Sodium Dihydrogen Arsenate: A Comprehensive Perspective
Historical Development
People first explored sodium dihydrogen arsenate as part of the wider quest to understand arsenic compounds. Mines and early laboratories in the nineteenth century saw scientists reacting arsenic oxides with sodium bases, mainly for agriculture and pigment production. These efforts weren’t just about curiosity—arsenic’s biological power pushed research forward, especially where control of weeds and pests held direct promise. As the world learned about chemical fertilizers, sodium dihydrogen arsenate surfaced in research discussions, sometimes praised for its strong effects and other times flagged for dangers. Earlier decades lacked clear rules about toxicity, yet interest in this compound kept rising in places where soil chemistry needed tweaking or analytical chemistry called for a solid arsenate standard. My own time in a university chemistry lab drove home just how wide its influence stretched—from history books to reagent shelves.
Product Overview
Sodium dihydrogen arsenate hits shelves for several uses, often as colorless crystalline granules or powders. Labs keep it as a reference material, while industry once saw it as a pesticide ingredient. Its chemical formula, NaH2AsO4, places it among inorganic sodium arsenates. Each bag or bottle carries a signal: this isn’t just another salt, but a tool requiring care and clear purpose. Suppliers pay attention to purity because even tiny contaminant levels can throw off experiments or processes. Some use the product for standardization in analytical chemistry, especially methods that test water or agricultural samples for arsenic, where accuracy determines safety and credibility.
Physical & Chemical Properties
Anyone who examines sodium dihydrogen arsenate notices classic crystalline forms. Most samples draw moisture from air, a property called hygroscopicity. The substance flows well as a powder, dissolves fast in water, and forms clear solutions—not much odor, and little taste in the concentrations labs use for calibration. On the shelf, this property set shapes handling and storage routines: glass bottles, dry rooms, clear labeling. Chemically, it acts as an oxidizer under some conditions and fits into complex arsenate chemistry, where pH, temperature, and other ions decide what happens next. Knowledge of these details moves beyond simple curiosity—it guards against missteps in synthesis, whether someone’s running a classroom experiment or handling larger-scale reactions.
Technical Specifications & Labeling
The label on a sodium dihydrogen arsenate drum stands out with a host of technical data: molecular weight (156 grams per mole), melting point near 60°C (as a hydrated form), solubility figures, sometimes even heavy-metal impurity levels below one part per million. Grade matters here. Reagent-grade purity assures labs their arsenic measurements won’t pick up phantom readings. Sellers include UN hazard codes and National Fire Protection Association diamonds, because this isn’t a casual compound—it calls for gloves, goggles, and a locked storage cabinet. In my work, I would always check the batch certificate, since even a reputable label means little without confirmation that someone tested what’s inside.
Preparation Method
Manufacturers turn arsenic-containing ores into sodium dihydrogen arsenate through controlled chemical steps. The process starts by roasting arsenic-rich minerals to generate arsenic trioxide. That gets dissolved in water and treated with sodium carbonate or sodium hydroxide, then acidified with mineral acid—often hydrochloric. Filtration, evaporation, and crystallization follow. Each stage aims for maximum yield and minimum contamination, as waste streams demand careful disposal to limit environmental harm. Old field guides describe how early chemists handled less-refined techniques, but modern routes focus on product purity and safe working conditions. Any preparation generates strict protocols because both the fumes and the finely divided dust threaten health and local ecosystems.
Chemical Reactions & Modifications
Sodium dihydrogen arsenate plays well with others in the lab. Its solutions can act as weak acids, and it participates in substitution or redox reactions with everything from iron and copper salts to organic ligands. Water chemistry experts use it to simulate arsenic movement in soil and groundwater, offering a predictable source when studying how arsenic travels or transforms in complex environments. People with expertise in minerals or soil science know how quickly an arsenate solution can shift under changes in pH or ionic strength, forming dimeric or polymeric species, precipitating out, or turning into other arsenate salts. These reactivity patterns help researchers model arsenic environmental behavior, especially for remediation projects after mining or pesticide use.
Synonyms & Product Names
Chemists sometimes call sodium dihydrogen arsenate "monosodium arsenate" or "sodium acid arsenate" in catalogs or safety documents. Older texts may use Latinized versions, but the modern marketplace favors short, direct synonyms—sometimes just abbreviated as "SDA" in industrial supply lists. Part numbers help distinguish hydrate content, as hydration changes weight and application. Product names shift across languages, yet the core chemical meaning stays the same, ensuring users know what they are buying—or handling in research or process work.
