Sodium Dimethylarsonate: Tracing Its Journey From Lab Bench to Real-World Impact
Historical Development
Sodium dimethylarsonate stands out as a product of both necessity and scientific ambition. Decades ago, researchers started searching for compounds capable of managing problems in agriculture and forestry where traditional solutions came up short. The pursuit of organoarsenic chemistry found fertile ground with the need for more effective herbicides and growth regulators. Scientists discovered new ways to bind arsenic to organic groups, hoping to tame its infamous toxicity and reap practical benefits. As scientists began to understand that tiny tweaks to chemical structure could change how a substance behaves, sodium dimethylarsonate became one of those stories: a chemical born from experiments and shaped by both laboratory curiosity and commercial pressure. Even today, its story runs parallel to discussions about balancing innovation with safety and sustainability.
Product Overview
This compound appears as a white, crystalline solid, soluble in water and often packed in bags that keep out moisture. Formulators rely on sodium dimethylarsonate thanks to properties that make it stable under normal handling—unlike some of its more reactive relatives. This chemical earned its place as a specialty herbicide, especially where plants or weeds proved resistant to other management methods. It’s not a garden-variety product, but gets frequent attention from experts in turf management and agriculture, packed into solutions, granules, or powders, depending on the application.
Physical & Chemical Properties
In the lab, sodium dimethylarsonate doesn’t look particularly flashy, but its impact lies in its composition. Each molecule consists of arsenic, oxygen, and methyl groups paired with a sodium ion, which affects how the compound dissolves and interacts in soils or water. It handles well under a range of temperatures, and it rarely surprises its handlers with violent reactions under normal conditions. Its structure creates a stability that’s attractive but also underlines the reason policymakers and scientists continue to keep a close eye on this class of materials: arsenic can be both tool and threat.
Technical Specifications & Labeling
Technical details do matter. Producers specify sodium dimethylarsonate content by percentage, ensuring its purity falls within narrow limits so users can predict how it will perform in the field. Labels often carry more than just composition—they include warnings about handling to protect both workers and the environment, mirroring growing community concern around chemical safety. Agriculture professionals and distributors push for transparent information so the field crews and regulators never have to guess what’s in the package.
Preparation Method
Chemists synthesize sodium dimethylarsonate by reacting dimethylarsinic acid with sodium hydroxide, a process that demands care from start to finish. Each step involves measuring, temperature control, and filtration to remove impurities. Experience tells that even minor changes during these reactions introduce impurities or reduce yield, so manufacturers build in checkpoints. Unlike bulk chemicals where impurity doesn’t make much difference, sodium dimethylarsonate’s users depend on predictability—anything less can affect results in surprising ways, or even create new safety risks.
Chemical Reactions & Modifications
After manufacture, researchers have tinkered with chemical modifications and analogs to see if safer or more effective versions could be developed. Sodium dimethylarsonate reacts under certain conditions, particularly with strong acids or oxidizing agents. These reactions in the environment influence how long the material persists in soil and whether it breaks down into other arsenic compounds. As technology advances, that old familiar question comes up again and again: can a tweak minimize risk or unlock new possibility? Laboratories across the world continue to explore this territory, sometimes stumbling onto something novel, sometimes facing setbacks.
Synonyms & Product Names
This compound masquerades under several names, which sometimes tricks those unfamiliar with chemical jargon. Terms like DSMA, disodium methanearsonate, and dimethylarsinic acid sodium salt overlap with trade names that have lived long lives in catalogs and on safety reports. The use of these names arose from historical conventions as much as chemical logic, and keeping track is a challenge for anyone digging through decades of regulatory or scientific papers. This naming jumble is more than a nuisance; it drives misunderstandings in cross-border discussions about use and restriction.
Safety & Operational Standards
Hard lessons from the past mold today’s rules on handling and application. Regulatory frameworks across North America, Europe, and Asia continue to grow stricter as toxicology advances. Sodium dimethylarsonate, given its arsenic backbone, falls under dangerous goods classifications in many regions. Operators don personal protective equipment, stick to prescribed application rates, and follow local restrictions. Waste disposal is tightly controlled, not only to protect individual users but also to prevent environmental contamination. Companies train their personnel regularly, update procedures after every incident, and invest in monitoring air, soil, and water—covering every angle because history taught the industry what can go wrong if care slips.
