Sodium Hexanolate: Past, Present, and Where It’s Going
Historical Development
My first encounter with sodium hexanolate gave me a glimpse into how chemical progress often goes overlooked outside specialist circles. Decades back, researchers dug into the family of alkoxides, eyeing ways to transform everyday organic compounds. Out of this surge in curiosity, sodium hexanolate surfaced as a valuable bridge between more basic sodium alkoxides and longer-chain substances that could adapt to new uses. The chemical appeared out of practical questions: could tinkering with the chain length of sodium alkoxides unlock new physical properties? Chemists in the mid-twentieth century didn’t just isolate sodium hexanolate out of theory — they looked for ways it could serve industry where shorter or bulkier alkoxides fell short. Over time, its unique handling characteristics and reactivity sparked further study, often paving the road for niche applications in synthesis, where other bases or nucleophiles struggled.
Product Overview
Walking through a chemical storeroom, sodium hexanolate doesn’t jump out with an extravagant label or much fanfare. The substance stands out for its calm reliability; it brings value for people mixing solutions that need a decent, predictable base without introducing clunky side-products. For industries making surfactants or custom organic molecules, sodium hexanolate usually flies under the radar, but those who work with it respect its straightforward performance. Researchers in custom synthesis appreciate the way it behaves — not overly eager to react where it’s not needed, but strong when put to the test in the right scenarios.
Physical & Chemical Properties
As someone who has weighed and watched sodium hexanolate dissolve, I’ve noticed the clear, white-solid appearance — it’s unremarkable until you factor in its solubility in polar solvents and its tendency to react rapidly with water. A dry, controlled environment matters because of its easy hydrolysis. The smell, faintly alcoholic, hints at its ties to hexanol. Its melting point sits above room temperature, giving some leeway in storage. Reactivity with carbon dioxide and moisture remains an ongoing challenge; you get sodium carbonate and hexanol as soon as the solid absorbs water, teaching anyone working with it to keep desiccators in steady service.
Technical Specifications & Labeling
Instead of elaborate marketing, the real worth comes from purity and freshness. In labs, clear labeling communicates its molecular formula, batch number, and sometimes the recommended storage environment. Chemists often opt for quality brands that pack sodium hexanolate under inert gas in air-tight bottles to safeguard against accidental degradation. Commercial users watch for specifications like sodium content and residual starting alcohols. Certifications confirming absence of heavy metals or other alkali impurities often accompany bulk shipments. This traceability not only ensures product safety but also acts as a silent partner to avoid batch-to-batch surprises in sensitive syntheses.
Preparation Method
Most who’ve handled sodium hexanolate know the drill: synthesis usually starts with a careful reaction between sodium metal and 1-hexanol. The sodium slices hissing their way through alcohol never lose their edge — one missed step, and you’ll end up with an impure mess or worse, an unsafe reaction. Once the reaction dies down, vacuum drying pulls out leftover hexanol, leaving a solid that can be scraped or powderized. The simplicity is deceiving, but for anyone who’s run the process, attention to dryness and cleanliness matters more than high-tech equipment or patent secrets.
Chemical Reactions & Modifications
Sodium hexanolate makes a handy launching point for etherification or esterification, passing along its hexyl group in reactions that tweak molecules for custom surfactants. Pair it with alkyl halides, and you’ll get a path to new ethers; add acid chlorides, and specialty esters aren’t far off. This breadth of use stems not from any headline-grabbing reactivity, but the gentle, almost cooperative behavior it shows in the presence of numerous organic substrates. And for making sodium-based catalysts or modifying surfaces to become more hydrophobic, the hexanolate group plays well with silanization and surface grafting, giving coatings that resist water or oils more evenly.
Synonyms & Product Names
Over time, sodium hexanolate has picked up alternative names, like sodium caproate or sodium 1-hexanolate. Industry veterans know these stem from the naming conventions of the parent alcohol, hexanol, and the sodium salt nature of the product. In research circles, journals may call it sodium hexanol-1-ate or even sodium n-hexanolate, each trying to clarify its source and structure. On chemical supplier websites, users search for any of these names and often find overlaps with related sodium alkoxides used in minor but crucial reactions.
