Sodium Chlorate: More Than Just an Industrial Staple
A Look Back at How Sodium Chlorate Entered the Scene
Sodium chlorate’s story starts in the 1800s, when folks first figured out you could make it by mixing saltwater with electricity. The process didn’t just pop up out of curiosity—demand for cheap, powerful oxidizers drove the science. Early manufacturers needed something reliable to use in dyes, matches, and eventually, the wood pulp industry. By the early 20th century, papermakers wanted an economic way to bleach wood pulp white, and sodium chlorate gave them exactly that. Production methods trickled from smaller workshops into major chemical plants, changing how things worked on a global scale. Because it used basic raw materials like salt and water and harnessed electricity, plants could get running wherever there was power and infrastructure. This shift from manual labor to engineered chemistry led to higher output and more stable quality, which only increased its popularity wherever industrial scale meant staying ahead.
What Sets Sodium Chlorate Apart as a Product
You find sodium chlorate in a white, odorless crystalline powder—easy to recognize once you know what you’re looking at. Unlike some industrial chemicals, it dissolves smoothly in water, which makes it easy to handle in large-scale processes. Its strong oxidizing power lets industries push reactions that wouldn’t go otherwise. People who haven’t seen sodium chlorate up close might underestimate it, but in factories, people rely on its ability to break down stubborn organic matter and regenerate things like chlorine dioxide, which ends up in various clean-up and synthesis steps. Names crop up in trade as Chlorate of Soda and Sodium Salt of Chloric Acid, but the working crews mostly call it sodium chlorate and keep things moving.
Physical and Chemical Properties Drive Its Uses
Sodium chlorate shows off a melting point around 248°C and carries a hefty molecular weight due to the three oxygens packed onto that central chlorine. Anyone who’s worked in a plant has probably seen its quick dissolving action in water—just a quick stir and there it goes, ready for action. Dry powder handles well unless moisture gets involved, so warehouses keep it sealed up tight. Oxygen released during decomposition makes this compound explosive in mixtures with organics—folks who have seen a bin of sawdust catch fire after a spill know this lesson too well. Its role as an oxidizer isn’t theoretical—people see it push reactions in pulp bleaching, weed control, and specialty chemical synthesis, showing practical value with every load tipped into a tank.
What Goes on the Label Tells a Lot
Labels might not grab the average person's attention, but anybody who runs a warehouse pays attention to technical specs. Manufacturers list assay purity, moisture, and bulk density, and point out the explosive risk. Even the size of the crystal matters; fine product hits hard and fast, while larger chunks meter out slower. Correct labeling means regulators, operators, and users don’t mix up bags or store them near combustibles. Old-timers love to see the purity above 99 percent—it means less sludge left over, less risk of surprise reactions, and easier control over downstream steps.
People Still Rely on a Straightforward Preparation
Most production still spins around electrolyzing brine in controlled, high-current cells. You pump saltwater, hammer it with electricity, and sodium chlorate forms at the anode. Chlorine gasses off first—dealt with by scrubbers—while the caustic finishes turning into chlorate in steps that look simple but demand tight controls. This routine method grew because it gave steady quality, scaled up easily, and fit well into chemical plant infrastructure. No one reinvented the wheel here: price pressures keep companies from making things any harder than they need to be.
Chemical Reactions and Modifications Keep Factories Humming
In application, sodium chlorate steps in as an oxidizer, turning tough lignin into soluble fragments during pulp bleaching. It serves in weed control too, burning off plants by releasing oxygen faster than tissues handle. Modifications show up through reduction: you push sodium chlorate with acid to get chlorine dioxide, the prized product in chlorine-free paper bleaching. In organic synthesis, chemists harness sodium chlorate not just for oxidation but for making special derivatives—work that pushes chemistry forward in medicine and specialty materials. Anybody who tinkered with it in a lab sees how quickly it drives change compared to other oxidizers. Despite the risks, people keep using it because newer chemicals rarely match its price-to-benefit ratio.
What’s in a Name: Synonyms and Trade Names
The name “sodium chlorate” sticks in most regions, but packaging in international trade sometimes lists Chlorate of Soda or Soda Salt of Chloric Acid. In local jargon across the pulp and paper industry, you just hear “chlorate.” No matter what you call it, buyers and users care less about the label and more about the assay on the spec sheet. Synonyms don't mean much once it’s through the warehouse doors—it all works the same in the vat.
