Sodium Nitrite: A Close Look at Practical Value, Risks, and Future Opportunities
Historical Development
Folks working with chemicals know sodium nitrite’s story goes way back. In the 19th century, saltpeter’s cousin started popping up in industrial cookbooks. Chemists noticed what happens when heat or strong acids hit sodium nitrate—nitrite appears, showing off pink curing color in meats and keeping bacteria in check. Food preservation used to be complicated, and adding nitrite to a curing brine was a leap forward. By the 1920s, food and chemical outfits used it for dye synthesis, laboratory work, and medical antidotes, as nitrite could counter cyanide poisoning. Industries built infrastructure around mass production because sodium nitrite opened doors to new chemical processes. Over time, this compound left its mark both behind the scenes and at kitchen tables.
Product Overview
Sodium nitrite stands out as a white, sometimes faintly yellow powder. In bag or drum, it looks plain but packs a punch. Among its many hats, sodium nitrite acts as a corrosion inhibitor, food preservative, textiles’ partner, and a staple in chemical syntheses. It pops up in sausage factories, cooling systems, and laboratories where engineers and chemists use knowledge and caution in equal measure. Shippers, grocers, and plant managers keep an eye on bulk purity for predictable results. Because of its strength and possibility for harm, regulations closely follow its route from factory to shelf.
Physical & Chemical Properties
The white crystalline powder tastes salty and melts around 271°C, showing it can take the heat in many industrial settings. It dissolves easily in water, a key feature when companies make solutions for metal treatments or food curing mixes. When exposed to air, sodium nitrite draws moisture—a sign of its hygroscopic nature. Mix it with strong acids, and it churns out toxic nitrogen oxides, warning anyone working with it to be deliberate and cautious. The chemical formula NaNO2 reflects a balance of sodium, nitrogen, and oxygen, packaged with enough reactivity to keep microbes in check but also catalyze chemical reactions in large-scale manufacturing.
Technical Specifications & Labeling
Chemists and industry buyers check sodium nitrite specs closely. Purity typically falls above 99%, as lower grades can spoil precision in processes or put food batches at risk. Grain size can influence solubility and mixing, while heavy metal traces need to be minimal for food and pharma grades—Lead, arsenic, and mercury get flagged fast. Containers carry sharp hazard pictograms, chemical abstract numbers, and storage advice. Folks shipping this compound face strict labeling and paperwork rules. International agencies like the UN and local food safety authorities treat it as hazardous, requiring hazard statements and safe handling tips on every delivery. Buyers in food or pharmaceutical lines check compatibility with food contact laws and national food codes.
Preparation Method
Large plants churn out sodium nitrite using a straightforward but heat-intensive method. Sodium nitrate, another staple, takes a spin in rotary kilns—heated to over 400°C in the presence of a reducing agent, like coal or small iron filings. This knocks oxygen out, reducing nitrate to nitrite. Some companies use gaseous methods, letting ammonia or natural gas work the reduction. The resulting molten salt gets cooled and ground to a white powder or granules, then washed to remove byproducts like unconverted nitrate. Operators keep ventilation running, since nitrogen oxides show up as a nasty byproduct. Purification steps pull out impurities, with filtration and careful crystallization delivering the trademark white nitrite to the market.
Chemical Reactions & Modifications
Sodium nitrite brings plenty of reactivity to the table. Dropping an acid in the mix liberates nitrous acid, which can turn into nitrogen oxides—a serious workplace hazard. In organic synthesis, sodium nitrite turns amines into useful intermediates, kicking off diazotization reactions that make dyes, specialty chemicals, and medicines. When paired with other nitrites or oxidizers, it can join ozone formation chains or nitrate production. Engineers looking to strengthen rust protection blend it with sodium benzoate. Newer research labs have found ways to anchor nitrite onto polymer frameworks, giving coatings with slow-release corrosion control. Handling the raw stuff, people avoid mixing with ammonium salts or strong reducing agents as it can set off dangerous explosions or toxic fumes.
Synonyms & Product Names
Textbooks and shipping records list sodium nitrite under names like Nitrous acid, sodium salt, and Chile Saltpeter’s cousin. Food industry circles know it as Food Additive E250—chefs and microbiologists run into this number when checking sausage and ham labels. Workers stocking chemical stores see brands like “Nitrochem Sodium Nitrite,” “ACS Sodium Nitrite,” or even “SpeedCure” in auto coolant supply catalogs. Misspelling as “sodium nitrate” happens often, but folks who know their chemistry quickly point out the big difference. In global trade and online shops, the right term means the difference between safe delivery and serious incidents.
