium periodate’s strong oxidative properties limit its fit for certain sensitive molecules and processes, creating a steady challenge for those adapting protocols or controlling unwanted side reactions. In my experience, patience runs thin when purification steps multiply, so faster, in-line monitoring promises big gains in both yield and safety.
Toxicity Research: Risks and What’s Still Unclear
The dangers tied to sodium periodate come from its ability to disrupt living cells. Tests in animals and lab assays show it’s moderately toxic, with irritant effects on the lungs and eyes, and it can pose risks if inhaled as dust or mist. Occupational limits on exposure reflect findings from both rodent studies and observational reports among workers. Chronic exposure data remains patchy, so risk assessments stay on the cautious side. Drinking water contamination isn’t a front-burner issue, but spill protocols remain strict due to uncertainty over long-term health impacts, especially for populations with underlying conditions.
The Road Ahead: What Comes Next for Sodium Periodate
Looking forward, the real question centers not on whether sodium periodate holds value—its selective oxidation has proven itself over decades—but rather on whether production and use can keep evolving with tighter environmental standards. Tougher limits on waste, new regulations for worker safety, and community concerns about chemical runoff keep the pressure on to find alternatives or at least make the process cleaner. Researchers chase catalysts that mimic periodate’s unique capabilities without the same hazard profile, but nature hasn’t handed out many easy swaps. For now, the future of sodium periodate rests on making its power available in safer, more sustainable ways, pairing old-school chemistry with a new world of green mandates and transparency.
What is sodium periodate used for?
A Powerful Tool for Chemists
Sodium periodate finds its way into labs not because it's flashy, but because it works hard. I remember the first time I watched an organic chemist use it; the glittering white powder seemed plain enough, yet it kicked off reactions with real purpose. Chemists lean on it as an oxidizing agent, which means it helps pull apart certain molecules by adding oxygen. This skill, breaking up complex molecules like sugars, comes in handy in all sorts of fields, especially in the search for new medicines and in analyzing biological samples.
Medicine, From Research to Diagnosis
People often overlook how much of the medical world depends on specialized chemicals. Sodium periodate plays a big role in preparing tissues and cells, particularly through something called periodic acid–Schiff (PAS) staining. If a pathologist wants to see details inside a cell or spot early disease, they often rely on this stain. Imagine trying to diagnose a rare blood disorder or finding signs of kidney damage—the ability to stain subtle structures with sodium periodate can make a real difference in catching problems before they grow.
Quality Control in the Food Industry
Food scientists also give sodium periodate a regular workout. Sugar content and structure matter a lot in everything from baked goods to energy drinks. By breaking down polysaccharides, sodium periodate helps determine exactly what types of carbohydrates are present and in what quantity. I've talked to manufacturing specialists who say their daily quality checks would be tougher without chemicals like this. They count on accurate analysis for consistency, taste, and, crucially, for meeting health regulations.
Materials Science and Technology
Materials science isn’t just about inventing new plastics or improving screens—it’s also about controlling exactly how materials behave. Sodium periodate comes into play when scientists want to modify the surface of cellulose fibers, which can improve water absorption or glue bonding. In a world where every little edge matters, companies experiment with sodium periodate to give textiles or packaging new features. This approach supports innovation in sustainability, since altering plant-based fibers can reduce dependence on synthetic chemicals.
Safety Concerns and Handling Practices
As useful as sodium periodate can be, no one should take risks with it. This substance can cause burns and irritation, so safety gear stays non-negotiable. Training and proper storage prevent most accidents. Regulators enforce strict labeling, and companies invest in waste treatment to avoid polluting water systems. Cutting corners never pays—I've seen mishandling lead to costly shutdowns, not to mention danger for workers.
Pushing Toward Greener Chemistry
Some folks worry about the impact of strong oxidizers, and that conversation matters. Research now targets ways to recycle or neutralize leftover sodium periodate. Alternative “green” oxidizers continue to pop up, yet sodium periodate hasn’t lost its place for special jobs requiring precision. The challenge now is to use it efficiently, capturing benefits while cutting down on waste.
Keeping Science Moving
The world keeps asking for smarter health tests, tougher materials, and safer foods. Each of those goals leans on chemicals like sodium periodate. It quietly bridges the gap from scientific discovery right into things people use every day. The real trick, as experience keeps showing, is to strike a balance—driving science forward while considering safety and the planet.
What is the chemical formula of sodium periodate?
Exploring the Basics of Sodium Periodate
Every science classroom usually holds stories of spilled chemicals and confused faces, but one compound’s formula always seemed to cause a special brand of head-scratching: sodium periodate. Its chemical formula can show up in more than one form, adding to the confusion. Sodium periodate exists as either sodium metaperiodate (NaIO4) or sodium orthoperiodate (Na5IO6). Most laboratories use it in the metaperiodate form because it dissolves readily in water and packs a punch as an oxidizing agent.
