Sodium Hydroxymethylglycinate: A Deep Dive
Historical Development
Back in the mid-20th century, chemists were searching for preservatives that could keep cosmetic products safe but also avoid the headaches of parabens or formaldehyde donors. Sodium hydroxymethylglycinate (often abbreviated as SHMG) was developed as one solution to this puzzle. Its roots connect closely with the growing demand for mild, broad-spectrum agents during the 1970s. Over time, this preservative found a home in many personal care products, thanks to a combination of stability and low usage levels. As cosmetic safety standards tightened across Europe and North America, the compound’s use became a steady, if sometimes debated, presence in ingredient lists.
Product Overview
You won’t see sodium hydroxymethylglycinate sold in shiny packaging on store shelves. Most folks encounter it under other names in lotions, shampoos, and cleansers, where it works out of sight, preventing spoilage. Suppliers ship it as a solid or sometimes a concentrated solution. Many manufacturers prefer it because even a small dose can help products meet shelf-life expectations. The primary draw for formulators is its clear compatibility with water-based products. Not everyone recognizes it at first glance, since ingredient panels can list it as Suttocide A or simply as “preservative.”
Physical & Chemical Properties
Look at a batch of SHMG and you’re likely to see a white, crystalline powder. It dissolves quickly in water, which ranks as both blessing and curse depending on the finished formula. Odor runs faint to undetectable, which pleases formulators aiming for unscented products. Its molecular formula, C3H6NNaO3, points to its origin as a glycine derivative. In practical use, it keeps stable under a pH range of about 3 to 12, but preservatives work best near neutral pH. Temperature resilience matters in manufacturing, and this compound remains sturdy up to usual processing conditions for cosmetics—so there’s little worry about breakdown during mixing or filling.
Technical Specifications & Labeling
Manufacturers track quality using typical purity standards of 98-102%. Regulatory agencies force a close look at labeling transparency. Finished goods that use sodium hydroxymethylglycinate carry CAS number 70161-44-3. Many product technologists keep careful records of preservatives used—especially since consumer interest in “clean” labeling means full disclosure. Restrictions on maximum concentration tighten as regulatory reviews continue; Europe’s Scientific Committee on Consumer Safety set the ceiling at 0.5% for rinse-off and leave-on products. Brands must navigate both regional and international rules when deciding how to label, and audits by regulators won’t overlook undocumented preservative claims.
Preparation Method
Production relies on reacting glycine and formaldehyde in the presence of sodium hydroxide. Unlike direct mixing, success depends on getting just the right temperature and pH to steer reactions toward SHMG and not unwanted byproducts. After synthesis, the batch gets filtered, neutralized, and dried to leave the tidy crystals chemists and supply chain managers want. Companies running these reactions invest decades of know-how into minimizing leftover reagents and optimizing yield, which holds down costs and keeps safety risks lower. Even so, handling formaldehyde during manufacture means employees follow strict protocols, both for quality and personal protection.
Chemical Reactions & Modifications
Sodium hydroxymethylglycinate’s real-world action traces back to its slow release of formaldehyde under certain conditions—giving it preservative punch without dumping free formaldehyde into the formula. When it encounters microbes in a lotion bottle, it disrupts proteins and enzymes that microbes use for life processes. This trick grants manufacturers longer shelf stability but has drawn the attention of safety agencies worried about formaldehyde exposure. Chemists experiment with modified versions or combinations with other preservatives to dial back any concerns, trading off between keeping molds and bacteria away and avoiding any unwanted breakdown products.
Synonyms & Product Names
The personal care industry loves aliases. You’ll find sodium hydroxymethylglycinate on ingredient lists as Glycine, N-(hydroxymethyl)-, monosodium salt, or Suttocide A. Trade names differ, as seen in catalogs from international suppliers. This doesn’t just trip up consumers; even experienced chemists pause to double-check ID numbers and alternative names to avoid batch errors or regulatory mix-ups. End users rarely know the difference but rely on consistent labeling to stay informed.
