Sodium Pentachlorophenoxide: A Commentary on its Development and Role in Industry
Historical Development
The story of sodium pentachlorophenoxide traces back to early studies in the field of chlorinated phenols. Through the decades spanning the 20th century, chemists recognized that modifying natural aromatic compounds produced agents with potent biocidal properties. Agricultural expansion and wartime logistics during the mid-century years pushed the demand for stubborn disinfectants and pesticides. Sodium pentachlorophenoxide, formed by treating pentachlorophenol with a strong base, offered a tool that could address fungicidal needs efficiently. Government agencies rolled out regulations as soon as toxicological data emerged, and manufacturers responded with additional efforts toward standardizing output and mitigating environmental risks. The scientific backstory reminds us that every chemical innovation stands on layers of testing, regulatory pushback, and real-world learning.
Product Overview
Sodium pentachlorophenoxide takes shape as a white or slightly off-white crystalline powder, distinct from its more famous cousin, pentachlorophenol. It draws attention in both the wood preservation and industrial sectors. Most folks in the field know it isn’t simply a derivative or substitute; its reactivity and solubility mean it excels in applications that demand strong, lasting antimicrobial effects. The product has no trouble dissolving in water, and it outperforms non-ionic phenolic compounds in deployment speed. From experience, workers prefer compounds that mix quickly and deploy predictably, which gives sodium pentachlorophenoxide strong appeal where fast action matters.
Physical & Chemical Properties
With a molecular formula of C6Cl5ONa, sodium pentachlorophenoxide boasts a notable density and stands out for its stability under a range of ordinary storage conditions. At room temperature, it keeps a solid form and does not emit significant odor—though it tends to release a noticeable phenolic tang if left in humid spaces. In simple terms, its high chlorine content drives why it does not degrade easily or react haphazardly with environmental agents. In laboratory conditions, it holds up under moderate heating, but it starts to decompose if exposed to very high temperatures or strong acids, evolving toxic gases such as hydrogen chloride and related chlorinated organics. Handling the powder directly, you feel the fine, slippery texture—a reminder of its purity and tight particle size distribution. Water brings the crystals into solution rapidly, and in my experience, this feature proves vital for technicians who do not want to waste time with sluggish reagents.
Technical Specifications & Labeling
Reputable suppliers provide sodium pentachlorophenoxide meeting stringent technical standards. Assay values typically surpass 98% on a dry basis, and allowable moisture content stays well below 1%. Labels call out storage instructions, including preference for sealed containers and dry, well-ventilated warehouses. Chemical safety statements focus on the dangers of inhalation and skin contact. Labels identify the substance with other trade or regulatory names—an important step for avoiding confusion at points of use or inspection. Manufacturers also display hazard pictograms and recommendations for emergency response, as labelling integrity directly affects workplace safety.
Preparation Method
Sodium pentachlorophenoxide usually forms by reacting pentachlorophenol with sodium hydroxide. In practice, the reaction proceeds in aqueous media or after suspending pentachlorophenol in an inert organic solvent. Experienced chemists add sodium hydroxide in slow increments, controlling temperature to prevent runaway reactions or unnecessary splashing. Reaction mixtures turn clear quickly, signaling complete neutralization. Filtration removes any solid traces, and rotary evaporation or simple drying yields the powder. Plants producing at scale often automate temperature control and agitation to minimize hazards and maximize yields. Facility audits in this industry commonly focus on tank integrity and vapor scrubbing, since spills or fumes can threaten both workers and nearby communities.
Chemical Reactions & Modifications
The phenoxide ion within sodium pentachlorophenoxide serves as a launching point for further reactions. Chemists may esterify the molecule or coax it into forming metal complexes to tailor its biocidal scope. Oxidation and reduction experiments create families of related chlorophenolic substances, some of which bear distinct biological signatures. Reference labs run long-term studies on the breakdown patterns of the base compound in soil and water matrices, hoping to get a handle on environmental persistence or secondary toxification. My own experience working with related phenolic derivatives shows that seasoned chemists handle these procedures with utmost care, appreciating the variability and sometimes unpredictable behavior of organochlorine chemistry in scale-up conditions.
Synonyms & Product Names
Sodium pentachlorophenoxide goes by an assortment of aliases in chemical catalogs and regulatory filings. Pentachlorophenol sodium salt remains the clearest alternative name, with regional importers sometimes using local abbreviations. In older literature, you may spot it listed under sodium pentachlorophenate or, less frequently, as pentachlorophenate of soda. Trade names occasionally pop up, but most users focus on the core scientific moniker to avoid confusion in procurement and usage documentation. Every industry participant, from researchers to logisticians, gains from keeping a watchlist of synonyms to catch regulatory updates or shipping restrictions as they emerge.