Operational and Safety Concerns Hold Back Trouble
Industry veterans never let operators get lax on safety. Mixing sodium chlorate with anything flammable means risking fire or explosion; keeping it far from sawdust, oil, or grease comes standard. Eye protection and gloves show up as minimum requirements, with ventilation a must for any large-scale operation. Anyone who’s dealt with plant mishaps knows how unforgiving sodium chlorate acts. Storage stays dry, sealed, and isolated from anything reactive. Operators follow stringent procedures for handling spills—water to dilute, never sweep up dry, and never use organic cleaning pads. These protocols grew out of hard lessons, not overzealous regulation. Most workers would rather see another meeting than another incident.
Sodium Chlorate Paints Broad Strokes Across Application Fields
Main demand for sodium chlorate comes straight from the pulp and paper sector. Bleaching non-chlorine dioxide pulp gives whiter and stronger paper without the toxic waste associated with early chlorine methods. Herbicide use peaked decades ago, but some regions still cycle through sodium chlorate floods for total foliage clearance on railways and industrial land. In laboratories, sodium chlorate gives a trial run in oxidation reactions nobody else wants to risk. You see it in fireworks too—its role as an oxidizer creates the distinct colors and bang that spectators love at big shows. Demand doesn’t stop with traditional sectors; specialty chemical companies continue to test what sodium chlorate can pull off in new synthetic pathways.
Research and Development Keeps Pushing the Limits
Researchers build on sodium chlorate’s core chemistry, focusing on cleaner production and lower waste. Big strides have come through improved electrolysis cell design—using new electrodes or altered current profiles to boost yield and lower power demand. Some labs run pilot-scale tests with renewable energy, hoping to decouple sodium chlorate’s carbon footprint from fossil electricity. Out on the application frontier, teams explore lower-impact methods in paper bleaching, aiming to minimize byproducts and effluent toxicity. This matters as environmental standards tighten up across developed regions. Efforts go into making downstream processes greener, including tweaks to minimize AOX (adsorbable organic halogen) compounds in effluent streams. Even when regulations change, sodium chlorate proves sticky—no easy, cheap replacement works as well at scale in the roles it fills.
Toxicity and Health Research: Lessons Learned Over Time
Workplace exposure to sodium chlorate doesn’t cause much trouble unless you breathe dust or end up with repeated skin contact. Still, companies saw problems with long-term exposure in early factory settings—like kidney effects and respiratory irritation. Studies recorded hazards from chronic intake, often traced to improperly sealed containers or accidental ingestion. These findings changed how plants manage air and personal hygiene. In accidental releases, aquatic life suffers most, as sodium chlorate strips dissolved oxygen out of water systems, leading to fish kills. Research driven by disaster, not prediction, shows that sodium chlorate in upstream discharge leads straight to downstream problems—nobody argues about isolating process waste now. Health and ecological tracking keeps running year after year, with major findings pushing engineered controls and continual checks rather than relying on good fortune.
Future Prospects: Sodium Chlorate’s Line in the Sand
Looking ahead, sodium chlorate’s place depends on industry’s willingness to build greener processes and regulators’ stance on environmental risk. Alternatives for paper bleaching haven’t matched sodium chlorate’s efficiency or cost, so you see it holding ground. Technological tweaks—cleaner electrolysis, recapture of waste, and better operator protections—promise to keep it in play for at least another generation of pulp and industrial chemical workers. Research into new oxidizing agents or process modifications keeps its competitors honest, but for big plants with sunk infrastructure, switching costs plenty. The big question hangs over how quickly global regulations tighten around effluent and air emissions. If energy supply goes greener, sodium chlorate production could align more closely with low-carbon goals. In my own experience, every time the industry forecasted sodium chlorate’s decline, it instead adapted, found another niche, or held off the challengers. History doesn’t repeat itself, but patterns echo strongly in the steady demand for this workhorse chemical.
What is Sodium Chlorate used for?
Breaking Down Sodium Chlorate's Everyday Impact
Sodium chlorate might not be a household name, but it certainly plays a part in many things people use and see. Its history stretches back to the early 1900s, and today, most folks connect it with weed control and the paper industry. Most of the time, sodium chlorate ends up in big batches at lumber mills, where it helps produce chlorine dioxide—a chemical that makes paper sheets look clean and bright. Without it, many paper products would bring a dull finish to the table.