Safety & Operational Standards
Each year, regulatory agencies review old safety data, new toxicity studies, and emerging consumer complaints. Production lines feature ventilation and personal protective equipment when formaldehyde is in use, and waste from manufacturing passes through strict treatment steps. Finished products must pass skin sensitization and irritation tests. Several regions restrict or cap SHMG use: the EU, Japan, and parts of Asia insist on lower maximum limits, especially as consumer advocacy groups keep tabs on all formaldehyde-releasing agents. Brands test not just their formulas but also packaging interactions, making sure no ingredient increases unwanted reactions over time.
Application Area
Most uses of sodium hydroxymethylglycinate cluster around rinse-off and leave-on personal care. Shampoos, face cleansers, body lotions, and even some wet wipes rely on it. Producers value it in products for sensitive skin, since it doesn’t tend to trigger allergies in most people at allowed concentrations. There’s occasional use in industrial fluids, but the bulk of demand still comes from consumer-facing brands looking for preservatives that keep fungal and bacterial contamination down while holding the line on claims of skin-friendliness. The shift toward paraben-free and formaldehyde-free marketing means formulators keep one eye on alternatives at all times.
Research & Development
Current research tracks toward better understanding of how low-level preservatives interact within complex formulas. Scientists keep pushing to find preservatives that last longer, work at lower doses, and avoid label red-flags for educated consumers. Modified derivatives of sodium hydroxymethylglycinate challenge the dominance of legacy preservatives, and in-house R&D teams run shelf-life studies against clones, generics, and combos. Teams often pull from industry-wide databases and toxicology journals, shaping decisions about which versions qualify for new product launches. They sometimes collaborate with dermatologists to look at rare sensitizations and reduce reporting risks.
Toxicity Research
Scientists ran early studies with high doses that flagged the risk of skin reactions—mostly redness or itchiness in rare cases. Toxicologists measure the release rate of formaldehyde, since that chemical can cause problems for people with allergies or asthma. Large-scale epidemiological evidence so far points to SHMG as less risky than many past preservatives, but nobody calls it risk-free. Animal studies at typical use concentrations didn’t show organ toxicity, but all chemical preservatives get routine re-evaluation, especially in Europe, where regulators set precautionary lower limits just to play it safe. Safety data grow each year, driven by both industry and academic scrutiny, and the debate over “allowable limits” almost never cools off.
Future Prospects
The future of sodium hydroxymethylglycinate depends on consumer trust and scientific advances. As more folks read ingredient lists and demand “cleaner” products, brands face pressure to keep reducing both the presence and the percentage of synthetic preservatives. Research teams look to tweak the molecule or combine it with plant-based preservation systems that skirt formaldehyde entirely. Digital tracking of adverse events could force new studies or stricter caps. Still, unless truly revolutionary preservation chemistries arrive, SHMG keeps a foothold as a go-to ingredient for many brands balancing cost, effectiveness, and regulatory hurdles. Experts in both cosmetics and chemical manufacturing won’t stop looking for safer, greener, or more transparent options, but for now, sodium hydroxymethylglycinate isn’t leaving the stage.
What is Sodium Hydroxymethylglycinate and what is it used for?
Understanding the Ingredient
Sodium hydroxymethylglycinate sounds technical, but most of us have brushed up against it somewhere in daily life. It’s a preservative made from glycine, a natural amino acid. Chemists developed this compound to keep mold, yeast, and bacteria out of products like shampoo, lotion, moisturizer, and even some liquid soaps. Clean bottles, fresh scents, and long-lasting creams often owe a debt to this preservative.
Why It Matters
Nobody picks up a face cream hoping bacteria have set up shop inside. If you’ve ever left fruit on the counter too long, you know what happens when microbes take over. Skincare isn’t much different—give bacteria a warm, moist bottle, and they’ll thrive. Sodium hydroxymethylglycinate keeps things safe and fresh for weeks or months. For people who want to avoid preservatives altogether, it’s easy to say "just use natural ingredients," but products without any kind of preservative spoil quickly. For stores and brands that want to guarantee safety and shelf life, finding something mild but dependable makes a big difference.