Why Knowing the Formula Matters
Someone working in research or manufacturing often needs to keep exact chemical formulas in mind. Mistaking NaIO4 for Na5IO6 can lead to ruined experiments, wasted funding, and occasional safety mishaps. Chemical formulas carry more than letters and numbers—they show the ingredients, the recipe. In one project, I learned this lesson when a mix-up between sodium periodate and sodium perchlorate forced our team to redo a week’s worth of work. The fallout wasn’t just wasted time; it came with the embarrassment of presenting the wrong findings to the department lead. That sting still hangs around, highlighting how small notational details shape much larger consequences.
How Sodium Periodate Gets Used
Few people outside research circles realize sodium periodate earns its keep well beyond test tubes. Medical diagnostics and organic synthesis both lean heavily on its oxidizing power. In biochemistry, scientists rely on sodium periodate to cleave sugars in glycoproteins—a step that’s central to mapping these complex molecules. In industrial processes, it delivers a cleaner alternative to harsher oxidizers, supporting safer protocols and cleaner waste streams. These roles tie directly to its formula, since the number of oxygens determines just how reactive and selective the agent will be.
Reliable Information Sources Build Trust
Students and professionals both get hit with conflicting data online about these compounds. I’ve watched newcomers to chemistry head for online forums, only to come away more confused after sorting through unverified guesses. Reliable, peer-reviewed sources like those from the National Center for Biotechnology Information or the Royal Society of Chemistry keep facts straight. For sodium periodate, these trusted databases consistently cite NaIO4 for the most commonly used variant. Trust builds over time when you return to these sources; you stop second-guessing every detail and start building deeper understanding.
Addressing Misinformation and Improving Chemistry Education
Many mistakes around sodium periodate and similar compounds start with unclear teaching and poorly designed reference material. In some schools, outdated textbooks and underfunded science labs slow down learning and spark confusion. I’ve seen students light up once the right resources or simple demonstrations clear up a stubborn chemical puzzle. Stronger basics, backed by up-to-date materials, make every formula carry meaning. Teachers benefit from direct access to reliable, interactive sources—and students can then work through problems without falling for common traps. Over time, these shifts close the knowledge gap, making complex substances like sodium periodate less intimidating and much more accessible.
Is sodium periodate hazardous to health?
The Hidden Risks in the Lab
Sodium periodate isn’t a household name. In the research world, though, it shows up in chemistry spaces, particularly for organic synthesis and analytical tests. Through years in the lab, I’ve watched people treat substances like this with casual acceptance, often glossing over safety data sheets. This shortcut puts people at risk, since sodium periodate offers more than a slight nuisance to human health.
Physical Contact and Inhalation Dangers
Direct skin contact rapidly leads to irritation—sometimes redness, itching, or mild burns. Once, a lab mate spilled a tiny scoop and laughed it off, only to regret it minutes later. This chemical, in powder form, floats into the air easily. Breathing in the dust leaves a burning sensation in the nose and throat and can trigger coughing fits. In more severe exposures, there’s risk of lung tissue damage. A Cambridge study highlighted increased hospitalization rates for people suffering acute respiratory exposure to similar oxidizers. Simple goggles and gloves can spare a lot of discomfort here.
Toxicity Beyond the Obvious
Ingestion brings a different level of hazard. Swallowing even small amounts damages tissues in the mouth, esophagus, and stomach, and can lead to vomiting and diarrhea. Most frustrating about chemicals like this is how symptoms sometimes lag behind the exposure. Folks eat or drink after touching it, thinking it’s fine until stomach trouble kicks in hours later. Laboratories should keep eye wash stations handy—not just for periodic checks, but because the risk is real. In a documented case out of Texas, delayed medical attention after an exposure led to several days in the hospital for one unlucky student.
Chemical Burns Impact More than Skin
Sodium periodate’s greatest hazard lies in its powerful oxidizing abilities. It reacts violently with certain organics or reducing agents, which means storage and handling protocols matter. Mixing with everyday items like paper towels, alcohols, or even a dirty benchtop may release toxic gases or cause fires. A published incident in a research lab pointed to an overheated trash can as a source, all because periodate waste mixed with cleaning rags. That incident, though minor, forced a full building evacuation. So, waste should go in designated containers—and never in open bins or regular trash.
Health Impacts: What Science Says
Data from the CDC and international health agencies show that chronic exposure, even in low doses, weakens tissues over time. My colleagues working in analytical labs underwent routine skin and lung exams, as recommended by occupational medicine experts. Ionizing chemicals can impair lung function and lower immune response. Some early-stage research in mice even suggested a link to changes in liver and kidney enzyme levels. Keeping records, offering regular health screening, and following strict clean-up routines help keep workers safer. It costs pennies to prevent what costs thousands in medical bills.
