Sodium Chromate: Beyond the Yellow Powder
A Walk Through History
Sodium chromate carries a long and complicated story, touching everything from the chemistry classroom to the heart of heavy industries. Its creation in the 19th century kicked off an era when chemists-masters of fire, flask, and curiosity-learned to extract value from the colorful world of chromium ores. Early manufacturers eyed chromium-rich minerals like chromite, treating them with sodium carbonate and air at elevated temperatures. This roasting process unlocked sodium chromate, a bright yellow crystalline salt. From the beginning, its strong oxidizing nature attracted interest. Back then, chemical innovation didn’t always come paired with safety, and only later did public health keep pace with industry. Once sodium chromate appeared on the market, industries began using it for everything from dye production to metal finishing. Watching its applications expand over the decades offers a stark reminder of how science shapes industry, and sometimes, the world’s priorities.
Understanding the Chemistry
Pick up a vial of sodium chromate, and you have a yellow, water-soluble powder with the formula Na2CrO4. With a melting point over 792°C and good solubility, it finds its way into many chemical setups. The chromate ion in this salt packs a serious oxidizing punch, which explains why it participates in reactions that strip electrons and color from other substances. Chemists often use sodium chromate as both a reagent and a tool for making other chromium compounds, its reactivity forming the backbone of its utility. It’s attacked by reducing agents to yield green chromium(III), and under acidic conditions, shifts chemically toward dichromate, making it versatile in multistep reactions. Researchers continue to explore its transformations, extending the inventory of what sodium chromate can do.
Labels and Specifications: The Real-World Details
Practical handling always beats memorizing numbers from glossy brochures. Sodium chromate comes labeled with hazard warnings – not just because authorities require it, but because history has highlighted the stakes. Its crystalline grains may look innocuous, but the risk of inhalation or skin contact pushes labs and factories to double-check PPE, ventilation, and container sealing. Specification sheets call out particle size, assay percentage, and moisture content, but in everyday practice, the most important specs arise in how workers apply safety routines. Labels translate chemical knowledge into real boundaries — signal words, hazard pictograms, and boldface warnings make the difference between a safe procedure and a trip to occupational health.
Getting from Ore to Product
Making sodium chromate starts deep in the earth with chromite. Refineries roast this mineral with sodium carbonate and oxygen, then leach the mixture with water. What you get is a solution of sodium chromate, which, after careful crystallization, forms the solid yellow salt. Chemists learned to tweak conditions for finer yields, but the core hasn’t changed much. Each ton produced brings with it questions about waste, purity, and environmental handling, forcing facilities to walk a tightrope between efficiency and responsibility. Better handling methods and tighter controls have developed, but the fundamentals still rely on solid chemical principles: heat, reactivity, and separation science.
How Sodium Chromate Changes and Reacts
Open any chemistry textbook, and sodium chromate shows off its shifting roles through a series of classic reactions. Add acid, and that familiar yellow turns orange as dichromate forms. Tack on a reducing agent, and it reverts to chromium(III), dropping from solution as a green precipitate. It can oxidize alcohols to aldehydes and ketones in organic labs, or serve in corrosion-inhibiting paints. This capacity for transformation makes sodium chromate more of a toolkit than a one-trick compound, and anyone working with it has to understand not just one reaction, but a whole family of possibilities. This fundamental reactivity brought about centuries of use and still invites innovations, both in process chemistry and in environmental management.
Names in the Field and on the Street
Sodium chromate wears several aliases: disodium chromate, chromic acid disodium salt, and even sodium monochromate, depending on the user’s angle. Regulatory agencies and chemists alike keep a careful register of these names, as supply chains and safety data rely on precise identification. Early commerce often involved ambiguous naming, but as regulation matured, so did the insistence on accuracy. Modern compliance—especially in global markets—demands clear labeling, since even a slip in product name can land a shipment in customs limbo or put workers at risk.
Staying Safe With a Tough Chemical
Industry veterans know sodium chromate as much for its hazards as its usefulness. Direct skin contact causes burns, while inhalation can damage lungs and irritate mucous membranes. Long-term exposure links to serious health concerns, including carcinogenic risks. Safety procedures go beyond checklists; they mean adopting a culture where every container is suspect until proven secure, and every cleanup is a potential emergency. Best practices grew out of tragedy and hard-won lessons—ventilation, spill protocols, regular medical checks, and substitution where safer options exist. Worker education proves critical, just as much as high-tech safety showers and containment systems, with everyone on site sharing responsibility for keeping standards high.
