Sodium Metaperiodate: More Than a Chemical Reagent
Historical Development
Long before sodium metaperiodate landed on the benches of today’s analytical labs or chemical plants, the story began with the search for strong oxidizers that could make other reactions tick. Years back, people looked for ways to break carbon bonds gently, and periodates, including sodium metaperiodate, offered that power. This compound didn’t simply drop out of the sky; its rise tracks back to the beginning of modern organic chemistry. Chemists noticed its knack for splitting diols, opening the door for advances in carbohydrate analysis, structural biology, and even fine-tuning synthetic pathways. It stands as one of those quietly transformative substances that roll behind milestone discoveries—something you only miss after using it for a while.
Product Overview
Sodium metaperiodate is a white, odorless solid, easy to recognize if you’ve run through a shelf of oxidizing agents. Often found as a crystalline powder, its sharp, clean look doesn’t match the punch it delivers in a flask. This material, chemically marked as NaIO4, doesn’t hog headlines, yet it sits at the crossroads of analytical chemistry, molecular biology, and a good chunk of fine syntheses. In the industry, the story rarely centers on showpiece molecules like metaperiodate, but its presence in the background ensures that more recognizable names can shine.
Physical & Chemical Properties
This isn’t a tough compound to spot or store. Sodium metaperiodate dissolves quickly in water, which gives it a real edge in lab settings where time counts. You won’t need to worry about strong odors, fumes, or heavy residues. It reacts as a strong oxidizer, switching up oxygen states, breaking chemical bonds in sugars and other targets. If you drop it into a reaction with a glycol, for instance, it will cleave those carbon bonds cleanly. It resists breaking down under normal temperatures, but bringing it into contact with the wrong materials—think strong reducing agents—can start trouble quickly.
Technical Specifications & Labeling
The real detail here lies in purity rather than just weight or grade. Chemists look for sodium metaperiodate with minimal impurities, since small contaminants can steer a reaction off-course. Most suppliers label product by percent purity, moisture content, and batch analysis data, since these figures matter more than fancy packaging. The win for end users comes with clear, honest labeling—knowing what the jar truly contains, no surprises when reactions run.
Preparation Method
Chemists have refined the way sodium metaperiodate is made. The classic route involves reacting sodium iodate with chlorine or hypochlorite in water under carefully controlled conditions. This process avoids wild swings in temperature or uncontrolled releases, yet it does demand attention to safe handling of reagents. As with most oxidizers, anyone making or storing it sticks to rigid safety routines. Controlling water content and temperature makes the difference—sloppy prep can mean low-purity product or unexpected hazard.
Chemical Reactions & Modifications
Where sodium metaperiodate thrives is selective oxidation. Scientists often point to its skill in breaking vicinal diols—splitting those pairs into aldehydes or ketones. This precise cut opens a floodgate of possibilities in carbohydrate work and in mapping complex biomolecules. Use this compound, and you can dial in the oxidation without sending the entire molecule into chaos. It is tricky with reducing agents because the reactions can run out of control if mixed carelessly. In biochemical fields, it tags sugar groups on proteins for conjugation, enabling targeted research and drug development. Over decades, chemists have tweaked the recipe—sometimes you’ll see sodium metaperiodate paired with other gentle reagents to get special selectivity or improved yields, especially in protein chemistry and advanced materials research.
Synonyms & Product Names
You won’t find sodium metaperiodate under too many street names, but “sodium salt of periodic acid,” “sodium orthoperiodate” (though this is technically distinct), and “NaIO4” pop up. Lab veterans know to watch for confusion with sodium periodate in general, because the “meta” and “ortho” versions wield different chemical strengths and solubility.
Safety & Operational Standards
Experience shapes your respect for strong oxidizers. Sodium metaperiodate doesn’t blast fumes or leave scars on your bench, but direct contact can burn. Inhalation or accidental spills may irritate the skin, eyes, or breathing passages, especially for those already sensitive to dust. Storage demands cool, dry shelves away from combustible or reducing agents, since mixing can unleash violent changes. Protective gloves and careful dust control are a must—nobody enjoys a trip to the eyewash or shower because of careless handling. Many labs rely on local guidelines, but anyone familiar with international safety literature or workplace best practices echoes the same message: treat it with the discipline given to any potent oxidizing salt, and things stay smooth.
