Sodium Chlorite: Deep Dive into Its Science, Safety, and Future
Historical Development
Sodium chlorite turned into a game changer for water purification and textile treatment industries in the twentieth century. Early chemists struggled with scaling its production due to the instability of chlorine dioxide, but by the 1920s, new electrolytic processes made commercial production practical. Once manufacturers unlocked reliable synthesis, sodium chlorite started making serious waves in both public health and specialized manufacturing. The chemical took on multiple new jobs with the growing importance of water safety and industrial bleaching, proving its place alongside classic oxidants.
Product Overview
Sodium chlorite comes as a pale-yellow or white, crystalline powder with a salty, slightly chlorine-like odor. Companies sell it in various grades: water treatment, textile use, and specialty chemical markets. It travels and stores well thanks to its stability, especially compared to volatile chlorine dioxide gas. By keeping it dry and tightly sealed, handlers cut the risk of breakdown or unwanted reactions, giving everyone a lot more confidence on the plant floor.
Physical & Chemical Properties
Chemically, sodium chlorite sits at the intersection of chlorates and hypochlorites. It carries an NaClO₂ formula with a molecular mass just north of 90 grams per mole. The compound dissolves in water to form a basic solution, which reflects its mildly alkaline nature. Its decomposition releases oxygen along with chlorine dioxide—both big players in oxidation. Heat or strong acids can accelerate this process and cause a runaway reaction. Above 175°C, decomposition can get out of hand, so keeping things cool and dry matters a lot. The powder doesn't ignite on its own, but contact with organic material sometimes causes fires or explosions due to rapid oxidation.
Technical Specifications & Labeling
Regulators want clear product labeling. Most packaging lists sodium chlorite as at least 80% pure, with sodium chloride as the main impurity. Moisture content stays below 5% in quality batches. Bags and drums should display hazard pictograms and carry the UN 1908 code for sodium chlorite. Manufacturers must follow strict storage protocols under the Globally Harmonized System (GHS), using corrosion-resistant containers and keeping product away from acids, combustible materials, and direct sunlight. Labels display emergency guidance and signal words like "Danger," especially for industrial-strength shipments.
Preparation Method
Making sodium chlorite at scale usually starts by running chlorine dioxide into a sodium hydroxide solution. Chlorine dioxide forms upstream from sodium chlorate—a process that demands expert handling and monitoring. The reaction between chlorine dioxide and sodium hydroxide produces sodium chlorite plus sodium chlorate byproducts. Once filtered, the mixture dries under vacuum or at low temperatures to avoid kicking off any decomposition. Some companies tweak the process with catalysts or recovery steps to recycle materials and keep waste in check. Technical staff train up to spot yellow-green off-gassing, a sign that something needs quick correction.
Chemical Reactions & Modifications
Sodium chlorite plays as an oxidizer in water, textiles, and pulp. Adding acids steers it to release chlorine dioxide, one of the most effective and selective oxidizing agents for killing microbial life and bleaching without degrading essential fibers. In basic environments, sodium chlorite shows compatibility with many co-oxidants and can form a variety of intermediate-chlorine oxyanions like chlorate or hypochlorite. Modifiers sometimes blend the product with stabilizers to target specific uses, keeping decomposition low and effectiveness high over long storage. In research labs, chemists often react sodium chlorite with organic substrates to carry out advanced oxidation, and it’s routinely explored for breaking down persistent environmental pollutants in advanced oxidation processes (AOPs).
Synonyms & Product Names
Sodium chlorite goes by many labels: Chlorous acid, sodium salt; Alkali sodium chlorite; and for regulatory filings, simply NaClO2. Suppliers sometimes offer brand names like "Chloroxilite" or "Sanidate" for water sanitation lines. Many importers and customs officials recognize its chemical Abstracts Service (CAS) number 7758-19-2. Despite its simple structure, every country maintains its own set of trade names and safety documents, and product handlers learn to check paperwork carefully to avoid mix-ups with related oxidants.
