Sodium Cerium Nitrate: A Closer Look at a Modern Compound
Historical Development
Curiosity tends to push science forward, and sodium cerium nitrate is a good example of what happens when the rare earth elements catch a chemist’s eye. As the periodic table filled out in the late nineteenth and early twentieth centuries, folks started mixing these lesser-known metals with common salts, carving out a new family of compounds. Sodium cerium nitrate began as more than just an experiment; it turned into a workhorse salt for labs and industry, right around the time rare earth chemistry had its boom thanks to new mining technologies and practical uses for cerium-based mixes. This compound started as something of a scientific oddity, but soon after, its vivid color and reactivity drew attention from environmental chemists and material scientists. It spoke to the intersection of tradition—pulling rare earths from notoriously difficult ores—and new possibilities for medicine, glass, and catalysis. Every new use for cerium-based salts like this one has roots in decades of tough extraction, clever purification methods, and plenty of scientific curiosity about what these elements could really do.
Product Overview
Sodium cerium nitrate stands out with its pale yellow tint, almost lemony when you peer at a beaker in the right light. This compound walks a line between being a research staple and a toolkit item for specialty processes. It owes a lot to its parent elements: sodium keeps things water-friendly, cerium brings the hefty atomic weight and quirky chemistry, and nitrate cranks up the solubility. Most labs get it in granular or powder form, sealed up tight because it draws moisture quickly. Open a fresh container and the slightly metallic scent is noticeable, almost marking it out among other rare earth salts. Buyers tend to check for purity levels, usually topped off above 99%, because even small impurities can throw off experiments or industrial runs. Being sensitive to environmental shifts, it calls for dry storage and careful handling—sloppy storage turns it into a clumpy mess, and nobody enjoys scraping chalky deposits from flasks.
Physical & Chemical Properties
The beauty of sodium cerium nitrate lies in its crystalline makeup. At room temperature, it holds its shape as a glassy yellowish powder, dissolving quickly in water, leaving barely a trace behind. This compound boasts a decent melting point, hovering around 70–80°C, with decomposition triggering before a full liquid state arrives. Its hygroscopic nature means it hungrily draws in water, swelling if exposed long enough. Chemically, cerium is a showoff here: switching oxidation states, catching and releasing electrons with surprising ease. In nitrate form, the compound behaves as an oxidizer, helping kick-start redox reactions with organic and inorganic materials alike. Mix it with acids and it holds steady, but throw a strong reductant in, and the cerium toggles states, sometimes settling into white cerous or orange ceric products, depending on who wins the chemical tug-of-war.
Technical Specifications & Labeling
Every bottle of sodium cerium nitrate worth buying lists its chemical formula—NaCe(NO3)4—front and center. Common lot certificates outline not just purity (often >99% trace metals basis), but also moisture content, color, and sometimes trace element profiles, usually reported by weight percent down to the ppm. Labels flag hazard statements clearly: “Oxidizer,” “Irritant,” and water-reactivity warnings. Regulatory compliance sits on the label too, whether it’s a CAS number or an EC code, plus the recommended storage—cool, tight-sealed, moisture-free. MSDS documents, required by reputable suppliers, walk through pictograms and first-aid steps with less formality than federal regulations might dictate, but they get the job done for busy researchers. Each batch sports a unique identification code, making it easier to track product recalls or troubleshoot a suspicious set of results.
Preparation Method
Making sodium cerium nitrate doesn’t take arcane alchemy, but it needs a solid grasp of wet chemistry technique. Most production runs start with cerium oxide—the raw stuff from monazite or bastnäsite ore. First, acid digestion converts cerium oxide to cerium nitrate with a strong shot of nitric acid. Once the ceaseless fizzing and warmth from neutralization settle, sodium nitrate gets added in the right stoichiometric amount. Careful evaporation crystallizes out the target compound. The powder gets rinsed, sometimes under vacuum, to drive off stray nitrate or acid. Drying in a desiccator wraps up the job. This multistep process turns ore material—once little more than mining tailings—into a pure, research-grade salt, all in the space of a few days. Every step demands skill, since a wrong pH or temperature slip leaves behind a mess of insoluble residue and wasted reagents.
Chemical Reactions & Modifications
Sodium cerium nitrate jumps into redox reactions without much coaxing. In organic synthesis, chemists use it as an oxidant to flip alcohols into aldehydes and ketones, saving time compared to fussier, less selective reagents. When paired with strong acids, it keeps its color and form, but bring in a reducing agent—one with teeth like sulfites or ferrous salts—and it drops the tell-tale yellow, leaving behind colorless cerous byproducts. It gets along poorly with strong bases, leading to hydrolysis and loss of nitrate groups, sometimes forming stubborn cerium oxides. Researchers tinker with this compound, combining it with other rare earth nitrates, hoping to unlock unique magnetic or electrical properties in custom crystal lattices. Even a simple adjustment, like switching sodium for potassium, shifts reactivity enough to change how it anchors in glass or ceramics, showing just how tunable rare earth salts can be.