Application Area
The reach of sodium metaperiodate keeps expanding. In biochemistry, it splits sugars or unlocks targeted modifications in glycoproteins, enabling drug companies and researchers to shape complex pharmaceuticals in ways that simply weren’t possible before. Histology uses its oxidation power to prep tissues for advanced staining or analysis, pushing the limits of what microscopes can reveal. Analytical labs look to it for fingerprinting carbohydrates and mapping natural products. Beyond science, some niches tap it for personal care products or specialty manufacturing, though that sits at the edge of bulk use.
Research & Development
The push for greener chemistry leads researchers to rethink how sodium metaperiodate gets used and made. Environmental concerns, with a sharp eye on waste and byproducts, drive tweaks in synthesis and cleanup steps. Scientists develop more targeted reaction setups, sometimes immobilizing this oxidizer on solid supports to recover or recycle it after each round of use. Drug makers and diagnostics experts chase more specific modifications of proteins and sugars, since precise control lets them build better therapeutics or track biological changes minute by minute. Current R&D spins around making the use of sodium metaperiodate safer, less wasteful, and tuned for new synthetic challenges.
Toxicity Research
No strong oxidizer enters constant use without scrutiny. Over years, researchers have studied sodium metaperiodate’s toxicity profile in cells and animals, checking for acute or chronic effects. Small, controlled exposures usually result in local irritation—nothing catastrophic—but careless spills or inhalation can mean real trouble for skin, eyes, or lungs. Some investigations suggest that long-term workplace exposure needs attention, since even mild oxidative stress over time chips away at cell health. Environmental studies pick up concerns about aquatic life, especially where spent reagents wash down drains or hit freshwater systems. This nudges chemical users to lock down waste streams and handle residues responsibly.
Future Prospects
Every year, labs pitch new ways to bring sodium metaperiodate into synthetic and analytical chemistry. As the hunt for more selective, greener, and cost-effective methods continues, this reagent doesn’t see retirement anytime soon. Trends point toward more responsible sourcing, sharper applications in medical research, and lower-impact processes that make better use of each gram. For generations of chemists, it remains one of those quietly essential substances—a helper standing just outside the spotlight but never leaving the stage.
What is sodium metaperiodate used for?
Behind the Name
Sodium metaperiodate sounds like something out of a high school chemistry lab, tucked away in a dusty jar with a faded label. But inside that bottle, you’ll find a reagent that quietly powers innovations in medicine, biotechnology, paper manufacturing, and environmental research. What catches my attention most is how a single compound ends up affecting so many corners of everyday life. Having worked in a clinical diagnostics lab, I've seen firsthand how small chemicals play big roles.
From Lab to Hospital: Medical Applications
Labs use sodium metaperiodate for modifying or analyzing carbohydrates and glycoproteins. Think about it: identifying different blood groups and diagnosing diseases turn on this compound’s strong oxidizing power. In immunoassays, sodium metaperiodate helps label antibodies so machines can detect diseases ranging from HIV to cancer. Without reliable reagents, doctors would face enormous challenges in providing clear answers fast. According to research in Analytical Biochemistry, periodate-based modifications increase sensitivity in many medical tests, directly impacting patient outcomes.
Champion of the Research World
Researchers value sodium metaperiodate for its ability to split certain sugar rings in carbohydrates, helping them study cell surfaces. Science journals like Carbohydrate Research often cite this reaction as a backbone for mapping out glycan structures. Scientists use these methods to explore everything from virus-host interactions to how cancers grow. This research lays the groundwork for new treatments down the line. My time working with graduate students reminded me just how often chemicals like this show up as “unsung heroes” behind breakthrough papers.