Safety & Operational Standards
Handling sodium dihydrogen arsenate pushes safety to the front. Heavy gloves, chemical splash goggles, dust respirators, and strong ventilation form the foundation of any serious operation. Exposure risks aren’t vague: ingestion can kill, inhalation damages lungs, and skin contact brings the threat of absorption or burns. People I’ve worked with respect the arsenic label above all else, because training once saved us from a careless spill. Disposal calls for hazardous waste protocols—waste drums shipped out on regular schedules, with clear labels and full tracking to an approved disposal facility. Standard operating procedures include spill kits, emergency eye-wash stations, and regular health monitoring for those who use the compound as part of their daily jobs. No shortcut feels worth it in a world where regulators and, more importantly, people’s lives are on the line.
Application Area
Sodium dihydrogen arsenate has played unique roles across analytical laboratories and some backward corners of agriculture. As a primary arsenic standard, scientists measure it to calibrate spectrometers, ensuring arsenic contamination gets caught before water or food heads to the public. Historical pesticide use marked a different age, now banned or strictly regulated in much of the world, but the compound’s analytical power remains. I’ve used it in graduate research to spike test soils or solutions, testing analytical methods aimed at lower and lower arsenic limits. Outside the chemistry lab, environmental engineers sometimes add tiny amounts to demonstration aquifers, teaching the next generation how to spot and remove arsenic from contaminated sources.
Research & Development
Scientists hold a sharp lens to sodium dihydrogen arsenate because its behavior in water and soil offers lessons on how to tackle global contamination crises. Environmental chemists use it to map the fate of arsenic compounds, compare treatment technologies, or to simulate real-world conditions in tightly controlled experiments. Research keeps pushing the envelope, searching for safer alternatives and designing rapid tests for arsenic levels in remote settings. Technical advances have produced sensors using color-change strips and portable analyzers, but sodium dihydrogen arsenate often sets the benchmark for these new gadgets. Universities and research institutes join forces with government labs, using the compound as a reference to build arsenic removal technologies that run from rural Asia’s groundwater pumps to city water plants in North America.
Toxicity Research
A look inside any environmental toxicology report reveals the heavy costs of arsenic exposure. Sodium dihydrogen arsenate provides a model toxin for laboratory studies, gauging how arsenic shifts between different forms as it travels through plants, animals, or bodies of water. Chronic exposure links clearly to cancers, vascular disease, and neurological problems—a fact that drives public health guidelines and sparks community action wherever arsenic levels rise in water or soil. The compound's toxicity provided early warning signs that shaped regulatory controls, and researchers now dig deeper into low-level, long-term exposure, especially for children and rural populations stuck with contaminated drinking water. Global studies drive the narrative, pointing toward stricter limits and improved technology, but the lessons learned in a lab with clear vials and record-keeping notebooks ripple outward to everyday health.
Future Prospects
Sodium dihydrogen arsenate stands at an intersection between scientific necessity and looming danger. On one hand, demand remains steady for analytical calibration, especially as detection requirements grow stricter in food and water testing. On the other, bans and restrictions on arsenic’s broader use tighten further every year. Companies innovate to find less toxic analogues, hoping to erase the poison but keep the analytical utility. Digital technologies empower scientists to model arsenic chemistry virtually, reducing how much real compound they use day-to-day. I see a future where sodium dihydrogen arsenate stays locked in lab cabinets, rarely released except in small, deliberate doses—essential for some research, yet edged out of applications where lives and the environment must come first. Regulatory changes, better detection, and a public push for clean water ensure that safe handling and responsible research determine the compound’s fate more than any chemistry textbook ever will.
What is Sodium Dihydrogen Arsenate used for?
The Chemical and Its Shadow
Sodium dihydrogen arsenate doesn’t show up in everyday conversations, but behind lab doors and in agricultural records, it’s left a real mark. This white, water-soluble powder once carried a reputation for getting deep into soil and disrupting ecosystems. Over the years, it’s been best known as an ingredient in some weed killers and insecticides. In the world of chemistry, sodium dihydrogen arsenate acts as a source of arsenic. That detail alone should set off alarm bells because arsenic isn’t something anybody wants lingering in their water, crops, or hands.