Safety & Operational Standards
Handling sodium chlorite means focusing sharply on safety. It doesn’t tolerate rough treatment, accidental mixing, or careless spills. Contact with acids in a closed room can poison the air with chlorine dioxide. Swallowing the powder or solution—or even breathing a misty fog—can burn airways, damage red blood cells, and trigger acute poisoning. Most plants restrict access with signage and train staff in chemical hygiene, using goggles, gloves, face masks, and lab coats every time. Industry standards like OSHA and European REACH guidelines require tailored risk assessments on exposure, clear labeling, and procedure documentation. Emergency showers and eyewash stations hang close to every work area.
Application Area
Public water systems count on sodium chlorite for its prowess in chlorine dioxide generation, smashing pathogens that sneak by other treatments. Food processing plants rely on diluted solutions for equipment and produce sanitization, slicing down the risk of outbreaks without leaving harsh chlorinated byproducts. Textile and pulp processors depend on it for bleaching delicate fibers where traditional oxidants break down too much of the base material. In oilfields, sodium chlorite finds use in downhole treatments, breaking up iron sulfide and stubborn organic slime. Even environmental remediation turns to this chemical, chewing through groundwater contaminants in emerging technologies like chemical oxidation.
Research & Development
Scientists keep exploring sodium chlorite's possibilities, playing with new catalysts to streamline chlorine dioxide production and searching for tailored stabilization methods to reduce storage hazards. Teams have tested new blending strategies to improve efficiency and diminish secondary waste during water purification. In agriculture, research pivots on whether diluted sodium chlorite could cut down fungal and bacterial spread on crops without lasting residue issues. Material scientists mix it with novel fibers, hoping to create effective sterilization masks and hospital wipes, especially since pandemics place fresh demand on disinfection technologies.
Toxicity Research
Toxicologists focus heavily on sodium chlorite’s balance between benefit and risk. Doses above trace levels can disrupt hemoglobin’s chemical structure, nudging it into methemoglobin and reducing oxygen movement in the blood. Laboratory animals suffered toxicity at lower doses than seen with regular drinking water exposure, yet real-world water systems keep worker and public exposure far below these levels through strict dosing and fail-safes. The EPA, WHO, and EU jointly review all new animal data every few years, ratcheting safety limits up or down as fresh results demand. Medical teams push for better antidotes and clinical guidance in the case of accidental ingestion or inhalation, since quick action remains the key.
Future Prospects
Emerging water purification challenges push sodium chlorite’s limits, especially as scientists hunt for versatile, green, and effective oxidants against resistant pathogens and chemicals. A new push for decentralized water treatment in developing regions promises growth for safer, easier-to-handle sodium chlorite blends. Research into slow-release formulations could stretch its application into long-term pipeline and well-maintenance. Climate change pressures, shifting pollution patterns, and fresh demands in the pharmaceutical industry keep chemists locked on optimizing this molecule and squeezing every last drop of benefit from a century-old compound, with watchdogs always keeping toxicity in sharp view.
What is sodium chlorite used for?
Where Sodium Chlorite Shows Up
Sodium chlorite does a quiet job in many parts of daily life, but most people don’t notice it. Take water treatment, for instance. Municipal plants and some private wells rely on it to keep drinking water safe. In combination with other chemicals, it produces chlorine dioxide, which attacks bacteria, viruses, and other unwanted particles. That means fewer people get sick from waterborne germs, which plays a big role in community health. Seeing this in action on the maintenance side of a small town’s water system showed me just how important that process is, even if the chemical itself never gets much attention.
Manufacturing and Cleaning
Many industries count on sodium chlorite for bleaching. In pulp and paper mills, it brightens wood pulp without the harsh byproducts that traditional chlorine leaves behind. Textile factories also lean on its bleaching strength, which lets them turn raw fiber into the white base for all kinds of colors and prints. Hospitals and food-processing plants use sodium chlorite solutions to sanitize surfaces and equipment, cutting down the threat of outbreaks that could sweep through vulnerable groups.
I've worked with teams scrubbing and prepping industrial kitchens. Safety instructions always came with those cleaning products, and sodium chlorite-based solutions had clear handling guidelines. Skin and eye contact cause serious irritation, and inhaling fumes in a closed room leads to coughing fits or worse. So while the chemical keeps salad bars clean, it doesn’t deserve blind trust. People deserve training and equipment if they're expected to use it daily.