Safety & Operational Standards
Working with sodium pentachlorophenoxide means laying down strong protocols for worker protection. At many plants, standard operating procedure may seem drill-heavy, yet every step matters because exposure brings real health risks, like chemical burns and long-term organ damage. Facilities enforce double gloving and fitted respirators. Air handling systems stay on high, with emergency eyewash stations and showers stationed within thirty paces of any chemistry set-up. Regular training drills reinforce best practices, especially for teams rotating between synthesis and waste management lines. Inspections target the storage of precursor materials as much as the finished product because leaks or losses at any point can lead to lasting consequences for soil and air quality. The safety culture here has grown from hard-earned lessons spanning decades of industrial mistakes and regulatory pushback.
Application Area
Sodium pentachlorophenoxide earns its stripes in areas where controlling mildew, mold, or destructive fungi matters most. Wood-protection specialists rely on it for treating railway ties, telephone poles, and fencing, ensuring those investments stand up to rot and environmental stress. In some producing regions, producers still use it for antitermite and general pest control, especially in timber yards under heavy weather. Ancillary uses pop up in dyes and pigment manufacture, and technical-grade product sometimes slips into specialized chemical synthesis as a strong base in aromatic substitutions. Soil researchers and field ecologists keep an eye on residual levels after major deployments. The compound’s ability to persist in treated materials brings pros and cons: on one hand, it protects assets far longer than “softer” options; on the other, its ecological footprint can ripple outward for years.
Research & Development
Researchers keep busy tracking how sodium pentachlorophenoxide breaks down across different environments. Longitudinal studies monitor whether treated wood releases harmful byproducts as it ages, and teams compare environmental loads from historic and current use. Analytical chemists push for better detection methods at lower thresholds, while process engineers test different blending and purification routines to cut trace dioxin formation. Development efforts sometimes focus on finding additives that neutralize the compound’s toxicity after use, or that enable recovery for safe disposal. Industry bodies host symposia pulling together toxicologists, regulatory scientists, and users from competing firms—a recognition that any chemical with such broad application stretches across disciplines and needs joined-up thinking to make progress.
Toxicity Research
Everything I’ve learned from working with chlorinated phenols underlines the importance of caution. Toxicology research attaches a heavy weight to sodium pentachlorophenoxide, cataloguing risks to workers, end users, and ecosystems. Laboratory rat studies show clear links to liver and kidney stress after both brief and longer exposure, and researchers continue to debate the exact cancer risk in humans. Fish and other aquatic organisms show acute sensitivity—one reason that spill response and wastewater treatment at production plants get a sharp focus. Field scientists carefully monitor for bioaccumulation, especially in marshes and riverine zones downstream from wood treatment facilities. Regulatory agencies demand regular review of published hazard data, and every credible company puts serious energy into tracking the latest findings and adapting safety sheets to reflect real-world experiences.
Future Prospects
Looking forward, sodium pentachlorophenoxide faces real tests from both policymakers and technology shifts. More substitute products compete for the wood treatment and preservative market, especially as societies demand solutions that limit persistent organic pollutants. Research into greener antifungal technologies spots every shortcoming in legacy chemicals, raising the bar for innovation and stewardship. At the same time, stakeholders with deep commitments to infrastructure and resource conservation argue that swinging too hard away from tried-and-tested agents triggers its own risks, from economic loss to premature decay in public assets. The smart path goes through transparent science, continued investment in alternatives, and policies that do not force short-term fixes at the expense of long-run health or sustainability.
What is Sodium Pentachlorophenoxide used for?
A Chemical Powerhouse in Industry
Sodium pentachlorophenoxide helps manufacturers produce chemicals for everyday products, yet it often flies under the radar. You most often find it in the backrooms of chemical plants. This compound plays a central part in making pesticides, especially pentachlorophenol, which workers use to protect wood from rot and pests. Farms, railroads, and telephone companies rely on these chemicals to keep wooden structures durable, clean, and resistant to weather. As a direct precursor to strong biocides, sodium pentachlorophenoxide supports the health of farms and forests by limiting the spread of invasive fungi and insects.