Why People Turned to Arsenic Compounds
Farms didn’t turn to arsenic out of sheer recklessness. Weeds and insects push up food prices, ruin harvests, and wear down the patience of generations. Synthetics like sodium dihydrogen arsenate promised control. In the early twentieth century, these compounds looked like game-changers. You could wipe out stubborn weeds and stubborn insects at once. For a while, many farm hands and field managers believed they’d just unlocked a safer, more predictable harvest season.
Those promises, though, came bundled with risks. Arsenic in any form can cause cancer, nerve damage, and a long list of ailments. It doesn’t break down easily in soil. It accumulates. That means runoff contaminates streams, kills off aquatic life, and moves its way through the food chain. I spent part of my childhood in a rural area where old arsenic pesticides haunted the groundwater. You could see the suspicion in every parent’s face when talking about what was safe to drink or plant in kitchen gardens.
Current Regulations and Real-World Impacts
Today, regulators in many countries don’t allow sodium dihydrogen arsenate as a pesticide. The United States Environmental Protection Agency points to the chemical’s toxicity and cancer risk. In the European Union, bans took root decades ago. Yet, legacy contamination remains. Arsenic still turns up in some old orchards and crop fields, and the process of cleaning those sites costs millions.
Those decisions reflect a growing respect for human health and the environment. According to research from the National Institutes of Health, just a small amount of arsenic over long periods weakens children’s intellectual development and raises the risk of skin and lung cancers. Farmers, workers, and even pets can’t avoid the risks. Allergy symptoms, rashes, chronic cough—these issues crop up in communities near old contamination zones.
What Comes Next?
The story of sodium dihydrogen arsenate shows that quick fixes can echo for generations. New pesticides go through stricter screening these days. There’s talk of biopesticides, crop rotation, and natural predators as smarter ways to manage pests without pouring toxins into the earth. Some farms now use groundwater monitoring and soil remediation to protect the next generation. Watching those efforts unfold, I keep thinking: simple, smarter solutions beat shortcuts and slow poisons every time.
Transparency from chemical makers, open data from government agencies, and strong local voices shape today’s choices. My experiences tell me that listening to people who work the land or drink the water every day leads to the safest innovations. If the tale of sodium dihydrogen arsenate teaches anything, it’s to weigh short-term gain against long-term loss, and to respect the invisible trails chemicals leave behind.
Is Sodium Dihydrogen Arsenate toxic or hazardous?
Digging Into the Details
User safety often depends on familiarity, and few people outside certain workplaces have even heard of sodium dihydrogen arsenate. This chemical pops up in settings where strict rules shape daily habits. My own years spent around laboratory benches taught me that some substances demand both caution and respect — arsenates definitely belong in that bucket.
Sodium dihydrogen arsenate, a salt of arsenic acid, isn’t something you want to handle without care. Arsenic, even in tiny doses, can unsettle the balance in living organisms. I remember the warnings from toxicology coursework: arsenate compounds can sneak into the bloodstream and disrupt enzymes vital to health. Symptoms from exposure range from stomach pain and vomiting to nerve damage and even cancer down the line. The Environmental Protection Agency and World Health Organization both set very strict limits on arsenic in drinking water, reflecting a body of evidence stretching back a century.
Practical Hazards and Real-World Examples
In the workplace, sodium dihydrogen arsenate usually falls under hazardous material guidelines. I’ve seen hazmat teams suit up for far less threatening substances. Even a single gram of an arsenic salt, when accidentally inhaled or swallowed, can set off a medical emergency. Farms and factories in the past sometimes turned to arsenic compounds as pesticides, only to discover lingering health and soil problems. Old textbooks rarely mention these stories, but I’ve heard neighbors worry about legacy contamination on their land years after these chemicals stopped being used.
At home, the risk drops, yet accidents can happen during amateur science experiments or home renovations in buildings with contaminated wood. Arsenic compounds often show no warning odors or colors. That makes handling them risky without the right equipment: gloves, masks, biosafety cabinets, and good ventilation. No shortcut replaces routine training, a lesson I picked up after seeing a colleague wind up in the ER from contaminated hands. Materials like sodium dihydrogen arsenate must be double-locked and clearly labeled for a reason.
Addressing Safety and the Bigger Picture
Pushing for education goes a long way toward prevention. Workers and students who get clear instructions, practical training, and regular health checks have much less risk for accidental exposure. Cleanup standards for arsenic compounds in soil or water come from decades of research, much of it driven by hard-won lessons. Hazardous waste rules require businesses to track exactly where and how these chemicals move. Continuous scientific review updates the rules as new data come in.