The Darker Side: Mischief and Misinformation
No discussion about sodium chlorite feels complete without mentioning the surge of online health claims. Some websites push “Miracle Mineral Solution” or similar products, urging people to swallow sodium chlorite to fight off illness. Stories from hospitals and poison control centers show how dangerous that advice turns out to be. Drinking sodium chlorite carries serious risks: vomiting, dehydration, kidney damage, and even death in high doses. In 2019, the U.S. Food and Drug Administration took action against companies making false medical claims, and other countries issued strong warnings.
It’s easy for misinformation to spread quickly online, but sifting through credible sources reveals the truth. Handing out advice about this compound without proper training or licensing threatens real lives. People who see claims about sodium chlorite working as a cure should look for evidence and ask medical professionals, not social media influencers. Public health agencies provide clear guidance based on data from real cases, not hype.
Moving Forward: Clear Rules and Awareness
Sodium chlorite itself isn’t the problem. How we use it and share information about it makes all the difference. Regulators hold companies to safety standards and issue recalls or warnings when needed, but education at every level helps keep both workers and consumers protected. Training, proper labeling, and easy-to-understand public information cut down on accidental poisonings and misuse.
In jobs that rely on chemicals, a bit of respect and caution goes a long way. A product that helps keep water clean or hospital rooms safe deserves to be used with care, knowledge, and responsibility. Seeing this firsthand on worksites and in small towns’ water plants left no doubt: sodium chlorite works best when handled with both science and common sense guiding the way.
Is sodium chlorite safe to handle?
Understanding Sodium Chlorite
Sodium chlorite shows up in various industries, from water treatment plants to textile cleaning. People usually talk about its power to disinfect, which gives it an edge in keeping spaces and products clean. Still, its chemical punch comes with real risks that many forget until they run into trouble.
Hazards Linked to Sodium Chlorite
Spills or even small mistakes with sodium chlorite make for a rough lesson. I remember the smell hitting my nose during a lab project in college, sharp enough to raise alarms. Skin burns became a topic in our safety brief that day, not just something in a manual. The National Institute for Occupational Safety and Health lists sodium chlorite as a strong oxidizer. If it touches skin, redness and irritation flare up quickly. Contact with eyes causes serious damage. Breathe in its dust and lungs start to protest. The chemical reacts with acids or organic matter — that means mixing it with common household cleaners or even a splash of vinegar could set off toxic chlorine dioxide gas. A small, unintentional mix at home can create a breathing emergency in seconds.
Why Some People Still Take Risks
The internet is full of wild stories. Reports claim sodium chlorite brings health benefits when ingested or inhaled, sometimes disguised under the label "Miracle Mineral Solution." This has never been proven safe. Both the U.S. Food and Drug Administration and the World Health Organization warn that swallowing even diluted solutions leads to vomiting, diarrhea, and kidney failure. Sales driven by social media exaggerate potential upsides while downplaying the damage. Some people trust these sources more than medical advice. This puts vulnerable groups — kids, older adults, people with immune issues — in harm’s way.
Safety Measures in Work and Home Life
Decent chemical safety comes down to respect and consistency. At my first job, old-timers insisted on gloves, goggles, and keeping the chemical locked away unless absolutely needed. The Occupational Safety and Health Administration (OSHA) marks sodium chlorite containers clearly with hazard labels, and the Safety Data Sheet stays posted for quick reference. Proper ventilation keeps fumes down. Emergency wash stations give immediate relief after exposure. Most accidents happen because folks let their guard down — rushing through a chore or reusing containers without a rinse. No one at that site ever handled sodium chlorite alone.
What Needs to Change?
Government rules help reduce danger, but casual handling outside industry continues. Education inside classrooms falls short. I rarely saw practical demos on real-world mistakes until I trained on the job. Retail outlets should keep sodium chlorite behind the counter. Fire departments urge safe storage far from combustibles, yet some homes pile chemicals together under the sink. Health warnings belong in plain sight, not hidden in small print. Community centers could run basic chemical safety workshops. As a reader, if you aren’t sure how to protect yourself, ask for a demonstration or skip the product altogether. Nobody needs to treat sodium chlorite with indifference.
