Sodium Chloroacetate: A Deep Look at an Ingredient that Shapes Industry and Research
Historical Development
Sodium chloroacetate traces its commercial roots back to the spurt of chemical process innovation that swept through the late 19th and early 20th centuries. The compound emerged as a versatile intermediate thanks to the growing demand for efficient, reliable methods for making herbicides, pharmaceuticals, and dyes. Industries found sodium chloroacetate handy for introducing new chemical groups to other molecules — a trait chemists couldn’t ignore. The postwar boom saw its use climb as plant pathologists and synthetic chemists searched for ways to address crop yield challenges and drive forward pharmaceutical breakthroughs. Over time, as governments and producers sharpened their attention on safer working practices, the production and application methods for sodium chloroacetate improved, making handling less hazardous and more predictable.
Product Overview & Synonyms
Sodium chloroacetate, known in many circles as monochloroacetic acid sodium salt or simply SCA, functions as a pivotal building block in chemical synthesis. It doesn’t float around in the consumer spotlight, but its outputs touch everyday life — from shampoos formulated with carboxymethyl cellulose, to common painkillers shaped in labs. Trade names differ between regions, reflecting slight variations in labeling and formulation, but the core product remains a pale, crystalline powder, recognized for its sharp, acetic odor. Other names might surface in literature or inventory sheets: sodium monochloroacetate, chloroacetic acid sodium salt. Each synonym points toward a substance able to modify, activate, or steer a complex reaction — not by accident, but by design honed through decades of use.
Physical and Chemical Properties
Sodium chloroacetate stands out due to its fine granular form, often white in color, and its high solubility in water. It dissolves easily, producing a mildly acidic solution, which can both support and speed up chemical processes. In the solid state, it remains stable at room temperature, resisting significant breakdown unless exposed to higher temperatures or strong bases. This reliability in storage and handling draws attention in industries where moisture control and chemical stability matter. Chemically, the compound features a chloro group attached to an acetic acid framework, a structure keen to take part in substitution or addition reactions. The presence of the chloro group primes it for broad reactivity, making it a favorite among chemists seeking efficiency in organic synthesis.
Technical Specifications and Labeling
Technical standards shape how sodium chloroacetate appears on a shipment slip or a warehouse label. Labs and factories list specifications like purity (usually above 98 percent for most synthetic work), moisture content, and pH. Color often tells its own story — discoloration hints at degradation. Packages carry UN numbers for safety during transit, and danger labels shout warnings against inhalation, highlighting the threat to skin, eyes, and lungs. Regulatory frameworks in the US, EU, and Asia call for detailed safety data sheets and handling information. Chemical suppliers today compete as much on the transparency of their labeling as on their ability to ship pure product, meeting customer demand for clarity amid tightening safety regimes.
Preparation Method
Industrially, sodium chloroacetate comes from treating monochloroacetic acid with sodium hydroxide in an exothermic neutralization reaction. This process takes place in controlled reactors, where engineers adjust temperature and reagent ratios to keep yields high while limiting byproducts. In practice, the scarcity of suitable disposal methods for chlorinated byproducts pushes plants to invest in scrubbing and recovery equipment, reducing environmental risk while keeping within government-mandated thresholds. The process produces a product with consistent reactivity, letting downstream users trust each new supply batch to perform as expected. As demand for greener methods grows, research looks for routes that minimize chlorine emissions and cut down on water consumption, aiming for more closed-loop processes.
Chemical Reactions and Modification
Sodium chloroacetate takes pride of place in laboratories seeking to build up or break down other chemical structures. Its most common role revolves around acting as an alkylating agent, allowing other molecules to “swap in” more complex groups onto its main carbon chain. This function sits behind the creation of carboxymethyl cellulose — a texture-changing polymer found in both foods and personal care products. The chloro group, a point of vulnerability in the molecule, makes it reactive enough to help forge bonds that might otherwise seem impossible. Its range of reactions ties it to antifungal agents, pharmaceuticals, and herbicides, meaning that advances in sodium chloroacetate chemistry ripple through sectors beyond the chemical plant. The diversity of its derivatives reflects ongoing experimentation, with scientists spinning out new families of glycine-based compounds, surfactants, and more. Every modification must balance efficiency, cost, and environmental safety, making the quest for improvements ongoing rather than settled.