Safety & Operational Standards
People who handle sodium nitrite wear gloves and goggles for good reason. Inhalation or skin contact causes headaches, irritation, or worse—blue lips from methemoglobinemia if it gets in the blood. Regulatory agencies demand well-labeled storage, locked cabinets, and restricted access, especially since small mistakes mean major accidents. Ventilation, spill kits, and emergency showers aren’t just extra steps—operators have seen workers sickened by vapor or spattered skin from poor handling. Wastewater must be treated to keep nitrite out of rivers. Transporters need licenses for toxic substances, and even food-grade nitrite must travel with paperwork and warning placards. Safety data sheets require regular updates, not just dusty binders in back rooms.
Application Area
Meat and sausage makers rely on sodium nitrite for more than just color—it stops deadly Clostridium from growing. Water treatment plants drop nitrite in cooling towers to corral corrosion, while textile firms use it in dye vats for colorfast fabrics. Pharma chemists use it to produce drugs, with careful controls on batch purity. Even pyrotechnic firms call on sodium nitrite for specific combustion effects. Soldiers carry antidote kits that work with nitrite in cyanide emergencies. Laboratories keep jars for test work, and scrap yards judge it essential for some metal processing washes. It crosses into so many industries, balancing safety, chemistry, and commercial need.
Research & Development
Labs worldwide are digging deeper into the chemistry of sodium nitrite. Food scientists in Europe and Asia run studies on alternatives, asking if plant-based extracts can fill the preservative role without health risks. Chemical engineers tinker with catalysts for cleaner nitrite manufacturing that emits less nitrogen oxide. Polymer chemists coat industrial pipes with nitrite-resin blends designed to slow rust for decades. Teams at national labs analyze interaction with new bacteria, watching for resistance or new pathogen suppression. Some researchers explore nitrite’s medical upside—examining its effects on blood pressure and tissue protection during surgery. Companies investing in research stress accuracy and reproducibility, with tighter quality controls and open peer review pushing discovery forward.
Toxicity Research
Toxicologists spend a lot of time on sodium nitrite studies because the compound straddles a line between helpful and harmful. Even low doses can flip hemoglobin into methemoglobin, starving tissue of oxygen. Epidemiologists link processed meat nitrite to higher cancer risk, as gut reactions sometimes form nitrosamines—strong carcinogens—in certain cases. Food authorities limit nitrite in bacon, ham, and deli products, keeping parts per million levels as low as possible. Medical experts have seen kids turn blue from accidental ingestion, and industrial accidents sent workers to hospital when airborne nitrite rose. These studies push industry and regulators to keep tighter controls, train workers better, and develop rapid detection tools to spot accidental contamination fast.
Future Prospects
Looking forward, sodium nitrite’s role will change—but won’t vanish. Next-generation food preservatives try to cut out cancer risks but need the safety margin nitrite brings. Cleaner, lower-emission production options will reshape factory tech. Lab-on-a-chip devices will help inspect finished food and factory streams for tiny nitrite traces in seconds, making food safety more proactive. Pharmaceutical research points to new uses for nitrite in heart and sepsis treatments. Regulations will keep tightening, especially where consumer health poses concerns. Alternative corrosion inhibitors may challenge its dominance, but old infrastructure and tested formulations mean sodium nitrite will stick around for years, just in safer, more tightly watched ways.
What is sodium nitrite used for?
Essential Role in Food Preservation
Sodium nitrite has become a familiar name on food labels, especially in cured meats like bacon, ham, and hot dogs. Many might not realize how this chemical shapes the taste and appearance of their favorite deli items. For generations, butchers have relied on sodium nitrite to help keep meat safe and fresh. It slows the growth of bacteria, including Clostridium botulinum, which can cause botulism. Not only does it help keep flavors consistent, but it also gives ham and salami that rosy pink color people expect. Without sodium nitrite, grocery shelves would look a lot different—less colorful, more prone to spoilage, with fewer options for cold cuts that hold up over time.