The Value and Danger of Powerful Chemicals
Products based on pentachlorophenol raised living standards in the twentieth century. Telephone poles lasted longer, rail ties stayed sturdy, storage sheds withstood the elements. Those changes made life safer and more productive. Yet it’s clear this chemical doesn’t just break down and disappear. Years in environmental testing show sodium pentachlorophenoxide’s legacy in rivers, in the ground, in wildlife, and in people’s bodies. This compound, along with its byproducts, can stay put for ages and enter drinking water, soils, and the food chain.
Workers who handle wood preservatives, industrial cleaners, or insecticides that rely on pentachlorophenol risk health problems without strong protection. Facts from long-term research show that exposure can harm the liver, kidneys, and nervous system. Some byproducts of these chemicals, like dioxins, receive attention because of their links to cancer, immune suppression, and hormone disruption. The evidence points toward the need for closer control and scrutiny, not just inside factories but out in the field.
Better Safeguards for Health and the Environment
To counter these risks, leaders in public health, policy, and industry talk about tighter limits and alternatives. Some countries have already placed sharp restrictions on pentachlorophenol-based products, focusing on safeguarding drinking water, soil health, and the people working along supply chains. I saw regulations get stricter after towns learned their water carried pesticide traces. Community groups, doctors, and regulators worked together to track and block contamination from large-scale wood treatment yards.
The push for cleaner sites and safer workplaces comes from steady pressure and solid information. Environmental monitoring, routine testing in occupational health, and transparent reporting standards all build trust. More research takes shape each year on practical, safer substitutes—organic preservatives, heat treatments, new coatings—that don’t carry the same baggage. Still, full change takes time, planning, and steady funding.
Learning from the Past and Shaping Better Practices
Stories of pollution and harm have sharpened our sense of responsibility. In my experience visiting old farm sites, I have seen firsthand how neglected wood-treatment areas can still affect soil and water after decades. Awareness on the ground keeps growing, not just in policy circles but in the next generation of engineers and environmentalists. Sharing research findings, pushing for public access to company records, and demanding worker protections help everyone learn from choices made.
In the end, sodium pentachlorophenoxide teaches a lesson found in many corners of modern life: powerful tools always come with real trade-offs. Watching what flows through our factories and yards, reevaluating what we spray and soak into the world, and replacing hazardous materials where we can—those steps matter for health, safety, and trust. That’s progress worth making.
Is Sodium Pentachlorophenoxide hazardous or toxic?
What Actually Is Sodium Pentachlorophenoxide?
Sodium pentachlorophenoxide isn’t a household word, but it shows up in discussions about chemical safety. It's basically a salt that forms when pentachlorophenol reacts with sodium hydroxide. Chemists know it for its use as a wood preservative and pesticide. I saw references to it in old farm supply catalogs and environmental hazard reports. Its chemical cousin, pentachlorophenol, already carries a heavy reputation for health risks. So it's natural to pay close attention to anything in the same family.
Known Dangers: Toxicity Is No Joke
Pentachlorophenol, and by association its sodium salt, has caused trouble for decades. The Environmental Protection Agency (EPA) lists pentachlorophenol as a probable human carcinogen. You don’t stumble across these chemicals in fruit bowls or playgrounds—most folks run into them through contaminated soil or through odd-smelling preservatives used on telephone poles or railroad ties. I once read about a mishap at a lumberyard where workers described chemical burns and breathing problems after a spill involving this stuff. Some of them developed rashes and had to be checked for liver function issues.
Lab results back up these fears. Studies show that sodium pentachlorophenoxide can damage the liver, kidneys, and nervous system. Some rodents exposed to high doses developed cancer. Human cases—thankfully rare—showside effects like headaches, weakness, and confusion. Children exposed to pentachlorophenol have, in the worst cases, died from severe poisoning.
Getting Real About Environmental Risks
This compound doesn’t easily break down in nature. It seeps into soil, drifts into groundwater, and can hang around in fish or farm animals. The EPA and European regulators both warn against eating food grown near contaminated locations. I grew up in a region with a pulp mill nearby. Anyone who drank water from shallow wells downriver from the plant would tell you the taste wasn’t just "off"—it burned the tongue, and local scientists tracked traces of persistent organic pollutants in the water. Years later, among the older folk, chronic illnesses like liver disorders and certain cancers seemed more frequent. While not every case links back to one chemical, sodium pentachlorophenoxide helped shape a pattern that communities ignored for too long.
How To Dial Down the Risk
People who work with wood treatment, manufacturing, or environmental cleanup should wear protective gear and follow proper disposal practices. Air filtration and sealed containment help keep the chemical out of breathing space. Handwashing after handling treated wood isn’t just a good idea—it should be a rule. For communities and homeowners, it pays to ask where treatment yards, old railroad ties, or contaminated wood have been dumped or buried. Testing well water and soil near these sites provides an early warning long before problems turn serious.