Solutions Aren’t Complicated
Effective ventilation, sealed containers, and well-trained staff do more than meet a checklist. In my own teaching, I stress that ordinary habits—washing hands after handling glassware, never eating near chemicals, reporting spills promptly—set the tone for lasting safety. Simple steps work better than over-engineered solutions. Workers deserve to leave their jobs each day with the same health they brought in that morning.
How should sodium periodate be stored?
Keeping People and Product Safe
Sodium periodate has a reputation for strong oxidizing power. Labs use it for breaking down molecules and creating new ones, but the story doesn’t end at the bench. It’s easy to overlook how important smart storage is until something goes wrong—a leaky cap, a splash, or worse. I remember working with oxidizers in a shared lab fridge, and one careless move put a row of reagent bottles at risk. Good storage habits make all the difference between a routine day and ruined research.
Moisture Doesn’t Mix Well
No one enjoys opening a bottle only to find clumps or crust in place of crisp white crystals. Sodium periodate attracts moisture like a sponge, and water kicks off chemical changes that weaken the reagent. Keep it tightly sealed, and make sure the lid is always clean and dry before closing. Even in humid climates, a sealed container guards against unwanted hydration and helps avoid waste.
Temperature and Light: The Low-Tech Solution
Heat and sunlight push sodium periodate toward decomposition, creating risks ranging from poor results to irritation or even fire hazards. A cool, dark cabinet delivers more than safety—reagents stay effective longer, and researchers don’t need to scramble for replacements. Flammable storage cabinets might feel like overkill, but they send the right message: here lives a chemical that can’t be treated like table salt.
Separation Matters
Too many stories start with someone shoving chemicals together on a shelf. Put sodium periodate near flammables, acids, or reducing agents, and disaster comes easy. I once saw bleach stored next to ethanol; it only takes a reaction between oxidizers and common solvents to create toxic fumes or fire. Assign a spot just for oxidizers and keep reducing compounds far away. Labels aren’t just red tape—they’re reminders that the safest routine is the one you never want to skip.
Original Containers: The Quiet Hero
Nothing beats the manufacturer's container. It’s designed for chemistry, with seals that keep out air and moisture. Switching to an unlabeled or worn-out bottle opens the door to confusion, cross-contamination, and accidents. If a transfer becomes necessary, new containers must be robust and labeled by hand, with clear dates and hazard markings. If mistakes sneak in here, even seasoned chemists fumble.
Handling Spills and Accidents
Despite all precautions, spills still happen. Because sodium periodate reacts aggressively with organics, water, and some plastics, standard paper towels or a swipe with a damp rag make the situation worse. Absorb spills with inert materials. Cleanup kits near the storage location make it easier for anyone to act quickly. Everyone in the lab or workplace should know the procedure—no one likes guessing in a crisis.
Why Details Matter in Practice
Storing sodium periodate sounds straightforward, but it’s the details that keep people safe. Learning the ropes early means fewer close calls, better science, and less money lost to ruined chemicals or cleanups. In any setting where this compound makes an appearance—research, industry, teaching labs—a culture of care and respect pays off day after day. Safe storage isn’t only about rules. It’s about protecting lives, research, and the peace of mind needed to solve real problems.
What safety precautions are needed when handling sodium periodate?
Why Respect for Sodium Periodate Matters
Anyone who's spent time in a lab knows mistakes can haunt you. Sodium periodate isn’t a friend you want to handle carelessly. With its strong oxidizing nature, simple missteps can spark chemical burns or start a fire, and exposure can punch your lungs or skin harder than you’d expect. I remember a story from my undergrad days, where a rushed colleague didn’t check for a clean work space during prep. An unnoticed spill mixed with leftover organics and ate through a bench mat before people caught the acrid smell. That mess ended an afternoon for half the department and led to some sharp reminders at the next safety meeting.
Personal Safety Means Real Barriers
Goggles don’t just sit on my forehead. I choose wrap-around eye protection, every single time. Chemical splash goggles make the difference between an ordinary cleanup and an ER trip. Gloves matter, too. Not just the standard latex – thick nitrile or neoprene stands a chance to keep hands untarnished when sodium periodate threatens. Some people assume sleeves will protect their arms, but cotton loves to hold onto chemicals. A long-sleeve lab coat made from flame-resistant fabric can prevent the kind of burns that take months to heal.
Air Matters More Than You Think
Fume hoods aren’t decorative. That faintly sweet, sharp smell tells you even a small quantity can drift, and that isn’t something anyone should breathe. Chronic exposure brings up warnings about possible lung injury. I’ve almost forgotten to turn on the blower during tedious setups, and the gentle guidance of a senior researcher stopping me never gets old. That’s not just caution—it’s a habit worth building.