Safety & Operational Standards
One gets a healthy respect for sodium alkoxides through handling sodium hexanolate. Its caustic nature demands gloves and goggles, not because it leaps at skin, but because even small spills can leave a rawness that takes days to heal. Inhalation shouldn’t be a risk with its low volatility, but dust generated in weighing or transfer asks for fume hoods and dust masks. Water contact generates heat and fumes, so fire protocols match those for other flammable sodium compounds. Training includes more than memorized instructions — sharing best practices and mistakes shapes the attitude toward safety much more effectively than labels or warning icons ever could.
Application Area
Sodium hexanolate rarely gets a starring role, but it sits in the toolkit for organic chemists, material scientists, and sometimes even enzyme researchers. In the search for base-catalyzed transformations, it steps up where sodium methanolate might react too quickly or where sodium octanolate resists dissolving. In surfactant research, hexanol-based precursors allow for tuning of hydrophobicity — manufacturers can stretch the role of sodium hexanolate into new detergents or specialty lubricants relevant in both industrial cleaners and machine maintenance. Biochemists sometimes probe the effect of such alkoxides on enzyme function, chasing both inhibition and activation trends that could point toward new understandings of metabolic pathways.
Research & Development
Watching grant proposals and patent applications over the years, one can spot a steady, if understated, flow of interest in sodium hexanolate’s role in advanced material creation. Polymer chemists look for new initiators, and sodium hexanolate sometimes fits as a milder base or nucleophile in polymerization reactions where more aggressive alkoxides ruin subtle features. As researchers design new hydrophobic coatings or want adjustable degradation rates in biodegradable plastics, sodium hexanolate’s moderate reactivity and handy solubility profiles come up for consideration. In applied research, the compound allows for creative synthesis paths; each breakthrough often feeds a niche but supports the expansion of toolkits for future makers and innovators.
Toxicity Research
Decisions about the safety of sodium hexanolate lean on studies of similar sodium alkoxides. My conversations with toxicologists reveal that acute toxicity stays low as long as precautions match the basic caustic hazard. Chronic effects haven’t emerged as a widespread problem, but incomplete data means the substance remains under gentle scrutiny, especially for workers with prolonged exposure. Responsible handling, clear workplace protocols, and the ongoing review of animal data keep the compound within safe margins for labs and industrial operators. The environmental side carries more weight; accidental dumping into water sources could upset local ecosystems, especially when coupled with other sodium salts.
Future Prospects
Looking forward, sodium hexanolate has yet to hit a ceiling. The growth in tailored surfactant and detergent production hints at a broader role for alkoxides with this chain length. The movement toward greener chemistry could push researchers to look at sodium hexanolate for cleaner synthetic routes, fewer by-products, and improved recyclability. New analytical techniques and automation in reaction development mean small players like sodium hexanolate can gain relevance in specialty synthesis and pilot plants pushing into renewable materials. Demand for specialty organosodium compounds will likely nudge industries to revisit the utility and versatility of “forgotten” substances — here, sodium hexanolate has a chance to shine, provided those in the field keep sharing their real-world experiences, successes, and setbacks.
What is Sodium Hexanolate used for?
Understanding Where Sodium Hexanolate Shows Up
Sodium hexanolate doesn’t turn heads in the headlines, yet it works its way through industries in unexpected places. From my time teaching high school chemistry, I learned how these chemicals shape everything from the gloss on our dinnerware to specialized coatings. Companies put sodium hexanolate to work in manufacturing, cleaning, and even a few food processes. This compound steps up as a building block, making things cleaner, tougher, or easier to handle.
Everyday Products That Rely on Sodium Hexanolate
I’ve seen the name pop up on the ingredient lists in soaps and detergents. This isn’t because it smells nice or kills germs—it helps mix oil and water, scraping away greasy messes. Imagine scrubbing a stubborn frying pan after a long dinner party. Sodium hexanolate helps break up the baked-on fat so hot water and elbow grease can finish the job. That matters in busy households where every minute counts and a cleaner kitchen means fewer worries about food safety.
