Sodium Fluoroacetate: A Deep Dive into Its Chemical Legacy and Modern Implications
Tracing the Trail: Historical Development
Salt and acids have shaped chemical industries for centuries, but sodium fluoroacetate brings its own story—a potent one, veined with human ambition and ecological consequence. Originally discovered within certain South African plants, this compound entered the industrial spotlight in the 1940s, leveraging its natural toxicity to combat pest populations. Governments and researchers saw promise in selectively managing species that threatened agriculture and native ecosystems. In Australia and New Zealand, sodium fluoroacetate still carries weight in the ongoing struggle against invasive mammals. Its historical path speaks to a time when society prioritized yield and conservation, attempting to use scientific insight to tip natural balances. Many developments built upon experiences, both tragic and triumphant, showing how chemistry often walks a razor’s edge between remedy and risk.
Examining Sodium Fluoroacetate Up Close
This chemical turns up as a colorless, odorless crystalline powder. It dissolves easily in water, making it feasible for baiting techniques in wildlife control. With a molecular formula of C2H2FNaO2, it stands out thanks to its fluorine atom, which is seldom seen in nature besides in this family of highly toxic plant toxins. Sodium fluoroacetate carries a low melting point around 200°C. Because it lacks strong taste or odor, it can blend easily into substrates—one key reason it calls for the most stringent handling protocols. As for chemical stability, it handles storage well under dry, cool conditions, but reacts rapidly in the presence of moisture or biological systems, which ties directly into its dangerous profile.
Understanding Technical Details: Specifications and Preparation
In its prepared form, sodium fluoroacetate demands accuracy. Industry standards enforce strict purity benchmarks, generally above 95 percent for pest control use. The technical labeling highlights acute toxicity, often flagged with symbols denoting "poison" and requiring clear usage instructions in both legal and operational contexts. Manufacture depends on fluorination chemistry—typically using chloroacetic acid and reacting it with hydrogen fluoride, yielding crude fluoroacetic acid, which then gets neutralized with sodium carbonate to form the salt. This process runs with significant safety hurdles; strong acids and toxic intermediates raise risks that only experienced chemical professionals manage. Storage containers require clear hazard labeling, secure closures, and restricted access to safeguard against accidental exposure or misuse.
Delving Into Chemical Behavior: Reactions and Modifications
Despite the simple structure, the molecule exerts complex effects in biological systems. In chemical terms, sodium fluoroacetate hydrolyzes quickly to fluoroacetic acid in water, which then enters the tricarboxylic acid (TCA) cycle in living organisms. Here lies the root of its toxicity. Fluoroacetate mimics acetate and becomes incorporated into metabolic reactions, ultimately blocking energy production and causing cellular malfunction. Modifications of the core molecule yield similar "suicide substrate" toxins, from methyl fluoroacetate to related compounds, yet the sodium derivative holds the dubious crown for both practical use and regulatory scrutiny. Major chemical reaction pathways revolve around either neutralization for waste management or attempted detoxification after accidental exposure.
Labeling Through the Years: Synonyms and Nomenclature
Sodium fluoroacetate travels the scientific literature under several guises. “Compound 1080” stands as its most famous pseudonym, embedding itself into safety manuals and legal literature worldwide. Others use “1080 powder,” or, in regulatory listings, simply “fluoroacetate, sodium salt.” The variance in naming comes from military, agricultural, and academic sources, reflecting decades of research, debate, and practical application.
Working Standards: Safety and Operation
Unlike household chemicals, sodium fluoroacetate earns its reputation among the most strictly controlled substances on any industrial site. Researchers and pest control professionals suit up in gloves, goggles, and respirators, often operating in ventilated chemical hoods. Legal standards restrict access, mandate training, and sometimes ban the product outright. Spill protocols involve immediate evacuation, absorbent neutralization, and in certain cases, hazmat team response. Disposal does not cross into municipal waste—it flows through incineration at high temperatures or chemical breakdown under regulatory supervision. In countries like Australia, 1080 baits must be dyed and flavored to repel non-target species, tying safety to every stage from transport to application. Ongoing national debates challenge whether chemical tools like this do more harm than good, pushing for greater transparency, contingency planning, and public education.