Safety and Worries
There’s no perfect answer with preservatives. Sodium hydroxymethylglycinate works at low concentrations, so you don’t need a lot to protect a product. That helps lower risk, but it doesn’t wipe it away. The main worry stems from its chemistry. This preservative releases a little formaldehyde as it breaks down. Formaldehyde can trigger allergies and skin irritation, especially in sensitive people. In extreme cases, long-term exposure to high levels can cause cancer. In personal care, the amounts used fall far below hazardous levels recommended by health authorities around the world. Modern manufacturing keeps testing these limits. Still, those with very sensitive skin sometimes notice redness or itching with creams that contain this ingredient. I’ve seen plenty of folks argue about safe numbers, but personal differences matter—some people break out, others don’t react at all.
Alternatives and Choices
People who want natural ingredients often turn to products with short ingredient lists or those bragging about being “preservative-free.” Some brands use plant extracts or ferment-based solutions, though most still rely on something synthetic to keep things safe for longer than a week or two. Potassium sorbate and phenoxyethanol gain fans for similar reasons—they work and tend not to irritate most skin types. For folks who need gentle formulas, fragrance-free and hypoallergenic labels help steer clear of known troublemakers, including sodium hydroxymethylglycinate.
Consumer Responsibility
Product labels carry clues. If a lotion lasts months at room temperature, some form of preservative lives inside, even if it sounds “natural.” People with sensitive skin or a tendency for allergies benefit from patch tests. You just dab a little product inside the elbow, then wait a patch or two of days for a reaction. If you break out, it’s a sign to adjust your routine or find brands that skip certain preservatives.
What Companies Can Do
Transparency matters. Brands build trust by listing what’s inside and not hiding behind “complex formulations.” Companies that pay attention to feedback add value, whether through clearer labeling, safer blends, or continuous testing. Keeping customers informed builds confidence. As people learn more about what goes into everyday products, honest information helps everyone make safer choices.
Is Sodium Hydroxymethylglycinate safe for use in cosmetics and personal care products?
What is Sodium Hydroxymethylglycinate?
Sodium hydroxymethylglycinate often shows up on product labels as a preservative. It’s known for keeping creams and lotions from growing bacteria. This synthetic ingredient comes from glycine, a simple amino acid, and contains a bit of formaldehyde in its structure. Many skin creams, shampoos, body washes, and even makeup removers list it as part of their formula. Its main role is to extend shelf life, something most of us appreciate if we leave a jar of face cream in a warm bathroom for months at a time.
What Science Says about Safety
The big concern centers on that small amount of formaldehyde lurking in the chemistry. Scientists have noted that, under certain conditions, sodium hydroxymethylglycinate can release formaldehyde—a problem because formaldehyde can irritate skin and eyes, and, with longer exposure and higher concentrations, even act as a carcinogen. The American Cancer Society and World Health Organization both list formaldehyde as a probable cancer-causing agent when inhaled repeatedly or in large amounts.
In practice, cosmetic products in the United States and Europe use this preservative in very low concentrations—often well under 0.5 percent. At these levels, most healthy adults tolerate it well. Organizations like the Cosmetic Ingredient Review (CIR) and U.S. Food and Drug Administration (FDA) have reviewed studies and haven’t found enough evidence to ban it outright in rinse-off or leave-on products, as long as companies stick to recommended concentrations. European regulators limit its use in leave-on products even further due to their stricter standards on anything that can release formaldehyde.
Who’s at Risk?
Some people feel the sting more than others. Folks with sensitive skin, existing allergies, eczema, or who often experience contact dermatitis need to pay attention. I’ve seen messages in skincare forums where users report redness, itchiness, or tiny bumps after trying a new face cream, only to spot sodium hydroxymethylglycinate on the label. Even at low amounts, reactions can happen—especially after repeated use.
Balancing Risks and Benefits
Preservatives aren’t just filler. Without them, moisturizers, sunscreens, and hair gels would turn into petri dishes of mold and bacteria. Homemade remedies that “go bad” quickly illustrate why brands turn to chemicals like this for safety. Well-formulated products keep our skin protected from more than just dryness or blemishes; they keep microbes away.
The challenge comes in picking the right preservative. Brands using sodium hydroxymethylglycinate need to keep levels low and test for stability and safety over months and years. Consumers who know they’re prone to reactions benefit from patch testing new products before slathering them on their faces or bodies. Labels deserve a close look, especially for those who have dealt with skin allergies in the past.