Safety and Operational Standards
Handling sodium chloroacetate demands respect for both its power and its dangers. Acute exposure causes irritation or even tissue damage, particularly to the skin, eyes, and respiratory system. Chlorinated organics like this have drawn strict attention from occupational health bodies due to their toxicological profiles. Protective equipment — gloves, goggles, respirators — becomes non-negotiable, and companies face fines or even shutdowns if safety lapses occur. Facilities invest in rigorous training, careful containment systems, and real-time monitoring for leaks or accidental releases. International transportation companies comply with hazardous materials codes, requiring traceability from factory gate to end user. Ongoing education supports a culture of safety, especially as new workers enter the field. Production plants weigh not only the direct danger of the chemicals, but also the risk of secondary reactions if sodium chloroacetate spills or mixes with incompatible materials. That keen attention to detail and strict adherence to regulatory requirements reflects both past hard lessons and a growing public accountability for chemical safety.
Application Area
Chemists depend on sodium chloroacetate far beyond the confines of their own discipline. In agriculture, it sits at the base of many herbicide syntheses, tipping the balance in the never-ending contest between farmers and weeds. The medical world taps the compound to develop active ingredients for medications, some of which break fevers or target infection. Water treatment experts reach for it to tweak the structure of specialty chemicals that clean up municipal supplies. Even paper and textile technologists work sodium chloroacetate into their supply chains, using it to adjust fiber properties or introduce new functional groups for advanced processing. Each field finds unique reasons for using the compound, but all depend on its steadfast performance and predictable behavior in chemical reactions. Environmental science teams look at it as both a tool and a hazard, weighing productivity against risk. No other single intermediate blends into so many product lines while still holding challenges when it comes to safe use and disposal.
Research and Development
Scientific interest in sodium chloroacetate grows as researchers chase lower-toxicity substitutes, greener chemistry, and higher product yields. University labs build up advanced reaction pathways, hoping to unlock new pharmaceuticals by refining or replacing portions of the sodium chloroacetate molecule. Production process teams work to cut emissions or recover waste from neutralization steps to satisfy ever-tighter environmental standards. A recurring challenge lies in the need to balance efficiency against both worker safety and environmental protection — a point hammered home by regulators and consumer advocates alike. Each research milestone, whether it’s a safer derivative or a more targeted synthesis, gets stress-tested in both pilot facilities and commercial production runs before reaching the real world. Publications highlight not just new chemistries, but also improved containment methods, smarter recycling, and the integration of digital control systems that catch problems before disaster looms. The discipline pushes everyone — from academic postdocs to plant engineers — to spend as much time on cleanup and control as on raw productivity.
Toxicity Research
People working with sodium chloroacetate learn quickly that exposure comes with clear risks. Toxicology studies point to marked irritation and corrosive effects, both through skin contact and inhalation. Scientists have observed acute toxicity effects, with thresholds that prompt strong guidance from agencies like OSHA and the European Chemicals Agency. Chronic exposure patterns show up in studies of workers handling the chemical over long periods, sometimes linking repeated contact to neurological symptoms or organ damage in laboratory animals. Modern research concentrates on quantifying these risks and identifying exposure pathways that matter most in manufacturing environments. Some groups catalog breakdown products formed during improper disposal, mapping how sodium chloroacetate fits into wider chemical hazard networks. Regulatory authorities use these findings to shape workplace standards, demanding not just safe handling instructions, but also investigation into alternatives and mitigation when plant discharges threaten public health or sensitive ecosystems. Wherever it enters the picture, sodium chloroacetate serves as a reminder that all chemical advances need regular re-examination under the spotlight of worker and community health.
Future Prospects
Looking into the future, sodium chloroacetate faces both opportunity and scrutiny. Its position in the supply chain stays strong in markets where low-cost syntheses of pharmaceuticals and specialty chemicals matter. Companies look for ways to manage risks while unlocking new chemistries that stretch its utility. At the same time, pressure grows to swap out hazardous precursors for safer, bio-based alternatives, especially in agrochemical production. Green chemistry movements spark waves of innovation, pushing for solvents that produce less waste and synthetic routes that consume less energy — each shift testing the grip sodium chloroacetate holds over current manufacturing methods. Real progress comes down to whether new approaches can match historical performance without exposing workers or surrounding communities to fresh hazards. Educational programs and stricter regulation nudge the next generation of chemists toward transparency and safety, setting the stage for an ingredient that either adapts to cleaner, smarter use or steps aside for something safer. Communities, regulators, and industries keep watch — not to halt progress, but to make sure it delivers more than short-term gains.
What is Sodium Chloroacetate used for?