Environmental and Industrial Applications
On paper, sodium fluoroacetate answers a perennial challenge: controlling mammals that threaten crops or native animals. Ranchers and conservationists deploy it with deliberation against foxes, wild dogs, rats, and invasive species in landscapes where other interventions fall short. The goal isn’t convenience, but necessity—in heavily modified ecosystems, chemical management has emerged as a last guard for biodiversity. Some reports cite dramatic population drops in targeted pests, protecting native birds and mammals. At the same time, critics document heartbreaking by-catch, as pets and non-target wildlife sometimes fall victim, raising questions about ethics, oversight, and alternatives. Where it matters most, voices from farmers and ecologists call for research-backed, carefully bounded, and constantly reviewed applications. Subtle shifts in regulation and technology evolve strategy, pointing toward more humane or ecologically sensitive pest control systems.
Pushing Science Forward: Research and Development
Curiosity frames much of the ongoing research. Chemists probe the fine details of metabolism, looking for ways to block or reverse the toxic effects inside the body. Biotechnologists have mapped antidotes for fluoroacetate exposure, though with mixed success. Ecologists track breakdown products and ecosystem outcomes, testing new formulations that limit risks to native wildlife. Laboratories refine delivery methods—less palatable baits, improved targeting, and “smart” systems that minimize suffering and exposure. Geneticists investigate resistance in target species, while regulatory scientists model community impacts and disease risks. Across every field, transparency and open data improve outcomes, with an eye toward better dialogue between science, public agencies, and affected communities.
The Grim Chemistry: Ongoing Toxicity Studies
Some chemicals announce their hazards with warning smells or bitter tastes. Sodium fluoroacetate offers none of that—its danger unfolds quietly, blocking the Krebs cycle in mammals. Even tiny doses prove lethal, manifesting as seizures and respiratory collapse in animals and people alike. In Australia alone, accidental poisonings of farm dogs drive layers of regulation and education. Toxicity tests on rodents and livestock confirm minute lethal doses compared to other compounds in routine use. Researchers map the substance’s journey through soil, water, and food chains, gauging both immediate risks and lingering residues. Scenarios of wildlife, pet, or even human exposure keep toxicologists pressing for improved safety nets, robust antidotes, and clearer on-the-ground protocols. With so much at stake, chemistry operates not in a vacuum but alongside strong frameworks for training, monitoring, and redress.
Looking Ahead: New Prospects and Better Solutions
No one in conservation or agriculture settles for business as usual. Sodium fluoroacetate’s story does not end with its introduction decades ago. Researchers visualize new stains, tracking agents that allow baits to self-identify predators, and even gene drives that promise population-level management without repeat chemical dosing. Biodegradable matrices lower the risk to non-target organisms, while scientists pursue less toxic, more selective pesticides. Integrated pest management—combining habitat restoration, fencing, and community outreach—shifts the focus away from broad-spectrum poisons. Regulatory bodies revisit risk assessments as scientists learn more about persistence in waterways and food webs. Public consultation amplifies the voices of indigenous communities, farmers, pet owners, and conservationists, leading to evolving policy. New generations of learners inherit both the hopes and the cautionary tales of sodium fluoroacetate, ensuring its lessons shape the balance between technological power and ecological responsibility.
What is sodium fluoroacetate used for?
The Main Use: Pest Control in Rural Areas
Sodium fluoroacetate, often called 1080, pops up most in conversations about pest control, especially in places where livestock and native wildlife need protection. Australia and New Zealand use it more than anywhere else. Farmers and government agencies rely on this compound to keep invasive animals—like foxes, wild dogs, and possums—from damaging crops, hurting livestock, or pushing native species toward extinction. This bait gets placed out in pellets, carrots, or even gel capsules. Animals eating the bait stop producing energy at the cellular level. Death is certain and, unfortunately, drawn out.
Why Some See It as a Necessary Tool
I've watched how destructive invasive pests can get. Foxes destroy lambs in the night. Possums wreck bird nests and eat baby birds. You walk into a forest that should be loud, but it's silent except for the wind. Entire native bird populations have disappeared in New Zealand because of stoats and possums, which didn’t exist there before people arrived. For ranchers, losing lambs to wild dogs wrecks confidence and pockets. Desperate for a fix, landowners often don’t see another option besides tough poisons like 1080.