Medical Uses and Complexity
Beyond the butcher’s block, the medical field finds uses for sodium nitrite. Hospitals stock it as an antidote for cyanide poisoning. In emergencies, fast-acting chemicals matter, and sodium nitrite reacts with hemoglobin in the blood, making it harder for cyanide to choke off oxygen. Poison isn’t common, but knowing doctors have a tool handy gives some peace of mind. That being said, sodium nitrite isn’t a casual cure-all—it requires careful handling due to the risk of side effects like low blood pressure or even methemoglobinemia, which interferes with the blood’s ability to carry oxygen.
Industrial Applications and Environmental Risks
Factories also tap into sodium nitrite’s power. You’ll find it often in coolant systems to protect metal pipes from rust. It pops up in chemical manufacturing processes, too. Paper makers, dye plants, and even pharmaceutical companies all have a stake in what sodium nitrite can accomplish. Chemical plants value it for its role in making other compounds. Removing it from industrial work would bring production lines to a crawl.
Still, large-scale use doesn’t come free of challenges. Accidental spills or poor storage can pollute groundwater or harm aquatic life. Everyday workers might handle it safely, but oversight is critical. Some companies have faced hefty penalties after sodium nitrite leaks led to environmental damage. In my experience covering environmental stories, local communities always pay close attention when chemicals are nearby, and for good reason.
Health Concerns and the Push for Safer Foods
Sodium nitrite’s presence in food has sparked many debates. Studies link high intake to an increased risk of colorectal cancer, especially after nitrites form nitrosamines under high heat or in the acidic conditions in the stomach. International agencies, such as the World Health Organization, classify certain processed meats as possible carcinogens—not because sodium nitrite alone causes cancer, but because the risks rise with heavy consumption and improper cooking.
Shoppers have started to demand more transparency. Some food companies switched to “natural” sources of nitrates, usually from celery or beet juice, which still convert to nitrites during curing. The label might read differently, but the chemistry tells a similar story. The safest bet often comes down to moderation, informed choices, and listening to researchers who sort fact from fear.
Looking Ahead: Safer Handling and Transparency
People deserve to know what goes into their food and local water. Strong regulations and responsible industry practices remain key. Regular monitoring at food plants, clear labeling, and worker training should be priority. Simple steps—like storing sodium nitrite away from acidic chemicals and using the right personal protection—can make a difference.
Everyone benefits when science leads the conversation rather than rumor or panic. Balancing safety, flavor, and economic reality is a tough challenge, but good information brings smarter decisions. That's what counts most when stories about food safety hit the headlines.
Is sodium nitrite safe for human consumption?
What Exactly Is Sodium Nitrite?
Sodium nitrite often shows up on ingredient labels for bacon, hot dogs, ham, and deli meats. It keeps these foods looking pink and slows the growth of bacteria that can cause botulism—a pretty dangerous situation in foods stored for a long time. It’s easy to see why food makers choose it. By slowing spoilage, sodium nitrite means meats last on the shelf and don’t turn brown or gray before reaching your sandwich plate.
How Sodium Nitrite Works
This compound helps meat stay fresh and kills some harmful germs before they grow strong enough to make people sick. But what’s meant to keep food safer also has a risky side. Once exposed to heat—think about frying bacon or roasting a ham—sodium nitrite can react with amines in protein to form nitrosamines. That’s where trouble starts. Studies stretching back for decades show nitrosamines can cause cancer in animals, according to the National Cancer Institute. Human studies have spotted links between high processed meat intake and some cancers, especially colorectal cancer.
Real-Life Risks and Benefits
Food scientists say eating a couple of nitrite-containing hot dogs on the weekend won’t immediately send someone to the hospital. Bodies process small amounts of nitrites pretty efficiently; leafy vegetables like spinach and lettuce actually provide more nitrites than a typical serving of cured meat. The difference often lies in the details: vegetables carry antioxidants like vitamin C, which block the chemical flip into nitrosamines that’s more likely in cooked meats.
In my own family, I noticed that traditions revolve around foods like sausages and jerky. Nobody checks for sodium nitrite content on the label. For years, eating these foods felt normal. News stories about cancer risks shifted my outlook. Cutting back got easier after exploring alternatives—chicken salad, grilled fish, or roasted chickpeas—without the lingering worry about cancer.
Supporting Facts and Guideline
Regulators keep a pretty close eye on sodium nitrite. The U.S. Food and Drug Administration and the European Food Safety Authority both put tight caps on how much can go into food. Strict monitoring aims to keep average intakes below the level considered dangerous. The World Health Organization classified processed meats as carcinogenic because of the links with cancer, but these warnings often refer to long-term, high consumption rather than the odd pastrami on rye.
