Sodium Dichromate: More Than Just an Industrial Chemical
Historical Development
Sodium dichromate stands as one of the core chemicals that marked the evolution of heavy industry and environmental awareness. In the nineteenth century, it helped textile mills color fabrics and supported leather tanneries aiming for fast results. Its bright orange color made it easy to recognize on factory shelves, but few thought about its impact beyond what it did for business. Later, large-scale mining and chemical manufacturing in Europe and North America relied on sodium dichromate, usually produced from chromite ore. For decades, workers handled it without gloves or masks, only learning much later about the risks. The chemical fueled bigger growth in pigments and metallurgy, but every period of progress carried a price; rivers near old factories still tell stories with their altered sediments.
Product Overview
Sodium dichromate’s deep orange crystals deliver more than just color. Industrial suppliers sell it as both dihydrate and anhydrous forms, depending on process needs. Tanks, barrels, and bags marked with its name traveled roads lined with smokestacks and rail yards. This chemical often acts as an oxidizer and is now closely tracked in shipping, storage, and reporting. Knowing exactly what’s inside matters, as purity and hydration level shift how it interacts. In industrial labs, keeping track of suppliers’ batch numbers and certification paperwork backs up safe use and outcome predictability—this comes from hard lessons after batches that failed or varied mid-production.
Physical & Chemical Properties
Sodium dichromate’s crystals don’t just catch the eye; their melting point sits high near 356°C, showing rugged thermal stability. It dissolves in water better than many salts, fueling its value in wet chemical processing. Reach for a strip of litmus paper, and you’ll see its solutions run acidic, which shapes how it gets handled in reactors or wastewater pipes. Chemically, it packs a punch: the chromate ion (Cr6+) lends strong oxidizing power, stripping electrons from everything from iron to organic dyes. That same reactivity delivered the bright yellow in road paints and aircraft primers, yet challenges any user to keep it contained and neutralized at the end.
Technical Specifications & Labeling
Labels on sodium dichromate containers aren’t just formalities—every detail from content percentage to batch ID keeps operations safe. Years back, I visited a lab where an unlabeled jug left staff guessing, slowing down work and raising worries. Modern specs usually expect purity upwards of 99% for most applications, with water content under tight control. Large operations demand Certificates of Analysis matching each delivery to process needs. Labels highlight the UN number (UN 3288), hazard pictograms, and GHS codes. Nobody in the business trusts a container without clear, compliant labeling—precision here directly protects workers and bystanders alike.
Preparation Method
Production still draws heavily on chromite ore, dug from the ground and trucked to chemical plants. Treatment with sodium carbonate and air drives out unwanted metals, leaving behind a melted mass rich in sodium chromate. Later, acid converts chromate to dichromate. This two-step method hasn’t changed much since industrial chemists first refined it, mainly because efficiencies in yield and safety took priority. Everywhere I’ve seen this process in action, the same approach holds: careful control of temperatures and acidity, with scrubbers and water treatment installed to tackle the runoff. Later on, crystalizers and centrifugal dryers prep the saleable product, a testament to years of innovation addressing air and water pollution risks head-on.
Chemical Reactions & Modifications
A sodium dichromate solution meets metal powders or strong acids and quickly launches redox reactions. Watch as chromium gets reduced from hexavalent to trivalent state—a major goal for safer disposal. Mixing it with sulfuric acid and organic compounds gets you oxidations used in everything from dye synthesis to lab reagents. React with sodium hydroxide, and dichromate shifts back to sodium chromate, showing a reversible dance in response to pH. Some researchers chase safer chemical substitutions, but the power and reliability of dichromate reactions underpin historic and modern techniques alike. I remember the fully vented fume hoods we relied on in student labs, always watching for color changes that spelled reaction progress or needed intervention.
Synonyms & Product Names
Walk into any factory, and you might hear sodium bichromate, bichromate of soda, or just dichromate. International shipments sometimes carry alternate names or translations. Every chemist or warehouse manager remembers the synonym used during their first training sessions, and mistakes about names once led to wrong deliveries—something manufacturers and pharmacists now work hard to prevent by sticking with standardized names and clear CAS numbers.