Application Area
Most folks who encounter sodium dimethylarsonate do so out in the fields or on golf courses. Dealing with stubborn weeds remains its claim to fame, and managers of green belts or sports facilities saw it as a solution during years when alternative herbicides failed. Governments now carefully weigh environmental value against weed control, sometimes dialing back use depending on soil sensitivity, groundwater risks, or wildlife needs. Even so, some specialists swear by its targeted action where other products have underperformed, arguing that options remain limited for some hard-to-kill species.
Research & Development
Ongoing experiments scrutinize every aspect of sodium dimethylarsonate’s behavior—from how fast it moves through soil to what trace residues remain in harvests and water tables. Institutions and independent scientists test alternative application methods, hoping to harness benefits while cutting exposure. New research investigates how plants metabolize or resist this and similar chemicals, informing next-generation weed management. Meanwhile, digital agriculture platforms layered with AI and precision monitoring supply new tools for reducing overall chemical footprints, promising to draw sharper lines between problem-solving and creating fresh risk.
Toxicity Research
Public trust turns on clarity about risk—a lesson drilled home by tragedies tied to unregulated use of arsenic in past centuries. Comprehensive studies show sodium dimethylarsonate delivers acute and chronic risks if people don’t respect its directions. Specialists monitor its presence in soil and runoff, measure residues on edible crops, and keep a running tally of arsenic uptake into animal and human food chains. Resulting data directly shape policy, not only on limits for agricultural use but also on site remediation and cleanup. Despite the inherent risk, careful management and decades of toxicological research limit adverse outcomes, although emerging studies continue to probe for subtle health and ecosystem effects missed by earlier generations of scientists.
Future Prospects
Looking forward, the future of sodium dimethylarsonate will hinge on how effectively science, industry, and regulators respond to new evidence of both risk and opportunity. As sustainable agriculture transforms into a priority, pressure on organoarsenic compounds will only intensify. Some innovators pour resources into nonchemical weed control, biological agents, or smarter technologies that cut down use of legacy products. Yet global need for efficient food production and land management means sodium dimethylarsonate’s journey is not ready to end just yet. Finding the right role for it demands constant vigilance: weighing benefits, monitoring impacts, and ensuring every batch produced meets requirements laid down by both regulation and public expectation. Combining historical lessons, cutting-edge research, and practical wisdom can chart a path that reaps agricultural reward without passing a toxic burden to the next generation.
What is Sodium Dimethylarsonate used for?
A Closer Look at a Controversial Chemical
Sodium dimethylarsonate pops up mostly in stories about farming. This compound fits into a group called organoarsenicals—arsenic paired up with carbon. Long before concerned parents checked food labels and organic markets took off, farmers reached for whatever would protect crops. That’s how this chemical got its place in agriculture.
People turned to sodium dimethylarsonate for weed control. Weeds choke out crops. If you’ve ever tried growing a few tomatoes and lost half your plants to pigweed or crabgrass, you understand the draw of strong herbicides. Unlike some alternatives that washed away with rain, this one stuck around, making it tough for weeds to bounce back. Cotton and rice fields were especially common sites for its use. Arsenic-based weed killers could work on stubborn weeds that ignored regular methods, so fields kept looking clean and yields stayed high.
Health and Environmental Shadows
Arsenic sounds more at home in crime novels than in a tank used for spraying plants. That's the rub: sodium dimethylarsonate stood out for weed control but raised eyebrows for safety. Arsenic, even in organic form, collects in soil. Not long after fields received repeated applications, researchers started spotting higher levels in groundwater and crops. Drinking water in certain farm communities sometimes pushed past recommended arsenic limits. Cases of skin irritation, digestive troubles, and long-term disease, including some cancers, grew more common in areas where this kind of product saw heavy use. The World Health Organization and local health agencies joined the conversation, pressing for limits and alternatives.
