Sodium Hydrogen Sulfate: More Than a Chemistry Class Staple
Historical Development
Long before big industry figured out what sodium hydrogen sulfate could offer, people discovered that mixing various minerals from the earth sparked powerful chemical changes. Chemists started paying attention to this white crystalline powder during the Industrial Revolution. Back then, folks wanted better glass windows and textiles that held color. Someone saw potential in reacting sodium chloride with sulfuric acid, kicking off commercial production. Compared to now, laboratory setups were crude, but that process led to the mass production of both hydrochloric acid and sodium hydrogen sulfate. By the 20th century, the chemical’s versatility opened doors in food, household cleaning, and heavy industry. Past discoveries shape today’s approach, especially as environmental rules tighten.
Product Overview
Anyone who has cleaned a pool or balanced the pH in a commercial kitchen likely brushed shoulders with sodium hydrogen sulfate, often called sodium bisulfate. Most people know it as a granular, acidic powder that dissolves pretty well in water, unlike some bulkier chemicals that cause clumping. Companies ship it in tons, not teaspoons, because so many applications rely on its simple acidity. You see variants in the market geared toward pool care, household cleaning, and even specialty food processing. So, it isn’t just another lab curiosity—it’s part of everyday life for farms, city water plants, and restaurant suppliers.
Physical & Chemical Properties
Sodium hydrogen sulfate usually comes as a colorless, odorless solid. Though it won’t set off alarm bells with a pungent smell, its low pH packs a punch. Water loves to dissolve it, which makes it practical for dosage control. Unlike sulfuric acid, which can eat through metal and clothing, sodium hydrogen sulfate offers a more manageable, user-friendly acidifier. Its stability at room temperature, non-flammability, and low volatility make it popular in both home and industrial settings where precision and safety matter. High temperatures may push it toward decomposition, releasing sulfur oxides, but under normal use, it behaves consistently.
Technical Specifications & Labeling
Regulatory labels don’t exist just to check a box—they play a key role in safe handling. Suppliers have to spell out purity, particle size, and upper concentration of trace compounds. Labels reference hazardous nature and recommend storage away from alkalis and oxidizers. In food-grade cases, the percentage of sodium hydrogen sulfate tends to be quoted outright, sometimes with certification badges for organic processing or compliance with food-safety agencies. As enviromental regulators update lists of restricted or monitored compounds, labeling practices keep evolving. That makes it easier for downstream users to handle and store it without running afoul of safety standards.
Preparation Method
Making sodium hydrogen sulfate looks deceptively simple: combine sodium chloride with sulfuric acid under controlled conditions. At industrial scale, this reaction forms sodium hydrogen sulfate and hydrochloric acid as primary products. Some facilities go with sodium sulfate converted further with more sulfuric acid, but the main driver remains economic feasibility and local environmental policies. Temperature and mixing speed influence product quality as well as conversion efficiency. Automation stepped up in recent decades, and remote monitoring has cut accidental releases or product variability. Large-scale batch production typically feeds into continuous lines for granulation and bagging, reducing workforce exposure to fumes or dust. While chemists often discuss green chemistry, this reaction keeps generating large tonnages with relatively fewer by-products compared to more exotic synthesis pathways.
Chemical Reactions & Modifications
Chemistry geeks know sodium hydrogen sulfate acts as both a reactant and as a pH modifier. Pouring it into water triggers immediate acidification. Add a base like sodium hydroxide and you get sodium sulfate, a table staple in chemical labs. Heat the powder up, and decomposition kicks in—sulfur trioxide leaves, creating more sodium sulfate. For specialty applications, you can blend sodium hydrogen sulfate with other acids or buffer agents, tweaking the end products as industries see fit. This flexibility powers everything from textile dye baths to in-situ cleaning of food-processing equipment. Its behavior around temperature and concentration changes keeps process engineers on their toes, particularly where by-products might affect downstream processes or regulatory compliance.
Synonyms & Product Names
Walk into any supplier's catalog and you’ll find sodium hydrogen sulfate under names like sodium bisulfate, sodium acid sulfate, or monohydrogensulfate. Food industry types might see “acid salt” or “E514(ii)” printed on spec sheets, especially in Europe. Pool maintenance teams recognize trade names that emphasize acidifying action or safety over sulfuric acid. Terminology shifts depending on industry, but the underlying chemistry stays the same. Staying familiar with synonyms matters because regulations or import/export rules sometimes call for reporting under specific names, causing headaches for anyone tracking materials in big facilities.