How the World Uses Sodium Chromate
Walk through history, and sodium chromate shows up in dye works, leather tanneries, corrosion prevention for aviation components, and laboratory syntheses. Its role as an oxidizing agent in organic chemistry stays relevant, but tighter regulations now force industry to scrutinize every gram. In metal finishing and enamel production, sodium chromate properties produce tougher, brighter products. Water treatment plants once relied on it for controlling corrosion, though environmental rules have shifted much of that demand toward less hazardous options. Its utility keeps the compound on the shelves, but in smaller, more controlled quantities than in the mid-1900s. Many who have handled sodium chromate recall its dual nature: the way it advances technology but also brings environmental and health risks that push for constant vigilance.
Where Research Pushes Boundaries
Current research stretches in two directions: some chemists fine-tune sodium chromate’s applications, while others design ways to replace it altogether. Analytical chemists study its role in trace-level testing, measuring environmental release and plotting its migration through water and soil. Industrial scientists still chase improvements in process efficiency, aiming for lower waste and easier recycling. Environmental scientists, meanwhile, scrutinize its fate after use, developing remediation technology for contaminated sites. Much of this work unfolds in large teams, blending practical knowledge with computational simulations or new detection methods. Researchers publicly share findings on exposure limits, environmental persistence, and safer substitutes, responding to growing public concern and strict regulatory oversight.
Tackling Toxicity: A Constant Battle
The reality of sodium chromate’s toxicity remains central to every conversation about its use. Extensive studies linked long-run exposure to increased cancer risk and severe environmental contamination. Medical and environmental data fueled sweeping bans and substitution efforts in many countries, especially for open-process applications. Toxicity research rarely stands still; new studies dig deeper into mechanisms of harm, doses, and vulnerable populations. This data drives regulators to tighten legal limits and demands sophisticated monitoring in the workplace. For chemists and manufacturers, this vigilance means relentless improvement—less waste, better PPE, lower exposure, safer substitution—anchored by the very real costs of getting it wrong.
Looking Ahead: Future Possibilities
Sodium chromate’s role in industry won’t vanish overnight, but the future looks different from its past. As green chemistry principles move to the fore, many sectors trade traditional chromates for less hazardous alternatives. Research teams both public and private devote resources to finding new corrosion inhibitors, dyes, and reagents, balancing performance with health and ecological safety. In some frontier labs, sodium chromate still delivers insights, especially where its specific chemistry can’t yet be matched elsewhere. Transparency, innovation, and ethical handling will define its twilight era, turning lessons learned into safer, cleaner practice. Each breakthrough in alternative chemistries and waste reduction shines a light on what industry can learn: respect for chemical power, and a responsibility that extends far past the factory gate.
What is Sodium Chromate used for?
The Real Uses Behind Sodium Chromate
Sodium chromate stands out in the world of chemicals, grabbing the attention of both industry veterans and environmental watchers. Most people probably don’t realize that this yellow crystalline compound, which doesn’t show up on supermarket shelves, quietly powers critical sectors, especially manufacturing, metalworking, and the treatment of wastewater.
Keeping Equipment Strong
Inside refineries and processing plants, machines face brutal conditions. Pipes and boilers need to push through punishing cycles of heat and wear. Here’s where sodium chromate makes its mark. Regular steel equipment rusts and weakens, so companies rely on corrosion inhibitors. Sodium chromate gets pumped into water systems to stop rust dead in its tracks. I remember working in a factory where downtime from a corroded pump meant missed deadlines and tense meetings. This compound can be the difference between a smooth operation and a month of headaches.
Manufacturing and Pigments
Dipping into manufacturing, sodium chromate carves out a spot for itself in the creation of pigments and dyes. In the past, factories used it for making chrome-based pigments, which turn up in paints, inks, and plastics. Bright colors matter more than we’d guess—they keep school buses visible, ensure highways get marked, and inject life into everyday products. Even though demand for chromium pigments looks different these days due to safer alternatives, plants still use sodium chromate as a raw material when nothing else will do.