Regulators stepped up and put tight limits on pentachlorophenol and sodium pentachlorophenoxide use in recent years. In several countries, only trained professionals can apply these chemicals, and treated wood gets clear labeling. People can push for more transparency and regular site testing, not just for peace of mind but to catch lingering pollution before it becomes a crisis.
Safer Substitutes and Smarter Choices
Manufacturers now turn to chemicals with fewer health concerns. Borate compounds, heat treatment, and even some plant-based preservatives offer real alternatives to harsh chlorinated chemicals. Builders and homeowners can start by looking at options before buying treated wood. I’ve made it a rule never to burn or chip old railroad ties or telephone poles in backyard fires—those fumes contain more than just smoke. Even aging swing sets, if built from older preserved wood, should be replaced or covered with inert weatherproof materials.
Sodium pentachlorophenoxide's dangers shouldn’t fall into the background. Staying informed, demanding disclosure, and making safer choices keeps families, workers, and the planet healthier in the long run.
How should Sodium Pentachlorophenoxide be stored?
Storing Chemicals Beyond the Rulebook
My first run-in with industrial chemicals didn’t come from a sleek lab or a fancy textbook. It came from a dusty storeroom where someone thought a cardboard box would stand up to leaks. Folks often approach chemical storage with the attitude of “this should work,” but cutting corners can cost people their health and livelihoods. Sodium pentachlorophenoxide isn’t your average kitchen cleaner—this powder packs enough punch to irritate skin and mucous membranes, and it shouldn’t get anywhere near your coffee mug.
Risks That Don’t Wait for Paperwork
In my own work, half-hearted safety turned near disasters into lessons. Sodium pentachlorophenoxide brings risks—think corrosive fumes, flammability, toxic residue. Even old packaging failures can ruin storage rooms and threaten the people who work in them. Cases reported by the Agency for Toxic Substances and Disease Registry show that leaks often happen because containers were left open or stacked near incompatible materials. OSHA blows the whistle on this all the time: improperly stored chemicals put not just property, but lives, on the line.
Sturdier Steps Protect Real People
Packing sodium pentachlorophenoxide into a tight-lidded, chemical-resistant drum seems basic, but harsh compounds eat through weak plastics and rust cheap metals. I once saw a steel canister corrode from the inside out, unnoticed until a worker brushed against it and chemical seeped onto his skin. Solid polyethylene, reinforced fiberglass, or high-grade stainless steel keep hazardous reactions at bay.
Cool, dry locations keep the powder stable. Heat or sunlight turn seemingly quiet bins into sources of vapor or fire. Humid storage rooms also invite clumping and chemical change. I learned to steer clear of sinks, pipes, or windows. Storage shelves should stand far from passages, drinking water, or food areas. Spacing cuts down on accidental mixes—never stack pentachlorophenoxide near oxidizers or acids; this isn’t just a rulebook demand, but a barrier between safety and chaos.
Label Everything, Respect Training, and Keep Eyes Open
Caution grows from labeling every container with chemical names, hazard warnings, and the date received. People working with these materials must know exactly what they’re holding. Regular training brings up real-world scenarios—spills, mistaken grabs, faulty packaging—so workers don’t just parrot rules but spot real trouble before it grows. Instilling habits like checking containers for bulges or damage catches problems early. Tracking what comes in and goes out covers blind spots.
Building Reliable Backup—What If It Goes Wrong?
In the real world, things do go wrong. Emergency spill kits and access to eye-wash stations make a difference. Emergency plans in plain language, not buried in binders, save minutes when alarms sound. Good ventilation keeps the worst risks at arm’s length.
Room for Better Solutions
I’ve seen positive change in warehouses that moved from old metal cabinets to purpose-built hazardous storage rooms, equipped with alarms, sensors, and controlled access. These places run drills and keep inspection logs, never treating sodium pentachlorophenoxide as just another box with a warning label. Industry reports show incidents drop when companies treat storage as a living process, not a checklist.
The point isn’t to make people nervous about every chemical. It’s about making sure that after everyone clocks out, they still get home healthy. Storing sodium pentachlorophenoxide isn’t just about obeying the law, but about respecting the people whose hands touch it every day.