From Land Management to Paper Production
Talking from experience in rural communities, sodium chlorate shows up wherever there’s a war on weeds. Farmers and property managers favor it to clear out plants that keep growing back with a vengeance. They trust its power to wipe out stubborn growth by stopping plants from making food through photosynthesis. Towns, counties, and railroad companies also use it to clear brush near roads and tracks. That kind of targeted use saves labor and keeps operations safe and smooth.
Beyond agriculture and forestry, sodium chlorate’s reach goes further into chemistry labs and factories. It helps produce dyes, explosives, and even plays a part in water treatment. That says a lot about the versatility of this salt and the way industrial chemicals shape so many corners of daily life.
Safety First in a Powerful World
Sodium chlorate packs a punch, and that brings concerns that deserve real attention. Farmers know firsthand that careless handling burns skin, damages eyes, and harms lungs. It’s a strong oxidizer, which means mixing it with flammable materials invites trouble—sometimes even explosive reactions. That’s not just a worry for factory workers; it matters for everyone who lives near chemical storage sites or shipping routes. The U.S. Chemical Safety Board and EPA keep a close watch, pushing for strong safety standards and clear labeling.
Stories circle in farming towns about livestock deaths or waterway contamination. Even the World Health Organization says runoff from fields poses risks if sodium chlorate drifts into drinking water. Most people don’t see this side, but it’s the kind of challenge that shows why precautions and environmental stewardship need constant attention.
Searching for Smarter Practices
Many companies and researchers work on finding safer alternatives or smarter ways to use sodium chlorate. In the pulp and paper world, some mills invest in recycling and advanced bleaching methods that cut down on chemical demand. Farmers use GPS-guided sprayers to keep chemicals on the field and out of nearby streams. Retail sales of sodium chlorate-based weed killers have dropped in many areas, with some brands switching formulas in response to health and environmental studies.
Local governments hold collection and disposal events for old chemicals and launch outreach programs to teach safer storage and handling. These ingredient swaps and updated rules show a real effort to balance the need for effective tools with the call for healthier communities and cleaner environments. As the science keeps evolving, communities find new ways to get the job done without giving up safety or risking their water supply.
Is Sodium Chlorate dangerous or toxic?
A Compound with a History
Growing up in a farming community, everyone knew someone who had handled weed killers or “brush burners.” Sodium chlorate always turned up in those stories, usually with a mix of respect and caution. This white, crystalline chemical earned its reputation honestly. It’s used for killing unwanted plants, bleaching paper, and sometimes in fireworks. Still, ask anyone with hands-on experience, and they’ll tell you: sodium chlorate is dangerous stuff.
Not Your Everyday Chemical
Sodium chlorate works by breaking down cell structure in plants, essentially destroying what it touches. That same trait isn’t friendly to people or animals. Swallow it — even a few grams — and the body starts reacting quickly. Nausea, vomiting, and stomach pain show up first. The real trouble starts as it hits the bloodstream, where it can break apart red blood cells and damage organs. Too much means a race against time to save kidney function, secure oxygen, and stop the domino effect of cell damage.
Growing up, we were always told to store any weed killer up high, far from kids and animals. There’s good reason for that. Reports show accidental poisonings can be fatal. Signs appear fast: bluish lips, trouble breathing, even seizures if enough is swallowed. Hospitals have no specific antidote, so treatment means supporting breathing, flushing out the chemical, and hoping for the best.
Fire Risks and Explosions
Many chemicals present fire risks if mishandled, but sodium chlorate lifts that danger to a higher level. It reacts fiercely with anything flammable — sugar, wood, oil, sawdust. High school experiments mixing sodium chlorate with sugar sometimes led to unintended fires. Even a small spill, swept up with a dusty broom, can spark a flash. Food processing plants and warehouses storing this compound follow strict fire code standards. They separate it from anything that could catch fire, keep the storage dry, and limit the quantities stored in one place.
Environmental Effects
Sodium chlorate runoff can travel far. I remember fishing after a big rain and hearing friends complain about strange weed die-offs around creeks. It accumulates in the soil and water, and at high enough levels, threatens fish and soil organisms. Cities and states have responded by restricting use on roadsides and parks, and switching to safer weed control methods when possible. Farmers lean on modern techniques like targeted herbicide sprayers and only break out sodium chlorate as a last resort.