Cured meats taste great and come with a dose of nostalgia—think of backyard BBQs and holiday tables—but health doesn’t stay untouched by memories. Public Health England, the American Cancer Society, and other groups urge people to limit their intake, pushing for less bacon at breakfast and more fresh produce instead. Some brands now use celery powder, which naturally contains nitrite, rather than synthetic sodium nitrite. But the chemical reaction is the same, and the risks don’t disappear just because the label says “natural.”
Where We Go from Here
The issue doesn’t have a single, simple answer. Someone who eats processed meats every day faces higher risks than someone who picks them up once a month. Focusing meals on fresh ingredients, saving bacon or pepperoni for special occasions, and reading ingredient labels empowers anybody who wants to eat with less worry.
Sodium nitrite extends shelf life, but the healthiest shelf probably stands in a home full of fresh vegetables, beans, eggs, and lean meats—foods rarely found on a cured meat label. Science hasn’t outlawed sodium nitrite, but smart choices and some healthy skepticism go a long way. Change doesn’t have to mean giving up flavor or tradition; it’s about tipping the balance toward food that supports a long, healthy life.
What are the potential health risks of sodium nitrite?
A Familiar Additive with Unfamiliar Risks
Sodium nitrite pops up in a lot of foods—think cured meats, bacon, ham, hot dogs, that sort of thing. It shows up because it keeps food looking pink and fresh, fights off life-threatening bacteria, and boosts shelf life. For anyone who’s reached for a quick lunch or enjoyed hot dogs at a ballgame, sodium nitrite made those moments possible. What’s less easy to spot is what happens long-term when you get regular doses of it from your meals.
How Sodium Nitrite Reacts in the Body
This compound doesn’t linger as sodium nitrite in your stomach. During digestion, acidic conditions help turn nitrites into chemicals called nitrosamines. Science has known about nitrosamines for decades. Many studies—Harvard School of Public Health and the World Health Organization among them—connect nitrosamines to increased risks of certain cancers. The American Cancer Society notes a link between processed meats containing sodium nitrite and colorectal cancer. That story runs deeper for people who find nitrite-rich foods on their plate every day.
Beyond Cancer: Other Health Concerns
Cancer isn’t the only concern. Sodium nitrite can mess with how the blood carries oxygen. High doses can trigger a rare but real condition called methemoglobinemia, where blood struggles to carry enough oxygen to cells. A handful of cases pop up every year, most often from accidental overdose or mislabeling. Pregnant women, babies, and folks with certain genetic conditions stand at greater risk. Public health agencies keep an eye on food safety regulations for this reason.
Why It’s Still Used
Pulling sodium nitrite entirely from the food system isn’t so straightforward. Those glowing pinks in deli cases and pepperoni pizza slices are more than just visual—they indicate the food’s been protected from bacteria like Clostridium botulinum, which can cause deadly food poisoning. That said, some traditional methods like dry curing and refrigeration pull their own weight in keeping food safe, though not all manufacturers use them.
Finding a Better Balance
Plenty of people, including me, have cut down on cured meats or picked up natural alternatives. Sticking to fresh meat and fish, going heavy on vegetables, and varying protein sources lowers your exposure. Some companies have switched to “naturally cured” meats using celery powder, which still introduces nitrites, just from a different source. Choosing these with your eyes open matters more than chasing a marketing claim.
Possible Steps Forward
Regulation keeps changing. The U.S. and EU both set limits for how much sodium nitrite can end up in food. Scientists are still pushing for more clarity on risk, especially for kids. The food industry can do more, too. Clearer labeling gives power back to shoppers. New tech may one day let us ditch chemical additives while still keeping our food safe.
Taking Charge of What We Eat
A lot of the risk boils down to how often sodium nitrite lands on your plate. Tossing leftover ham in sandwiches, grilling hot dogs by the dozen at every cookout—it adds up. Fresh food, cooked at home, sidesteps most of the trouble. Anyone who cares about health can make big strides just by paying attention to ingredient labels and limiting processed meats. At the end of the day, information empowers us all to eat better, and push for safer food on store shelves.
How should sodium nitrite be stored?
Understanding the Risks at Home and in Business
A lot of folks don’t give much thought to sodium nitrite unless they work with food preservatives, chemicals, or science classrooms. Yet, it grabbed news headlines because of poisonings linked to careless storage or misuse. I’ve grown up around places that kept odd things like fertilizer and pool chemicals in the garage, so I know how easy it is to shrug off warnings and store chemicals within easy reach. That can create a recipe for trouble—especially when you’re dealing with something that endangers health so quickly.