As someone who grew up near cotton country, I’ve heard older farmers talk about “lead arsenate days” with a sort of grim nostalgia. They wanted easy weed control but often worried about wells turning bitter, fruit trees withering, or kids feeling unwell after playing in runoff water. These worries didn't just stay in the café—science started backing it up. The U.S. Environmental Protection Agency flagged sodium dimethylarsonate, listing it as a restricted-use pesticide. Other countries banned it outright or set tighter controls.
New Paths for Farming and Public Safety
Cotton and rice growers can’t just pull up and walk away from their land. Farmers began shifting tactics, learning about crop rotation, cover crops, and non-arsenic herbicides. Some switched to methods that manage weeds without chemicals: mechanical cultivation, smarter planting schedules, or biological controls.
Research keeps moving forward. Universities put time into finding weed killers that break down faster and don't linger in the soil or water. Testing for arsenic in produce and water grew much more common. In my own life, getting involved in local food co-ops made the idea personal—people care about how their dinner is grown and where their water comes from. It’s no longer just about killing weeds; it's about weighing the health of soil, water, and communities.
Sodium dimethylarsonate tells a complicated story of progress and setbacks in agriculture. A solution for one problem led to others. The lesson stands clear: tools for modern farming carry big consequences, and balancing human needs with environmental realities takes more than just finding the next quick fix.
Is Sodium Dimethylarsonate toxic or hazardous?
What Is Sodium Dimethylarsonate?
Sodium dimethylarsonate, usually called DSMA, comes up most often in agriculture. Its use as a herbicide dates back decades. The compound’s main purpose is to control weeds, particularly in cotton, turf, and some crops. DSMA belongs to the family of organoarsenicals, tools once promoted as alternatives to the more toxic inorganic arsenic products. As with anything carrying arsenic in its chemical makeup, questions pop up about its safety.
Health Concerns Linked to Arsenic Compounds
Stories about arsenic poisoning fill history books. Chronic exposure puts people at risk of a long list of health issues: skin lesions, cardiovascular problems, and increased risks of several cancers. Arsenic has come into cancer research discussions for decades. The U.S. Environmental Protection Agency (EPA) and the International Agency for Research on Cancer (IARC) both place arsenic compounds in their top carcinogen categories. This concerns anyone working with substances containing arsenic, even those rebranded as “less harmful.”
Toxicity and Environmental Impact of DSMA
Scientists have looked at DSMA’s toxicity with care. Animal studies show DSMA gets absorbed when swallowed or when it makes contact with skin. The body breaks it down and eventually excretes it through urine. Exposure at high enough levels causes headaches, nausea, diarrhea, muscle pain, and, in rare cases, liver or kidney damage. Over long periods, risks climb—especially for farm workers and communities near treated fields. One big issue is that DSMA breaks down into inorganic arsenic in the environment, the type well known for poisoning groundwater, crops, and soil.
Arsenic in water or food often goes undetected without proper testing. Even at low levels, it builds up in the body. The EPA’s drinking water limit for arsenic stands at 10 parts per billion—a measure reflecting years of research on what keeps communities safe. Yet, surface runoff from farms using DSMA helps arsenic seep into streams. The risk doesn’t stop at the farm gate. Birds, fish, and frogs mistake contaminated water for safe drinking water with no way to tell the difference. Studies show arsenic can make its way up the food chain, posing ongoing concerns for public health.
Regulation and Safer Alternatives
In the United States, use of DSMA faces tight controls. California banned the sale of most arsenic-based herbicides, and other states have restrictions. The EPA reviews these compounds every few years. Sometimes they cancel registrations outright if new evidence suggests bigger dangers or if companies can’t address environmental risks. Weed control strategies have shifted over the years. Integrated pest management programs and less persistent chemicals have stepped in where organoarsenicals used to dominate.
Certain crops still attract DSMA applications because few options work as well, especially with herbicide-resistant weeds. But chemical companies, extension agents, and researchers push for safer formulas whenever possible. Rotating crops, using mechanical removal, and improving soil health keep weed problems in check with much less risk to people and the planet.