Safety & Operational Standards
Hexavalent chromium compounds—sodium dichromate included—come with a heavy safety legacy. Agencies like OSHA, ACGIH, and the European Chemical Agency set tight exposure limits due to proven risks. Handling this compound without gloves, goggles, and fitted respirators lets the dust reach lungs and skin, causing irritation, ulcers, and cancer over long-term exposure. Absorbing best practices means locking down processes to keep airborne dust at bay, enacting strict personal hygiene, and treating spills with reducing agents like sodium thiosulfate. Employers who rushed safety controls in the past now pay for it with medical claims and restricted site licenses. Industry groups keep pushing safer storage, fast cleanup protocols, and substitutions wherever feasible. Every new process review starts with exposure monitoring and contingency planning—the costs for neglect always outweigh a proper safety setup.
Application Area
A century ago, sodium dichromate ran through tanneries, dye houses, and pigment works. Later, its role expanded to surface treatment for metals, producing corrosion-resistant coatings for aircraft parts and automotive gear. Many plants still rely on this chemical for etching glass, coloring ceramics, and photoengraving plates. Water treatment professionals once used sodium dichromate to fight scale and biological fouling in industrial boilers, a use curtailed by stricter discharge rules. Modern applications survive in specialty chemicals, battery manufacture, and laboratory research where strong oxidizers are needed. Some regions push for chromium-free replacements, but legacy equipment and processes often force companies to adapt rather than abandon dichromate chemistry altogether.
Research & Development
Scientists and product safety engineers dive into alternatives, searching for ways to replace or detoxify sodium dichromate on a molecular level. Polymer chemists look for greener materials in pigment development, and corrosion experts study chromium-free alloy coatings. At every symposium I’ve attended, teams present newer reducing agents and encapsulation techniques aimed at cutting workplace exposure. Environmental labs devote acres of bench space to testing remediation methods for contaminated sites, from bioremediation using fungi to advanced precipitation and filtration. Progress happens stepwise as regulatory agencies demand more evidence and measured improvements, and peer-reviewed journals keep pressure high by sharing both successes and setbacks.
Toxicity Research
Decades of health studies link sodium dichromate to skin ulcers, lung cancer, and DNA damage. Animal testing in the mid-twentieth century drove home its risks and now fuels rules on workplace exposure and effluent discharge. National health databases and local clinics track adverse incidents long after plants shut down. One thing that comes clear: even trace exposures add up. Companies running aging machinery or waste handling processes face tough decisions balancing legacy output against clear and present health costs. Public health researchers argue for stricter enforcement and frequent updates of allowable limits. Efforts to build safer processes rely not just on chemical substitution but on worker training, real-time monitoring, and transparent reporting, principles now embedded in modern chemical handling guides.
Future Prospects
Looking ahead, sodium dichromate’s role keeps shrinking as industries step up environmental and worker protections. Manufacturers seek non-chromium-based routes for pigments, corrosion inhibitors, and oxidizers. Engineers building new plants often leave space for rapid phase-out as regulations catch up globally. Still, legacy uses and low-cost supply keep it in certain processes. Startup labs test machine learning models to predict chromium contamination flows, aiming for pinpointed, cost-effective remediation. Investment in green chemistry opens doors for start-ups with safer oxidizing agents. The conversation now includes a broader mix of voices—regulators, communities, and workers—each pressing for meaningful, measurable reductions in toxic exposure. As experience accumulates, lessons learned from sodium dichromate shape the attitudes and practices driving twenty-first-century chemical stewardship.
What is Sodium Dichromate used for?
Behind the Bright Orange Crystals
Walk into a factory that makes stainless steel, dyes, or even airplane parts, and you might find containers marked with a warning sign and filled with bright orange granules. That’s sodium dichromate, a substance that plays a role in a surprising number of everyday products. It’s easy to overlook chemicals like this because the finished things—a new car, clean stainless sinks, vibrant textiles—get all the attention. Dig just beneath the surface, though, and you’ll find sodium dichromate working quietly in the background.
Industrial Muscle in Action
Sodium dichromate brings muscle to manufacturing. Steel plants rely on it to add a protective layer of chromium to metals through a process called chrome plating. This results in gear that resists rust and lasts for decades. Every time you spot a shiny chrome bumper or grab a sturdy steel wrench, sodium dichromate has touched it in some way.