Finding Responsible Solutions
Bigger companies now invest in worker training and spill response teams. Some even audit their own storage each month. At home, safer cleaning methods — borax, vinegar, baking soda — offer good enough results for most chores without risking a trip to the hospital. State health departments publish up-to-date advice and emergency contacts. By sharing honest stories and clear facts, people can make better calls on chemical handling. Personal experience and scientific evidence both point to this: stay cautious around sodium chlorite and seek help fast if something goes wrong.
How should sodium chlorite be stored?
Practical Storage: What Works and What Doesn't
People who handle chemicals know that some compounds demand respect, and sodium chlorite stands out as one of those. I remember working in a small water treatment plant, watching how a single slip—leaving a bag of sodium chlorite open near the moisture-prone floor—could easily turn one chemical into a real problem. Safe storage here isn't just about following checklists. It's about knowing how the stuff behaves and respecting its risks.
Sodium chlorite, a strong oxidizer, reacts aggressively if you get moisture or organic material anywhere near it. Once, we found an old container that had sucked in some humidity. The caking was the least of our worries compared to the risk of accidental mixing. Even a little contamination brings out its dangerous side. That's why storage places must stay bone-dry and clean. Air-tight containers made from plastic or glass keep sodium chlorite separate from the outside world. Avoiding metal containers keeps rust from getting involved in the game—metal invites unwanted chemical reactions and leaks.
Temperature and Placement Matter
Even seasoned workers overlook where they set the drums. I recall a colleague who left a barrel near a sunny window. After just a few hot days, the warning labels started curling. Hot or freezing conditions mess with sodium chlorite. Extra heat can lead to pressure buildup, while cold can make it lumpy and hard to handle. A cool, dark, and well-ventilated room with steady temperatures is the safest bet. Never put storage near acids, fuels, or anything that could trigger a reaction if there’s a leak or spill.
Why Labeling and Access Controls Aren’t Optional
Accidents don't just happen in the lab. Poor labeling caused a janitor at our site to mistake a sodium chlorite drum for floor cleaner. He didn't open it, but the scare was real. Bold labeling in clear language—with hazards spelled out—prevents these kinds of mix-ups. Locks and limited access keep well-meaning but uninformed staff from stumbling into trouble. Only trained folks should even touch this stuff, and training isn’t just a one-time box-check. Rules and routines help avoid complacency, which sneaks up after years without an incident.
Handling Leaks and Spills
Even the best plans face hiccups. Spills often come from carelessness during handling, or bad containers. Clean-up kits, absorbent materials that don’t react with sodium chlorite, and personal protective equipment (gloves, safety goggles, right masks) sit close by for good reason. Quick, calm action keeps small mistakes from growing. The area stays off-limits until fully cleaned and aired out.
Stopping Trouble Before It Starts
Routine checks decide if containers have cracks, faulty seals, or evidence of moisture. Companies should build inspection into their daily flow instead of leaving it to emergency drills. Regular record-keeping—tracking how much sodium chlorite comes in and leaves—helps spot errors before someone notices a leaking puddle. A few minutes each week looking for rust, leaks, or tampering cuts risk down to almost zero.
Responsible Disposal
Old or unneeded sodium chlorite shouldn’t just get tossed down the drain or dumped in the trash. Licensed disposal professionals handle the end-of-life for these chemicals so the environment and waste handlers stay safe. I’ve seen companies fined or shut down over careless dumping—and the community always remembers who put their water or air at risk.
Safe storage isn’t theory or paperwork. It's a habit that grows from respecting both the chemical and everyone working nearby. Cutting corners never ends well; even experienced teams mess up if they forget why these safety basics matter.
What are the potential health risks of sodium chlorite?
Knowing What Sodium Chlorite Does in the Body
Sodium chlorite might show up in stories about water treatment or cleaning solutions, but some internet corners push it as a supposed miracle cure for everything from infections to chronic diseases. Before anyone considers drinking or ingesting substances containing this chemical, facts about its dangers demand real attention.