Storage Matters. Labeling Matters More.
Sodium periodate and organic materials don’t mix nicely. Give this stuff its own labeled, chemical-resistant container. Even a crumb of mishandled powder next to solvents can ruin your day. In the research spaces I’ve visited, the bottles get double-sealed and marked in plain handwriting, not faded labels from two years ago. Good storage means cool temps, dry shelves, and clear warning signs.
Spills and Disposal
Quick action beats panic during a spill. Loose powder needs gentle sweeping into dedicated containers — no improvised dustpans that contaminate garbage down the line. Liquid waste never joins the regular trash stream. Specialized containers, and calls to hazardous waste team, keep toxic byproducts away from public water supply. I watched one hasty undergrad dump diluted periodate into a normal drain; their explanation didn’t fly, and a supervisor spent hours sorting out consequences with environmental compliance officers.
The Importance of Training and Repetition
Nothing builds muscle memory better than repeated, honest training. Relying on a quick scan of a safety sheet won’t cut it. Refresher courses prevent the small shortcuts that lead to big accidents. Institutions with zero injury records usually invest the most in regular drills and visible reminders. Spending five minutes walking through protocols keeps both nerves and skin intact — for every beginner and old hand alike.
Practical Fixes and Real Accountability
Handling sodium periodate safely depends on culture, not just equipment. Supervisors setting good examples, regular checks on safety gear, and zero tolerance for shortcutting protocols make a visible difference. Even small roles — someone double-checking the inventory, labeling a replacement bottle, or calling out a lapse — reinforce a culture that keeps everyone safe. Industry-wide data supports the idea that real improvements follow tough, detailed oversight combined with honest feedback from staff in the trenches.
Caring for every step of sodium periodate use — from PPE to proper labeling, to disposal and training — matters because folks working around hazardous materials deserve to head home safe each day. Facts back it up: even seasoned chemists make mistakes, but teams investing in basics keep disaster out of the lab and off the news.
| Names | |
| Preferred IUPAC name | Sodium periodate |
| Other names |
Sodium metaperiodate Sodium salt of periodic acid Disodium periodate Sodium orthoperiodate |
| Pronunciation | /ˌsəʊdiəm paɪˈrɪə.deɪt/ |
| Identifiers | |
| CAS Number | 7790-28-5 |
| Beilstein Reference | 4039889 |
| ChEBI | CHEBI:75144 |
| ChEMBL | CHEMBL1200867 |
| ChemSpider | 51874 |
| DrugBank | DB13428 |
| ECHA InfoCard | 100.028.897 |
| EC Number | 231-890-0 |
| Gmelin Reference | 1102 |
| KEGG | C00642 |
| MeSH | D011086 |
| PubChem CID | 24816 |
| RTECS number | SD9800000 |
| UNII | 9D95Q8R1LO |
| UN number | UN1479 |
| Properties | |
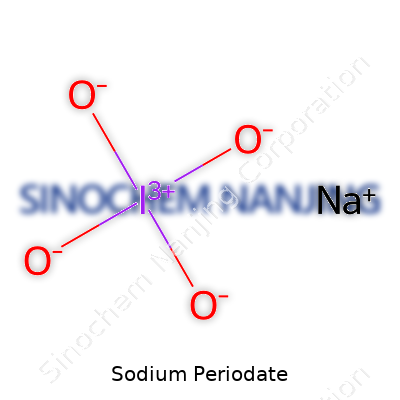
| Chemical formula | NaIO4 |
| Molar mass | 213.89 g/mol |
| Appearance | White crystalline powder |
| Odor | odorless |
| Density | 3.865 g/cm³ |
| Solubility in water | soluble |
| log P | -4.2 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 6.0 |
| Basicity (pKb) | 11.35 |
| Magnetic susceptibility (χ) | +79.0e-6 |
| Refractive index (nD) | 1.741 |
| Viscosity | Viscous liquid |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 214.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –298.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -295.7 kJ/mol |
| Pharmacology | |
| ATC code | V03AB52 |
| Hazards | |
| Main hazards | Oxidizer, harmful if swallowed, causes severe skin burns and eye damage |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H271, H302, H318, H335 |
| Precautionary statements | P210, P220, P221, P260, P264, P280, P301+P310, P305+P351+P338, P306+P360, P370+P378, P403+P233, P404, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-2-OX |
| Lethal dose or concentration | LD₅₀ (oral, rat): 1,890 mg/kg |
| LD50 (median dose) | 250 mg/kg (rat, oral) |
| NIOSH | SN1225000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 35-40 |
| IDLH (Immediate danger) | IDLH: 250 mg/m3 |
| Related compounds | |
| Related compounds |
Sodium chlorate Sodium bromate Sodium iodate Potassium periodate |