Factories that make pigments use sodium dichromate as a starting point for all kinds of colorants and dyes. These provide the yellow on road signs and the bright red in artists’ paints. The process gets complicated, with chemical reactions transforming sodium dichromate into color-rich compounds that hold fast under sun and rain.
Tanning leather once relied heavily on sodium dichromate. Old-school tanneries counted on chromate salts to harden and preserve animal hides, creating shoes, belts, and bags that last through years of use. Some countries still use it this way, though concerns over health and safety are shifting the industry toward alternative options.
The Challenge of Safety
Sodium dichromate’s power comes with serious risks. It releases hexavalent chromium—a toxic and cancer-causing compound—when handled carelessly or when dust escapes into the air. Think of the story behind the famous Erin Brockovich case, where water contamination with hexavalent chromium led to health crises in nearby communities. Even workers in well-ventilated factories need heavy-duty gloves, masks, and rigorous training to keep exposure in check.
Regulators in the United States and Europe keep a close watch on how sodium dichromate gets stored, used, and disposed of. Factories that cut corners on safety pay hefty fines or wind up shut down. The direct risk to everyday people is much lower, since almost nobody keeps sodium dichromate at home. Still, factories must follow strict rules to keep the environment safe and clean.
Looking for Better Solutions
There’s a growing push to replace sodium dichromate in many industrial processes. Science hasn’t delivered a perfect substitute for every use, and some industries still struggle to find new methods that match the performance, cost, and durability of chromium-based options. Water treatment professionals now use it far less than in decades past, choosing safer alternatives whenever possible.
As manufacturers keep searching for greener and less dangerous chemicals, the future of sodium dichromate seems tied to tighter regulation and better protective equipment. Making those changes takes effort and careful investment, but the potential health benefits for workers and communities are hard to ignore.
Trust Built on Information
Living in a world driven by heavy industry and shiny products, it’s easy to overlook the chemicals hidden in the process. Sodium dichromate carries risks that demand respect, but it’s also plugged into the backbone of manufacturing. Learning what’s in use and how it gets managed supports informed choices, whether you’re in the lab, on a factory floor, or at home reading the news.
Is Sodium Dichromate hazardous to health?
Plain Facts About Sodium Dichromate
Sodium dichromate doesn’t show up much in daily life for most folks, but it appears regularly in certain industries – metal finishing, pigments, wood preservation, and even leather tanning. If you ever took shop class or worked in construction, you might have crossed paths with it in rust-proofing or paint stripping. In industrial settings, safety data sheets flag sodium dichromate as intensely toxic, not the sort of chemical you touch bare-handed or breathe in without protection. This stuff turns bright orange, which feels like a warning in itself.
How It Gets Into Your System
Most people face risk through inhaling dust or fumes or by skin contact. Breathing in airborne sodium dichromate at work or through careless storage at home carries some of the biggest health risks. I remember reading about a factory in which a single spill sickened workers because dust got everywhere. My own uncle lost some lung function after years of working without a fitted mask near chromium chemicals.
What Exposure Does to the Body
This isn’t the kind of exposure that leaves you sore for a few days and lets you recover. Sodium dichromate acts as a powerful oxidizer and causes severe irritation. Even low-level contact can trigger skin rashes, nosebleeds, and headaches. It’s corrosive – burns show up within hours on exposed skin. Worse, chronic contact builds up in the body over time. Medical research has nailed down the link between hexavalent chromium compounds and cancer (particularly lung and nasal). Evidence from major occupational health studies confirms it — the risk isn’t theoretical.
The Environmental Protection Agency and the International Agency for Research on Cancer both label sodium dichromate as a human carcinogen. Repeated exposure also attacks kidneys and liver. I’ve heard of workers losing their sense of smell permanently due to tissue scarring in the nose. Drinking water contaminated with hexavalent chromium – similar to what happened in Hinkley, California, years ago – put entire communities at risk, not just industrial workers.
Why It Still Matters
Regulations exist, but keeping up enforcement isn’t a walk in the park. Even the best rules don’t always get followed on shop floors or in warehouses. Personal protective gear matters, but busy workers skip it in the rush sometimes. I spent a summer at a plating facility and watched as managers cracked down on safety for periodic inspections, then let things slide once the coast was clear. Even today, older buildings sit with contaminated soil or dust, putting neighbors at risk if anyone disturbs it.