Solutions on the Horizon
Many companies listen to the growing interest in gentler alternatives. There’s rising demand for preservatives made from natural sources, like certain essential oil components, fermented extracts, or organic acids. These options usually cost a bit more and add complexity to the manufacturing process, but the tradeoff is worth it for people seeking a cleaner label.
Thicker regulation and thorough testing will always matter more than clever marketing. Clear ingredient lists and honest conversation between brands and shoppers can help catch issues early. If someone struggles with repeated reactions and can’t seem to find the culprit, consulting a dermatologist and asking about patch testing for preservatives can pinpoint sensitivities.
Summary
Sodium hydroxymethylglycinate works as a preservative in low doses. Most people won’t run into major problems, but those who know their skin’s history need to stay alert. More transparency and better alternatives open the door for safer, more reliable skincare for everyone.
Are there any side effects or risks associated with Sodium Hydroxymethylglycinate?
What’s in Our Products?
So many personal care and cosmetic items get their long shelf life from preservatives. Sodium hydroxymethylglycinate is one of those chemicals you’ll spot in ingredient lists—lotions, shampoos, cleansers, and even baby wipes. It’s derived from glycine, an amino acid, but labs tweak it so it gets antimicrobial strength. This preservative stops things like bacteria and mold from taking over the bottle before it runs out.
Unpacking the Side Effects
Most folks use these products every day and never give preservatives a second thought. Sodium hydroxymethylglycinate doesn’t attract as much attention as parabens or formaldehyde, but dermatologists and scientists see it show up in patch testing often enough. One thing that stands out: this compound can release tiny amounts of formaldehyde over time, especially if exposed to heat or stored for a while.
For people with sensitive skin, eczema, or chronic allergies, this can mean burning, irritation, rashes, or worse. European studies have pinpointed cases where sodium hydroxymethylglycinate triggered contact dermatitis. Even those without usually reactive skin might notice flare-ups after using products that stick around all day—think face creams or body lotions—right along hairlines, eyelids, or hands. The U.S. Cosmetic Ingredient Review panel put limits on its use for this reason. In the EU, regulations cap the concentration at lower levels to keep risks down.
Who’s Most at Risk?
It’s the new ingredient on the block that not everyone’s skin recognizes. Babies, toddlers, older adults, and immune-suppressed folks have skin barriers more likely to react. Repeated use ups the odds of sensation and redness. Before I tried a new moisturizer loaded with sodium hydroxymethylglycinate, my own sensitive skin felt fine. A couple weeks in, though, I developed an itchy patch along my jawline. Only when I ditched that bottle did the rash go away. After that, I checked ingredient packs much more closely.
There’s also the bigger question of formaldehyde exposure. This compound gets labeled as a “donor,” which means it releases formaldehyde as it breaks down. Formaldehyde carries a link to certain cancers and can trigger bad asthma flares or allergies, especially in workplaces where it’s used heavily. Cosmetic companies keep their preservative concentrations low to avoid most of the danger, but if you already react to conventional formaldehyde, you probably want to skip products with sodium hydroxymethylglycinate.
What Can Shoppers Do?
Folks with tricky skin would do well to patch test new products before using them on large areas. Reading a label for long, unpronounceable chemical names gets exhausting, but the effort pays off if you dodge a two-week rash. Look for brands honest about what’s in their stuff and call customer service if ingredients aren’t clear.
Companies can help by improving labeling and offering preservative-free options or using alternatives shown to be gentle in studies. Dermatologists and allergists recommend keeping track of reactions in a journal. For salon professionals and others exposed at work, good ventilation and gloves lower exposure risks.
With so many safer preservatives on the market, it makes sense for people to push for formulas that keep skin comfortable and lower chemical exposure. Keeping a close eye on research helps everyone choose with confidence.
Is Sodium Hydroxymethylglycinate a natural or synthetic preservative?
Looking at Ingredient Labels
Grocery shopping for skin or hair products used to mean trusting the brand on the bottle. Over time, I started squinting at the labels, especially as I learned about how some chemicals help with shelf life but can raise health questions. Sodium hydroxymethylglycinate keeps popping up in moisturizers and shampoos. Some bottles boast “naturally derived” next to it, others say “paraben-free, safe and mild.” The ingredient’s origins matter because more people want safe but effective preservatives from natural sources.