Environmental Science and Industry
Not all of sodium metaperiodate’s work happens in medicine. In paper manufacturing, this reagent breaks down cellulose fibers, giving manufacturers more control over texture and strength. These small tweaks help recycled paper keep up with quality demands, cutting down on virgin wood use. On the environmental side, scientists rely on its oxidizing strength to analyze toxic organic pollutants in soil and water. By helping identify contamination, sodium metaperiodate plays a part in local cleanup efforts. My own community lived through a groundwater contamination scare, and swift chemical testing made a world of difference.
Handling Risks and Moving Ahead
Of course, strong oxidizers bring hazards. Mishandling can damage living tissue or spark a fire. Industries that use sodium metaperiodate require strict training and strong safety protocols. Sustainable solutions matter. Teams are already researching greener alternatives and ways to minimize waste, as highlighted by the Journal of Cleaner Production. I appreciate seeing companies push for safer, cleaner production without sacrificing reliability, since long-term health and environmental stewardship matter more with each passing year.
Looking Forward
Sodium metaperiodate may not make headlines often, but it shapes results from clinics to classrooms. Reliable sources, transparent handling, and innovative minds all play a role in maximizing its benefits while limiting risks. It teaches us to pay attention—not just to big innovations, but to the quiet science supporting them. As new uses appear, involving a broader conversation between scientists, consumers, and regulators becomes essential. The path forward demands not just technical know-how, but responsibility and trust, rooted in evidence and experience.
What are the storage requirements for sodium metaperiodate?
The Tough Stuff About Handling Sodium Metaperiodate
Sodium metaperiodate comes with a reputation for being powerful—an effective oxidizer in organic chemistry labs, and a staple for researchers breaking down complex sugars or preparing DNA samples. This kind of strength carries a risk. I’ve watched new lab members quickly realize the rules around storage aren’t just a suggestion. Years back, an acquaintance thought a simple plastic container on a top shelf would do, missing how easily this compound pulls in moisture and reacts with other chemicals nearby. The mess that followed wasn’t pretty, and it could have caused real harm.
Humidity: The Hidden Enemy
Anyone who’s worked with sodium metaperiodate knows moisture gets under its skin. Leave the lid off by accident, or use a subpar container, and the white powder clumps fast and changes composition. The reaction with water isn’t dramatic at a glance, but it doesn’t take long before you've got an unstable material that’s impossible to weigh accurately for experiments. Using a desiccator—one of those sealed chambers with drying agents—has always made life easier. Keep things dry, and you keep risk down.
Not Any Old Storage Spot Will Cut It
Location matters a lot. It truly pays off to put sodium metaperiodate away from direct sunlight, strong heat, or areas where equipment throws off sparks. I once saw a careless example where an oxidizer was shoved above a sink. More humidity, more accidental splashing, and a guarantee something would go wrong. Instead, a cool, well-ventilated cupboard at bench level removes a lot of the guesswork. It’s not just about following a manual. It’s about walking into the lab the next morning without worrying if a chemical reaction ruined your weekend.
The Company It Keeps
Storing hazardous materials gets personal when you see what can go wrong. Sodium metaperiodate doesn’t play well with organic materials or acids nearby. Even small fumes can spark trouble. Keeping strong oxidizers away from anything flammable or reactive makes sense. There’s a strict “no mixing” rule in every organized lab I’ve ever worked in. Color-coded shelves and clear labels sound basic but prevent the sort of mistakes that stories get written about later.
Container Choice: Small Details Make Big Differences
Ask anyone who’s had to clean up after a chemical spill—they’d rather spend twice the time picking the right container than five minutes fixing a leak. Glass bottles with airtight seals or heavy-duty plastic containers keep air and moisture out. Big containers stay in the stockroom, with smaller amounts weighed out for the week’s work. Everything gets double-bagged, too. Not fancy, just common sense from years of seeing what sweat and inattention can cost.
Being Ready for Trouble
Knowledge matters. Most places I’ve worked in require clear signs, regular checks, and accessible material safety data sheets. People know where to find spill kits, and they always use eye protection and gloves. It only takes one incident to hammer home the idea that safety isn’t just paperwork. Having routines and explaining them—especially to new hires—keeps surprises to a minimum.