Some communities face higher arsenic risks because of factors outside their control. Natural groundwater in several parts of the world holds arsenic above safe levels. Here, tools like water filters with iron oxide or activated alumina media step in, and community health programs offer affordable testing. Public health groups and scientists working together have brought down poisoning rates in affected areas by sharing lessons openly and responding quickly to new findings. Regulations only work when people on the ground trust and understand the need for them.
Focusing on Trust, Transparency, and Science
Real safety with sodium dihydrogen arsenate grows out of honest conversations, sound research, and old-fashioned attention to detail. I’ve learned over the years that conversations about chemicals gain power not through alarm, but clear facts and patient teaching. By sharing real stories and scientific evidence — and making sure everyone from kids to experts knows what’s at stake — we protect not only our own health, but the future of everyone who shares the environment with us.
What are the storage and handling requirements for Sodium Dihydrogen Arsenate?
Looking at Hazards First
Sodium dihydrogen arsenate is not your everyday chemical. It carries serious toxicity, both from arsenic and its reactivity. Exposure brings risks of poisoning, so there’s no room for guesswork. Personal experience working in labs tells me that underestimating arsenic compounds can end up badly for both people and the environment. This one asks for strict respect and sharp attention to detail, so its storage space and the habits around it matter every single day.
Choosing the Right Storage Location
Direct sunlight, heat, and moisture get in the way of safe storage. A dry, cool, locked room, with no direct contact with the outside air, works best. You don’t want a spot that can flood or one prone to wild temperature swings. Rust and humidity speed up container breakdown, leading to leaks. Dedicated shelves labeled for toxic chemicals, ideally in a chemical fume room, help prevent accidental mix-ups. Stick to non-corrosive, shatter-proof containers—usually high-density polyethylene or glass with tight seals. Never cut corners by reusing old food jars or open-topped bins. The slightest slip turns a minor oversight into a real hazard.
Isolation Stops Trouble Before It Starts
Arsenic compounds require segregation from common chemicals, especially any that might trigger a reaction. Keep away from foods, feed, water, acids, and bases to reduce risk. Separate poisonous powders from flammables and organics, since cross-contamination can cause dangerous byproducts. Once during graduate work, I saw a careless stacking method; someone put arsenic near ammonium compounds. A fume hood caught the reaction, but if unchecked, that mix would have threatened the entire building. Safety barriers or secondary containment trays make clean-up and spill control far easier. It’s never wasteful to invest in extra storage boxes or trays—one contaminated rag left sitting out can mean disaster.
Handling: Gloves and Vigilance
Nobody touches sodium dihydrogen arsenate bare-handed. Nitrile gloves, safety goggles, and long sleeves block dust and splashes. Respiratory protection fits the situation—sometimes a simple mask, sometimes a full-face respirator for weighing and mixing. Every chemical worker I know insists on using disposable bench paper and meticulous daily cleaning. After each handling, wash hands and any exposed skin with plenty of soap and water. Never eat, drink, or touch your phone while working with this poison. Once a lab mate forgot to wash mid-shift and wound up with contaminated lunch gear; luckily, he got checked in time, but that sort of scare stays with you.
Disposal and Emergency Planning
Spills and leaks follow Murphy’s Law, so having a spill kit means you don’t improvise with paper towels. Kits should include absorbents, neutralizing agents, and waste bags for safe pickup. In the worst cases, knowledge of emergency procedures saves lives—this means clear labels, posted phone numbers, and regular drills. Disposal routes pass through licensed hazardous waste handlers. Never send arsenic waste down the drain or into regular trash. Proper record-keeping tracks every gram in and out, which helps in audits and protects everyone from accidental misuse.
Building a Culture of Respect
Strict storage and handling set the baseline, but culture cements those habits. Training everyone involved, keeping detailed instructions on hand, and fostering a climate of double-checking form a protective net. Shared stories—those close calls and reminders—keep comfort and complacency at bay. Sodium dihydrogen arsenate’s danger never fully fades, and only steady respect keeps the harm away.
What is the chemical formula of Sodium Dihydrogen Arsenate?