Controversy: Safety, Ethics, and the Bigger Picture
Risk always comes in as a huge concern. 1080 isn’t just deadly for pests—it's just as dangerous for dogs and other mammals, including pets and working dogs. Dogs die yearly in the outback from swallowing or even sniffing a piece of tainted meat. No antidote exists for accidental poisoning. Vets can only try to flush the poison quickly, but the odds are never good.
Another issue involves environmental impact. The chemical breaks down quickly in water, but not all non-target species stay clear of bait. Some folks worry about birds, reptiles, or even livestock picking up the poison. Waterways in treated areas get monitored to avoid contaminated drinking water, but mistakes happen.
People debate the ethics, too. Watching a dog or wild animal die from 1080 isn’t quick or painless. Some animal welfare groups claim this kind of poison causes needless suffering. I’ve seen local communities split over aerial bait drops—farmers see protection, others see cruelty. The push and pull between conservation and animal welfare lights up social media feeds whenever a new poison drop gets planned.
What Can Be Done: Seeking Alternatives and Better Methods
Science keeps looking for better solutions. Genetic research and fertility control may offer hope far ahead, but not in most farmers' lifetimes. Some research goes into creating targeted baits that only attract pests using scent and flavor cues that native animals ignore. Even then, nature tends to surprise.
Old-school methods like fencing and trapping can take the edge off pest numbers, but they struggle to keep up in big, rugged backcountry. Aerial drops hit thousands of acres that trappers and shooters could never cover on foot. Some regions run campaigns warning hikers and dog owners to stay away during drop seasons.
Communities deserve open conversations about trade-offs. Experts from ecology, farming, and animal welfare bring smart perspectives. Keeping the public looped in about risks, plans, and research wins trust, even if a perfect solution doesn’t exist yet. If alternatives get tested and investment keeps flowing to new ideas, maybe someday we’ll see the end of cruel, broad-spectrum poisons—without losing native wildlife or the livelihoods of those who depend on the land.
Is sodium fluoroacetate toxic to humans and animals?
What Is Sodium Fluoroacetate?
Sodium fluoroacetate, known as 1080, comes up in wildlife management and pest control. It’s a chemical that packs a punch. Producers use it to tackle invasive species—mostly pests that damage crops or native animals. The simple truth is that this stuff can cause harm not only to targeted critters but also to the ones people want to protect, including pets, livestock, and even ourselves.
The Science Behind the Toxicity
It’s not just another harsh-sounding chemical. Sodium fluoroacetate messes with the way living things generate energy. Cells try converting food to fuel, but 1080 hijacks this system, causing a build-up of toxic substances and eventually shutting the body down. All mammals, birds, and even some insects can be affected. Humans, pets, and livestock catch a dose of poison right alongside pests if they’re unlucky enough to eat it—or eat something that has.
Human Risk is Real
Accidental poisoning isn’t science fiction. Reports from Australia, New Zealand, and elsewhere show people have stumbled across baits in the wild or mishandled the chemical unintentionally. 1080 doesn’t just wash away after a spring rain, either. That persistence means long-term caution gets woven into any plan using it. Symptoms in humans range from headaches and nausea to seizures and, in serious cases, death. For a parent, a dog owner, or a rural neighbor, this knowledge lives in the back of the mind.
Pets and Livestock Are Often Victims
Farmers and ranchers can list stories about lost sheep or dogs. Some farmers I’ve talked with won’t let their working dogs near areas where 1080 baits have gone out. One Australian vet described losing two farm dogs within hours after a stray bait was eaten. Studies back this up: even trace exposures can poison non-target animals; there is no specific antidote. Everything rests on quick action, which is tough in a remote pasture or forest.
Is There a Justification?
In some parts of the world, native wildlife face extinction because imported predators wreak havoc on eggs and nests. Conservation groups sometimes argue there’s no better option right now. Yet weighing convenience against risk can’t erase tragic consequences for families or whole farms. Recent court cases in New Zealand show just how passionate both sides of the debate stay, especially as communities push for safer alternatives.