What Can Reduce Risks
Mostly, preventing harm means using strict controls in workplaces — high-quality ventilation, gloves, respirators, and ongoing medical checks make a difference. The focus should shift toward using safer alternatives in manufacturing, not just controlling the problem after it’s made. Schools and local governments need to raise public awareness about hidden sources of industrial chemicals. Old sites with sodium dichromate residues need thorough cleanup before redevelopment starts. The science is pretty clear — the less exposure people have, the safer the outcome. Anything that cuts the use or the spread of sodium dichromate goes a long way in protecting health.
What are the proper storage and handling requirements for Sodium Dichromate?
Why Worry About Sodium Dichromate?
People in manufacturing and labs know sodium dichromate for its power in treating metals and producing pigments. Alongside its uses, this compound brings plenty of risk. It’s a strong oxidizer and throws serious health concerns your way—breathing it in or getting it on your skin can cause big trouble, like allergic reactions, ulcers, and cancer. Chromium (VI) compounds rank high on the list of workplace hazards. I spent years working around industrial chemicals—mistakes or neglect in storage and handling often meant real harm. Bad news only gets worse when folks cut corners on these basics.
Storing Sodium Dichromate Right
You want a storage spot built to limit contact. Think dry, cool, and away from sunlight. Moisture makes sodium dichromate clump, leak, and start chemical reactions with whatever it touches. I once saw a rusty drum fizz from contact with a cleaning liquid—a classic case of bad storage. Keep this chemical away from wood, paper, and anything organic. Fire risk ramps up if sodium dichromate meets flammable stuff.
Labeled, locked cabinets make sense. Store containers made from materials that stand up to corrosion. Make sure workers see warnings where they need them. Regulatory rules, like OSHA and the EPA, require secure labels and restrict access to trained people. I always made sure storage areas came with warning signage and those handy Material Safety Data Sheets, right on the door.
Handling: Suit Up and Train Up
Every time someone opens a drum, particles and dust could float up. The answer lies in personal protective equipment—gloves, goggles, a real respirator (not just a dust mask), and clothing that resists leaks. No shortcut here. Washing stations stuck close by make it easier to respond if powder spills or splashes.
Engineered controls like exhaust hoods make a difference. Spend the money on good ventilation—nobody wants to breathe this stuff. Training every new hire or temp was standard at my job. The company had us run drills for spills and splashes, hammering in the habit to never eat or drink around toxic chemicals. Good managers set up checklists and spot checks. This kind of culture, where everyone flattens the risk, starts with leaders who care.
Disposal and Emergency Steps
Improper disposal creates nightmares. Sodium dichromate doesn’t just vanish; it leaves behind dangerous chromium waste that wrecks water and soil. Licensed waste handlers pick it up and treat it before sending it to landfills lined by law. Proper paperwork stops shortcuts and illegal dumping. Emergency plans prep teams for spills. Absorbent pads and neutralizing agents on hand save time and protect everyone.
Fact-Based Solutions for a Safer Workplace
Solving sodium dichromate risks comes down to preparation. Proper containers, training up each team member, investing in safety equipment, and following strict disposal rules—these steps pull people out of harm’s way. After years in chemical plants, I believe companies and crews do best with hands-on training, regular safety reviews, and easy-to-follow signage. Safety works best as a habit, not a one-time thing. It’s not just about rules—it’s about bringing everyone home in one piece.
How should Sodium Dichromate spills or exposures be managed?
Recognizing the Real Risk
Sodium dichromate spells trouble for skin, lungs, and the environment. The bright orange crystals don’t look menacing, but the story changes fast if you’ve handled a spill or seen someone exposed. Hexavalent chromium compounds hit hard with their toxic punch. Breathing the dust, getting it on bare skin, or letting it settle in soil or drains can cause much more than a workplace headache. Anyone who’s read up on chromium toxicity or spent time in a chemical plant gets it. There’s a real reason safety pros lose sleep over this stuff.
Clear Steps Save Health
Clearing up a sodium dichromate mess takes no shortcuts. Every rule comes from tough lessons, not paperwork. Personal protective equipment isn’t optional. Chemical-resistant gloves, goggles, full coveralls, and a solid respirator keep harm at bay. People who skipped even one step in real-life scenarios ended up with chemical burns or worse. During a spill at an old plating facility I worked with, a missing glove led to weeks of medical checks. Direct contact wrecks health both short-term and long-term. Hexavalent chromium exposure brings higher cancer risks, severe skin damage, and even ulcers.