Perspective From the Ground
Years working on and visiting farms taught me something about chemical hazards: the risks go far beyond labels. Some farmers remember dusting fields with DSMA with none of the modern safety gear anyone expects today. The smell and taste lingered after work, and the idea of residues drifting into a neighbor’s creek or a child’s sandbox didn’t cross anyone’s mind. It takes vigilance, regular soil and water tests, and a willingness to invest in safer solutions to keep arsenic risks low.
What is the chemical formula of Sodium Dimethylarsonate?
Digging Into the Roots: What Is Sodium Dimethylarsonate?
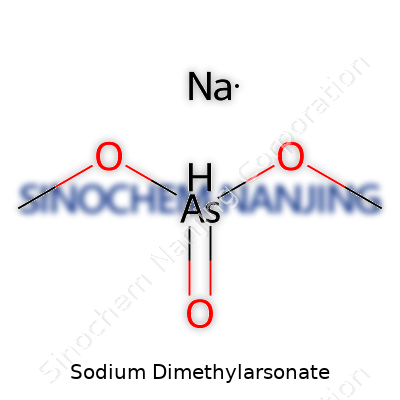
Sodium dimethylarsonate isn’t some stranger to those who work in fields like chemistry or agriculture. Its formula, CH6AsNaO4, packs more meaning than a string of letters and numbers. A breakdown reveals carbon, hydrogen, arsenic, sodium, and oxygen, fitting together to produce a salt used for more than textbook examples.
Why the Formula Matters
In labs, a correct formula means the difference between success and a batch wasted. Researchers value sodium dimethylarsonate for the arsenic atom it carries. While arsenic can't be called a friend to human health in high amounts, its chemistry brings benefits when handled right. Synthetic chemists look at its arrangement—two methyl groups attached to arsenic, which links to a sodium salt. This structure determines how the substance dissolves, reacts, and, crucially, interacts with other molecules.
Agriculture’s Past With Arsenic Compounds
Someone raising crops a few decades ago would have known this chemical through its historical use as a herbicide. In those days, sodium dimethylarsonate often went by the name "cacodylate," battling weeds on golf courses, rights-of-way, and in cotton farming. Smart use meant following precise formulas and application rates. The formula, CH6AsNaO4, helped companies design safer labels and recommendations.
Stories from retired agronomists often include respect for the power in these compounds—plenty saw risks firsthand. Prolonged misuse led to concerns about environmental build-up, showing how once-accepted chemicals gained scrutiny. Both soil and groundwater contamination turned into real issues, especially in regions with heavy arsenic herbicide use. Residents near treated land faced health concerns that drew attention from regulators.
Modern Perspective—Knowledge Shapes Better Practice
These days, labs and public safety boards look hard at any material that contains arsenic. The molecular formula guides hazard evaluation: a sodium salt like this one dissolves easily, increasing the risk of transport in water. The right formula lets environmental scientists trace the source of contamination or track chemical pathways. Work experience has shown me how formulas, too, become tools for public safety and clean-up crews. Testing programs focus on regions where residues build up, using knowledge of structure and solubility.
Removal from wide-scale use doesn't erase the need for understanding. Old stockpiles can linger. Sometimes, property transfers bring up questions about legacy contamination. Those details demand professionals who know not just the name, but the essential nature that CH6AsNaO4 describes. This awareness protects new generations, especially where water sampling and land reclamation are constant efforts.
The Path Forward
Technical facts matter. Respect for the formula of sodium dimethylarsonate makes a difference in real-world outcomes. Wherever people encounter legacy arsenic compounds, precision in knowledge acts as a safeguard. Doing soil tests, remediating contaminated ground, or evaluating imported herbicides gets easier with accurate chemical information. Sharing this kind of experience—and talking openly about formula, risk, and safe handling—helps fix old mistakes and keeps today’s users from repeating them.
How should Sodium Dimethylarsonate be stored and handled?
Understanding the Real Risks
Many people glance at a bag of sodium dimethylarsonate and don't realize just how hazardous it can be. Coming from a farming background, I remember working with chemicals where a little mishap could lead to big problems. Sodium dimethylarsonate isn't something ordinary folks regularly encounter, but its dangers cannot be understated. As an arsenic-containing compound, this chemical can threaten both human health and the environment if handled or stored carelessly.