Behind the Chemical Name
Sodium chloroacetate sounds technical, but its impact connects closely with many daily products and important industries. Most people never hear about it, yet those working in crop protection, pharmaceuticals, and chemical research, know it as an essential ingredient. The compound comes from a mix of sodium hydroxide and chloroacetic acid, forming a white powder that brings real value in how it reacts with other substances.
The Role in Modern Agriculture
Farmers depend on effective herbicides to protect their crops. Sodium chloroacetate serves as a building block for these chemicals. Think of popular weed control products in fields — many begin as sodium chloroacetate before being turned into finished sprays. This foundation lets companies produce vital agents that handle weeds without harming the crops. That delivers better yields and steadier food supply. Without such chemistry, food producers would have a tougher time keeping fields productive, and costs would climb.
Beyond the Farm: Medicines and Research
Medicine also leans on sodium chloroacetate. Pharmaceutical labs convert it into key ingredients that fight infections or address certain health conditions. Broad-spectrum antibiotics often start with a compound like this, giving researchers a versatile starting point for targeting bacteria or parasites. My time studying chemistry showed me how foundational ingredients like sodium chloroacetate guide the process — from petri dish to prescription. It enables chemists to design molecules that survive stomach acid or target specific tissues.
Other Everyday Products
Manufacturers use this chemical in dyes, flavors, and even cosmetics. For instance, producing some color pigments and perfumes relies on sodium chloroacetate to change the way molecules behave. Many food additives and scents in lotions trace their roots back to this simple substance. While consumers rarely recognize the name, the results show up every day, from brighter clothing to enticing fragrances.
Concerns and Safe Use
With every powerful tool, safety becomes a top priority. Sodium chloroacetate causes irritation to skin and eyes, and inhaling its dust may lead to health issues. Strict guidelines control storage and handling so factory workers stay safe. Environmental impact also matters. This compound can harm aquatic life if improper disposal happens, so regulators set tight waste controls when plants manufacture or process it. From my own experience in a lab, safety measures aren’t just about rules — they protect people’s health and build trust in what we use and eat.
Steps Forward: Responsible Chemistry
To improve safety, companies now turn toward better containment, robust employee training, and waste recycling. Monitoring exposure helps track risks early. Investing in alternative, greener chemicals could slowly reduce reliance on traditional compounds like sodium chloroacetate. Connecting expertise from toxicologists, chemists, and farmers brings practical progress.
Final Thoughts
Everyday reliance on sodium chloroacetate rarely catches public attention, but it keeps progress moving behind the scenes. Understanding its uses, risks, and future possibilities makes clear the importance of transparency and innovation, making sure both people and the environment remain protected as we rely on chemistry to solve real-world problems.
What are the safety precautions when handling Sodium Chloroacetate?
Recognizing the Hazards
Sodium chloroacetate is no ordinary workplace chemical. It can cause skin burns, poison the body if inhaled, and damage eyes on contact. Even a quick spill can bring harm. Years of reporting from chemical labs and manufacturing sites have shown that those who stay alert avoid trouble most often. You don’t want to treat this compound with a relaxed attitude; multiple case reports detail rushed experiments ending in emergency care. This isn’t just a rulebook issue—it’s about going home healthy.
Getting the Right Gear
Personal safety gear means more than a pair of gloves grabbed from a box. Choose chemical-resistant gloves, not simple latex. Polyvinyl chloride (PVC) or neoprene gloves block skin contact far better. Don’t skimp on splash goggles—regular prescription glasses can’t stop the tiniest dust or mist from getting through. Throw on a face shield if there’s a chance of splashing. A fitted lab coat, long sleeves, and sturdy, closed shoes round out the basic outfit. Fit matters; gaps invite accidents. I’ve been in spaces where skipping just one item led to injuries, no matter how experienced the handler.
Work Environment
Ventilation slows the risk of fumes. A fume hood works best for weighing, mixing, and transferring sodium chloroacetate. Keeping the compound sealed unless in use helps control air quality—no one wants a faint sweet odor warning them too late. Emergency eyewash stations and safety showers should always sit within a few steps of the worksite. It’s shocking how often companies overlook this simple step, only noticing during drills or real emergencies.
Safe Handling and Storage Habits
Open containers slowly. Any rush turns a controlled transfer into a hazardous release—keep motions smooth and deliberate. Scoop with plastic or corrosion-resistant tools to avoid unwanted reactions. Never return unused sodium chloroacetate to the original bottle, which reduces the chance of contamination.