Searching for Alternatives
Research into safer, more precise solutions continues. Some scientists engineer pest-specific baits, or work on genetic tools to reduce animal populations without poisoning everything in reach. Advocates and land managers highlight education—clearer signs, locked baits, stricter control of distribution—to shrink the odds of non-target deaths. Personal experience teaches me that prevention really starts long before anyone puts out a single dose: locals should be warned, supplies must stay locked, and pets kept far from risky ground.
The Case for Transparency and Listening
People who live and work close to baiting areas want their concerns answered, not brushed aside with technical language. The public deserves plain discussion about the effects, both intended and not. Only with open debate, strong safety checks, and solid new research can communities make informed calls about what risks they can accept. At its core, this is about keeping people, pets, wildlife, and food supplies safe—because a mistake with sodium fluoroacetate is one lesson best learned from others, rather than the hard way.
How does sodium fluoroacetate work as a poison?
How It Affects the Body
Sodium fluoroacetate, often called “1080”, causes trouble in a sneaky way. It slips into the body and blocks a basic function inside cells that lets them turn food into energy. Once swallowed or absorbed, the compound combines with natural chemicals to form fluoroacetyl-CoA. This trickster then slips into the process that breaks down sugars, known as the Krebs cycle, and jams up the machinery. The process stops at a roadblock called fluorocitrate, which ends up halting energy production. Organs that burn a lot of fuel for their work—brains and hearts—take the hardest hit.
That sudden energy shortage hits animals fast. Rats, possums, and even bigger creatures like dogs start staggering, shivering, or panting. Within hours, convulsions, paralysis and, often, collapse set in. Death usually follows within a day. People, on rare occasions, have been poisoned too. Symptoms show up after a few hours: nausea, confusion, and trouble breathing. Modern medicine has no magic cure for this poisoning. Doctors can pump stomachs and try to flush out the chemical, but they mostly have to support failing hearts and help people breathe.
Why This Is a Real-World Problem
Sodium fluoroacetate has made headlines, not because it’s common in everyday life, but because land managers use it to control pests such as invasive possums, foxes, or rabbits. It works well against animals plaguing farms and wild spaces, but its power has a big downside. It doesn’t choose its victims—native wildlife, farm dogs, and other unintended animals can die if they eat poisoned bait. Critics call the approach reckless, warning about harm to biodiversity. Supporters point to fewer invasive animals attacking crops, rare birds, or grazing land. Nobody wins outright. Communities around the world still argue about the value and risks.
My own experience with pest control campaigns in rural areas proved just how polarizing this tool has become. Farmers desperate to protect herds see it as the only option. Conservationists fear losing precious, endangered birds. Pet owners live in dread after hearing stories about dogs dying from half-eaten bait. Once the chemical gets into a local food web, tracking its movement turns complicated. Even scavengers and insects sometimes pick up traces. Careless handling spreads danger further.
Looking for Solutions
A safer world means fewer broad-brush poisons lying around. Some researchers keep looking for antidotes, but progress stays slow. I’ve watched new bait designs use special coverings or scents to attract only the target pests. These tricks work on a small scale—sometimes native creatures do avoid them, sometimes not. More effective fencing, habitat restoration, or targeted hunting help too, but cost more in time and money. Nobody’s come up with a simple answer.
Teaching local communities—especially kids—about the risks makes a difference. I’ve seen schools in farming districts run classes on what poisoned bait looks like and who to call if an animal eats something suspicious. Spreading that kind of practical knowledge saves lives, both animal and human. Honest labeling, strong regulations, and keeping chemicals away from curious hands matters. The debate around sodium fluoroacetate won’t disappear anytime soon, given the tightrope walk between protecting farms and sparing nature. But voices from science, local knowledge, and real experience add needed light to every decision made.
What are the safety precautions for handling sodium fluoroacetate?
A Chemical That Demands Respect
Sodium fluoroacetate turns most people cautious the moment its name pops up in a safety meeting. It’s not some household cleaner you left under the sink; it’s a poison with a history in pest control, known for being effective and, frankly, unforgiving if things go wrong. My first lesson about this chemical didn’t come from a textbook, but from a wrinkled supervisor with stories about near misses and clear rules burned into memory.