Industrial folks embrace it, too. Paints and coatings need chemical helpers to prevent streaks and clumps. Sodium hexanolate helps paints spread evenly on surfaces—with fewer weird patches and less waste. People want a smooth finish, whether painting a shed in the backyard or operating industrial spray booths for vehicles.
Hidden Role in Food Processing
I once worked in a bakery during college. Food safety feels personal—you never want to serve anything questionable. Sodium hexanolate shows up not as a flavor booster, but as a processing aid. Think anti-caking or stabilizing textures in certain foods. The U.S. Food and Drug Administration allows limited use for specific tasks, and oversight stays tight. Bakers and snack producers rely on it to keep products fresh for longer and improve quality.
Safety, Health, and Environmental Concerns
News stories highlight health, and sodium hexanolate deserves scrutiny. It shouldn’t touch bare skin or eyes, especially in industrial settings—workers need gloves, goggles, and training. Environmental regulations restrict dumping or emissions that might hurt waterways. Getting these rules right protects the people handling chemicals, as well as communities downstream. A mistake doesn’t just disrupt production; it can put local environments at risk.
Open dialogue improves outcomes. Trade groups and advocacy organizations keep pressure on manufacturers to test chemicals for safety. If my years in the classroom taught me anything, it’s that kids want to know “why.” That question—why safety rules exist—keeps accident rates down and builds a culture where factory workers, managers, and brand leaders all look out for one another.
Looking Ahead: Smarter Solutions
Safer alternatives attract attention. Research teams explore bio-based and biodegradable substitutes to replace conventional surfactants like sodium hexanolate. Some big brands support these innovations, thinking about the bigger picture—from supply chain carbon footprints to rivers in their own backyard. Governments balance innovation with safety by updating standards and watching for emerging trends.
People deserve transparency about chemical use. A clear ingredient label empowers families to choose what feels right at home. As more manufacturers open up about their processes, consumers gain confidence and options. In this way, sodium hexanolate isn’t just a behind-the-scenes player; it’s a spark for conversations on health, product quality, and environmental responsibility.
Is Sodium Hexanolate safe for consumption or external use?
What Is Sodium Hexanolate?
Sodium hexanolate isn't a well-known name on food labels or skincare products. This compound forms when hexanoic acid reacts with sodium, creating a salt used in labs and research settings. Companies sometimes consider it for industrial uses because it's cheap and easy to produce. That doesn’t mean it belongs on our skin or in our food.
Looking Into the Research
I always check reliable databases and science journals before I put trust in any ingredient. Sodium hexanolate doesn’t have piles of studies behind it like some acids or preservatives do. The FDA doesn’t list it as approved for food additives. There’s barely a whisper about it on the European Food Safety Authority’s resources. ScienceDirect and PubChem flag it as a chemical of interest, but no one gives it a safety pass for ingestion or topical use. That raises a red flag for me as someone who cares about what goes in and on the body.
Potential Risks and Gaps
If an ingredient hasn’t gone through proper toxicity studies, nobody can say it’s safe for your salad or your face. Chemicals can behave one way in a test tube and another way on our skin. Some sodium salts show up as food preservatives or cleaning agents, but each one has to pass its own safety checks. Hexanolate’s long-term effects on human cells, hormone balance, or sensitive skin haven’t been mapped out as far as I can see. People who react to fatty acids or have sensitive skin might risk burns, rashes, or irritation from compounds their bodies don’t recognize.
Importance of Safety Data
I’ve seen what can happen when untested chemicals sneak into cosmetics or wellness trends. A rush to innovate sometimes means skipping the boring steps of repeated testing and peer review. That ends poorly for folks who end up with allergies, hormone disruption, or even chronic illness over time. Ingredients like parabens and phthalates were once given a free pass before research caught up to the risks. That lesson sticks in my mind whenever a new compound appears without a long list of safety studies. Experts at the American Contact Dermatitis Society and EPA both say if no one’s tested a chemical for personal care or food, don’t roll the dice.
What Consumers Can Do
Labels don’t always make it easy to spot chemical names. Anyone concerned about what touches their skin or goes in their mouth has the right to demand transparency. Product makers who want real trust need to back up every ingredient with data from reputable labs. Dermatologists and dietitians can serve as helpful gatekeepers in spotting safe ingredients. I’d rather pay a little more for a product that posts its full safety reports instead of settling for mystery compounds.