Store the chemical in a dry, cool spot, away from acids, bases, and flammable substances. Clear labels make all the difference; nobody profits from a guessing game in an emergency. Safety Data Sheets (SDS) need to be on hand and familiar—not buried in a binder nobody reads. During my early lab years, confusion often sparked from unclear labeling or forgotten paperwork, and that’s where small mishaps grew into chaos.
Dealing With Spills and Exposure
If a spill occurs, keep untrained folks away. Use dedicated spill kits—typical cleaning rags don’t work. Neutralizing the chemical may require specific agents, so check instructions from hazard sheets. Cleanup teams should wear complete protective gear, not just gloves and a mask. Any skin contact should be rinsed with water—keep it running for at least fifteen minutes. Inhalation means fresh air, medical help, and not just waiting it out.
Regular Training Makes a Difference
Everyone’s skill with safety gear and emergency steps will fade without practice. Refresher courses help staff recall exact protocols. I’ve sat through both dry and engaging trainings—the effective ones always included real scenarios or firsthand accounts from those who had close calls. Remember, accidents don’t announce themselves. Preparation isn’t just company policy, it’s a decision to protect everyone involved.
What is the chemical formula of Sodium Chloroacetate?
Looking Beyond the Letters: NaCH2ClCO2
If you work in a lab, mix chemicals at industrial scales, or spend afternoons reading up on molecules, the chemical formula NaCH2ClCO2 might show up more often than you’d expect. This is sodium chloroacetate, a compound born from the meeting of sodium and chloroacetic acid. Pretty straightforward: swap the hydrogen on chloroacetic acid with sodium, and the result is sodium chloroacetate.
Sodium Chloroacetate: Not Just Letters and Numbers
People usually think of chemical formulas as sterile or detached from daily life. In reality, compounds such as sodium chloroacetate play a direct role in more places than most realize. Think about the herbicides that keep crops safer and food shelves more predictable. Sodium chloroacetate steps up as one of the basic building blocks for glyphosate and 2,4-D, two names that crop up constantly in farming and weed management. Without it, that chain breaks down, and suddenly, farmers lose reliable tools for keeping fields productive.
It’s not just about weed killers. The compound paves the way for a range of industrial chemicals. Take carboxymethyl cellulose, for example—easily dismissed until you realize it thickens toothpaste, lends texture to ice cream, and helps pharmaceuticals take shape. Chemists rely on sodium chloroacetate in the conversion routes that shape flavors, pharmaceuticals, dyes, and more.
Recognizing the Risks: Mishandling the Molecule
The world isn’t only composed of formulas and benefits. Handling sodium chloroacetate demands a healthy dose of respect—this is not a kitchen-shelf ingredient. Exposure easily leads to skin irritation, and inhaling even a small amount could trigger serious health problems. During my research, I saw incidents where confusion between this compound and safer sodium salts led to emergency room visits. Once, a small chemical company near my hometown mixed up their sodium salts during a shipment, causing a scare that led to an extensive safety review before business resumed.
Sodium chloroacetate’s production, too, comes with baggage. The process requires chloroacetic acid, which itself brings environmental and safety challenges. Safe containment, responsible waste management, and routine monitoring keep accidents at bay. It’s easy to overlook these steps at small facilities when margins tighten, but regulation must remain tight. No shortcut can justify putting workers or neighbors at risk.
Industry and Community: Room for Improvement
Better labeling and clear documentation go a long way. Mistakes fade when labels include bold font, color-coding, and international chemical identifiers. Training matters even more. Newcomers to chemical handling lose their hesitation around sodium chloroacetate when taught how to respect its power and see the safety measures in practice. Small and mid-scale companies should not skimp on this investment.
On a policy level, incentives for adopting greener production routes could ease the hazards connected with making and using sodium chloroacetate. Investment in research could uncover catalysts and conditions that sidestep some of the most problematic steps. The chemical itself won’t disappear, but the risks it brings can drop with the right approach.
In the Lab, on the Farm, and in Everyday Products
Sodium chloroacetate (NaCH2ClCO2) is more than a set of elements stuck together in a formula. Each time researchers test a new herbicide, each batch of carboxymethyl cellulose, each safety manual rewritten with real-world clarity, the story of this compound unfolds a little more. Clear information, respect for its hazards, and commitment to smarter manufacturing make all the difference between catastrophe and quiet success.
How should Sodium Chloroacetate be stored?