Textile and Tanning
Textile mills and leather tanneries have depended on this chemical for its oxidizing muscle. During the dyeing process, sodium chromate kicks off chemical changes that fix vibrant colors to fabrics. It helps keep colors bold, even after washing. In leather tanning, small doses let hides resist decay. That means boots, belts, and bags last longer. Of course, these industries have come under fire for pollution, but sodium chromate’s role in their process has played a key part in product quality.
Environmental and Health Concerns You Can’t Brush Off
Working with sodium chromate certainly isn’t child’s play. This compound packs toxicity; even a tiny amount in water can harm fish and poison streams. I’ve seen reports from environmental enforcement teams showing just a drop can set off alarms. Breathing its dust or touching the powder brings health risks—chromium compounds have a reputation for causing cancer if mishandled. Workers and companies take these risks seriously, decking out in masks, gloves, and keeping tight controls on any spills or leaks. Rules laid out by OSHA and the EPA guide the entire lifecycle, from use to disposal.
Choosing Safer Paths and Looking Forward
Many industries keep searching for less hazardous replacements. Green chemistry and smarter processes draw attention, but shifting old systems always takes time and investment. Some places have made real progress, using organic corrosion inhibitors or recycling systems that catch the worst runoff before it escapes. Still, for some critical uses—like nuclear power or specialized parts—engineers trust sodium chromate because nothing else stacks up quite the same.
The story of sodium chromate isn’t a simple good or bad—it’s about weighing performance against persistent risks. Companies, regulators, and communities face choices every year about how much to use, how to handle it, and what protection looks like in practice. For now, this old chemical keeps a strong grip in industry, serving up valuable lessons about both innovation and responsibility.
Is Sodium Chromate hazardous to health?
Straight Talk About Sodium Chromate
People sometimes use chemicals every day without thinking too hard about what happens if something leaks, spills, or gets on the skin. Sodium chromate stands out in the crowd of industrial chemicals because this stuff carries real risks if not handled with serious care. Growing up with a family member who worked in metal finishing, I’d hear stories about the caution required on the job. Sodium chromate showed up on warning signs in the factory and the anxiety around it came up at the dinner table more than once.
Why It Matters
Sodium chromate gets used for a range of tasks: dye production, corrosion control, and leather tanning. The problem comes from one of its main ingredients—hexavalent chromium. This is the same type of compound that became infamous in real-life environmental scandals. Breathing in dust or fumes, or letting it touch the skin, does not just trigger mild irritation. Over time, health problems can pile up. People exposed at work might notice skin ulcers, nosebleeds, or chest tightness that never really go away. I read a Centers for Disease Control (CDC) investigation that linked chromate exposure to increased risk of lung cancer, kidney damage, and chronic respiratory problems in workers over long service years.
Chromate’s corrosive qualities have caused concern for years among safety officials. Skin contact may cause open sores known as “chrome ulcers.” Getting it in the eyes or breathing it can end badly, too—sometimes resulting in vision problems or asthma-like attacks. Not all chemicals are equally hazardous, but it’s a fact that hexavalent chromium compounds like sodium chromate belong on the “cancer-causing” list, confirmed by the International Agency for Research on Cancer (IARC).
Protecting the People Who Use It
Factories can go months without a reported incident, but all it takes is one splash or dust cloud for things to change. Stories from those who handle this compound stick with me—engineers who remember a glove tearing, workers rushed to an eyewash station, old-timers who never quite shake their coughs. U.S. standards, like those set by OSHA, try to keep things in check. Regulations cap the amount allowed in workplace air and demand gear: gloves, goggles, heavy-duty masks, and proper ventilation. In honest terms, these rules often come into play only when someone raises a fuss, or after an inspection turns up a problem.
Communities living near factories that use sodium chromate aren’t always out of harm’s way either. One famous lawsuit in California made sure everyone remembers the dangers of groundwater and air contamination from careless handling of this compound. Health risks don’t stay boxed up behind factory gates.
Finding a Better Way
Sodium chromate’s dangers call for a few basic steps. Any company using it owes workers thorough safety training—demonstrations, not just posters. Regular medical checkups make difference, since catching a problem early gives workers a better shot at avoiding permanent damage. If possible, industries could replace sodium chromate with something less toxic. Though that’s often easier said than done, safer options exist for some applications.