Setting the Standard in Storage Safety
Keeping sodium metaperiodate safe and stable isn’t just good lab hygiene—it’s good science. By respecting its hazards and making smart decisions about storage, researchers protect lab members, research, and the building itself. It comes down to experience, preparation, and not getting careless, even once. For anyone handling powerful chemicals, the habits you keep are more crucial than the warnings on the label.
Is sodium metaperiodate hazardous to handle?
The Real Risks in the Lab
Most lab techs have cracked open bottles labeled with long chemical names, but sodium metaperiodate tends to get attention from even the most relaxed. Its sharp, crystalline look says “be careful” before the label does. The chemical carries real weight in research, especially for oxidizing sugars and preparing advanced bioconjugates. But just because it’s common doesn’t make it harmless.
What Makes It Hazardous?
You catch a whiff of sodium metaperiodate dust, and the nose starts to tingle; it acts as an oxidizer, and that’s a big tipoff it can react violently. Bumping it into the wrong chemicals sparks accidents—think combustible organic material or strong acids. The oxygen-rich nature isn’t just good for chemistry—it kicks up the risk of fires in places usually safe from flame.
Spilling some on your skin leads to burns, sometimes worse than expected for a white powder. Breathing in the dust can irritate, and it’s not an irritant you brush off two minutes later. Swallowing even a little by mistake? That’s an ER visit, no debate. Many of the chronic risks aren’t well understood; studies haven’t cleared up the long-term effects, but the immediate hazards are enough.
The Voice of Lab Experience
Anyone who’s prepped a batch of PAS stain for histology, or done periodate oxidation of carbohydrates, knows the drill: gloves, coat, solid eye protection, and the faint hum of a ventilation hood. Rushed work, shortcuts, or mixed-up containers lead to injuries. A friend once ignored a tiny residue on her palm after cleaning up, only to run to the sink minutes later. The red streak didn’t fade for days.
Fact Check: The Data
The CDC lists sodium metaperiodate as a strong irritant and a dangerous oxidizer. The European Chemicals Agency (ECHA) rules call for signal words like “Danger” on every package. Case studies from labs report skin rashes, eye injuries, and two fires in the last years from improper storage near paper towels and ethanol. Hazard rating charts from Sigma-Aldrich and Fisher Scientific flag it at the top in reactivity.
Practical Solutions
Risks drop fast once people get serious about controls. Latex or nitrile gloves aren’t enough; thicker neoprene styles hold up better. The chemical pops open a glove after ten or fifteen minutes of exposure, especially if you’re handling it wet. Fume hoods make a difference, and sealed containers keep dust out of the air. Storage next to flammables or acid wastes invites disaster, so locked cabinets matter.
Education helps too. Training sessions boost awareness far more than unread safety sheets. Adding story-based sharing—real incidents, not just generic warnings—resonates with newcomers. Creating standard operating procedures, posting emergency rinse stations, and tracking chemical inventories keeps surprises down.
Waste handling also deserves attention. Sodium metaperiodate waste doesn’t mix well with anything else, and tossing it down the sink risks pipe corrosion or unexpected reactions. Following disposal rules protects the building, and people in it, long after the experiment runs its course.
Sodium Metaperiodate Demands Respect
It’s easy to see this as just another jar on the shelf, but too many people pay the price for routine. Proper gear, smart storage, and serious cleanup habits turn a risky job into a safe one. It’s not about avoiding the chemical—it’s about handling it like your health depends on it.
What is the chemical formula of sodium metaperiodate?
A Closer Look at Sodium Metaperiodate
Chemistry shapes a lot of what happens in daily life. Sodium metaperiodate, with the formula NaIO4, gets used in scientific laboratories all over the world. While it may seem like another long chemical name, it plays a crucial role in a lot of procedures, and its imprint shows up from research to industry. I remember my own early days in a teaching lab, where this white crystalline compound turned a dull routine into a lesson about how even a single element’s position changes the use and safety of a material.