Better Paths Forward
If manufacturers want to introduce substances like sodium hexanolate, the best approach runs through full FDA or EFSA evaluation. That isn’t just paperwork – it’s basic respect for the consumer. Until then, people can stick to ingredients with public track records and plenty of science to back up their place in the shopping cart.
What are the main properties of Sodium Hexanolate?
Chemical Structure Matters
Sodium hexanolate, a sodium salt produced from hexanol and sodium, shows some surprising features once you look past the dry chemical equations. It carries the formula C6H13ONa. The molecule itself boasts a straight six-carbon chain with sodium dangling off the oxygen—a structure that sets the stage for its reactivity and use. Placing sodium onto that alcohol group flips the game: suddenly the compound turns from a mild, neutral alcohol into a strong base ready to snatch protons and jump into chemical reactions.
Physical Traits You Notice Right Away
The white, powdery appearance of sodium hexanolate doesn’t give much away, but handling it tells another story. Think of it as a soap-like material that dissolves well in water and alcohol. It feels slippery because it breaks down oils, much like you’d expect from a heavy-duty cleaning product. In labs, its strong base properties can irritate skin and eyes, demanding careful handling.
Compared to common sodium hydroxide, sodium hexanolate stands out with its longer hydrocarbon tail. This detail impacts both solubility and the way it interacts with other organic molecules. Because of this structure, it often shows more compatibility with oils and greases—useful in organic chemistry and cleaning up tough residues.
Reactivity: More Than Just Basic
Some chemicals just sit there on the shelf, but sodium hexanolate almost begs for action. Drop it into water and you’ll get a quick reaction, forming hexanol and sodium hydroxide. This reaction reveals its core personality: a strong base eager to swap out sodium for hydrogen when given the chance.
Working with this compound, I've seen it act as a catalyst, nudging along reactions that would take forever otherwise. It doesn’t just sit idle in bulk; it pulls double duty in both lab synthesis and industrial processes. Used to strip fats from surfaces or to introduce hexyl groups in organic synthesis, sodium hexanolate acts fast and thorough.
What Makes It So Useful
The oil-soluble nature means sodium hexanolate finds a place in detergents, especially for breaking down tough, oily messes. Its presence in manufacturing, particularly of specialty soaps, often goes unnoticed—but you spot the difference in the effectiveness of cleaners meant for greasy equipment.
In research settings, this base helps drive forward specific chemical reactions, where mild bases just won’t get the job done. A chemist’s toolkit doesn’t work without reliable, strong bases to coax molecules into forming new bonds. Sodium hexanolate does this reliably, especially when working with fatty acids or other non-polar compounds.
Potential Issues and Safer Handling
Strong bases cause burns. Sodium hexanolate can be worse if you get careless, as the oily nature means it sticks around longer on skin or equipment compared to pure sodium hydroxide. Gloves, eye protection, and proper ventilation become non-negotiable. In my own work, I store it in airtight containers, away from moisture, since the powder likes to grab water right out of the air, clumping up into a greasy mess if you leave it exposed.
Disposal also needs attention. Flushing large amounts straight down the drain invites problems, since sodium salts and fatty byproducts can cause blockages and harm water systems. Small quantities dilute well, but big operations need neutralization and responsible chemical waste management.
Room for Smarter Solutions
Innovations in green chemistry call for less hazardous alternatives. Recent research tracks ways to use milder, more biodegradable bases where possible. For settings demanding sodium hexanolate’s specific performance, better packaging and safer handling instructions could cut down on accidents and waste. Some companies already work on coated forms that reduce dust and accidental contact, which I see as a step in the right direction.
Are there any hazards or precautions when handling Sodium Hexanolate?
Understanding the Substance
Sodium hexanolate doesn’t appear in everyday conversation outside of chemical labs. It’s a specialized compound, mostly used by chemists who need strong organic bases in their reactions. This stuff packs a punch, not just in chemical reactions but also in the risks tied to its use. If you walk into a lab and see this labeled on a bottle, it tells you one thing: caution matters just as much as accuracy.