Facts back the care: OSHA calls sodium dichromate a cancer hazard. Inhalation limits run strict for a reason. The National Institute for Occupational Safety and Health flags chronic exposure as a real cause of respiratory illness and kidney damage. Every facility that stores or uses sodium dichromate should drill emergency response plans until muscle memory kicks in. Delayed decisions increase both contamination and medical fallout.
Containing the Problem
The spill cleanup starts with containment. Nobody wants to see dust kicked further or runoff reaching a drain. Dry methods like HEPA vacuuming or wet mopping with minimum disturbance make a difference. Someone might figure a broom works in a pinch, but that just spreads the nightmare. On bare ground, shovels and dedicated disposal bins stand ready—then immediate sealing and labeling for hazardous waste disposal follows. These contaminated materials never go near regular trash. I once saw a local lab face major fines after waste made it into the wrong bin, leading to costly soil remediation.
Water contamination turns a workplace event into an ecological threat. Sodium dichromate destroys aquatic life rapidly. Facilities near storm drains or rivers face added pressure. Open communication with local environmental agencies can prevent small mistakes from ballooning into regional health warnings. In 2017, a Midwest warehouse avoided that nightmare by alerting fire services and wastewater pros right away after a spill. Fast action slashed downstream chromium levels below regulatory alarms.
Prevention Outweighs Damage Control
The best way forward centers on less reliance on high-risk chemicals and strong training. Substitutes matter; the science community continues to search for safer corrosion inhibitors and pigments. A few industries have cut sodium dichromate use through updated technology. Where nothing else works, robust controls—ventilation hoods, health checks, and chemical-specific training—keep danger at arm’s length.
Facilities storing sodium dichromate in sealed, clearly marked containers along with eye wash stations and spill kits set the gold standard. Regular audits with real penalties for shortcuts create a culture that values health over speed. In my experience, the companies that embrace this approach suffer far fewer incidents and set themselves up for fewer regulatory headaches down the line. Protecting workers, communities, and waterways always justifies the cost and effort.
What is the chemical formula and physical appearance of Sodium Dichromate?
Chemical Formula and Structure
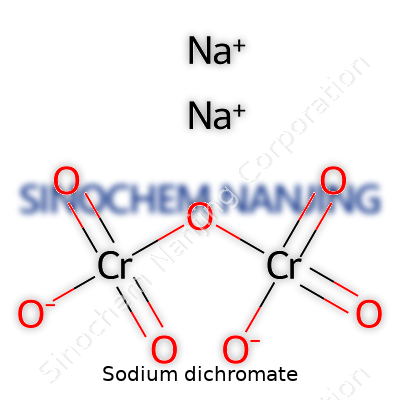
Sodium dichromate holds the chemical formula Na2Cr2O7. The numbers stand for its core composition: two sodium atoms, two chromium atoms, and seven oxygens. In standard conditions, it stands as one of the most recognized chromium compounds. Its formula gives away its aggressive oxidizing property, which shapes its role across multiple sectors.
Physical Form You Can Recognize
If you have ever opened a chemistry set or looked over pictures of lab chemicals, sodium dichromate likely stood out with its bright orange or reddish-orange crystalline appearance. It forms granular crystals or powder, easy to spot from a distance. Unlike many industrial powders, this color catches your eye and makes accidental exposure easier to see, although the safety risk remains real. The physical feel is gritty and somewhat coarse against the fingertips, which sets it apart from the flowy texture of table salt or sugar. It isn’t easy to forget this compound after handling it with gloves in any high school or industry lab.
Significance in Industry and Everyday Life
Sodium dichromate plays a major part in tanning leather, producing pigments, and as a vital oxidizing agent in manufacturing. The chemical not only brings color to dyes and textiles but also helps in corrosion control for metal industries. Every time you look at vivid colored textiles or well-preserved factory pipes, odds are sodium dichromate played a hidden role in their processing. For glassmakers, adding small quantities can yield green-tinted glass because of its chromium content. In the world of chemistry labs, its strong oxidizing ability provides an effective way to study unique reactions.