Immediate Health Effects from Exposure
Contact with sodium chlorite has never been harmless. Inhalation, skin contact, or swallowing even a small amount triggers quick symptoms—nausea, vomiting, and severe stomach pain hit almost immediately. Start with the basics: this compound acts as a strong oxidizer. It irritates the skin, eyes, and mucous membranes. If inhaled, it burns the respiratory tract, leading to throat swelling, coughing, or wheezing. Accidental splashes in the eye cause burns and sometimes vision loss.
I’ve read poison control data and seen cases where kids landed in the emergency room because a parent thought “miracle” health products offered a shortcut to wellness. Those young patients came in dehydrated and weak. Sometimes, their blood work showed hemolysis—where red blood cells break apart before they should. This sets up the body for kidney damage and even organ failure. The FDA and CDC have received similar reports for years. Parents poured supposed wellness solutions down the drain, then drove to the ER in regret and panic.
Long-Term Risks Involving Major Body Systems
Sodium chlorite doesn’t just irritate the body for a day or two. Swallowing it puts major organs through more risk. Doctors know it can damage red blood cells, leading to a condition called methemoglobinemia. The oxygen carried in the blood drops so low that a person’s lips turn blue and organs falter. No one has to look far for real numbers—CDC records document patients needing intensive care and blood transfusions after drinking “health” tonics containing this chemical.
Some manufacturers promote sodium chlorite solutions under names like “Miracle Mineral Supplement” despite safety warnings. These products pose risks to kidneys, liver, and the immune system. Immune cells don’t bounce back fast after being exposed to strong oxidizers. Recent research confirms that continued exposure puts both children and adults at risk for chronic problems, including digestive and kidney dysfunction.
How to Stay Safe: Practical Steps and Policy Change
Clear, honest education remains one of the best defenses against these risks. Educators, healthcare providers, and parents need to spread facts close to home and online. Social media has amplified bad advice, so public health campaigns must become just as visible. The FDA has already cracked down on companies pushing sodium chlorite as a treatment, but stricter online enforcement and accurate information make a big difference. When people know a chemical can burn through skin or blood vessels, they stop seeing it as a health trend.
Safe drinking water and cleaning don’t rely on shortcuts with unknown or dangerous chemicals. Listening to health professionals goes further than taking cues from online videos. Talking about sodium chlorite in community meetings, school health classes, and with friends keeps more families out of the emergency room. Those who look for miracle cures deserve full, honest information and protection from false promises—because everyone deserves the chance to make truly safe choices for their health.
Is sodium chlorite the same as sodium chloride?
Looking at the Names: Close Isn’t Always the Same
People hear “sodium chlorite” and “sodium chloride” and sometimes think they’re talking about the same stuff. Their names sound similar, so the mix-up happens pretty easily. But there’s a world of difference between table salt sitting in your kitchen and the chemical agents used in industrial cleaning or water purification.
Table Salt: The Everyday Staple
Most folks have grown up around sodium chloride. It’s just plain old salt, made up of two everyday elements: sodium and chlorine. We sprinkle it on food, it keeps our bodies running right, and it’s even crucial for athletes or anyone sweating buckets to avoid cramps. Science calls it NaCl. Medical experts say it's essential, but too much can lead to health issues like high blood pressure. Grocery store shelves are stacked high with it, and health guidelines usually focus on how to manage salt intake.
The Other Guy: Sodium Chlorite
Sodium chlorite tells a very different story. Its chemical formula is NaClO2. Just slipping in an oxygen atom turns this into an entirely different chemical. You find sodium chlorite in places that need strong disinfectants—think treatment plants, paper mills, or industrial laundry facilities. It releases chlorine dioxide when used right, which works as a powerful bleaching agent or disinfectant.
No nutritionist will ever recommend sodium chlorite as a dietary supplement. The US Food and Drug Administration warns against using it as a cure or cleanse in drinks. Just swallowing a small amount can lead to serious health problems like kidney failure, severe dehydration, or low blood pressure. News reports have covered cases where people landed in hospitals after confusing the two chemicals.
Why This Mistake Gets Dangerous
Marketing tricks and online rumors sometimes push sodium chlorite as a miracle fix for various ailments. These products show up with names like “Miracle Mineral Solution,” and shoppers with little background in chemistry can fall for the sales pitch, mistaking it for harmless salt.