Safety & Operational Standards
Anyone in a lab or pool-supply warehouse understands the importance of handling sodium hydrogen sulfate with respect, not fear. Direct skin exposure is unpleasant; dust clouds cause eye irritation and trouble breathing. Regulators demand sturdy gloves, goggles, and dust masks in any operation involving bag breaking or large-scale mixing. Spill protocols focus on containment and neutralization, not panic. Fire departments know it won’t fuel flames directly, but in a tightly packed warehouse it can amp up corrosiveness if water lines break. Guidelines handed out by OSHA and similar agencies globally ensure that both storage and shipping stay in line with public health. Periodic training refreshers remind workers about proper ventilation, dry storage away from bases, and emergency cleanup. These are practical steps any operation could implement, offering real-world protection rather than empty paperwork.
Application Area
Sodium hydrogen sulfate lands everywhere from swimming pools to dinner tables. Pool owners rely on it to rein in unruly alkaline levels, keeping water safe for swimmers and equipment. Breweries and food processors tweak pH without harsh acids, meeting quality standards for cheese, canned veggies, or wheat flour. Metal finishers strip scale with it, giving new life to machine parts and cookware. City water managers use sodium bisulfate for pH adjustment to protect infrastructure and keep water tasting right. Professional cleaning teams rely on its slipstream effect, busting mineral deposits in commercial kitchens and dishwashers. In agriculture, the compound shows up as a soil conditioner, breaking apart alkali deposits and keeping crops healthy. Its reach touches pharmaceuticals as a precursor or buffer, reflecting its status as a go-to for diverse industries.
Research & Development
Research has shifted lately toward eco-friendlier uses and production practices. Environmental engineers look at sodium hydrogen sulfate as a less hazardous acidifying agent compared to hydrochloric or sulfuric acid. Researchers examine how precisely it interacts with common water contaminants and whether hybrid blends could unlock new industrial cleaning routines. The food sector invests in trials exploring microbial stability and product shelf life, all while keeping ingredient lists clean and simple. Lab teams track potential synergies with new green surfactants in detergents, aiming to curb waste and boost energy efficiency. Scientists are asking hard questions about end-of-life impacts too, seeking ways to bring sodium hydrogen sulfate full circle through recycling or benign degradation. It feels like every field using the compound faces a push for more sustainable, transparent processes, which should benefit industry and consumers in the long run.
Toxicity Research
Unlike some industrial chemicals, sodium hydrogen sulfate rests on a solid base of toxicological studies. Acute toxicity seems low for most short-term, incidental exposures, but concentrated dust or solution burns unprotected skin and eyes. Repeat exposure studies outline upper safety limits—mostly to protect factory workers or waste-treatment teams. Animal tests helped nail down safe commercial dosages for food-use, with years of review by international agencies. Environmental scientists watch for runoff issues, as excessive acidification harms aquatic life or soil microbes. Their research feeds directly into modern risk assessments and product labeling. Chronic-exposure studies continue, especially for people working near hydrometallurgy plants or commercial-scale food processing. Community health projects add to the pool of data, balancing industrial progress against basic safety.
Future Prospects
Looking down the road, sodium hydrogen sulfate finds itself in the mix for more sustainable industry solutions. As water treatment facilities worldwide tighten output standards, the chemical gains favor for controlled acidification with lower safety risks. Food manufacturers want simpler, more recognizable ingredients, which makes “acid salt” an easy sell compared to unknown preservatives. Green manufacturing drives research into new synthesis pathways that reduce by-products and energy costs. Engineers see potential for its integration into closed-loop processes—recycling both the product and its reaction partners. Tech developers aim to automate safe handling, reducing human exposure and waste during bulk transfer or packaging. As both consumers and regulators zero in on transparency and reduced ecological impact, sodium hydrogen sulfate’s versatility positions it as a practical tool for decades to come.
What is Sodium Hydrogen Sulfate used for?
Cleaning Up, Quite Literally
Anyone who’s spent time scrubbing away mineral stains from a bathroom or cleaning pool tiles has probably benefited from sodium hydrogen sulfate. This compound, with its punchy acidic quality, steps in where soap and water back down. Removing hard water stains and scaling from surfaces—sinks, toilets, dishwashers—often means reaching for a cleaner containing sodium hydrogen sulfate. It gets right to the source of build-up, breaking down calcium and mineral deposits. It’s a common ingredient because it works fast, rinses easily, and doesn’t leave a strong chemical smell behind.