What’s Behind the Name?
The ingredient traces its roots to glycine, which is an amino acid found in many living things, including people and plants. Companies make sodium hydroxymethylglycinate by reacting glycine with formaldehyde under controlled conditions. This process transforms glycine, but it doesn’t just squeeze it out of a plant or animal and pop it in a bottle. Chemical reactions and lab equipment play a significant role before a cosmetic company even sees the preservative.
What’s the Definition of “Natural?”
Plenty of personal care companies stretch the meaning of “natural.” Some shoppers expect natural to mean something taken straight from nature—like rosemary extract or honey. Others are okay with “naturally derived,” which often points to a lab synthesis that starts from a natural source. Sodium hydroxymethylglycinate fits more in the “derived” camp. Even if the process begins with glycine, adding formaldehyde in a lab doesn’t happen in nature.
Does the Origin Matter?
Plenty of consumers do care. Labels play a big role in choosing what goes on our skin, especially for those looking to avoid preservatives seen as risky. Parabens and formaldehyde donors have caught bad press for potential links to irritation or allergic reactions, and in rare cases, more serious health problems. What raises questions for sodium hydroxymethylglycinate is the use of formaldehyde—a compound some see as a red flag, despite its low release levels in finished products.
Safety Record
Cosmetic chemists point to tests from groups such as the Cosmetic Ingredient Review and regulatory limits set by the European Union and US FDA. These studies set usage amounts considered safe for rinse-off and leave-on products. Use in products complies with these levels. That said, some people with very sensitive skin have reported reactions. One study in the Contact Dermatitis journal pointed out rare cases of allergic contact dermatitis from this preservative. These reports push people to read labels and look for milder options.
Finding Solutions for Cleaner Labeling
Companies have a chance to clarify their labels so people know exactly what’s inside. Using clearer language—such as “synthetic preservative, starts from an amino acid or glycine”—helps shoppers make choices that align with their priorities. Product safety teams could invest in alternatives with long safety records and fewer links to skin reactions, like potassium sorbate or phenoxyethanol. Transparency builds trust, especially as younger buyers value honesty more than the promise of “all-natural.”
Thinking Before Buying
People should weigh their own tolerance and priorities—sodium hydroxymethylglycinate in low concentrations brings fewer risks than going without a preservative altogether. If a product is labeled “natural preservative,” consider reading further to understand the process behind it. The deeper we dig, the better choices we can make for ourselves and our families.
Can Sodium Hydroxymethylglycinate cause allergic reactions or skin irritation?
What is Sodium Hydroxymethylglycinate?
Sodium Hydroxymethylglycinate pops up in lots of personal care products—moisturizers, shampoos, and cleansers. Companies use it as a preservative to keep products fresh. It’s synthetic, and it gets made from glycine, a simple amino acid. The cosmetic industry picked this ingredient up because it’s effective at controlling bacteria and fungi, which can spoil a product fast.
Allergic Reactions and Skin Irritation
Anyone with sensitive skin has probably learned to scan for parabens and fragrances, but new names keep popping up. Sodium Hydroxymethylglycinate rarely makes headlines, but stories about stinging, redness, or rashes can still happen—even in formulas labeled as gentle.
I remember testing a “clean” moisturizer a few years ago. About an hour after slathering it on, I felt a burning itch across my cheeks. Sodium Hydroxymethylglycinate was lurking in the ingredient list, and that lesson taught me to pay attention to more than just the bold claims. Dermatologists confirm this: Most people tolerate products with this ingredient, but those with eczema or a history of contact allergies should be careful. Medical research backs this up. There’s a documented risk of irritation, especially at higher concentrations. Some patch tests show 2–4% of people can react to it with redness or even blisters. Children’s skin and people overusing leave-on products often respond more strongly.
Why Reactions Happen
Sodium Hydroxymethylglycinate doesn’t just act as a preservative. It also releases tiny amounts of formaldehyde during its shelf life. The beauty world debates about this a lot. Formaldehyde can trigger allergies, and people with a pre-existing sensitivity notice issues sooner. The United States and European Union limit how much formaldehyde any preservative can contribute to personal care products—current limits try to keep exposure safe, and routine oversight happens, but laws can’t protect everyone.
