Sodium Iodate: More Than a Chemical, A Pillar of Progress
Historical Development
Most people walking past a chemical storeroom would never think about sodium iodate. Yet, this compound has helped power a chapter of human progress with quiet reliability. Since its introduction in the mid-19th century, sodium iodate found a niche in industrial chemistry well before the word “innovation” became a boardroom buzzword. Back then, folks in factories worked long shifts handling everything by hand, mixing this salt as part of early efforts at water purification, textile dyeing, and even stabilizing food supply chains. It’s striking to realize just how long scientists and engineers have turned to this salt for its oxidizing qualities. Sodium iodate’s journey started well before my own career in research labs, yet every generation finds new ways to put such legacy chemicals to work. Experiments in the early days often focused on getting it pure enough for reliable use in qualitative analysis, which paved the way for a chain of discoveries that stretched from medicine to manufacturing.
Product Overview
Sodium iodate looks about as unremarkable as any fine, white crystal can. Scoop some into your hands, and you’d have a powder that dissolves easily in water, ready to play a role in chemical labs and industry. It carries the formula NaIO3, marking it out as both a salt and a powerful oxidizing agent. These characteristics make it a staple reagent for analytical chemistry, yet the wider value emerges in practice—manufacturers depend on sodium iodate for iodizing table salt, while researchers appreciate its stable reactivity for redox titrations. It’s not an everyday pantry item, but anyone who values clean water or consistent iodine intake benefits from it, whether they realize it or not.
Physical & Chemical Properties
A bottle of sodium iodate on the shelf tells just part of the story. The crystals glint, odorless and solid, with a melting point that far outpaces the boiling point of water. Toss it into a glass beaker and it dissolves swiftly, releasing ions that interact with the solution. As an oxidizer, it carries significant chemical power, which explains both its usefulness and the care needed during handling. Its stability at room temperature offers peace of mind in most storage situations, but introduce it to combustible materials and things become risky fast. This blend of stability, solubility, and strong oxidation sets sodium iodate apart from many similar salts and explains why it persists across a shifting chemical landscape.
Technical Specifications & Labeling
Walk the aisle in any chemical supply warehouse and the labels on sodium iodate tell a clear story. Purity levels usually reach analytical grade, often above 99%, to serve labs where results matter. Package labeling must follow regulations for hazardous materials, warning users about its oxidizing nature. Its clear chemical identity and batch-specific data let researchers trust that experiments will run as expected. Over the years, changes in labeling standards have aimed to reduce workplace accidents and ensure safe logistics, reflecting the evolving approach to chemical safety and traceability.
Preparation Method
Crafting sodium iodate takes more than mixing ingredients. Manufacturers start by reacting sodium hydroxide or sodium carbonate with iodine or elemental iodine under controlled conditions. The process demands attention—a poorly monitored reaction yields unwanted byproducts or low purity. Efficient synthesis avoids contamination and conserves resources, which matters more today with growing environmental and economic pressures. I’ve stood alongside chemists fine-tuning these reactions, watching them balance temperature, concentration, and timing for the purest result. This work looks monotonous to outsiders, but the smallest adjustment can mean the difference between a flawless product and a shelf full of waste.
Chemical Reactions & Modifications
Anyone who’s worked with sodium iodate knows its strongest asset comes during reactions where a reliable oxidizer is needed. In the lab, it helps detect small changes in chemical systems. Add sodium iodate to reducing agents, and it shifts quickly to form simpler iodide ions, releasing oxygen along the way. This reaction forms the backbone of important titration methods, especially in water analysis. Beyond the textbook, sodium iodate can be modified for use in advanced materials science, and research keeps uncovering new ways to adjust its reactivity through combinations with stabilizing agents or functionalizing additives. Real progress comes from these persistent experiments on variations, not just from innovation in the abstract.
Synonyms & Product Names
Chemistry isn’t just about formulas—they often sound like foreign languages outside a lab. Sodium iodate also goes by names like soda iodate, sodium salt of iodic acid, or simply NaIO3. These names show up in catalogues, research papers, and regulatory texts, but all point back to the same familiar white powder. This variety can trip up new researchers, but better communication and unified labeling standards do help cut through the confusion.