Public Awareness and Safer Alternatives
Regulations grew stronger because people spoke up about accidents and dangerous spills. Many countries now require labels outlining severe risks. Training became part of farming and landscaping jobs. People with years in the field can tell you that safety is about more than gloves and goggles — it’s about knowing when to call in help and how to store things safely.
Other options exist for weed and brush control: mechanical removal, mulching, or using less persistent herbicides. Safe handling guides can help keep people and animals safe, and proper disposal means fewer chemical traces in the food chain or groundwater.
Nobody should treat sodium chlorate lightly. It brings real risks, but information, training, and discipline go a long way in keeping it from turning a job into an emergency.
How should Sodium Chlorate be stored?
A Chemical with a Temper
Sodium chlorate looks like an ordinary white powder, but anyone who’s worked around chemicals knows that appearances fool. Its tendency to react with organic material or even a bit of grease means poor storage choices lead to real disaster. There’s a reason schools teach about oxidizers with dramatic lab stories—one careless person in a warehouse mixes sodium chlorate dust with a bit of oil, and the explosion makes headlines.
Physical Locations Matter Most
People sometimes treat chemical storage as a technical afterthought. They tuck bags in a cramped storeroom next to janitorial supplies, hoping paper warning labels do all the work. With sodium chlorate, luck runs out fast. Dry, cool, and well-ventilated rooms make the difference between a quiet shelf and a pile of scorched debris. Humidity causes clumping, and more dangerous, can set off slow decomposition that puts toxic gas in the air.
Metal drums with tight-fitting, non-reactive lids guard the substance from accidental moisture and unplanned contact. I remember visiting a pulp mill’s chemical storage. No wooden pallets or sawdust in sight—just clean concrete floors and every drum stacked away from walls, clear space to walk, every container labeled with hazard symbols bigger than my hand.
Keep It Far From Flammables
Storing sodium chlorate next to diesel cans or paint buckets equates to gambling with people’s homes. Oxidizers feed fire the way wind stokes dry grass. Stuffing fuels and oxidizers together turns one spark into a tragedy. One plant in Texas learned that in the worst way after a minor spill set off a blaze the length of a basketball court. Storing sodium chlorate away from anything that burns shows real respect for co-workers’ safety.
Containers Count
Decent storage starts with containers designed for harsh chemicals. Polyethylene barrels work well, because sodium chlorate eats right through most metals except stainless steel. I’ve seen plastic bins sagging from rough handling; that sort of wear turns into cracks, leaks, and eventually, emergencies. Inspecting containers before stacking them—just being nosy about every new shipment—stops accidents before they start. It’s not complicated, just routine vigilance.
Keep the Documentation Close
Labels and Material Safety Data Sheets shouldn’t live in a file cabinet or a boss’s locked desk drawer. I watched firefighters scramble in the dark once, trying to figure which chemical started a blaze because no one could find the product sheet. Quick access to the right hazard codes can save lives, and regular drills keep teams sharp.
Training Beats Comfort Zones
Even people with years of experience get complacent around familiar chemicals. Regular training resets everyone’s mental clock. Reminders about personal protective equipment—gloves, eye shields, and good boots—stick best through hands-on sessions. No one wants to stand through more safety lectures, but nobody wants permanent scars either.
Accountability Through Record-Keeping
Logs showing exactly when shipments come in, how many containers stand in storage, and who handles them aren’t bureaucratic busywork. They help track down inventory in an emergency and spot patterns that warn about dangerous habits. I saw a manager once catch a missing drum before anyone noticed a spill—just by matching delivery logs to his storage chart.
Every Rule Backed By Hard Lessons
Every step of sodium chlorate storage comes from someone’s hard-won experience. Firefighters, warehouse workers, and chemical engineers all want to clock out with all their fingers and the same set of eyebrows they started with. Those rules might seem strict, but they’re shaped by real stories, not just paperwork. Safety lives in the details—and everyone on the job deserves that respect.
What are the safety precautions when handling Sodium Chlorate?
The Risks Behind That White Powder
Sodium chlorate has a reputation that goes before it. Most folks have never handled this chemical directly, but for people who work with herbicides, pulp and paper bleaching, or even fireworks, the dangers are more than just warnings in a manual. The white crystalline powder looks harmless but can turn life upside-down with one careless mistake.