Trying to Stay Safe
Reading labels gets tiring. Still, for those with sensitive skin, it makes a real difference. Product batches can vary, and some companies don’t make concentrations obvious. Consumer advocacy groups call for clearer labeling and lower thresholds for chemicals that might trigger skin reactions. As a consumer, I try to stick with brands that share their full ingredient list and list the percentage of preservatives used. It also helps to choose products tested for sensitive skin or those under dermatologist control.
Scientists continue searching for safer, effective preservatives. Some brands rely on packaging that reduces contamination risk, cutting down the need for strong preservatives. The less the product gets exposed to air or fingers, the lower the risk of bacteria multiplying—and the fewer harsh chemicals needed.
What Dermatologists Recommend
Those struggling with rashes or breakouts after using a new product should stop use immediately and check in with a dermatologist. Bringing the product along helps because patch testing may identify if Sodium Hydroxymethylglycinate, or something else, did the damage. Most dermatologists suggest patch testing every new product before use, especially on sensitive or inflamed skin.
Most people won’t have a problem with Sodium Hydroxymethylglycinate at the low levels found in reputable brands. But anyone with a compromised skin barrier, a background in allergies, or those using many different products each day stands at higher risk. Trusting your skin’s reaction and advocating for transparency from brands only makes everyday personal care safer.
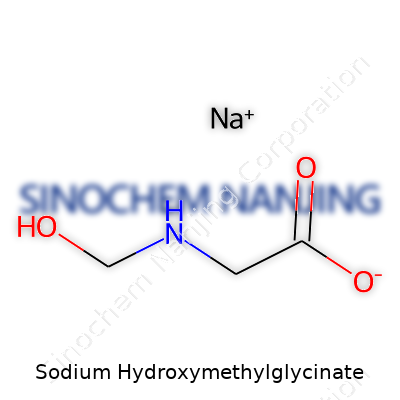
| Names | |
| Preferred IUPAC name | Sodium 2-(hydroxymethylamino)acetate |
| Other names |
Sodium Hydroxymethylglycinate Sodium salt of Hydroxymethylglycine Hydroxymethylglycinate sodium Sodium N-(Hydroxymethyl)glycinate |
| Pronunciation | /ˌsoʊ.di.əm haɪˌdrɒk.siˌmɛθ.əlˈɡlɪ.sɪ.neɪt/ |
| Identifiers | |
| CAS Number | 70161-44-3 |
| Beilstein Reference | 1771383 |
| ChEBI | CHEBI:85145 |
| ChEMBL | CHEMBL3301063 |
| ChemSpider | 234203 |
| DrugBank | DB11157 |
| ECHA InfoCard | 03-2119980889-23-0000 |
| EC Number | 701-089-9 |
| Gmelin Reference | 84938 |
| KEGG | C14230 |
| MeSH | D000079568 |
| PubChem CID | 22259 |
| RTECS number | PB0680000 |
| UNII | 6FZ7V9276T |
| UN number | UN2810 |
| Properties | |
| Chemical formula | C3H7NO4Na |
| Molar mass | 111.09 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.31 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -3.0 |
| Vapor pressure | 0.01 mmHg (25°C) |
| Acidity (pKa) | 11.5 |
| Basicity (pKb) | pKb = 3.0 |
| Magnetic susceptibility (χ) | -13.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.510 |
| Dipole moment | 4.04 D |
| Thermochemistry | |
| Std enthalpy of formation (ΔfH⦵298) | -619.6 kJ/mol |
| Pharmacology | |
| ATC code | D08AX12 |
| Hazards | |
| Main hazards | Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS05, GHS07, Danger, H314, H317 |
| Pictograms | GHS05, GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H318 |
| Precautionary statements | Precautionary statements: P261, P280, P305+P351+P338, P310, P337+P313 |
| NFPA 704 (fire diamond) | 1-0-0 |
| Lethal dose or concentration | LD50 (oral, rat): 2,680 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 2.61 g/kg |
| NIOSH | UUJ3C9E6YM |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.5% |
| Related compounds | |
| Related compounds |
Glycine Sodium glycinate Hydroxymethylglycine Formaldehyde N-(Hydroxymethyl)glycine Methylol glycine |