Safety in Swimming Pools
Swimming pool owners recognize the name from another chore—balancing water pH. Pools attract all sorts of contaminants, including sunscreen, sweat, and leaves. Over time, these things push pool water towards the alkaline end. A pH that runs too high can turn chlorine into a bystander, leaving swimmers at risk for eye irritation or worse. Sodium hydrogen sulfate, labeled “pH reducer” at pool supply shops, brings pH back down where it belongs. This prevents cloudy water, scaling, and lets chlorine do its job. Backed by studies, balanced pool water helps protect both equipment and skin. That’s a win on both health and wallet.
Quiet Role in Food and Industry
Food factories rely on sodium hydrogen sulfate to tweak acidity. Certain foods—especially candies and canned items—use it to achieve that mouth-puckering tang. It controls flavor, but more importantly, acidity slows spoilage and keeps food safer longer. The U.S. Food and Drug Administration green-lights this use, provided strict limits are followed. Ingredient lists may hide it under technical names, but its role remains. Focusing on safe levels and regular review of research helps keep risks in check.
On factory floors, sodium hydrogen sulfate helps in dyeing textiles, cleaning metal, and stripping rust. Paint booths use it for preparing metal surfaces, getting rid of old paint or corrosion before a new coat goes on. It steps into these jobs as a safer substitute for harsher acids. Workers need training and proper gear, but compared to concentrated sulfuric acid, it lowers accident rates and environmental hazards.
Staying Safe Matters
Sodium hydrogen sulfate has teeth. Swallowing it, splashing it on your skin, or breathing its dust can cause burns or serious irritation. Gloves and goggles aren’t optional in the workplace or in the home. Keeping it locked up away from curious kids and pets is common sense. People with asthma should steer clear of dust or fumes. Poison control centers offer advice if accidents happen, but the best course relies on careful use and respect for chemicals.
Finding Balance
So many modern conveniences, clean showers, sparkling pools, safer canned foods, owe something to an unassuming powder. Sodium hydrogen sulfate might never earn a place on the red carpet, but it strengthens consumer safety, food freshness, and manufacturing productivity. Better instructions on packaging, accessible safety data, and honest ingredient disclosure empower everyone. It’s worth asking questions and looking up information before using products containing any chemical, even trusted ones.
Is Sodium Hydrogen Sulfate safe to handle?
Understanding the Risks
Sodium hydrogen sulfate often shows up in cleaning products, swimming pool chemicals, and some food processing environments. At home, I once saw it in a pool pH decreaser sold at a big-box store. Truth is, it isn’t some exotic lab curiosity, but it isn’t table salt either. Its crystals sting skin, bite open cuts, and don’t get along with eyes. A splash makes itself known fast.
According to the U.S. National Library of Medicine, sodium hydrogen sulfate draws water and reacts with moisture on skin, leaving burns and blisters. Inhaling its dust feels rough on the nose and throat. Eye exposure has landed folks in the ER. Direct contact deserves respect, not curiosity.
Storage and Handling, Done Right
I always favor well-labeled containers. Leave this stuff near heat or in a humid spot, lumps form, and handling gets even messier. I keep bags of chemicals clearly away from other cleaners. Spills on concrete or wood floors can eat away at finishes, and swept dust flies into the air with ease.
Safety isn’t just about skill—it's about gear. Gloves, a pair of goggles, and maybe a basic dust mask get the job done for small use. Never use bare hands. Sodium hydrogen sulfate feels dry, so it’s easy to think it’s harmless, but it sneaks through unprotected skin. At my local pool supply shop, I once saw a worker pour it into the water using nothing but a plastic scoop. I’ve watched the splatter on their fingers—you can tell fast who respects their job and who just wants to get through it.
Why Precautions Matter
The Centers for Disease Control and Prevention (CDC) and OSHA mark sodium hydrogen sulfate as an irritant—classified under hazardous materials. Too many folks get complacent. Some homeowners throw chemicals together, causing unwanted reactions. Mixing sodium hydrogen sulfate with chlorine-based pool shock can trigger dangerous fumes. Each year, poison control centers deal with chemical burns and breathing injuries that could have been avoided.