Direct Exposure Poses Real Risks
One of the things that stands out about sodium hexanolate is how it reacts to moisture and skin contact. Just a dash of water kicks off a strong, caustic reaction. Splashes on skin or eyes can cause burns—think of what strong drain cleaner can do and double it. I remember a colleague at university who got careless during a synthesis project, thinking gloves alone would do the trick. The compound seeped through a small tear and gave him a nasty burn, forcing him to seek medical attention. This isn’t a risk to wave off—handling such chemicals without proper barrier protection is a gamble that doesn’t tend to end well.
Air, Water, and Fire Don’t Mix Well
Open air isn’t friendly to sodium hexanolate. Exposure pulls moisture out of the air, sparking off reactions that can degrade the compound—and spread caustic fumes in the process. These fumes can irritate airways within minutes. Breathing in just a bit without a mask can leave you coughing and red-eyed for hours. It’s not just about comfort; it’s about protecting lung health.
Few people realize that sodium hexanolate also creates flammable hydrogen gas when it touches water or acids. Even a small spill near a sink can fill a tight space with invisible gas. A spark, a pilot light, anything hot enough becomes a real hazard. The lab safety sheets say it clearly, but until you’ve heard the pop and hiss or felt the heat from an unexpected spark, it feels like theory.
Storage and Personal Protection
Dry, air-tight containers keep sodium hexanolate steady and safe. The smallest crack in a seal can attract moisture, building up pressure or leading to surprise reactions inside storage cabinets. Chemical-resistant gloves, goggles, and lab coats are standard for a reason, and anyone who’s tried to skip the goggles learns fast that sodium hexanolate is unforgiving. Chemical splash goggles trump regular glasses. Fume hoods aren’t a workplace luxury; they’re the buffer between a controlled procedure and an accident that calls for the safety shower.
Finding Better Methods
Lab teams have started looking for alternatives to harsh sodium alkoxides like sodium hexanolate. Some processes stick with it for efficiency, but safer chemistry is always in demand. Automated handling systems, where possible, remove people from direct contact. Industry push for improved packaging—double-sealed containers, precise spout bottles—makes a difference in both labs and transit. Regular safety training rooted in real incidents reminds both rookies and veterans that familiarity breeds carelessness. Sharing those stories builds a culture where safety beats speed.
Responsibility in Action
Sodium hexanolate isn’t one for cutting corners. Whether you’re a seasoned chemist or a student learning the ropes, taking risks with this compound can change the course of a whole project—or ruin a career with one mistake. Personal safety in the lab doesn’t rest on policies alone; it grows from respect for what these substances are capable of doing, and from a work culture that values coming home unharmed at the end of the day.
Where can I purchase Sodium Hexanolate and what are the packaging options?
Scarcity and Legitimate Supply Chains
People asking about buying sodium hexanolate often face a tough road. This isn’t just another reagent one stumbles upon in a corner shop or on popular online marketplaces. Call any regular chemical supplier, and you’ll likely hear a puzzled pause. Sodium hexanolate does not see much demand in routine industry or consumer spaces, which means only a handful of specialist chemical distributors stock it – mostly those serving research labs, not general public hands.
I’ve seen researchers lose days, sometimes weeks, thumbing through catalogs and sending out desperate emails. Even in leading academic circles, sourcing rare reagents usually means contacts, paperwork, and patience. Before considering a purchase, you’ll be asked for your purpose, proof of credentials, and a shipping address that matches an approved institution. This kind of compound rarely slips through regulatory cracks.
Packaging is Not Like Your Typical Purchase
A big expectation gap trips up those used to simple e-commerce. Chemicals come in a range of pack sizes, but sodium hexanolate rarely appears in small, consumer-friendly portions. Instead, suppliers prepare requests based on order minimums, lab needs, or project scales. Most will not consider anything below 25 grams, and it's often packed in tightly sealed glass or high-density polyethylene bottles. The extra care limits the chance of moisture, contamination, or uncontrolled reactions. No one wants loose white powder arriving in a Ziploc. Not in this field.
Bulk options may run from 100 gram jars to kilogram canisters, but you’ll need documented institutional use to unlock those volumes. Pricing sits all over the place, depending on purity, batch source, and geographic location. Most sales stay locked to professionals or companies with a record of ethical use, vetted by sales staff trained to sniff out shady intentions.