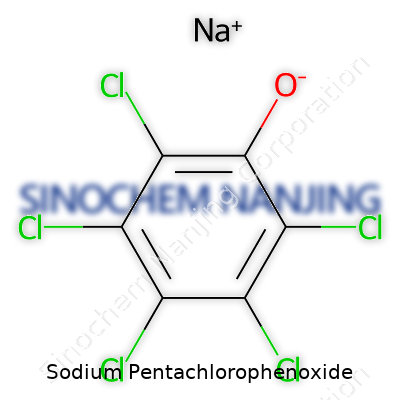
What is the chemical formula of Sodium Pentachlorophenoxide?
Understanding the Formula: C6Cl5ONa
Sodium pentachlorophenoxide, with the formula C6Cl5ONa, shows up in chemistry and industrial spaces more often than you might guess. This compound comes from pentachlorophenol, where the hydrogen on the phenol group gets swapped for a sodium atom. The change may sound small, but it rewires the substance’s behavior. Visualize the structure: a benzene ring packed with five chlorine atoms, each ready to influence reactivity, paired with an oxygen and sodium duo. After years of hands-on chemical work, these “modifications” always mean something real for reactivity or safety down the line.
Practical Importance: More Than Just a Chemical Curiosity
The manufacturing world has leaned on sodium pentachlorophenoxide for wood preservation and pest control. Years ago, while working with wood preservation teams, I saw that workers watched these treatments closely—this wasn’t a backroom afterthought. The chemical’s role in stopping fungi and insects helped push durable wood construction, especially where humidity invited decay. The chlorine-heavy structure made sure no biological intruder felt welcome.
It’s crucial to mention this chemical isn’t an everyman’s tool in the shed. Regulatory bodies such as the Environmental Protection Agency and the World Health Organization keep tight reins on its use. The persistent nature of these chlorine compounds means they don’t just break down in the soil and water. Instead, they stick around, seep into groundwater, and enter food chains. Nothing about this risk feels vague or distant—residents in agricultural regions have spoken up about health concerns more than once.
Risks: Real Lives, Real Impact
The health side of sodium pentachlorophenoxide cannot be brushed aside. Handling it in a lab or industrial setting each day hammered home the need for preparation. Workers exposed to pentachlorophenol or its sodium salt can face respiratory troubles, headaches, and, with long-term contact, more severe problems. These aren’t abstract dangers; they affect families and communities. Once, during an environmental assessment in a farming town, people shared firsthand accounts of chronic symptoms correlated with exposure to treated wood materials.
The fate of this compound in the environment is just as concerning. Water runoff from sites using the treatment can ferry residue into streams and lakes, affecting aquatic life. Fat-soluble chemicals like this build up in organisms, climbing up the food chain and amplifying risk at each step. This is not just a textbook lesson but a reality environmental scientists keep tracking in rural wetlands and fisheries.
Finding a Safer Way Forward
There’s no need for alarmist thinking, but cutting-edge research and better oversight hold promise. Some countries have begun restricting or phasing out sodium pentachlorophenoxide use. Environmentally safer wood preservation techniques—like borate-based treatments or thermal modification—show up more frequently in new construction projects. These substitutes don’t pack the “staying power” in the environment, reducing long-term harm. Continuing to develop and adopt alternatives, combined with stricter handling protocols, keeps safety and sustainability on firmer ground. A hard lesson from years past: playing catch-up to environmental damage comes with a steep cost, so staying ahead matters for everyone involved.
What are the safety precautions when handling Sodium Pentachlorophenoxide?
Understanding the Risks
Sodium pentachlorophenoxide is not an everyday chemical. It lands squarely in the category of compounds demanding respect—a lesson anyone working in a lab or maintenance setting figures out fast. Breathing in dust or vapors can irritate the respiratory tract and cause more serious health problems down the line. Even a small splash on your skin can prompt an intense reaction. With enough exposure, people can get dizzy, develop headaches, or experience persistent nausea. If you’ve spent any time working with hazardous chemicals, you know cutting corners here invites real trouble.
Personal Protective Equipment
No glove, no touch—that’s the rule. Nitrile gloves work best since they ward off the substance more reliably than latex. Splash goggles stay on your face, not perched on your head. People handling powders or mixing the stuff should wear lab coats or coveralls. Closed-toe shoes win out over sandals, and a face shield adds a layer of safety for bulk transfers or cleaning spills. Respirators matter, especially if ventilation can’t keep up; those dealing with powders know how quickly the air can turn problematic.