Practical Steps for Safe Storage
Every good operator begins by keeping chemicals in their original containers, sealed tight to avoid leaks or spills. A good storeroom offers solid shelves, dry air, and plenty of space. Stacking containers too closely can spell trouble. Humidity triggers clumping, leaks, or worse, so a dehumidifier pays for itself over time. Stick a label with large, bold writing and clear hazard warnings on every container, so nobody pretends they didn't see it.
Never keep sodium dimethylarsonate near food, drinks, or common break spots. Store it away from heat and direct sunlight. Sparks, open flames, or simple static can bring out the worst in chemicals like this one. My mentor always taught me to separate incompatible substances—never stack pesticides with oxidizers or flammables. It's not about following a rulebook; it's about keeping people and property safe. Walls and floors built to resist spills and corrosion also reduce the odds of a small accident turning into a disaster.
Simple Habits When Handling
Wearing the right gear feels awkward at first, but a good respirator, gloves, goggles, and a protective apron will save a lot of heartache. This isn't just about following regulations. Over the years, I've seen skin burns and respiratory problems from those who got careless or trusted cheap gloves. Never touch your face, eyes, or mouth while working.
Work in a well-ventilated spot with good airflow. Ventilation fans should point away from break rooms, offices, or livestock pens. Anyone handling sodium dimethylarsonate needs access to running water, eye wash stations, and emergency showers. If powder spills, sweep gently into sealable containers rather than hosing it down. Water can spread the chemical, seeping into the ground and risking contamination.
Why This Matters
Sodium dimethylarsonate doesn’t just disappear after use; it lingers in soil, water, and even dust. Exposure can cause symptoms from headaches to more severe nerve and organ damage. Protecting workers and neighbors starts with clear procedures, regular reviews, and open communication. Training isn't about filling out a checklist. It's about making the dangers real so mistakes feel less likely. Younger workers, in particular, learn best from direct stories and seeing what happens without proper caution.
Building a Safer System
Store inventory records, safety data sheets, and emergency contact numbers close to the entrance of the storeroom. Let local fire departments and medical responders know if you stock large quantities. In case of a spill, a written action plan beats improvising every time. Recycle or dispose of old containers and expired chemicals through licensed hazardous waste contractors.
Good storage and handling of sodium dimethylarsonate aren't just about ticking off legal boxes—they keep farms, workers, and communities safe. Every step taken now means fewer health and environmental headaches down the road.
What are the safety precautions when working with Sodium Dimethylarsonate?
Understand What You’re Handling
Working with chemicals like sodium dimethylarsonate calls for real respect. This compound has found its way into agriculture as a herbicide, but its true nature revolves around arsenic. That word alone should get attention. Arsenic doesn’t forgive sloppy habits. I’ve seen too many folks brush off safety briefings, and looking back, the luckiest outcome was only a ruined glove.
Personal Protective Gear is Non-Negotiable
Safety goggles, chemical-resistant gloves, and a sturdy lab coat earn their keep on any day sodium dimethylarsonate comes out of storage. My own reminder came during a summer internship. A chemist reached up high for a bottle, lost control, and ended up with powder dust on bare skin. The irritation followed for days, and there was a real fear about longer impacts. Proper gloves, long sleeves, and splash-proof goggles avoid these scares. No one wins by skipping gear – even a minute of exposure can leave a mark.
Keep Air Moving—Ventilation Matters
Many labs cut corners. Too many times, I’ve walked into rooms with faint, strange smells—an easy sign the fume hood isn’t on, or improper techniques have crept in. Sodium dimethylarsonate doesn’t belong in spaces with stagnant air. Always measure and transfer this compound inside a working fume hood. Those fans and filters aren’t just for show; arsenic compounds can linger in the air, and the lungs are the quickest route for trouble.
Clean-Up Starts Before Spills Happen
Preparation beats panic every time. Keep spill kits stocked with material designed for chemical absorption, not just a stack of old newspapers. I’ve watched an unprepared team scramble for solutions with a spreading pool of unknown chemical on the floor. Sodium dimethylarsonate doesn’t leave room for improvisation. Lock up food, drinks, and any personal items. Clear labeling and a logbook offering up-to-date chemical locations help others who step into your workspace on another shift.