Where It Counts: Real Science, Real Risks
Ask most chemists about sodium metaperiodate, and the words “oxidizing agent” come up right away. This chemical holds strong power for breaking bonds in certain molecules, which is why it ends up in organic synthesis. Researchers rely on NaIO4 to open up complex sugars. This helps them discover new pathways in medicine and biology. Digging into how DNA or carbohydrates work wouldn’t move forward as fast without access to the right chemicals for the job.
Some people overlook how handling a formula like NaIO4 isn’t just about technical skill. Years ago, an accident at a university taught me to respect these white powders. Sodium metaperiodate can cause burns if it lands on your skin and its dust can hurt your lungs if you breathe it in. Clear protocols and personal experience enforce the habit of putting on goggles and gloves before anyone even begins measuring out a sample. The stories floating around labs make safety sound less like a rule and more like self-preservation. Each ingredient in the formula matters, both for chemistry’s sake and for health’s sake.
Applications Beyond the Lab
Outside chemistry class, sodium metaperiodate finds use in several specialized areas. Biochemists count on its ability to cleave vicinal diols, which helps in mapping the structure of molecules. In material science, modifying proteins and sugars can spark innovation in biometric sensors and drug delivery research. Analytical chemists dig into their kits for NaIO4 in high-performance experiments. Every breakthrough or diagnosis that sodium metaperiodate enables traces back to the precision packed inside a formula as short as NaIO4.
Addressing Safety and Environmental Responsibility
With so much power, sodium metaperiodate also brings a set of responsibilities. Strong oxidizing agents like NaIO4 can’t just wash down the drain. I’ve seen research groups plan their use from project start to finish, ensuring residuals get neutralized and disposed of following strict environmental guides. A single spill or ignored bottle can undercut months of good work and cause real harm. It’s a reminder in every safety training—protect people, then protect the planet.
Moving Toward Safer Practices
If the science community wants to reduce risks further, training stays key. Lab instructors today drill students on correct storage—keeping sodium metaperiodate dry and away from organic material cuts down on fire hazard. Clear labels, sturdy packaging, and accessible information help keep mistakes rare. Innovation might eventually shrink the need for tough oxidants in some cases, but until then, education and attention make the difference.
Sharp lessons from chemistry echo beyond the beaker. Formulas like NaIO4 don’t just solve scientific puzzles—they shape how we think about responsibility and progress.
How should sodium metaperiodate spills be cleaned up?
A Tough Job for a Potent Chemical
Sodium metaperiodate looks harmless on a shelf, but just one accident will show why it gets treated with respect in chemistry labs and manufacturing sites. I've seen the nervous faces when a bottle tips or a tray cracks. This white, crystalline powder doesn't just make a mess — it brings a bundle of hazards: strong oxidizing action, skin and respiratory irritation, and the risk of serious chemical reactions. Rushing in with a mop or tossing water at the problem only ups the danger. There’s no substitute for knowing what works.
Why Training and Quick Action Matter
Every cleanup story I’ve heard from experienced technicians ends the same way: prevention takes you farther than any heroic response. Still, even the careful ones get a leaky vial once in a while. The most important first move is to get people away from the spill. Sodium metaperiodate dust floating in the air creates a real breathing risk. Anyone nearby should grab a proper respirator, gloves, goggles, and a chemical-resistant apron before touching anything.
Waste Control: Don’t Let It Spread
Spilled sodium metaperiodate never stays in one place if left alone. Without barriers, airflow or careless shuffling kicks up more particles. The smart path means closing doors to the room, shutting off any vents, and picking up glass or containers with tongs or forceps. Piling loose paper towels or using shop-vacs only makes things worse by spreading fine dust.
Picking the Right Absorbent and Neutralizer
I learned early on that water does not help with this chemical — it releases strong acids that corrode pipes and surfaces. Labs use special inert absorbents like dry sand, clay-based spill pillows, or commercial chemical absorbents rated for oxidizers. Plenty of folks have stories of coworkers dumping paper towels on the spill, but that risks fire. Once the bulk of the powder is scooped, the next move depends on your facility’s plan. Sodium bisulfite or thiosulfate solutions sometimes get used to reduce leftover material. Always check local waste treatment rules: dumping anything unreacted into the sink or regular trash brings legal trouble and harms water systems.