Things Can Go Wrong Fast
Touching sodium chlorate without gloves stings right away. The skin gets dry and itchy. The real threat kicks in once this chemical mixes with organic stuff — sugar, sawdust, even a grimy rag — and starts a fire with no mercy. Smoke fills the air, and you’re left running for help. I saw ash rain from the rafters thanks to a single unnoticed spark in a shed. That’s all it took. Sodium chlorate doesn’t give second chances; it reacts hard the minute the balance tips.
What Works: Good Gear and Clean Workspaces
Gloves do more than keep hands clean. They block chemical burns and stop sodium chlorate from soaking deep into the skin. Goggles keep fine powder out of the eyes, since contact sends people straight to the emergency room. Cotton overalls help, but synthetic fabrics actually make things worse. Plastic melts on contact with chemical heat, making burns more severe, so old-school cotton rules the day.
Masks make a difference, especially where dust clouds or tiny particles get in the way. Breathing sodium chlorate dust hits the lungs hard. Protective masks, not flimsy paper ones, stand between you and real trouble. There’s no room for shortcuts. I learned that lesson once, coughing for hours after ignoring my own advice.
No Smoking, No Sparks, No Shortcuts
Open flames spell disaster in a sodium chlorate work zone. There’s no saving a fire started by this chemical. Smoking breaks belong outside, far from storage or work areas. Sparks from tools, electrical malfunctions, or static electricity combine with sodium chlorate and start fires that nothing can stop. Never store this chemical next to fuels, rags, or paper — even dust swept into one corner can rise up with the faintest spark.
Keeping Sodium Chlorate Where It Belongs
At the end of a shift, all traces need scooping up and sealing tight. Containers labeled with bold, waterproof tags keep things clear. I once visited a site where someone slapped a bit of masking tape on a bucket as a label — next day, rain erased the writing, and confusion reigned. Storage calls for sealed metal drums that block moisture. Keeping the chemical dry matters: even a few drops of water lead to dangerous reactions, so no leaky pipes or puddles get tolerated anywhere near the stuff.
Training: The Strongest Line of Defense
Workers handle sodium chlorate safely when training turns into habit. Every year brings new people on crew, and every one of them deserves a lesson in respect and caution. Real stories work better than endless manuals. I share the mishaps from my own career — close calls and lessons learned — to drive the point home that sodium chlorate doesn’t play favorites.
Smart Disposal Counts
Throwing sodium chlorate in the trash endangers more than just the worker. Like oil on water, it finds its way into places it never belongs. Local hazardous waste sites handle it best. Follow disposal rules without question, even if it takes extra time. The price for carelessness runs high — and for some, there’s no way to undo the damage.
Can Sodium Chlorate be mixed with other chemicals?
The Hidden Dangers Lurking in Simple Chemistry
Most folks see sodium chlorate as just another white powder on a chemical supply shelf. Many use it for bleaching pulp or killing tough weeds, but mixing it with other chemicals turns a regular weekday job into a ticking time bomb. I’ve seen people treat it far too casually—like salt on a kitchen counter—because the danger doesn’t always jump out at you. At home or in industry, basic chemistry lessons get skipped, and that’s when someone gets hurt.
What Happens When Chemistry Goes Sideways
Sodium chlorate loves to release oxygen. That’s where the trouble kicks in. When it comes into contact with organic materials, sugar, sawdust, or even clothing fibers, the combination can set off a fire or a violent explosion. In college, one of my lab partners nearly lost an eyebrow when his notebook paper got a bit too close during an experiment. Factories and farmers aren’t immune either—handfuls of cases make the news every year about explosions or chemical burns.
Then there’s the issue of fuel-based mixing. Tossing sodium chlorate in with any simple fuel—think sugar, charcoal, or even a stray drop of oil—starts a reaction that cooks up enough heat to light the mix in seconds. Stories run around farming communities, tales of someone’s back shed catching fire or barrels going up at landfill sites, always with the same ingredient list.
Mixing with Acids: A Recipe for Disaster
Adding acids into the picture, like hydrochloric or sulfuric acid, can punch things up a notch—chlorine gas forms faster than you can cover your nose. Chlorine isn’t just a pool cleaner; in high doses, it drops people to the ground gasping for air. Surviving a mix-up once is luck, not skill. Health records show sharp spikes in respiratory problems in towns where sodium chlorate use goes unchecked.