Safety & Operational Standards
Whoever scoffs at chemical safety hasn’t seen a lab after a spill. Museums and universities teach safety rules for a reason—sodium iodate can spark dangerous reactions if mishandled. It oxidizes fuels and organic material, so small samples and clean tools are the order of the day. Protective gear, from gloves and goggles to chemical aprons, guards users against dust inhalation or accidental contact. Storage demands dry, cool spaces, kept away from acids or organic carts. Every generation hears stories of accidents, and the lesson stays constant: you don’t cut corners with potent chemicals like sodium iodate. Industry standards set by occupational health agencies offer a common sense path for daily use, but culture and personal discipline build the strongest line of defense.
Application Area
Take a run through water treatment, food processing, and lab research—sodium iodate leaves its mark in each. Its role in iodized salt gets the most headlines, anchoring public-health successes like reduced goiter and improved brain development in children. Food industry operators rely on its stability to keep iodine levels consistent, for both shelf-life and nutritional labeling accuracy. In environmental science, the salt cleans up water supplies, acting as a standard for titrations and checking the integrity of purification equipment. Pharma researchers use it for synthesis and analytical testing, harnessing its oxidizing ability. From my own experience running comparative studies, sodium iodate rarely gets the spotlight, yet its reliability turns the wheel of progress.
Research & Development
Innovation can sound like hype, but with sodium iodate research, small improvements translate into big results. Ongoing work in academic and industrial labs explores greener synthesis paths, reduced energy consumption, and the design of safer handling protocols. Collaboration between academia and industry pushes these efforts, looking beyond old habits to find new efficiencies. Nanotechnology applications are emerging, expanding the list of uses and offering creative twists on a familiar chemical. Research teams focus on improving purity without excessive cost, so new applications become practical, not just theoretical.
Toxicity Research
Ask toxicologists about sodium iodate, and they’ll mention both occupational risk and environmental persistence. Exposure through inhalation or ingestion can damage the gastrointestinal tract, kidneys, and thyroid in humans, with higher doses raising red flags fast. Acute toxicity pushes for tighter workplace controls, especially where dust generation or accidental mixing with organic materials becomes likely. Over time, evidence suggests sodium iodate breaks down safely in water if handled with proper oversight, but accidental releases or mishandling can bring harm. These facts push regulators to demand routine training sessions for handlers and encourage companies to design storage solutions that minimize spill potential. Building safer labs and factories starts here—with hard-won experience and a determined attitude toward risk.
Future Prospects
No chemical, no matter how reliable, can rest on its laurels. Sodium iodate’s future relies on both tradition and change. As the push for sustainable chemistry intensifies, companies will rethink every facet of its life cycle—from sourcing raw materials to offering take-back programs for waste. Better process monitoring trims emissions and material use, while advances in green chemistry may one day transform its production entirely. Researchers hunt ways to increase selectivity and control up and down the supply chain, broadening its reach into clean tech and emerging fields like advanced sensors and smart food fortification. Policy shifts around environmental regulation and public health maintain pressure for safer, cleaner practices. Like many workhorse chemicals, sodium iodate’s best days might still lie ahead—built on a backbone of cautious learning and determined ambition, not quick-fix solutions.
What is Sodium Iodate used for?
A Chemical with a Purpose
Sodium iodate isn’t the kind of powder that finds a spot in your spice drawer or your medicine cabinet at home, but its impact stretches across industries. In my years reading the fine print on food and troubleshooting issues in the lab, I’ve come across the many hats this compound wears. It’s not famous like table salt or baking soda, yet it has earned trust from food scientists, medical experts, and even environmental workers for some very practical reasons.
Tackling Iodine Deficiency Head-On
A lot of us grew up seeing “iodized salt” on the kitchen shelf. The idea behind this is to help people get enough iodine, which keeps thyroid glands working and brain development on track, especially for kids. Sodium iodate plays a key role in the process. Food manufacturers use it to create iodized salt because of its ability to deliver stable iodine, which doesn’t lose its punch in humid conditions like potassium iodide sometimes does. I still remember sitting through a nutrition seminar where a public health official said that simple things like this saved communities from goiters and brain development issues that often sneak up in places where the soil is low in natural iodine.