Safe Disposal and Reporting
Sealed, labeled containers keep collected waste safe until a hazardous waste specialist picks it up. It’s easy for staff to slip up once the obvious mess is gone, but forgetting paperwork or leaving waste in public areas puts everyone at risk. Even a small spill has to be reported, not just for rules, but so teams learn from mistakes. In my time volunteering helping students, I’ve seen cleanup logs used in weekly safety meetings to spot trends and fix shortfalls in training. Small investments in communication go a long way.
Building a Stronger Safety Culture
It’s not just about one spill or one mistake — habits around chemical handling make all the difference in keeping people safe. I’ve found that the labs and warehouses with clear instructions, regular drills, and well-labeled safety kits bounce back much quicker from accidents. Industry studies back that up: injuries drop in places where people expect spills, know the cleanup steps, and keep supplies handy. Simple actions — sealing containers, double-checking storage, keeping spill kits ready — do more than any single protocol on a poster. Sodium metaperiodate keeps its reputation as a handful, but with careful habits, spills don't have to turn into disasters.
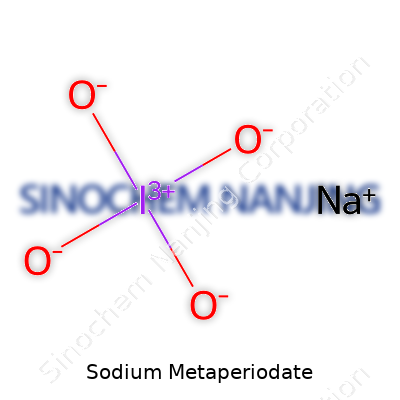
| Names | |
| Preferred IUPAC name | Sodium periodate |
| Other names |
Sodium periodate Sodium meta-periodate Disodium periodate Sodium metaperiodate(VII) |
| Pronunciation | /ˌsoʊdiəm ˌmɛtəˈpaɪ.əˌdeɪt/ |
| Identifiers | |
| CAS Number | 7790-28-5 |
| Beilstein Reference | 3592542 |
| ChEBI | CHEBI:76143 |
| ChEMBL | CHEMBL1350 |
| ChemSpider | 50455 |
| DrugBank | DB11047 |
| ECHA InfoCard | 100.028.859 |
| EC Number | 231-196-0 |
| Gmelin Reference | 118119 |
| KEGG | C06325 |
| MeSH | D017575 |
| PubChem CID | 24861414 |
| RTECS number | SD7250000 |
| UNII | 9QZT1S7M9D |
| UN number | UN1504 |
| Properties | |
| Chemical formula | NaIO4 |
| Molar mass | 213.89 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 3.865 g/cm3 |
| Solubility in water | soluble |
| log P | -4.3 |
| Vapor pressure | Negligible |
| Acidity (pKa) | ~3.6 |
| Basicity (pKb) | 11.6 |
| Magnetic susceptibility (χ) | +54.0·10^−6 cm³/mol |
| Refractive index (nD) | 2.346 |
| Viscosity | Viscous liquid |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 143.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -293.25 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V03AB38 |
| Hazards | |
| Main hazards | Oxidizer, harmful if swallowed, causes severe skin burns and eye damage, may cause respiratory irritation. |
| GHS labelling | GHS05, GHS07, GHS08 |
| Pictograms | GHS05,GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H271, H302, H314, H318 |
| Precautionary statements | P280, P264, P270, P301+P330+P331, P305+P351+P338, P310, P501 |
| NFPA 704 (fire diamond) | 2-0-2-OX |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 (oral, rat): 2,500 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 3,700 mg/kg |
| NIOSH | SDC |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Sodium Metaperiodate: Not established |
| REL (Recommended) | 2-8°C |
| Related compounds | |
| Related compounds |
Potassium metaperiodate Sodium periodate Potassium periodate |