Despite all its usefulness, the chemical cares little for safety on its own. Handling sodium dichromate without care can lead to serious health risks: skin burns, respiratory issues, and a high risk of carcinogenic effects through prolonged exposure. If someone in your household works near chromate compounds, the orange stains on clothes or equipment serve as a reminder of the need for strict personal protection.
Safety, Health, and the Path Toward Responsible Use
Regulatory agencies, such as OSHA and the EPA, have clamped down on improper handling of sodium dichromate. Morgan, a friend who spent ten years in waste management, always spoke about the limitations set by National Institute for Occupational Safety and Health (NIOSH). He described strict ventilation systems and locked storage, meant to keep workplace exposure well below harmful thresholds. Factories often switch to less hazardous alternatives, but sodium dichromate’s effectiveness means it still shows up where nothing else works as well.
Proper handling starts with personal protective equipment. Gloves, goggles, and fully sealed storage containers aren’t just recommendations — they form the foundation of safe work involving this compound. Wastewater treatments or production facilities must filter effluent for chromium before discharge, a lesson learned from hard experience with groundwater contamination near old plants.
Finding Better Solutions
Finding safer substitutes grows increasingly urgent. Some industries look to cerium compounds or organic-based oxidizers for less toxic options. Others explore changes in process chemistry to get the desired results without sodium dichromate. Sharing information and enforcing strong workplace controls continue to protect people today, while ongoing research could eventually minimize exposure risk. Watching new technologies emerge gives hope for a future where vibrant color and clean environments don’t demand a toxic trade-off.
| Names | |
| Preferred IUPAC name | disodium dichromate |
| Other names |
Bichromate of soda Disodium dichromate Sodium bichromate Sodium dichromate(VI) Sodium dichromate anhydrous |
| Pronunciation | /ˌsəʊdiəm daɪˈkrəʊmeɪt/ |
| Identifiers | |
| CAS Number | 10588-01-9 |
| Beilstein Reference | 3569907 |
| ChEBI | CHEBI:78031 |
| ChEMBL | CHEMBL1200876 |
| ChemSpider | 20756 |
| DrugBank | DB14539 |
| ECHA InfoCard | 100.266.859 |
| EC Number | 234-190-3 |
| Gmelin Reference | 790 |
| KEGG | C01357 |
| MeSH | D003708 |
| PubChem CID | 24586 |
| RTECS number | WC2450000 |
| UNII | X0G1D5B44Q |
| UN number | UN3288 |
| Properties | |
| Chemical formula | Na2Cr2O7 |
| Molar mass | 261.97 g/mol |
| Appearance | Orange red crystalline solid |
| Odor | Odorless |
| Density | 2.52 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -2.7 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 12.8 |
| Basicity (pKb) | 6.5 |
| Magnetic susceptibility (χ) | +16000.0e-6 cm³/mol |
| Refractive index (nD) | 1.382 |
| Viscosity | Viscosity: 2.23 mPa·s (at 25 °C, for a 50% solution) |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 190.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -2055 kJ/mol |
| Pharmacology | |
| ATC code | V07AV Source: WHO ATC/DDD Index |
| Hazards | |
| Main hazards | Oxidizing, toxic if swallowed, causes severe skin burns and eye damage, may cause cancer, may cause genetic defects, may damage fertility or unborn child, very toxic to aquatic life. |
| GHS labelling | GHS02, GHS05, GHS06, GHS08, GHS09 |
| Pictograms | GHS05, GHS06, GHS08, GHS09 |
| Signal word | Danger |
| Hazard statements | H350, H340, H372, H314, H317, H334, H302, H400, H410 |
| Precautionary statements | P210, P220, P221, P260, P264, P270, P271, P273, P280, P301+P310, P301+P330+P331, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P308+P313, P310, P314, P321, P330, P352, P362+P364, P363, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-2-OX |
| Autoignition temperature | 400°C (752°F) |
| Lethal dose or concentration | LD50 oral rat: 50 mg/kg |
| LD50 (median dose) | 50 mg/kg (oral, rat) |
| NIOSH | 0084 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Sodium Dichromate: 0.005 mg/m³ |
| REL (Recommended) | 0.05 mg/m³ |
| IDLH (Immediate danger) | 15 mg/m3 |
| Related compounds | |
| Related compounds |
Chromic acid Potassium dichromate Sodium chromate |