Why People Should Care About Proper Storage
Anyone who’s ever worked with chemicals in a lab or on the job knows neglect costs real money and leads to real hazards. I once watched a small spill become a stressful evacuation. That lesson sticks, especially with substances like sodium chloroacetate. This compound, often used for dyes, herbicides, and pharmaceuticals, poses health risks if handled improperly. So, putting thought into storage safeguards not just your product, but your people and the wider community. The Occupational Safety and Health Administration (OSHA) reported over 2,000 chemical exposure cases in a recent year—far too many from simple lapses.
Picking the Right Container Matters
You’ll find sodium chloroacetate as a white, crystalline powder. The stuff absorbs water from the air, then it starts breaking down. Problems follow—contamination, clumping, and chemical instability. I’ve seen companies use regular poly bags and regret it. Pipes clog, reactors jam up, and—worst—workers face toxic fumes. Air-tight containers with tight-sealing lids block out moisture. Polyethylene or high-density polyethylene drums stop chemical reactions dead in their tracks. Glass jars with lined lids work well for smaller amounts, though bulk users swear by steel-lined barrels that resist corrosion.
Temperature and Light Influence Safety
Chemical storage is rarely about just a shelf. Sodium chloroacetate reacts if it gets too warm. Most chemical suppliers recommend a cool, well-ventilated spot, out of direct sunlight. Those overhead lights add up: the stuff degrades much faster under harsh bulbs or UV. Most suppliers suggest a range no higher than 25°C (77°F). Even a basic wall thermometer saves money and headaches. I’ve used simple stick-on thermometers as a checkpoint near bins and barrels. It’s a minor expense that helps keep things on track.
Stay Away from Incompatibles
Good practice means keeping strong acids or bases well away from sodium chloroacetate. Reactions turn violent, releasing dangerous gases or causing pressure to build. A chemical inventory sheet posted right in the store area goes a long way—once, our team caught a risky shelf swap before disaster hit, just because someone spotted the posted guideline. This isn’t just best practice; it’s basic risk management.
Clear Labeling and Emergency Prep Save Time
Every drum, every jar, every bag needs a label. Not tomorrow—even one missed label leads to finger-pointing and time wasted in an emergency. Emergency kits with neutralizers and absorbents belong near storage areas. I’ve seen labs keep nothing but an old towel nearby and pay the price. Sodium chloroacetate can cause chemical burns. Stock eye wash stations and safety showers nearby, because minutes make the difference when things go sideways.
Review and Responsible Oversight
Schedule a quick monthly walk-through with a checklist. Regulations shift, and so do workplace needs. The best-run operations log each container: what form it’s in, how much remains, and when to replace it. Let people know who’s responsible. Experienced supervisors spot small issues before they grow. By paying attention, storage stays safe and reliable.
Moving Toward Smarter Handling
People sometimes ask why they need such diligence for “routine” chemicals. The answer comes in lost products, damaged equipment, and avoidable medical bills. By getting the basics right—dry, cool storage, verified containers, clear labeling—the workday stays safer and the bottom line looks healthier.
Is Sodium Chloroacetate hazardous to health or the environment?
Understanding the Basics
Sodium chloroacetate may not turn heads in a busy chemistry lab, but that little molecule packs a punch. It shows up in herbicides and is also used in dye manufacturing and the pharmaceutical world. Glancing at its clear crystals, most wouldn’t guess the danger lies in wait — danger to people and the planet.
Risks to Human Health
Handling sodium chloroacetate seldom gets people nervous until they check the safety data. Direct skin contact can produce pain, blisters, and sometimes deep burns. Inhalation hits even harder, triggering respiratory distress or, in unlucky cases, damage to the nervous system. The compound doesn’t just irritate; it can sneak through the skin, enter the bloodstream, and find its way to the organs.
Emergency medicine warns that ingesting even a small dose can bring on vomiting, severe weakness, and, without quick response, convulsions or even fatal poisoning. From my time working close to chemical storerooms, the rule was simple: gloves and eye protection always, no shortcuts. More than a few seasoned lab workers know colleagues who learned this lesson the hard way, leaving behind stories of hospital visits and internal injuries.
The U.S. Centers for Disease Control and Prevention calls sodium chloroacetate “highly toxic.” Long-term exposure connects to nerve damage among other chronic illnesses. Not a substance to ignore on any shelf.
Environmental Challenges
Factories often flush chemical residue away, and sodium chloroacetate pushes its harm into water and soil. Fish and amphibians end up in trouble even at low concentrations. I’ve seen reports from environmental watchdogs showing local streams losing insect life and fish populations after a leak. The chemical breaks down slowly; it clings to the landscape, finding time to reach bigger bodies of water or groundwater.