Laboratory Workhorse
Sodium iodate isn’t just for fortifying food. In chemistry labs, the compound features in redox titrations, those old-fashioned but reliable tests that help determine how much of a substance is present in a solution. It behaves predictably in these tests, making life easier for students and seasoned chemists alike. Back in college, frustration usually followed flunked titrations, but sodium iodate stood out for giving consistent results, time and time again. That reliability still means a lot in research settings where precision can make or break experiments.
Pigments, Dyes, and Water Treatment
If you look deeper into how colors get manufactured—whether for textiles, plastics, or inks—sodium iodate sometimes plays a role there too. It chips in as an oxidizing agent, taking part in chemical reactions that help form stable pigments. Environmental engineers sometimes rely on it to make certain contaminants easier to remove from water. Water safety isn’t always glamorous, but I’ve seen how batch treatment using little-known chemicals like this keeps municipal systems in the clear. Knowing that these basic compounds can help maintain clean water gives me a bit of respect for the quiet work they do in the background.
Safety and Moving Forward
Handling sodium iodate isn’t something to tackle without proper care. It stands on the list of chemicals that need gloves and goggles. Breathing in its dust or eating it can cause trouble. Groups like the World Health Organization keep an eye on how much iodine people should get, reminding both regulators and manufacturers to check their numbers before making recommendations. Safe use and accurate dosing help make sure the benefits show up without causing harm.
Finding Solutions for the Future
As world populations grow and food chains stretch across continents, the demand for basic nutrients will only climb. Leveraging compounds like sodium iodate for public good isn’t just about chemical know-how; it's a matter of social responsibility. Smarter use, better oversight, and public education can keep iodine deficiency at bay and make sure industries that depend on these building blocks don’t overlook their risks.
Is Sodium Iodate safe to handle?
Taking a Look at Sodium Iodate
Handling chemicals always comes with its own set of concerns. Sodium iodate gets used in labs, food fortification, and even photographic processing. Folks see a powder, read a technical label, and wonder if caution is necessary. From my time in science classrooms, experience tells me chemicals with strong oxidizing properties deserve respect—even ones labeled for general use.
Risk Factors and Exposure
Sodium iodate stands out because of its ability to react with organic matter. Touching it without gloves might not burn right away, but it does act as an irritant. Skin may turn red, eyes sting, and I’ve seen a classmate end up coughing after accidentally inhaling the dust. NIOSH and the CDC both agree: proper handling means personal protective equipment. I’ve seen experienced chemists skip goggles for “quick” measurements, but the one time powder blew off a scoop, lab rules mattered.
Environmental and Health Considerations
Ingesting sodium iodate on accident poses more than just short-term worries. Large doses disrupt the thyroid, and chronic exposure leads to health problems in lab animals. The EPA lists it as a substance that needs proper disposal, so I wouldn’t send it down the sink. Mixing with strong acids or reducing agents? Results include rapid reactions and fumes, experiences nobody asked for. The science backs it up—not so different from other oxidizers I’ve worked with, but not one to treat lightly.
Why Some Use Still Makes Sense
Food scientists fortify salt with iodine to support thyroid health. Sodium iodate makes the cut because it stays stable in table salt over months and years. In practice, the amounts average people encounter remain tiny, with exposure kept well below safety thresholds. But handling the pure chemical is a different story—a scoop in a food processing plant or chemistry lab needs more precautions than a sprinkle of iodized salt at dinner.
How to Handle It the Smart Way
Labs investing in safety gear rarely regret it. Hand-washing, gloves, eye protection, and ventilation go a long way. Good habits I picked up during internships pay off in peace of mind and cleaner inspection reports. Small spills demand immediate cleanup with wet paper towels, tossed in dedicated waste bins, not sinks. Teachers remind students to keep dry chemicals away from heat and organic materials—those warnings stuck after a minor fire in a storage closet years ago. Even one mistake puts people, property, and projects at risk.