Sodium nitrite looks harmless: just a white or light yellow powder. Toss it in with other shelf-stable powders and one wrong move could turn routine chores into a disaster. This isn’t a case for stuffing containers on an open shelf or leaving big sacks in the laundry room. The stuff is toxic if swallowed, can trigger headaches and nausea at low doses, and may harm the kidneys or blood system at higher exposures. Accidental spills have serious downsides—especially for curious kids or pets.
Common Sense, Not Just Labels
Clear labeling helps, but the way sodium nitrite gets stored counts for more in the long run. Chemicals with known risks need their own, locked cabinets—preferably somewhere dry, cool, and well-ventilated. Taking safety seriously means checking containers for cracks or damage before putting anything away, using original packaging whenever possible, and sealing bags tightly after scooping powder out. No one wants an easy route for moisture to sneak in and clump up the contents, raising the odds of mishandling.
Leaky packaging doesn’t just change how well sodium nitrite works; it draws pests and breeds confusion later on. Once chemicals get mixed up, mistakes follow, and mistaken identity has cost more than a few folks their health. In my time working in school labs, I saw labels fade off jars and leftover powders dumped into “temporary” tubs. These careless moves nearly resulted in the wrong ingredients being handed to students—an accident waiting to happen.
Safe Distances and Smarter Storage
Some rules never get old: keep strong chemicals away from food, drink, and anything you’d toss in a pan. Putting sodium nitrite in pantries or cupboards with sugar or flour is playing with fire. Store it with other hazardous chemicals, but also spaced apart from acids and organics because mixing can trigger fires or poisonous gases. Separating sodium nitrite from cleaning products also lowers the risk of unexpected reactions that catch people off guard.
Never underestimate the benefit of education, especially if you share living or working spaces. Grownups can’t assume others know the dangers just from a hazard symbol. I’ve had family members mix cleaning solutions without reading instructions, thinking “it’s just another powder.” Sharing even a short rundown of risks pays off, ensuring everyone understands why a cabinet stays locked, why gloves are essential, and why spills need a specific cleanup—not just water and paper towels.
Small Steps with Big Impact
It doesn’t take high-tech locks or complex protocols. Buy a lockbox. Place it somewhere out of sight but not next to anything edible or flammable. Record inventories so nothing gets lost or forgotten in the back. Post emergency numbers and basic chemical safety instructions on the door to the storage area. If you buy sodium nitrite for any reason, invest a few minutes to think about who else might come across it in your space.
The bottom line remains clear. Treat sodium nitrite—and any chemical with a track record for harm—like it matters. No warning or regulation does as much good as putting safety into everyday habits. People—especially young ones—learn by watching what others do, not what’s printed in a manual. That simple change in mindset makes all the difference.
What is the chemical formula of sodium nitrite?
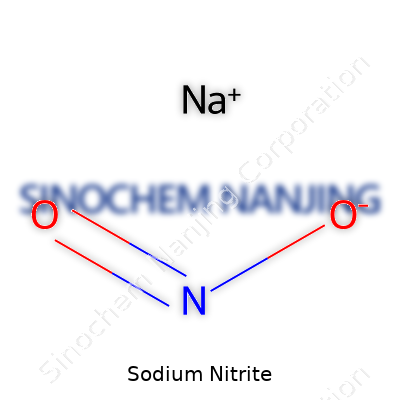
Understanding Sodium Nitrite: NaNO2
Sodium nitrite carries the simple formula NaNO2. It looks almost like table salt and dissolves easily in water. The mix of sodium (Na), nitrogen (N), and oxygen (O) creates a compound that does a lot more than just sit on lab shelves. Many people including those with backgrounds in food science, chemistry, and public health have crossed paths with it, sometimes without even realizing it.
Role in Food Safety and Science
Most people spot sodium nitrite on the ingredient label of cured meats, like bacon or hot dogs. It prevents the growth of dangerous bacteria such as Clostridium botulinum, which can cause botulism. If you’ve ever wondered why store-bought ham keeps its pink color, sodium nitrite lends a hand there as well. The nitrite reacts with proteins in the meat, leading to that distinct appearance and taste.