Understanding What Sodium Dihydrogen Arsenate Is
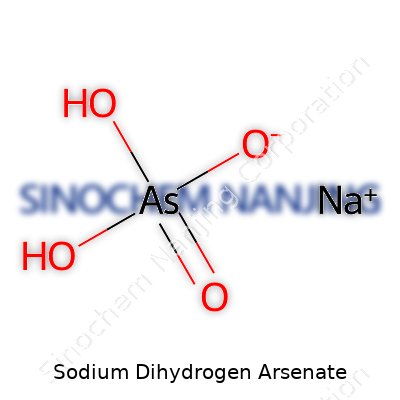
Sodium dihydrogen arsenate sometimes pops up in lists of chemicals, especially in industrial or academic labs. People who spend time in chemistry classrooms might remember seeing this compound during lessons about acids, bases, and salts. It carries the chemical formula NaH2AsO4. With one sodium atom, two hydrogen atoms, one arsenic, and four oxygens, it looks a lot like other arsenate salts, but the difference rests with those two hydrogens hanging onto the molecule.
Why This Formula Matters—Beyond Memorization
Getting the formula right isn't just an academic exercise. In the real world, chemistry labs handle substances with tricky, and sometimes dangerous, properties. Sodium dihydrogen arsenate is no exception, since arsenic compounds raise big red flags for health and environmental safety. If the chemical formula is misunderstood or ignored, someone might underestimate the risks or miscalculate the dose for a reaction. There's a personal angle too: as a student once struggling to get my head around chemical nomenclature, seeing the “dihydrogen” part made me pause. Does it mean two hydrogens are bonded somewhere unique? Turns out, these hydrogens connect to the arsenate group, and that changes everything about how the molecule behaves.
The Role of Sodium Dihydrogen Arsenate in the Bigger Picture
People don’t keep sodium dihydrogen arsenate under the kitchen sink. This compound usually serves as a reagent or intermediate in industry and research. Scientists sometimes use it to add arsenate ions to solutions. It’s also referenced as a comparison to other arsenate salts. Unlike the more notorious arsenic trioxide, this compound sits firmly under “highly regulated.” Even small spills must be taken seriously—decades of scientific research confirm that arsenic, in any form, enters the food chain easily and causes major health problems such as cancers, skin lesions, and neurological damage.
Balancing Use With Responsibility
Some people might think that only chemists or industrial workers need to care about the fine points of chemical formulas. That mindset leads to preventable mistakes. History tells stories of industries that ignored the warning signs—think of contaminated well water in Bangladesh or industrial spills in the United States. Both cases involved arsenic, and the damage didn’t spare local communities. Getting the formula right helps workers understand what they’re dealing with, and makes it easier to spot hazards if someone mislabels a container or mixes up similar-looking compounds.
Supporting Safer Practices Around Chemicals
Education forms the first line of defense. Everyone in a lab or workplace deserves regular training about what chemicals are on hand, the proper formulas, and what makes each one unique. This builds a positive safety culture. Regular audits and clear labeling back up the effort, reducing the risks of accidental exposure. Open communication helps—workers should feel confident about checking labels and asking supervisors if something looks out of place. Digital inventories and material safety data sheets also support tracking and emergency planning for hazardous substances like sodium dihydrogen arsenate.
Staying Informed Keeps Communities Safer
Chemistry isn’t just theory or rote memorization. These formulas impact real lives outside the classroom. Knowing the structure of sodium dihydrogen arsenate—NaH2AsO4—serves as a small but important step in building a safer environment, both in labs and in communities that live with the consequences of chemical use.
How should Sodium Dihydrogen Arsenate be disposed of safely?
Understanding the Substance
Sodium dihydrogen arsenate rarely appears in kitchen cupboards or local hardware stores, yet it crops up in labs and industrial processes, particularly as a reagent or in glass production. With arsenic at its core, this compound shows its true colors as a toxin, carrying serious risks for people, groundwater, and local wildlife. The compound dissolves easily in water, and just a little can travel far and linger. It brings high stakes to any decision about what happens to leftovers, spills, or waste. One mistake here goes beyond a personal hazard — it could poison a neighborhood.
Why Proper Disposal Matters
Since my school days in the chemistry lab, labels marked “arsenic” made hands steady and nerves buzz. Teachers drove home the point about how fast this stuff could harm. I’ve since learned arsenic compounds ranked among the top ten chemicals flagged by global health agencies for public concern, backed by data linking them to cancer, developmental delays in children, and organ damage. Getting rid of sodium dihydrogen arsenate responsibly isn’t about ticking boxes; it protects real people downstream.