Building a Culture of Safety
Safety guidelines only help if people follow them. From school labs to industrial settings, making good habits stick turns information into real protection. I’d rather spend an extra minute setting up than risk burns or a trip to urgent care. When storage, labeling, and PPE become part of the daily routine, sodium iodate isn’t some lurking danger—it becomes another tool, used with care.
What is the chemical formula of Sodium Iodate?
Understanding Sodium Iodate: More Than Just a Chemical Formula
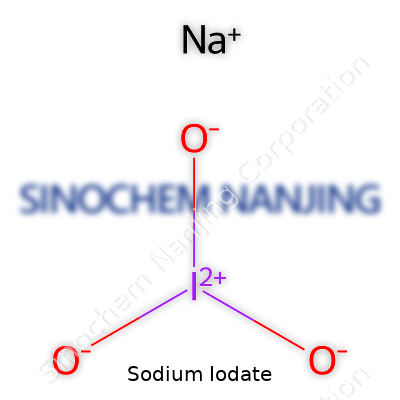
Sodium iodate shows up in both the classroom and the lab as NaIO3. Chemists pin down its structure easily: one sodium atom, one iodine, three oxygen atoms. The formula alone doesn’t say much without looking at where it fits in real life.
The Point of Knowing Chemical Formulas
Anybody reading food labels may have bumped into sodium iodate without realizing it. Food science relies on this compound whenever food companies enrich salt with iodine. Without understanding the precise molar makeup, food chemists run the risk of dosing either too much or too little. People need just the right amount of iodine for thyroid function—too much or too little swings health in the wrong direction. According to the World Health Organization, iodine deficiency affects close to two billion people. This tiny formula, NaIO3, packs a global impact in terms of public health.
Formulas and Trust in the Science
Mistakes in chemical notation sometimes slip into textbooks or websites. I still remember a classmate getting penalized for swapping a subscript somewhere on an exam. Seems trivial, but chemistry doesn’t forgive small errors. Mixing up NaIO3 with, say, sodium iodide (NaI) changes the game completely. Each compound has different properties and applications. Industry standards rest on getting the formula right the first time.
Real-Life Applications and Where Things Go Wrong
In industry, sodium iodate keeps popping up in analytical labs, photography, and even certain medical uses. Its oxidizing power makes it useful, but also a bit dangerous if handled carelessly. I once watched a simple mixing error in a student lab escalate to a small glassware incident—nobody listens to “measure twice, pour once” until the stuff foams over.
Mistakes with sodium iodate reach beyond school labs. In 2008, a surplus of sodium iodate in packaged table salt caused an over-iodization crisis in some countries, leading to cases of thyroid dysfunction. These slip-ups go all the way back to formula literacy. When scientists, regulators, or food processors don’t pay attention to the details, the public pays the price.
Solutions, Not Just Formulas
Stepping up chemical education helps cut down slip-ups, both for professionals and the public. Making molecular formulas part of early science classes fixes learning gaps before they show up at bigger scales. In workplaces, routine checks and peer reviews spot errors early. Developing digital labeling and batch tracking could offer extra insurance—companies would spot numbers out of place before products hit grocery shelves.
NaIO3 tells a story beyond the letters and numbers. Whenever chemistry leaves the page and enters daily life, knowing a simple formula becomes a safeguard against bigger troubles. People depend on accuracy, not just in the lab, but in the foods and medicines they take for granted.
How should Sodium Iodate be stored?
Understanding the Risks
Sodium iodate stays active as an oxidizing agent. That means it reacts easily with other chemicals, which can sometimes lead to hazardous situations. I remember reading about a laboratory fire in an academic setting, all because someone stashed sodium iodate in the same cabinet as organic solvents. This wasn’t just an academic mistake; it echoed a bigger problem—many underestimate the everyday risks in chemical storage. Mixing oxidizers with flammable items sets up a recipe for disaster.