I’ve seen small changes—using enclosed systems, investing in better air filters, or switching to alternate chemicals—make big differences for health and peace of mind. Reducing the risks from sodium chromate saves money in the long run, not just from lower injury costs but from keeping good workers healthy. People shouldn’t have to choose between a job and their health. With the knowledge we have, chemical hazards like these deserve respect and caution every single day.
What are the storage requirements for Sodium Chromate?
Sodium Chromate—What It Is and Why Storage Matters
If you ever worked in a lab or a plant, the bright yellow of sodium chromate usually stands for something potent and dangerous. This chemical gets used to make dyes, pigments, and sometimes even in treating metals. What may not be obvious to folks outside the industry: sodium chromate demands respect, especially once it’s time to put it away on a shelf. Mishandling creates real risks, not just to workers, but to the entire facility and even to surrounding neighborhoods.
Security and Isolation
Staying safe starts with separation. Storing sodium chromate in a space away from food, feed, acids, and anything organic keeps problems to a minimum. This means lockable cabinets, rooms with clear warning labels, and no shortcuts. Carelessness could lead to chemical reactions no one wants to deal with. I’ve seen the aftermath of a spill due to improper segregation, and cleaning up even a small mess requires hazmat suits and lots of patience. Health regulations and OSHA guidelines help guide good storage practices, including using corrosion-resistant shelving and double containment wherever possible.
Why Dry Conditions Matter
Anyone who’s opened a drum after a week in a humid warehouse knows chemicals don’t like moisture unless you want trouble. Sodium chromate is no exception. Humid air causes caking, turning free-flowing crystals into hard lumps. But the real threat comes from leaks and water exposure. This chemical reacts with water to form highly corrosive and toxic solutions. Storing it in tightly sealed, moisture-proof containers with clear labeling can’t be emphasized enough. Some facilities go a step further, investing in climate control and regular air quality monitoring around chemical storage rooms.
Temperature and Fire Precautions
Sodium chromate might not ignite on its own, but it can intensify fires if exposed to combustible materials. Never store it near paper, wood, or fuel lines. Heat from equipment or sunlight should stay well away. The right move? Pick a dry, cool area, shielded from direct sun, and nowhere near anything that burns easily. Many companies rely on temperature alarms and sprinkler systems certified for chemical use. Fire safety drills focusing on oxidizers help workers stay sharp and ready in case the unthinkable happens.
Environmental Concerns and Spill Response
Sodium chromate is nasty stuff for the environment. A small leak can travel through drains and damage ecosystems far beyond your fence. That’s why secondary containment—think sumps, spill pallets, and sealed concrete floors—makes a big difference. Having spill kits close at hand with materials meant for chromium is more than just a best practice—it can mean the difference between a minor incident and a reportable environmental disaster. Government regulations get strict here for good reason. Regular inspections and strong policies keep facilities from paying steep fines or causing long-term contamination.
Training and Organizational Responsibility
No storage setup covers every risk without proper staff training. Everyone from janitors to supervisors ought to know how to read hazard labels, handle containers, and act fast if trouble hits. Regular drills and mandatory safety reviews help reinforce good habits. I remember one supervisor who always told new hires, “Treat every chemical like it’s looking for trouble—because someday, it will be.” Helping people understand what they’re protecting—not just inventory, but lives and nature—keeps safety front and center, where it belongs.
How should Sodium Chromate be handled and disposed of safely?
What’s at Stake with Sodium Chromate
Anyone who’s ever worked in a lab knows bright yellow sodium chromate powders don’t just look menacing. They’re loaded with risks you won’t want to ignore, whether you’re weighing them for a test or cleaning up that infamous yellow dust. Sodium chromate contains hexavalent chromium, a toxic heavy metal known for its role in chromium-induced cancers. That’s not just scary on paper—people’s health and the planet are both in trouble if protocols get overlooked.
Personal Experiences in Labs
I’ve spent long hours pouring over chemical inventories and safety data sheets, then countless more teaching young scientists why they can’t treat sodium chromate like table salt. There’s no shortcut for personal protective equipment: thick nitrile gloves, goggles, and dust masks every single time. No bare hands “just for a second.” Exhaust fans and fume hoods must hum whenever anyone opens a container. I’ve watched more than one seasoned researcher try to shake out a corner of a weigh boat, only to send up a chromate cloud—not pretty, and not safe.