Why the Handling Rules Matter
Sodium fluoroacetate interrupts cell metabolism—the reason it’s so dangerous. A spill on your skin or, worse, a bit getting airborne and breathed in, doesn’t just fade away. The body doesn’t have an easy way to deal with it, so small accidents can take a big toll. Anyone touching the stuff owes it to themselves, and everyone around, to approach it with a certain humility. Utah Poison Control warns that even tiny doses bring on heart and nervous system symptoms, and there’s no straight-up antidote. Hospitals focus on symptoms, not reversal, so there’s no shortcut back if someone's been exposed.
Personal Protective Equipment: Non-Negotiable
I always remember the fuss over goggles and gloves, and there’s no overstating their value with sodium fluoroacetate. Nitrile gloves hold up better than latex, and lab coats or overalls keep clothing from soaking up any splashes. Respirators step in where powders or dust fly around, and I’ve never regretted pulling on a proper mask even if it felt overkill at the time. One slip without it could mean a trip to the ER, not just a scolding.
Workspaces That Don’t Cut Corners
Good safety with this chemical comes from workspaces built for the job. Closed systems that cap off fumes or dust, eye-washing stations right at arm’s reach, and warning signs for anyone not cleared linger in my mind after visiting tight, well-run pest control labs. Double-bagging leftovers and sealing every container isn’t overkill. Ventilation pulls any stray particulate out of the room, instead of into someone’s lungs. That extra layer pays off in peace of mind.
Training and Preparedness Make the Difference
The plain fact is, reading the safety sheet once and shoving it in a drawer leaves everyone vulnerable. Companies with real safety records don’t settle for an one-off demonstration; they circle back every season, walk everyone through spill drills, and check expiration dates on emergency kits. Emergency numbers go on walls, not just in binders. Mock drills may feel overdone, until real worry grabs hold during a scare and everyone moves in sync because they practiced. In my time, I’ve watched quick action make a difference after a tear in someone’s glove. It’s a team effort, not a solo show.
Solutions That Work in the Real World
Switching to bait stations or formulations that reduce dust can take the edge off some risks. Engineers and chemists I’ve met stay on the hunt for less hazardous alternatives—sometimes finding promising leads, other times admitting sodium fluoroacetate’s effectiveness is tough to match. Until the day someone cracks the code, those handling it need regular health checkups and solid support from management. The weakest link is always overconfidence, so a healthy respect and a willingness to speak up about safety rules go further than any one person’s experience ever can.
The lessons boil down to something simple: no shortcut is worth the cost with sodium fluoroacetate, not for yourself or anyone nearby. Smart handling pays off every shift, every time.
Is sodium fluoroacetate banned or regulated in any countries?
The Controversy Around Sodium Fluoroacetate
Ask anyone who’s dealt with pest control in Australia or New Zealand about “1080,” and the stories range from effective rabbit management to heated protests over water contamination. Sodium fluoroacetate, known mostly by its code number 1080, sparks debate from environmentalists, farmers, and public health advocates alike.
The Real-World Impact
I grew up among sheep stations dotted across the New Zealand high country, where conservation rangers sometimes relied on 1080 baits to tackle invasive possums and rabbits. With no native land mammals in New Zealand, a single possum can devastate native forests, so I witnessed wary support for 1080 from people who cared deeply for nature. But it stuck out that regular folks never felt settled about the risks. Even if the poison works against pests, the fear of poisoning dogs, livestock, or native birds never died down. Every time a hunting club dog wandered near an active drop zone, locals braced for bad news.
Across the world, sodium fluoroacetate belongs to a unique class of poisons with no simple antidote. The tiniest dose can kill a large mammal. This background has driven different countries to take widely different stances.
Bans Versus Controls: Patchwork Approaches
Sodium fluoroacetate remains legal in both Australia and New Zealand—though under strict controls. Aerial drops get monitored, and signs go up to warn the public in drop zones. In contrast, countries in Europe, as well as the United States and Canada, mostly turn their backs on this poison. It lost favor in the US back in the 1970s after public outcry over accidental wildlife deaths and water contamination. The Environmental Protection Agency restricted almost all its use, leaving minor exceptions for experimental or emergency purposes.
In Germany, Austria, and a string of other European countries, sodium fluoroacetate sits on lists of banned substances completely. Some bans reach back to the late 1960s, spurred by both wildlife protection movements and a lack of faith in truly safe application methods. Literature from the European Food Safety Authority highlights the risks: accidental poisoning of non-target animals, impossible removal from drinking water, and human health incidents among rural communities.