Storage and Handling Habits
Storage keeps risk at bay. Anyone who’s worked with reactive chemicals knows that a sealed, labeled container, stashed in a cool and dry cabinet, prevents more accidents than warning signs ever will. Sodium pentachlorophenoxide reacts badly with acids and strong oxidizers. Stashing it away from these is just common sense. Moisture and sunlight speed up its breakdown, so light-blocking containers in a dry spot make sense. Clear labels, easy-to-read hazard signs, and a tidy workspace stop mistakes from happening.
Spill Response and Disposal
Every crew or solo operator needs a spill kit built for chemicals, not just the generic type. Absorbent pads, neutralizing agents, disposable gloves, and waste bags should sit within arm’s reach. If a spill hits the floor, don’t grab the mop—use the appropriate absorbent and keep the mess contained. Ventilate the area, rope it off, and alert anyone nearby. Workers I’ve seen improvise here usually regret it. Waste disposal means following hazardous waste guidelines, not just tossing leftovers or wash water down the sink or in the trash. Incineration at approved facilities reduces risk to sanitation workers and the environment. Most shops work with certified contractors to haul it away safely.
Training and Emergency Preparedness
People can’t follow rules they don’t know. Regular training makes safety a habit instead of a hassle. Refresher briefings remind workers to replace cracked gloves, flush their eyes at the emergency eyewash, or shut down a spill before it spreads. Printed safety data sheets keep critical information close at hand—it’s surprising how often these go missing or out of date. Quick access to emergency showers and eyewash stations can mark the difference between a close call and a trip to the hospital.
Steps for a Safer Workplace
Clear communication and good workflow planning help everyone. Each person plays a role—managers by enforcing safety rules, technicians by checking protective gear, health officers by running drills, and the whole crew by staying alert. In my years on the floor, people respected and protected each other because no one wanted to see a coworker get hurt. Demanding safe practices does add time, but it saves lives, money, and plenty of headaches. Organizations prioritizing chemical safety end up with safer, more reliable teams.
| Names | |
| Preferred IUPAC name | sodium 2,3,4,5,6-pentachlorophenoxide |
| Other names |
Pentachlorophenol sodium salt Sodium pentachlorophenate |
| Pronunciation | /ˌsoʊdiəmˌpɛntəˌklaɪrəˈfɛnɒksaɪd/ |
| Identifiers | |
| CAS Number | 131-52-2 |
| 3D model (JSmol) | `3Dmol.js?&call=setMol('NaC6Cl5O')` |
| Beilstein Reference | 1073088 |
| ChEBI | CHEBI:91122 |
| ChEMBL | CHEMBL3786757 |
| ChemSpider | 21569939 |
| DrugBank | DB11136 |
| ECHA InfoCard | 05fa400c-e1e2-4ad2-baad-9db8b931c3ea |
| EC Number | 218-521-6 |
| Gmelin Reference | 1310787 |
| KEGG | C19239 |
| MeSH | D013002 |
| PubChem CID | 161112 |
| RTECS number | SN1225000 |
| UNII | 2Q6R7B81D5 |
| UN number | UN3155 |
| Properties | |
| Chemical formula | C6Cl5ONa |
| Molar mass | 246.320 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.65 g/cm³ |
| Solubility in water | soluble |
| log P | -0.15 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 7.5 |
| Basicity (pKb) | 7.47 |
| Magnetic susceptibility (χ) | -28.5e-6 cm³/mol |
| Refractive index (nD) | 1.588 |
| Viscosity | Viscous liquid |
| Dipole moment | 3.5 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 100.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -217.9 kJ/mol |
| Pharmacology | |
| ATC code | D08AJ02 |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled. Causes severe skin burns and eye damage. Suspected of causing cancer. Very toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P264, P270, P273, P280, P301+P312, P330, P391, P501 |
| NFPA 704 (fire diamond) | Health: 3, Flammability: 0, Instability: 1, Special: – |
| Flash point | Above 110°C |
| Autoignition temperature | > 520°C |
| Lethal dose or concentration | LD50 oral rat 1750 mg/kg |
| LD50 (median dose) | LD50 (median dose): 190 mg/kg (oral, rat) |
| NIOSH | SZ9800000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Sodium Pentachlorophenoxide: 0.5 mg/m³ (as pentachlorophenol), 8-hour TWA (OSHA) |
| REL (Recommended) | Not recommended. |
| IDLH (Immediate danger) | IDLH: 7.5 mg/m³ |
| Related compounds | |
| Related compounds |
Pentachlorophenol Sodium phenoxide Tetrachlorophenol Trichlorophenol Pentafluorophenol |