Environmental and Health Concerns
Once sodium chromate gets into soil or groundwater, cleanup grows expensive and slow. The compound doesn’t vanish over time; it sticks around and poisons whatever moves through the soil, including people. Inhaling dust or letting it touch skin won’t always cause pain or burning right away, but that won’t stop hexavalent chromium from increasing cancer risk or causing skin ulcers. Regular health monitoring for those who work with these chemicals matters, and so does honest reporting whenever spills happen.
Storing Sodium Chromate Safely
Chemical cabinets, even the locked ones, are only useful if people respect the rules. Sodium chromate belongs in sealed, clearly marked containers far from acids and organic materials. Humidity control helps keep powders from clumping or spreading. I always check for leaks or cracked lids every few weeks, and I recommend every lab add that to monthly safety checks. Labeling skips mean big trouble: everyone in your facility needs to know what’s inside yellow jars at a glance and how to grab the right spill kit if a container breaks.
Disposal Steps that Protect People and the Environment
It’s tempting for some organizations to treat older bottles as landfill trash or pour diluted solutions down the drain “just this once.” Regulatory fines pale next to the lifelong damage this chemical can cause. Sodium chromate solutions and residues belong in hazardous waste containers, labeled by type and date. Certified waste processors should pick up these containers and provide certificates of destruction or safe treatment. In my experience, partnering with reputable companies protects your team and your community—every dollar spent here is an investment in public trust.
Lab managers and technicians deserve better than vague memos about “universal precautions.” Some hands-on, scenario-based training helps new and seasoned staff build habits that last. I’ve seen workshops where people learn to spot signs of contamination—yellow stains along benchtops or dust in cracks—and those sessions make a real impact.
Culture of Responsibility
Safe handling and disposal depends on building a culture that values transparency, follow-through, and care for the next shift. Good habits don’t start with the chemical, but with the people using it. Consistency, vigilance, and treating every use like it matters—that’s the way to keep sodium chromate from becoming another chemical tragedy.
What is the chemical formula and appearance of Sodium Chromate?
The Make-up of Sodium Chromate
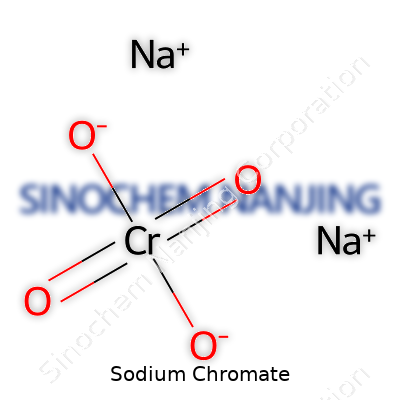
Sodium chromate belongs to a group of chemicals called chromates, which spring from chromium. Its chemical formula, Na₂CrO₄, reveals a clear story. You find two sodium atoms per molecule, along with a single chromium atom and four oxygen atoms all pulled together in a distinctive structure. For anyone with some background in basic chemistry, seeing "CrO₄" signals the presence of the chromate ion. Chemists and industry insiders spot this detail right away, knowing chromium sits at the core of this compound’s activity—lending unique properties, but also risks.
Real-World Appearance
Pulling sodium chromate off a storeroom shelf or out of a production bin, you don't need a magnifying glass or special equipment to identify it. The color jumps out immediately. Sodium chromate crystals usually appear as bright yellow, and they shimmer with a somewhat crystalline shape. Compared with the dull look of sodium chloride or other common salts, sodium chromate looks much more aggressive. This visual warning signals caution—chromium compounds like this have a record of being dangerous if handled carelessly.
Importance in Industry and Everyday Life
For industrial chemists and manufacturers, sodium chromate stays valuable because of its chemical behavior. It’s used for dye production, corrosion prevention in cooling systems, pigment making, and even in tanning leather. Its actions rely on that strong oxidizing power—which owes a lot to the structure of the chromate ion itself. The chemical’s popularity in different sectors comes from the way it reacts in solutions, breaking apart into charged particles. This helps speed up reactions and forms protective coatings on metals. Large-scale engineering and textile work have counted on it to deliver steady, predictable results.