Why Proper Storage Really Matters
People sometimes forget how humidity and direct sunlight speed up chemical breakdown, especially for something like sodium iodate. Moisture can clump up the powder or trigger reactions. I once opened a poorly sealed container and noticed caking, which can make weighing out doses tricky and less reliable. Those little mistakes chip away at lab safety and cause headaches during audits or inspections.
Storing Sodium Iodate the Right Way
Safe storage keeps everyone healthier and workspaces more productive. From my experience, the best place for sodium iodate is a cool, dry spot without much foot traffic. Use a sturdy, airtight container—something made of glass or certain plastics like polyethylene. Keep the vessel labeled clearly with the chemical’s name and hazard information. Ignoring labels confuses people and ramps up risk.
Never place sodium iodate near anything organic or flammable. Separation rules aren’t red tape—they prevent real harm. Labs usually dedicate cabinets for oxidizers, away from acids, bases, or combustibles. In my own teaching, I’ve seen students accidentally store incompatible chemicals together, only realizing the danger after a supervisor points out the problem. Early habits stick, so it’s important to set storage straight from the start and stick to the rules.
Personal Protective Equipment and Reporting
Strong safety culture doesn’t only depend on where you stash chemicals. If anyone spills sodium iodate, gloves and goggles come out immediately. Adequate ventilation is more than just a line in the manual; it keeps harmful dust out of the lungs. Anyone working with or around this substance deserves practical training, not just a quick rundown in a dusty booklet.
If you notice damage to a container—something as small as a cracked lid—it falls on you to report it, not wait for someone else. Leaks or spills demand swift, clear communication. Relying on others doesn’t cut it when health is on the line.
Paths to Safer Labs
Some problems just keep cropping up: mixing up containers, forgetting to relabel, skipping inspections. Fixing these means regular training refreshers and standardized labeling. Automated inventory systems can lessen human error, making it easier to track where chemicals actually sit. In my last job, digital logs helped everyone know at a glance what sat in each storage area and prompted checks for expired stocks.
Professional organizations like OSHA and the CDC update guidelines after real incidents, not guesswork. Following their advice links back to real-world consequences—keeping people safe and research reliable. Each step, from airtight containers to regular review, creates a small shield against unnecessary risk.
Why This All Matters
Treat sodium iodate with respect instead of routine. Safe storage isn’t just about compliance; it protects people, data, and careers. The best labs I’ve seen take chemical handling as seriously as the science they produce. Others could learn from their example and adopt practical changes sooner, not after something goes wrong.
What is the difference between Sodium Iodate and Sodium Iodide?
Getting Past the Labels
Folks have a habit of treating chemicals as dry facts in a textbook. For most of us, the difference between sodium iodate and sodium iodide never crosses our minds. Both have “sodium” in the name, both carry iodine, so it’s tempting to think they do the same job. Truth is, they carry different personalities—one that perks up a table salt with needed nutrients, another working as a powerful oxidizer.
What’s the Deal with Sodium Iodide?
Iodine sits among those minerals your body can’t live without. If you grew up using table salt that’s “iodized,” thank sodium iodide. This compound gets mixed straight into food salt because the human body easily pulls iodine from it. That detail keeps millions clear of thyroid problems. A lack of iodine causes goiter, a problem seen often before salt producers added sodium iodide into the mix decades ago.
Medical labs trust sodium iodide for even more important tasks. Doctors use it during scans of the thyroid. It’s also essential in some emergency situations—after nuclear accidents, for example—because it blocks the thyroid from grabbing radioactive iodine lurking in the air. The body grabs sodium iodide fast and puts it to work right away.
Sodium Iodate: A Different Player
Sodium iodate wears a different hat. This compound contains iodine, too, but the oxygen it holds makes it quite powerful. Chemically, sodium iodate acts as an oxidizer. Bakers sometimes use it to bleach flour or strengthen dough. Over in water treatment plants, you’ll find sodium iodate in processes that clean up contaminants. Those extra oxygen atoms make it risky for direct consumption; your body doesn’t handle it as smoothly as iodide. Toxicity comes into play if someone takes too much, which sets up a big red flag for food use.