Know the Consequences—Health Risks are Real
Direct contact with sodium dimethylarsonate means trouble for skin, eyes, and internal organs. Chronic exposure brings an even grimmer risk: arsenic poisoning doesn't announce itself until the damage is done. Symptoms like headaches, confusion, and abnormal skin changes creep up slowly and linger. According to the CDC, arsenic exposure increases the risk of cancer, respiratory issues, and nervous system problems. These aren’t “what ifs”—they show up in industry accident reports year after year.
Training and Teamwork Keep Everyone Safe
Chemical safety doesn’t live in a binder. Every worker must know emergency procedures, locate eyewash stations, and practice good hygiene after handling any hazardous chemical. After years in research and industrial labs, I’ve found that shared responsibility reduces mistakes—remind your crew to wash hands before touching anything outside the lab, especially before eating or using the restroom.
Disposal: More than “Throw it Away”
Hazardous waste rules forbid dumping arsenic compounds down the drain or into regular trash bins. I’ve seen serious fines for improper disposal—it hurts workers, neighbors, wildlife, and water sources. Accumulate waste in labeled, compatible containers and book prompt pickups with certified hazardous waste handlers. The environment isn’t just “out there”—everyone shares the consequences.
No Room for Carelessness
Sodium dimethylarsonate demands careful practices, teamwork, and an attitude that says, “not on my watch.” No one ever looks back after an accident and feels their shortcuts were worth it. Every glove, every label, every prompt clean-up, pays off in real safety—every day, for everyone.
| Names | |
| Preferred IUPAC name | Sodium methanearsonate |
| Other names |
Cacodylic acid Dimethylarsinic acid DMA Hydroxydimethylarsine oxide |
| Pronunciation | /ˌsoʊdiəm daɪˌmɛθɪlˈɑr.səˌneɪt/ |
| Identifiers | |
| CAS Number | 138-53-4 |
| Beilstein Reference | 1818738 |
| ChEBI | CHEBI:74854 |
| ChEMBL | CHEMBL285661 |
| ChemSpider | 17116 |
| DrugBank | DB01372 |
| ECHA InfoCard | 03b1e826-0e64-420a-82c8-72c3ebaf0e3c |
| EC Number | 214-939-5 |
| Gmelin Reference | 82882 |
| KEGG | C01255 |
| MeSH | D012542 |
| PubChem CID | 8551 |
| RTECS number | CN8110000 |
| UNII | D0TJT4NA8S |
| UN number | UN1437 |
| CompTox Dashboard (EPA) | DTXSID4024094 |
| Properties | |
| Chemical formula | C2H6AsNaO4 |
| Molar mass | 186.03 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 2.37 g/cm³ |
| Solubility in water | Soluble |
| log P | -4.48 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 8.5 |
| Basicity (pKb) | 11.55 |
| Magnetic susceptibility (χ) | -46.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.425 |
| Dipole moment | 4.29 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 185.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -995.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1299.4 kJ/mol |
| Pharmacology | |
| ATC code | V03AB04 |
| Hazards | |
| Main hazards | Toxic if swallowed. Causes skin and eye irritation. May cause respiratory irritation. Suspected of causing cancer. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS06, GHS08 |
| Signal word | Danger |
| Hazard statements | H301+H331: Toxic if swallowed or if inhaled. H373: May cause damage to organs through prolonged or repeated exposure. |
| Precautionary statements | P261, P264, P270, P271, P272, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P308+P310, P310, P330, P362+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-0-💀 |
| Autoignition temperature | Autoignition temperature: 690°C |
| Lethal dose or concentration | LD50 oral rat 765 mg/kg |
| LD50 (median dose) | LD50 (median dose): 700 mg/kg (oral, rat) |
| NIOSH | NA |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Sodium Dimethylarsonate: 0.5 mg As/m³ (as arsenic) |
| REL (Recommended) | 2 mg As/kg |
| IDLH (Immediate danger) | 100 mg/m3 |
| Related compounds | |
| Related compounds |
Cacodylic acid Disodium methyl arsenate Methylarsonic acid |