Chemists, doctors, and regulators alike stress that sodium chlorite and sodium chloride don’t play the same role in human health. World Health Organization guidelines cover sodium intake for sodium chloride, but call for absolute caution with sodium chlorite.
Clearing Up Confusion: A Need for Straight Talk
Educators and health professionals should help folks spot the difference, especially with so much information—and misinformation—circulating online. Clear labels on cleaning products, proper chemical education at school, and responsible journalism all play a part.
Anyone unsure about an unfamiliar product can ask a doctor or a licensed pharmacist for advice. Health hotlines and poison control centers often publish information about the dangers of ingesting chemicals never intended for human consumption.
Keeping Things Safe at Home and Work
If a label says sodium chlorite, keep it out of the kitchen. Store it with other cleaning or industrial products, far from food. Parents, teachers, and employers can keep communities safe by staying alert to possible mix-ups and sharing real facts, not rumors. Following well-established safety guidelines from government organizations protects families from accidental poisoning.
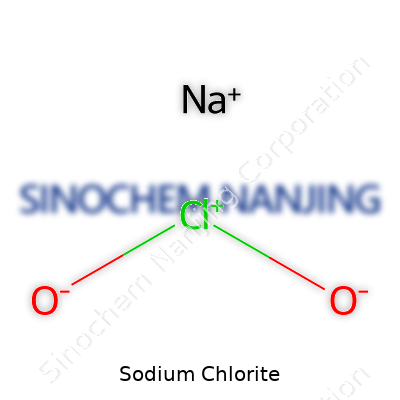
| Names | |
| Preferred IUPAC name | Sodium chlorite |
| Other names |
Chlorous acid, sodium salt Sodium chlorite (NaClO2) Textone Anthium Dioxide Slow-death Sodium oxidochlorate Greencide |
| Pronunciation | /ˈsəʊdiəm ˈklɔːraɪt/ |
| Identifiers | |
| CAS Number | 7758-19-2 |
| Beilstein Reference | 3569726 |
| ChEBI | CHEBI:9129 |
| ChEMBL | CHEMBL1201560 |
| ChemSpider | 14215 |
| DrugBank | DB13945 |
| ECHA InfoCard | 100.035.592 |
| EC Number | 231-836-6 |
| Gmelin Reference | Gmelin Reference: **1522** |
| KEGG | C13587 |
| MeSH | D017026 |
| PubChem CID | 24857 |
| RTECS number | VZ2450000 |
| UNII | 49GR9A0H0X |
| UN number | UN1908 |
| Properties | |
| Chemical formula | NaClO2 |
| Molar mass | 90.44 g/mol |
| Appearance | White or yellowish crystalline powder |
| Odor | Odorless |
| Density | 2.5 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -3.17 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 12.4 |
| Basicity (pKb) | pKb: 6.92 |
| Magnetic susceptibility (χ) | +22.0·10⁻⁶ |
| Refractive index (nD) | 1.466 |
| Viscosity | 10 - 15 cP |
| Dipole moment | 2.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 107.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −288.6 kJ/mol |
| Pharmacology | |
| ATC code | V03AB16 |
| Hazards | |
| Main hazards | Oxidizing, harmful if swallowed, causes severe skin burns and eye damage, toxic to aquatic life |
| GHS labelling | GHS05, GHS06, GHS09 |
| Pictograms | GHS03,GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H271, H302, H312, H314, H373, H400 |
| Precautionary statements | P220, P221, P261, P264, P270, P271, P273, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P363, P370+P378, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | Health: 3, Flammability: 0, Instability: 1, Special: OX |
| Autoignition temperature | 250 °C (482 °F) |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 Oral Rat: 165 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 165 mg/kg |
| NIOSH | 0458 |
| PEL (Permissible) | PEL: 0.1 ppm |
| REL (Recommended) | Sodium chlorite: 0.01 mg/m³ |
| IDLH (Immediate danger) | IDLH: 100 mg/m³ |
| Related compounds | |
| Related compounds |
Chlorous acid Chlorine dioxide Sodium hypochlorite Sodium chlorate |