Kids should never mess with pool chemicals, but I’ve heard stories from neighbors whose kids got curious. One time, a friend’s 12-year-old decided to “help clean the pool” and wound up with a bright red rash and watery eyes. Secure storage keeps curiosity accidents from happening.
Looking for Solutions
Education is the best solution. Retailers owe it to customers to hand out simple, readable instructions. Big warning labels help, but a short, clear talk at checkout goes further. Pool associations and community centers can host yearly safety demos.
For people working with this chemical regularly, real training matters more than any printed sheet. I’ve seen workplace accidents traced back to new hires never getting a full rundown. Nothing replaces a hands-on walk-through by someone who’s handled tough spills and knows the shortcuts to avoid.
Disposal ends up forgotten. Dumping leftover sodium hydrogen sulfate down household drains can corrode pipes. Most municipalities accept small amounts at hazardous waste collections. Following those local rules helps out the city and keeps waterways safer.
Respecting Experience and Evidence
Anyone who’s handled sodium hydrogen sulfate learns quick to treat it with care. It’s not complicated but demands attention. A little extra caution saves pain, property, and sometimes more. Backing each step by facts from the CDC, OSHA, or poison control builds real trust. Combining guidelines, personal caution, and clear explanations makes a tough job routine, and everyone goes home safe.
What are the storage requirements for Sodium Hydrogen Sulfate?
Understanding the Chemistry
Sodium hydrogen sulfate, sometimes called sodium bisulfate, packs a punch in cleaning products, pools, and plenty of industrial uses. This salt stands out for its ability to drop pH in water and clean scale off surfaces. I’ve seen it used in both home pools and big food processing operations. For something so handy, you’d think storage would be simple. The catch: this compound brings along some pretty clear risks if you get storage wrong.
Moisture: The Enemy of Safe Storage
I learned early on that sodium hydrogen sulfate easily absorbs water. Leave a bag open in a humid storeroom, and it’ll clump or even liquefy. In a warehouse I visited, staff left a sack too close to a leaky wall. The result was a sticky, corrosive mess that needed hazardous waste handling. To avoid this, store it in airtight containers, and use strong, water-resistant packaging. Polyethylene drums or sealed plastic tubs do the job well. Always keep sacks off concrete floors, since even trace dampness from below can seep up and cause trouble.
Separation: Keep It Away from Incompatibles
Sodium hydrogen sulfate does not play nice with some materials. Mixing with bases or alkalis creates a dangerous fizz and lots of heat—never a good thing inside a storeroom. Even worse, contact with certain metals may lead to corrosive reactions that damage shelving or contaminate stored chemicals. I once saw a rusty metal scoop dissolve after it spent the weekend in a drum of bisulfate. Warehouses should always store this product on shelves or pallets reserved for acids or acid salts. Metal storage bins or tools don’t belong anywhere near it.
Temperature and Ventilation
A storeroom doesn’t have to stay icy cold, but high temperatures speed up moisture absorption and breakdown. In warm climates, sweat in the air causes sodium hydrogen sulfate to clump. Decades of industry guidelines recommend cool—below 30°C—ventilated rooms. Ventilation stops any buildup of fumes and keeps the workspace pleasant for anyone handling bags or drums. From personal experience in large pool supply outlets, poor airflow turns storerooms stuffy and makes powder caking far more likely. Wall-mounted exhaust fans are an easy fix.
Labeling and Security: Safety Above All
It’s tempting to cut corners by stacking this product beside fertilizers or detergents, since it looks so much like them. Don’t. Mixing up sodium hydrogen sulfate with something alkaline can spell disaster if the wrong bag ends up in the wrong process. Clear labels, physical barriers, and signs colored for acid hazards make mistakes less likely. Companies with good storage protocols also keep spill kits close—a practical step that cuts response time if a bag breaks. I’ve seen well-organized warehouses recover quickly from small accidents, while sloppy ones spend half the day cleaning corrosive messes.
Waste and Long-Term Storage
Expired or caked-up sodium hydrogen sulfate doesn’t go in the dumpster. Local regulations treat it as hazardous waste, and disposal needs certified carriers. Long-term storage creates further risks because containers degrade. Annual checks and solid inventory control avoid forgotten, aging product stashed in dark corners. Responsible handling from receiving shipments straight through to disposal shows respect for both worker safety and the environment.
How should Sodium Hydrogen Sulfate be disposed of?