Many folks don’t realize: sodium iodate actually sticks around in some salt products outside the United States. In other regions of the world, it fortifies salt as a fallback for sodium iodide. Hot, humid areas—such as some parts of Africa—deal with iodide breaking down too quickly because of warmth and moisture, so they pick iodate-based compounds, even if they’re less bioavailable.
Understanding Impact Beyond the Lab
The difference between these two shows up in everyday life and carries real consequences. For example, people living in northern regions get plenty of iodine in their diets thanks to sodium iodide, and thyroid disease rates dropped because of it. Anyone traveling or living in subtropical areas can run into salt with sodium iodate, but they often need more thorough public health measures to make sure iodine deficiency doesn’t return.
What worries health workers is that food habits rarely stay the same. If a supply chain swaps sodium iodide for sodium iodate, people could end up with less usable iodine than they need. At the same time, storage conditions and quality standards demand constant attention. Stability matters, too—sodium iodate lasts longer on shelves but brings other complications.
Paths to Better Health
A little detail like the name of a compound in a salt box makes a difference for human health on every continent. Food makers and regulators have their work cut out making sure the right iodine source ends up in the right spot. Regular monitoring, clear labeling, and switching up minimum fortification levels all help keep communities healthy. As food traditions evolve and climate changes, it’s worth keeping an eye on what lands in the salt shaker.
Choosing between sodium iodide and sodium iodate looks simple on paper, but the best choice depends on climate, technology, supply stability, and how bodies process nutrients. The lesson is simple: sometimes science hides inside the most familiar things—like a pinch of salt at dinner.
| Names | |
| Preferred IUPAC name | Sodium iodate |
| Other names |
Iodic acid, sodium salt Sodium iodate(V) Sodium salt of iodic acid Iodate de sodium |
| Pronunciation | /ˈsəʊdiəm aɪˈəʊdeɪt/ |
| Identifiers | |
| CAS Number | 7681-55-2 |
| Beilstein Reference | Beilstein Reference: 3943933 |
| ChEBI | CHEBI:75834 |
| ChEMBL | CHEMBL1201146 |
| ChemSpider | 50171 |
| DrugBank | DB09416 |
| ECHA InfoCard | 100.029.171 |
| EC Number | 231-194-7 |
| Gmelin Reference | 27114 |
| KEGG | C02315 |
| MeSH | D013480 |
| PubChem CID | 24857 |
| RTECS number | WN3675000 |
| UNII | 9M83V0777Q |
| UN number | UN1479 |
| Properties | |
| Chemical formula | NaIO3 |
| Molar mass | 197.89 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 4.28 g/cm³ |
| Solubility in water | 8.7 g/100 mL (20 °C) |
| log P | -4.5 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 12.4 |
| Basicity (pKb) | pKb ≈ 11.8 |
| Magnetic susceptibility (χ) | Magnetic susceptibility (χ): −45.0·10⁻⁶ |
| Refractive index (nD) | 2.352 |
| Viscosity | 30 mPa·s (20°C, 10% aq. sol.) |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 230.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -295.27 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -295.0 kJ/mol |
| Pharmacology | |
| ATC code | V08AA06 |
| Hazards | |
| Main hazards | Oxidizing solid, harmful if swallowed, causes serious eye damage. |
| GHS labelling | GHS02, GHS05, GHS07, GHS09 |
| Pictograms | GHS05, GHS07 |
| Signal word | Danger |
| Hazard statements | H271: May cause fire or explosion; strong oxidizer. H315: Causes skin irritation. H319: Causes serious eye irritation. H335: May cause respiratory irritation. |
| Precautionary statements | P280, P305+P351+P338, P308+P313, P370+P378 |
| NFPA 704 (fire diamond) | 2-0-2-OX |
| Lethal dose or concentration | Oral rat LD50: 1,200 mg/kg |
| LD50 (median dose) | LD50 (median dose) Oral-Rat: 178 mg/kg |
| NIOSH | WN3675000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Sodium Iodate: "Not established |
| REL (Recommended) | 1.5 mg/kg |
| Related compounds | |
| Related compounds |
Sodium chlorate Sodium bromate Potassium iodate Iodic acid |