Once in the ecosystem, the compound can move up the food chain. Small invertebrates soak it up, then it keeps climbing: from worms to birds to mammals. Birds of prey picking off exposed rodents turn into unwilling carriers of high toxicity loads. That’s a tough blow for farmers trying to strike a balance between productivity and maintaining a healthy ecosystem on their land.
Preventive Action and Possible Solutions
Factories still play a pivotal role in keeping sodium chloroacetate away from natural water. Closed-loop systems cut leaks by capturing chemical runoff and recycling water, but adoption takes time and cash. Governments could push for stricter regulations, not just written rules but actual, regular audits, surprise inspections, and real-time water monitoring. Industry transparency helps—sharing accident details, not hiding them, reduces repeat mistakes.
Every worker who steps into a plant or research lab needs more than just a Hazmat sign; they deserve real safety training. I’ve found that ongoing workshops and open reporting systems catch problems earlier than one-time lectures or paperwork exercises.
Switching to less toxic substitutes helps, too. Some herbicides on the market promise the same weed control without the high health stakes. Market pressure and buy-in from farm cooperatives could speed up the shift, especially with public support behind them.
Final Thoughts
Sodium chloroacetate might look like just another industrial product, but its risks are very real. Keeping communities and ecosystems safe calls for action at every step—manufacturers, regulators, frontline workers, and everyday citizens all have a stake. History shows the cost of ignoring the warning signs runs higher than the price of prevention.
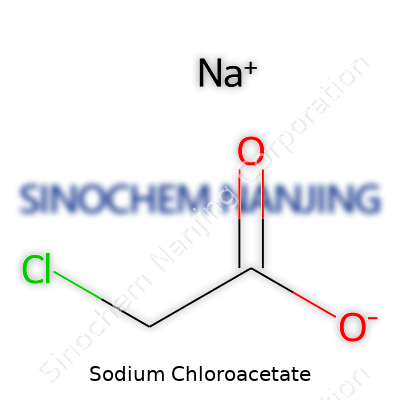
| Names | |
| Preferred IUPAC name | Sodium 2-chloroacetate |
| Other names |
Chloroacetic acid sodium salt Monochloroacetic acid sodium salt Sodium MCA Acetic acid, chloro-, sodium salt |
| Pronunciation | /ˌsəʊdiəm klɔːˈrəʊ.əˌsiː.teɪt/ |
| Identifiers | |
| CAS Number | 79-11-8 |
| 3D model (JSmol) | `/4OCH2Cl.Na` |
| Beilstein Reference | 1209224 |
| ChEBI | CHEBI:42261 |
| ChEMBL | CHEMBL50394 |
| ChemSpider | 21105718 |
| DrugBank | DB09462 |
| ECHA InfoCard | 03b1f6b0-c4a3-4d99-a39a-1e8e7fe86e27 |
| EC Number | 207-638-8 |
| Gmelin Reference | 7855 |
| KEGG | C18301 |
| MeSH | D013472 |
| PubChem CID | 7173 |
| RTECS number | AG1400000 |
| UNII | 1K78QR940N |
| UN number | UN2579 |
| Properties | |
| Chemical formula | C2H2ClNaO2 |
| Molar mass | 94.48 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.58 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -3.1 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 2.86 |
| Basicity (pKb) | pKb = 2.86 |
| Magnetic susceptibility (χ) | -38.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.424 |
| Viscosity | 500 mPa.s (20°C) |
| Dipole moment | 7.28 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 150.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -537.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -833.0 kJ/mol |
| Pharmacology | |
| ATC code | D06AX03 |
| Hazards | |
| Main hazards | Toxic if swallowed. Causes severe skin burns and eye damage. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS06,GHS05 |
| Signal word | Warning |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P261, P264, P270, P271, P273, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P310, P312, P330, P363, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-1健康-可燃性-反应性 |
| Autoignition temperature | > 580 °C |
| Lethal dose or concentration | LD50 oral rat 418 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 55 mg/kg |
| NIOSH | NA9395 |
| PEL (Permissible) | PEL: 0.5 mg/m³ |
| REL (Recommended) | 50 mg/m³ |
| IDLH (Immediate danger) | 50 mg/m³ |
| Related compounds | |
| Related compounds |
Chloroacetic acid Sodium acetate Monochloroacetic acid Dichloroacetic acid Potassium chloroacetate |