Health and Environmental Risks
Yellow crystals of sodium chromate are not just a visual warning—they signal a real hazard. Chromium(VI) compounds, with sodium chromate among the most common, have a well-documented toxic profile. Both workers and environmental health specialists keep a close eye on sodium chromate storage and disposal for important reasons. Studies published by agencies like the U.S. Environmental Protection Agency and the National Institutes of Health catalog how chromium(VI) compounds can cause cancer, trigger allergic reactions, and harm soil and water quality. Exposure—even at modest levels—can leave both wildlife and people worse for wear over time.
Workplaces using sodium chromate rely on engineering controls: strong ventilation systems, personal protective equipment, and careful monitoring through chemical sensors and health checks. Regulations tightly restrict its use. Proper treatment during disposal, such as reducing chromium(VI) to the much less dangerous chromium(III) form, keeps the worst of the contamination in check. Factories often lean on closed-loop recycling and sealed containment, which have proven their worth in keeping leaks and spills from creating an environmental mess.
Options for a Safer Path Forward
While sodium chromate fills essential roles, safer alternatives are drawing more attention as regulations stiffen. Researchers and industrial chemists stay busy searching for replacement compounds which can provide needed effects, like anti-corrosive protection or pigment color, but with less risk. Many alternatives rely on zinc, phosphate, or organic chelating agents. These substitutes struggle at first to match the performance of chromate, but with each passing year, improved formulas and coatings continue to close the gap. Progress comes through a mix of research, stronger oversight, and, just as importantly, listening to people closest to the hazards: workers on the factory floor and technicians who know the risks inside out.
| Names | |
| Preferred IUPAC name | Sodium tetraoxochromate(VI) |
| Other names |
Sodium chromate(VI) Disodium chromate Chromic acid disodium salt Sodium monochromate |
| Pronunciation | /ˈsəʊdiəm ˈkrəʊmeɪt/ |
| Identifiers | |
| CAS Number | 7775-11-3 |
| Beilstein Reference | 010873 |
| ChEBI | CHEBI:40919 |
| ChEMBL | CHEMBL1200896 |
| ChemSpider | 53644 |
| DrugBank | DB01386 |
| ECHA InfoCard | 100.029.203 |
| EC Number | 231-889-5 |
| Gmelin Reference | 828 |
| KEGG | C01141 |
| MeSH | D017805 |
| PubChem CID | 24594 |
| RTECS number | GB5425000 |
| UNII | 93428QYT9A |
| UN number | UN3288 |
| Properties | |
| Chemical formula | Na2CrO4 |
| Molar mass | 161.97 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 2.7 g/cm³ |
| Solubility in water | 64.7 g/100 mL (20 °C) |
| log P | -3.9 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 12.5 |
| Basicity (pKb) | 3.04 |
| Magnetic susceptibility (χ) | +1120.0e-6 cm³/mol |
| Refractive index (nD) | 1.484 |
| Viscosity | Viscosity: 1.23 cP (at 25 °C, 10% solution) |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 199.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -937.4 kJ/mol |
| Pharmacology | |
| ATC code | V09CX02 |
| Hazards | |
| Main hazards | Oxidizing, toxic if swallowed, causes severe skin burns and eye damage, may cause cancer, may cause genetic defects, very toxic to aquatic life |
| GHS labelling | GHS05, GHS06, GHS08, GHS09 |
| Pictograms | GHS05,GHS06,GHS08,GHS09 |
| Signal word | Danger |
| Hazard statements | H350, H340, H372, H301, H314, H410 |
| Precautionary statements | P201, P202, P220, P221, P264, P270, P273, P280, P301+P312, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P308+P313, P314, P362+P364, P391, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-2-OX |
| Lethal dose or concentration | LD50 oral rat 1150 mg/kg |
| LD50 (median dose) | 50 mg/kg (oral, rat) |
| NIOSH | 0014 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Sodium Chromate: "0.1 mg/m³ (as CrO₃), OSHA PEL (8-hour TWA) |
| REL (Recommended) | 10 mg/m³ |
| IDLH (Immediate danger) | 15 mg Cr(VI)/m3 |
| Related compounds | |
| Related compounds |
Potassium chromate Sodium dichromate Chromium trioxide Potassium dichromate Chromium(III) oxide |