Where Most Go Wrong
Many folks assume small quantities don’t count or that diluting with enough water means “out of sight, out of mind.” Water systems do not filter out arsenic easily. Flushing, burning, or mixing into local landfills turns a local problem into a shared disaster. There is no at-home fix here, and dumping on soil or down a drain could leave neighbors surprised by unexplained sickness or contaminated water.
Proven Steps for Safe Disposal
Trained professionals take the lead from the moment a jar of sodium dihydrogen arsenate wears the “waste” label. Sealed, labeled containers should store all forms—solid, liquid, or any mix—until handoff. Once at an approved hazardous waste facility, workers use barriers and containment systems built to handle toxic substances. High-temperature incinerators made for arsenic compounds or chemical treatments turn the waste less harmful, sometimes converting it into solid forms locked inside concrete or specialized glass. These methods stem from EPA and international guidelines informed by a century of chemical disasters and public health studies.
Getting Help and Making Choices
People who find containers of sodium dihydrogen arsenate at home or in the workplace shouldn’t try a DIY approach. Local environmental protection offices and hazardous waste contractors offer collection days and hotlines for just these situations. Costs, though sometimes steep, pale in comparison to lawsuits or community health impacts. Real trust comes from transparency, so sharing disposal steps with workers, neighbors, and regulators keeps everyone safer.
Better Solutions Ahead
Researchers keep working on ways to break down arsenic compounds more effectively and at lower cost. New filtration materials, such as iron-based gels, and recovery technologies show promise in grabbing arsenic from waste streams before they ever reach wells or rivers. Regulations now ask companies to rethink the design of chemical processes, aiming to swap in less harmful substitutes.
Conclusion: Responsibility Over Convenience
Working with or around sodium dihydrogen arsenate brings sharp consequences for shortcuts. Old habits led to countless exposures and ruined land. Real stewardship doesn’t mean perfection; it comes down to following strict, proven disposal routes, letting experts handle the hard work, and fighting the urge to cut corners for convenience. The next generation deserves land, air, and water safe from careless mistakes made today.
| Names | |
| Preferred IUPAC name | Sodium dihydroxidoarsenate |
| Other names |
monosodium arsenate sodium dihydrogen arsenate (1:1:2) sodium arsenate monobasic |
| Pronunciation | /ˌsəʊdiəm daɪˌhaɪdrəʤən ɑːˈsəniːt/ |
| Identifiers | |
| CAS Number | 10048-95-0 |
| Beilstein Reference | 1710785 |
| ChEBI | CHEBI:37138 |
| ChEMBL | CHEMBL1201738 |
| ChemSpider | 79632 |
| DrugBank | DB14048 |
| ECHA InfoCard | 03d0b86e-6192-474e-b52b-509fb74891e6 |
| EC Number | 231-993-5 |
| Gmelin Reference | 6366 |
| KEGG | C02344 |
| MeSH | D011461 |
| PubChem CID | 25970 |
| RTECS number | CG3325000 |
| UNII | 2G1U6V5S3Y |
| UN number | UN1871 |
| CompTox Dashboard (EPA) | DJ5E7L7V4S |
| Properties | |
| Chemical formula | NaH2AsO4 |
| Molar mass | NaH2AsO4: 141.92 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 2.36 g/cm3 |
| Solubility in water | soluble |
| log P | -4.3 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 1.99 |
| Basicity (pKb) | 11.54 |
| Magnetic susceptibility (χ) | -44.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.361 |
| Dipole moment | 2.24 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 145.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1236.6 kJ/mol |
| Pharmacology | |
| ATC code | V03AB04 |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled; causes damage to organs; may cause cancer |
| GHS labelling | GHS05, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H312 + H331: Toxic if swallowed, in contact with skin or if inhaled. H350: May cause cancer. H372: Causes damage to organs through prolonged or repeated exposure. |
| Precautionary statements | P260, P264, P270, P271, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P311, P314, P330, P501 |
| NFPA 704 (fire diamond) | 3-0-2 |
| Lethal dose or concentration | LD50 oral rat 40 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 48 mg/kg |
| NIOSH | TN1400000 |
| PEL (Permissible) | PEL: TWA 0.01 mg(As)/m³ |
| REL (Recommended) | 1 mg/m3 |
| IDLH (Immediate danger) | 3 mg As/m³ |
| Related compounds | |
| Related compounds |
Monosodium phosphate Arsenic acid Disodium hydrogen arsenate Trisodium arsenate |