What the Experts Say Matters—For a Reason
Government agencies and chemical safety boards send out warnings each year for a reason. The U.S. Chemical Safety Board tracked hundreds of factory incidents, and sodium chlorate pops up more often than you might think. The substance holds an oxidizing power strong enough to accelerate fires that regular extinguishers can’t handle. In one pulp facility, a chemical mishap spilled chlorate onto oily rags, setting off a fire that shut production for weeks.
Research from the National Institute for Occupational Safety and Health points out the material’s dangers too. Results show that just a small amount, added unknowingly or by mistake, can cause massive property and health damage.
Getting Real about Solutions
No fancy safety campaign replaces basic respect for chemistry’s risks. Labs and workplaces that store sodium chlorate need strict inventory checks and better training for younger staff. On farms, safer alternatives to sodium chlorate-based herbicides do exist, and local governments could do more to encourage their use and provide disposal programs for old stockpiles. Community training sessions from local fire departments help average people spot trouble before things heat up, literally.
Experience shows that chemical disasters never announce themselves—they show up fast and hit hard. Clear labeling, secure storage, and a hard rule against home mixing keeps more people safe than any warning label stuck to the side of a drum. Simple, grounded action beats cautionary tales every time. Those who work with sodium chlorate owe it to themselves and their communities to treat it with the seriousness it demands.
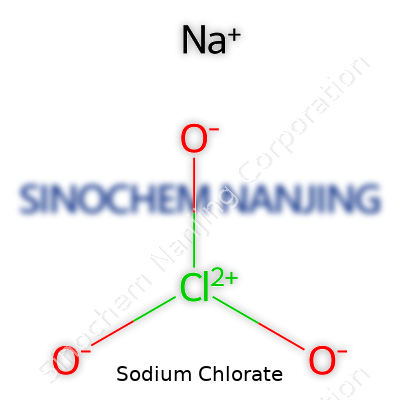
| Names | |
| Preferred IUPAC name | Sodium chlorate |
| Other names |
Chlorate of soda Sodium chlorate(V) Soda chlorate UN 1495 |
| Pronunciation | /ˈsəʊdiəm ˈklɔːreɪt/ |
| Identifiers | |
| CAS Number | 7775-09-9 |
| Beilstein Reference | 803134 |
| ChEBI | CHEBI:32144 |
| ChEMBL | CHEMBL1086634 |
| ChemSpider | 14129 |
| DrugBank | DB11081 |
| ECHA InfoCard | 100.028.760 |
| EC Number | 231-887-4 |
| Gmelin Reference | 22390 |
| KEGG | C07397 |
| MeSH | D017379 |
| PubChem CID | 24586 |
| RTECS number | SC8640000 |
| UNII | 9A2M9QSJ87 |
| UN number | UN1495 |
| Properties | |
| Chemical formula | NaClO3 |
| Molar mass | 106.44 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 2.49 g/cm³ |
| Solubility in water | Very soluble |
| log P | -6.42 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 1.15 (for HClO3) |
| Basicity (pKb) | -11 |
| Magnetic susceptibility (χ) | -42.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.445 |
| Viscosity | 0.89 mPa.s (at 20°C, for aqueous solution) |
| Dipole moment | 4.33 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 247.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –385.0 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | −364.8 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V03AB06 |
| Hazards | |
| Main hazards | Oxidizing solid, may intensify fire; toxic if swallowed; harmful if inhaled or in contact with skin; causes severe eye irritation. |
| GHS labelling | GHS02, GHS06, GHS09 |
| Pictograms | GHS03,GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H272, H302, H318, H410 |
| Precautionary statements | P210, P220, P221, P260, P264, P270, P273, P280, P301+P310, P304+P340, P305+P351+P338, P306+P360, P310, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-1-OX |
| Autoignition temperature | 250°C (482°F) |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 oral rat 1200 mg/kg |
| LD50 (median dose) | 1,200 mg/kg (oral, rat) |
| NIOSH | PY8050000 |
| PEL (Permissible) | PEL: 10 mg/m³ (as total dust) |
| REL (Recommended) | 30 mg/m³ |
| IDLH (Immediate danger) | 250 mg/m³ |
| Related compounds | |
| Related compounds |
Potassium chlorate Sodium chlorite Sodium perchlorate Sodium hypochlorite Sodium sulfate |