The Value of Local Solutions
Australia stands as an outlier because of its devastating record with introduced foxes and wild dogs. Farmers in Western Australia, New South Wales, and Queensland rely on 1080 because little else checks the numbers of hungry predators that threaten sheep flocks and small marsupials. Regulations demand tight controls on where and how the poison gets used, but critics say contaminated carcasses harm wedge-tailed eagles and dingoes alike. Indigenous communities have raised alarms around waterway safety, and news of pet deaths, despite warning signs, erodes public trust.
Charting a Smarter Course Forward
Locally adapted pest control matters, but real faith in these poisons only grows alongside strong safeguards and frank engagement with communities. Seeing the backlash up close, I learned that listening to residents—especially those who rely on wild lands for food or leisure—tends to yield more effective management. Investment in research on non-lethal methods, predator-proof fencing, and targeted camera traps provides families an alternative that doesn’t pit conservation against rural livelihoods.
Bans didn’t arrive lightly in most countries; they came after hard lessons about how difficult it is to contain such potent poisons. Where sodium fluoroacetate use persists, ongoing investment in best practices, community warning systems, and poison-free pest-management alternatives feels more urgent every year.
| Names | |
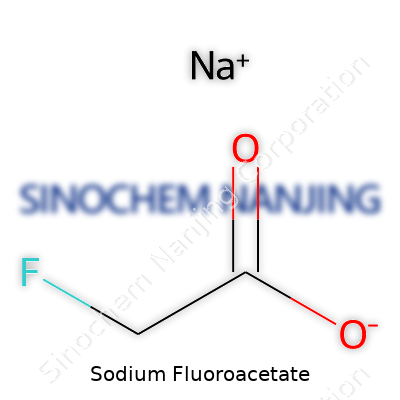
| Preferred IUPAC name | Sodium 2-fluoroacetate |
| Other names |
Compound 1080 1080 fluoroacetic acid sodium salt sodium monofluoroacetate sodium fluoroacetate (anhydrous) sodium monofluoroacetate (anhydrous) |
| Pronunciation | /ˌsəʊdiəm flʊəroʊˈæsɪteɪt/ |
| Identifiers | |
| CAS Number | 62-74-8 |
| Beilstein Reference | 1906947 |
| ChEBI | CHEBI:39021 |
| ChEMBL | CHEMBL1896 |
| ChemSpider | 8716 |
| DrugBank | DB01361 |
| ECHA InfoCard | ECHA InfoCard: 027-004-00-6 |
| EC Number | 206-197-5 |
| Gmelin Reference | 8838 |
| KEGG | C14314 |
| MeSH | D013406 |
| PubChem CID | 9547 |
| RTECS number | XW8225000 |
| UNII | 4UF4043G9V |
| UN number | UN0179 |
| Properties | |
| Chemical formula | C2H2FO2Na |
| Molar mass | 100.04 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | Density: 1.325 g/cm³ |
| Solubility in water | Soluble |
| log P | -0.3 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 2.6 |
| Basicity (pKb) | 9.42 |
| Magnetic susceptibility (χ) | -46.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.333 |
| Dipole moment | **2.06 D** |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 117 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -702.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –796.2 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V1CA01 |
| Hazards | |
| Main hazards | Fatal if swallowed. |
| GHS labelling | GHS06, GHS08 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H300: Fatal if swallowed. |
| Precautionary statements | P260, P262, P264, P270, P273, P280, P284, P301+P310, P304+P340, P310, P320, P330, P361+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-2 |
| Autoignition temperature | 540 °C |
| Lethal dose or concentration | LD50 (oral, rat): 0.2 mg/kg |
| LD50 (median dose) | 0.7–2 mg/kg (oral, rat) |
| NIOSH | UR8225000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Sodium Fluoroacetate: 0.05 mg/m³ |
| REL (Recommended) | 0.2 mg/m3 |
| IDLH (Immediate danger) | 0.1 mg/m3 |
| Related compounds | |
| Related compounds |
Methyl fluoroacetate Fluoroacetamide Chloroacetate Bromoacetate Monofluoroacetic acid |