For those passionate about cooking or nutrition, it’s important to know what goes into food. The U.S. Food and Drug Administration recognizes sodium nitrite as safe within specific limits. Excess use, though, causes health problems. Nitrites can form nitrosamines in the body, and research points out that these chemicals link to cancer risks. A 2015 report from the World Health Organization classified processed meat as a carcinogen, in part because of these compounds.
Sodium Nitrite Beyond the Kitchen
You can find sodium nitrite in industrial settings, too. Water treatment plants use it to combat corrosion in pipes, where the formation of rust puts drinking supply quality at risk. Medical professionals sometimes use it for treating cyanide poisoning. In those critical moments, sodium nitrite turns into a life-saving agent. Outside these benefits, it plays a role in dye production and analytical chemistry labs.
Balancing Benefits and Drawbacks
Striking a balance becomes the big challenge. Reducing risk takes effort at every stage—from factories mixing the powder to kitchen tables where it lands on tonight’s dinner. Food producers can switch to lower concentrations or look at natural alternatives, like celery juice, which also contains nitrites but comes paired with protective compounds. Families thinking about health swap out some processed meats for fresh options. These little changes matter.
Regulators and the scientific community keep a close watch on new research around nitrites. They update safety standards and share findings with the public, aiming for practical solutions rather than just sounding alarms. Sharing this information in plain language helps everyone make smarter choices at the grocery store and beyond.
Looking Forward
Sodium nitrite tells a story of trade-offs. It keeps food safer, supports industries, and even provides emergency medical benefits. Still, too much or careless handling leads to long-term health troubles. Education builds trust, and continued research shapes smart regulations. Every time we read a label or talk with a doctor about our diet, we bring that science into our own kitchens.
| Names | |
| Preferred IUPAC name | Sodium nitrite |
| Other names |
Nitrous acid sodium salt Sodium nitrite E250 Sodium salt of nitrous acid Nitrite of soda Sodii nitris |
| Pronunciation | /ˈsəʊdiəm ˈnaɪtraɪt/ |
| Identifiers | |
| CAS Number | 7632-00-0 |
| Beilstein Reference | Beilstein Reference: "3560256 |
| ChEBI | CHEBI:32145 |
| ChEMBL | CHEMBL19224 |
| ChemSpider | 6329 |
| DrugBank | DB00755 |
| ECHA InfoCard | 100.007.763 |
| EC Number | 011-037-00-0 |
| Gmelin Reference | 822 |
| KEGG | C01574 |
| MeSH | D009670 |
| PubChem CID | 23668193 |
| RTECS number | RA1225000 |
| UNII | 9Q994720L3 |
| UN number | UN1500 |
| Properties | |
| Chemical formula | NaNO2 |
| Molar mass | 68.995 g/mol |
| Appearance | white to slightly yellowish crystalline powder |
| Odor | Odorless |
| Density | 2.168 g/cm³ |
| Solubility in water | 820 g/L (20 °C) |
| log P | -3.7 |
| Vapor pressure | 1 mmHg (at 20°C) |
| Acidity (pKa) | pKa ≈ 3.0 |
| Basicity (pKb) | 10.07 |
| Magnetic susceptibility (χ) | +45.0e-6 |
| Refractive index (nD) | 1.341 |
| Dipole moment | 2.39 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 82.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -360.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -360.9 kJ/mol |
| Pharmacology | |
| ATC code | V03AB08 |
| Hazards | |
| Main hazards | Oxidizer, toxic if swallowed, causes methemoglobinemia, harmful if inhaled, may cause fire or explosion. |
| GHS labelling | GHS02, GHS03, GHS06 |
| Pictograms | GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H272, H301, H319, H335 |
| Precautionary statements | P210, P220, P221, P264, P270, P273, P301+P312, P304+P340, P305+P351+P338, P308+P313, P330, P370+P378, P403+P233, P501 |
| NFPA 704 (fire diamond) | 3-0-1-OX |
| Autoignition temperature | 550°C |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 Rat oral 180 mg/kg |
| LD50 (median dose) | LD50 (median dose): 180 mg/kg (oral, rat) |
| NIOSH | NN3575000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Sodium Nitrite: "Not established |
| REL (Recommended) | 200 ppm |
| IDLH (Immediate danger) | 100 mg/m³ |
| Related compounds | |
| Related compounds |
Sodium nitrate Potassium nitrite Potassium nitrate Ammonium nitrite Silver nitrite Calcium nitrite |