Looking at the Risks
Sodium hydrogen sulfate shows up everywhere from pool cleaning kits to classroom chemistry sets. It doesn’t look menacing—usually a white powder, sometimes in granules—but it carries a punch. Dropping it down the drain without thinking can knock household pipes and create dangerous conditions. Mixed wrong, it stings the eyes and throat, ruins your skin, and puts serious pressure on local sewage systems.
Why Proper Disposal Deserves Attention
I learned the hard way in a college lab that some chemicals can cause a heap of trouble in small amounts. Sodium hydrogen sulfate turns water acidic fast. That acidity won’t just burn human skin; it also throws off the pH in rivers and lakes, hurting fish and plant life. From what I’ve seen, places that allow chemicals to flow unchecked into drains end up footing big bills for water treatment, or worse, facing lawsuits after public health scares.
What the Experts Say
Environmental agencies across the US, including the EPA, mark sodium hydrogen sulfate as hazardous waste if you have more than a trivial amount. The substance lands on the list for its effects on aquatic environments. Pouring it away without thought is illegal in most states. Even small businesses and schools get checked for violations. Repeated reports show communities suffer from acid spills, even in small doses, with higher medical issues and damaged infrastructure.
Steps That Make a Difference
Old habits die hard but safer routines stick after a while. Diluting sodium hydrogen sulfate so that it mirrors the pH of tap water works for household leftovers—never dump it neat, always check with litmus paper and neutralize with baking soda. Larger amounts always call for hazardous waste collection. Towns usually announce drop-off days, and many schools keep a running log of hazardous waste collection. Homeowners can find local recycling centers online with a quick search.
Leaving chemicals labeled and in original containers curbs confusion for anyone handling waste later on. In workplaces, a trained person needs to sign off before anything leaves the premises. My first job in a community pool taught me to double-check labels and consult the local water board before calling the job done. Getting it right saved us from fines and locked gates.
Building a Safer Future
Nobody wants to scroll the news and read about local fish dying, kids getting rashes from swimming, or pipes melting in an apartment complex. Simple changes, like community education programs and stricter retail labeling, can change outcomes. Schools benefit from up-to-date disposal guides that staff and students both understand. Cities that spend on public information campaigns see better environmental scores and fewer accidents. Often, all it takes is someone like you or me, standing up and saying ‘let’s do this right.’ Small actions pile up—in the lab, at home, in the neighborhood—protecting water, health, and future budgets.
It’s easy to assume a simple powder can’t cause much harm. But chemicals stack up fast, and the cost of carelessness shows up in utility bills, health stats, and the spread of damage downstream. Respect for the process, a little bit of effort, and a willingness to teach the next person go a long way toward keeping our water clean and our communities safe.
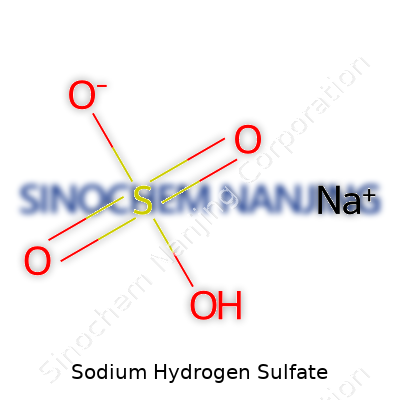
What is the chemical formula and properties of Sodium Hydrogen Sulfate?
What Is Sodium Hydrogen Sulfate?
Sodium hydrogen sulfate takes the chemical formula NaHSO4. Sometimes it crops up in older textbooks as sodium bisulfate, and a lot of folks recognize it in pool pH reducers or cleaning agents. It comes out of the production lines as a white, odourless crystalline powder, able to draw moisture right from the air. Touch some water to it, and the substance dissolves easily. Every lab stockroom I’ve seen holds a jar or bag of the stuff, since it serves all sorts of quick fixes for shifting pH.
Chemical Properties
In simple terms, sodium hydrogen sulfate acts as an acid salt. Water knocks loose a hydrogen ion, turning the solution acidic. A 1 M solution strolls in with a pH of about 1, so it demands the same care as other corrosive materials. Tipping some onto metal sees a bit of fizz as hydrogen bubbles escape. Sodium ions stay dissolved, while sulfate partners with whatever base gets mixed in.