Why Regulation and Documentation Matter
Sodium hexanolate doesn’t float around markets for a reason. Reactive chemicals like these can carry serious safety, liability, and legal risks. In my years working near labs, I’ve seen firsthand how improper sourcing can open the door for disaster—leaks, misuse, even uncontrolled reactions that put entire facilities at risk. Perhaps more crucial, regulators tighten access to prevent this compound from ending up in unqualified hands.
Expect to fill out forms about your intended application. These documents aren’t a hurdle, they’re a safeguard for everyone. Chemical dealers want credible labs, faculty researchers, and engineers with known projects on record. If you represent an institution, get your internal purchasing office ready to handle questions. Those working in regulated markets (education, medical research, industrial R&D) already recognize these channels and know which boxes to tick.
Possible Alternatives and Safer Practices
If you’re an amateur, rethink your approach. Plenty of educational-grade compounds serve basic tests or demonstrations without the headaches of controlled substances. For hobbyists, digging too deep into advanced materials can bring more worry than progress. Partnering with an institution—university, R&D lab, even a science-focused start-up—often opens new doors, both for sourcing and safe handling.
Ultimately, finding sodium hexanolate builds a case for working within established frameworks and trusted supplier networks. Agencies like Sigma-Aldrich, Alfa Aesar, and Thermo Fisher maintain strict eligibility checks for good reasons. These aren’t just hurdles, but the difference between safe practice and risky shortcuts. If you need it, lean on professional networks, and be ready to answer direct questions about your work.
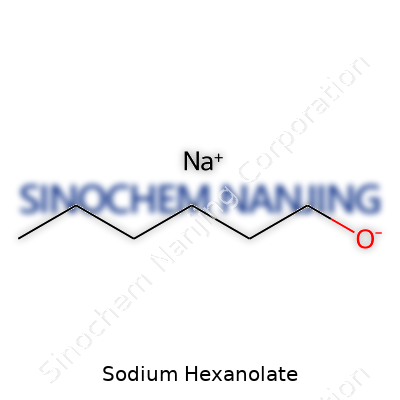
| Names | |
| Preferred IUPAC name | sodium hexan-1-olate |
| Other names |
Hexyl sodium Sodium caproate |
| Pronunciation | /ˈsəʊdiəm hɛkˈsænoʊleɪt/ |
| Identifiers | |
| CAS Number | [24304-26-5] |
| 3D model (JSmol) | `[Na+].CCCCCC[O-]` |
| Beilstein Reference | 3858736 |
| ChEBI | CHEBI:52016 |
| ChEMBL | CHEMBL514344 |
| ChemSpider | 23073442 |
| DrugBank | DB14163 |
| ECHA InfoCard | ECHA InfoCard: 100940-197 |
| EC Number | 213-633-2 |
| Gmelin Reference | 114388 |
| KEGG | C14325 |
| MeSH | D015550 |
| PubChem CID | 23665765 |
| RTECS number | MO2325000 |
| UNII | SYE18K89EA |
| UN number | UN2813 |
| Properties | |
| Chemical formula | C6H13NaO |
| Molar mass | 102.126 g/mol |
| Appearance | White powder |
| Odor | odorless |
| Density | 0.93 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -0.77 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 15.8 |
| Basicity (pKb) | pKb ≈ 0.4 |
| Refractive index (nD) | 1.401 |
| Dipole moment | 2.90 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 208.6 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | C05XA01 |
| Hazards | |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS05 |
| Signal word | Danger |
| Hazard statements | H314: Causes severe skin burns and eye damage. |
| Precautionary statements | P280, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 3-1-2-W |
| Flash point | 54°C |
| Lethal dose or concentration | LD50 oral rat 2,270 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2 g/kg (oral, rat) |
| NIOSH | NT8010000 |
| PEL (Permissible) | PEL (Permissible) for Sodium Hexanolate: not established |
| REL (Recommended) | Not recommended |
| Related compounds | |
| Related compounds |
Sodium pentanolate Potassium hexanolate Sodium heptanolate |