Heat changes the game for sodium hydrogen sulfate. Crank temperatures past 315°C, and it decomposes, breaking down into sodium sulfate (Na2SO4), sulfur trioxide (SO3), and water. That’s a detail schools and industrial plants alike care about, since hot equipment plus acid gas makes for a risky situation. For anyone prepping a lab demo, decent ventilation and a bit of caution beats stories told after the fact.
Where Sodium Hydrogen Sulfate Shows Up
People run into sodium hydrogen sulfate most often while treating pools, adjusting laundry water, or balancing commercial dishwasher cycles. Acid shines at knocking down stubborn mineral deposits, so automatic dish machines use it to keep nozzles clean and scale at bay. Homeowners like it for resin water softeners or when lowering garden pH beats replanting. Teachers mix it into chemistry lessons, since it brings acidity without the splash hazard of liquid sulfuric acid.
Handling and Risks
From years around labs, the bite of sodium hydrogen sulfate on skin feels like any strong acid. Quick rinses matter, especially on cuts or near eyes. Workers handling drums wear gloves and goggles, and no one stores it next to bases like sodium hydroxide — acids and bases react fast, so separation goes a long way toward workplace safety. Inhalation gets overlooked until the irritation kicks in, so proper masks and good airflow earn their keep in dusty spots.
Disposal runs pretty straightforward, as diluted sodium hydrogen sulfate and its salts don’t hang around in the environment. Wastewater treatment handles moderate spills. Runoff from high-use sites might increase local acidity or stress aquatic life — an occasional concern near pool contractors or commercial laundries. Regulations push for keeping large spills out of open streams, and that’s a rule worth following.
On Safe Use and Alternatives
Folks sometimes switch to milder acids when the job allows, but sodium hydrogen sulfate offers a middle ground between harsh mineral acids and weaker options. Many manufacturers explore using enclosed dosing systems or solid-state pH adjusters to cut down on airborne dust. Clear labeling and simple instructions help the average user avoid injuries or mix-ups, especially where kids or pets roam.
Sodium hydrogen sulfate fits a host of roles, but, as with any chemical, it rewards those who read the labels and respect its punch. For anyone keeping a bag on the shelf, a sharp focus on ventilation, storage, and basic safety gear sets the standard for good practice.
| Names | |
| Preferred IUPAC name | Sodium hydrogen sulfate |
| Other names |
Sodium bisulfate Monosodium sulfate Acid sodium sulfate Nitre cake Sodium acid sulfate |
| Pronunciation | /ˌsoʊdiəm haɪˈdrɒdʒən ˈsʌlfeɪt/ |
| Identifiers | |
| CAS Number | 7681-38-1 |
| Beilstein Reference | 1207086 |
| ChEBI | CHEBI:86163 |
| ChEMBL | CHEMBL1459 |
| ChemSpider | 61935 |
| DrugBank | DB09446 |
| ECHA InfoCard | 03b9b8c9-33c3-420a-8441-2d8e1b5b46dd |
| EC Number | 231-665-7 |
| Gmelin Reference | 754 |
| KEGG | C16236 |
| MeSH | D013502 |
| PubChem CID | 516919 |
| RTECS number | WB2200000 |
| UNII | X0O6L7X4WJ |
| UN number | UN3260 |
| Properties | |
| Chemical formula | NaHSO4 |
| Molar mass | 120.06 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 2.74 g/cm³ |
| Solubility in water | Very soluble |
| log P | -4.3 |
| Acidity (pKa) | 1.99 |
| Basicity (pKb) | Basicity (pKb) = 11.96 |
| Magnetic susceptibility (χ) | `-48.6·10⁻⁶ cm³/mol` |
| Refractive index (nD) | 1.335 |
| Dipole moment | 0 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 157.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -945.0 kJ/mol |
| Pharmacology | |
| ATC code | A09AB16 |
| Hazards | |
| Main hazards | Causes severe skin burns and eye damage. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H315: Causes skin irritation. H319: Causes serious eye irritation. H335: May cause respiratory irritation. |
| Precautionary statements | P264, P280, P305+P351+P338, P337+P313, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 2-0-1-acid |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 Oral Rat 2490 mg/kg |
| LD50 (median dose) | 2,800 mg/kg (rat, oral) |
| NIOSH | WC5600000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 15 mg/m³ |
| Related compounds | |
| Related compounds |
Sodium bisulfite Sodium sulfate Sodium sulfite Sodium metabisulfite |