Synonyms & Product Names
Trade names and synonyms flow freely in the chemical supply world. Sodium cerium nitrate pops up as sodium ceric nitrate or sodium cerium(IV) nitrate, depending on the local naming conventions. Some catalogs lean on “cerium sodium nitrate double salt,” a mouthful but useful in telling it apart from simpler mixtures. The CAS registry number—16774-21-3—keeps confusion down, though a handful of aging papers still cite outdated nomenclature. Manufacturers sometimes label it as “Nitrate, sodium-cerium(IV)” for compliance in export or regulatory filings. The naming shuffle means researchers double-check before starting large-scale syntheses or orders, especially when purity and hydration states matter so much for reproducible results.
Safety & Operational Standards
Handling sodium cerium nitrate calls for respect, not paranoia. The bright yellow warning stickers hint at the real risks: oxidizing power, skin and eye irritation, potential environmental harm if it escapes down a drain or onto soil. Most users reach for gloves and goggles before opening even a small sample, since the fine powder finds every small cut or damp patch on exposed skin, leading to rashes or discomfort. Good ventilation stops nitrate dust from lingering in the air, and any accidental spill gets tackled with damp cloths, not vacuums—nobody wants to ignite fine oxidizing powders inside a cleaning device. Lab safety sheets advise against eating or drinking near the compound, a rule that seems obvious until long shifts blur the line between coffee breaks and benchwork. Storage in powder-tight containers, away from acids and organic solvents, prevents weird reactions and keeps the salt ready for the next experiment. Disposal goes through hazardous waste channels, never mixed with regular garbage or sink waste.
Application Area
This salt earns its keep in glass polishing, ceramics doping, analytical chemistry, and even the odd clinical application. In glassmaking, sodium cerium nitrate replaces toxic alternatives, refining texture and adding a yellow hue that softens glare from fluorescent bulbs or smartphone screens. Analytical chemists trust it as a redox indicator, identifying subtle shifts in complex titrations involving iron, manganese, or other transition elements. Occasionally, it pops up in dental pastes or burn ointments, where cerium ions show mild antimicrobial action, though such uses stick to strict, regulated limits. Those working in catalysis appreciate how readily the cerium toggles oxidation states, boosting reaction efficiency in everything from fuel cells to water purification. The niche uses keep growing: researchers mix it with other rare earths to push boundaries in quantum dot manufacture, superconductors, or even magnetic memory storage.
Research & Development
Research on sodium cerium nitrate never sits still. Material scientists keep uncovering how the compound’s redox agility unlocks new reaction pathways or boosts energy storage density in custom battery chemistries. Studies funded by green chemistry grants focus on safer ways to handle and recycle cerium-based materials, banking on sodium cerium nitrate as a benchmark test reagent. Environmental chemists hope to harness its oxidizing chops for pollutant breakdown, potentially treating industrial wastewater streams with fewer secondary hazards. Teams across Europe and Asia compete to design ever-purer, more customizable versions, with particle sizes small enough for nanotechnology or colloidal suspensions. Medical research investigates its low-level antimicrobial activities, testing for safe use in wound care where traditional antibiotics struggle with resistant strains. The diversity of cerium chemistry just keeps fueling new grant proposals and out-of-the-box applications, and the published science climbs year after year.
Toxicity Research
Every new compound finds itself under the toxicology microscope. Sodium cerium nitrate’s risks fall on the moderate side: it irritates skin and mucous membranes, never as severe as mercury or chromium salts, but annoying enough to demand strict laboratory hygiene. Animal studies show low oral toxicity, yet higher doses or chronic exposure build up in vital organs, raising concerns about bioaccumulation. Environmental release causes trouble for aquatic life, turning cerium ions into stressors for certain fish and algae species. Researchers dig into long-term exposure risks for workers, measuring air and dust concentrations in manufacturing plants. So far, links to cancer or major systemic disease haven’t borne out, but regulatory agencies don’t let their guard down. Every new application triggers another round of safety reviews, driven by the basic principle: no benefit comes without a check for hidden costs. The compound’s fingerprint is now on various toxicology and risk databases, sometimes flagged for further research before use in food or medicine gets widespread approval.
Future Prospects
Outlook matters, especially for compounds straddling specialty chemistry and mainstream applications. Sodium cerium nitrate sits at a crossroads: glass and ceramics industries count on its color control and polish, while environmental sectors eye its potential for green remediation. Battery researchers test it as a model material for next-generation anodes; analysts lean on it for sharper, more accurate titrations. As the tech sector chases smaller, faster electronic parts, rare earth salts—including this one—may lead the way in advanced manufacturing or low-waste optical coatings. Medical innovators keep searching for sweet spots where antimicrobial properties outshine side effects, hoping to slip cerium-based blends into new therapies. The push for sustainability and supply security makes recycling and safer handling a big talking point, possibly fueling greener preparation routes and leaner industrial footprints. Progress won’t stall—each breakthrough promises a compound less known by the public but increasingly vital behind the scenes where high-tech meets practical daily life.
What is Sodium Cerium Nitrate used for?
Not Just Another Lab Chemical
Sodium cerium nitrate doesn’t land in the news cycle every week, but from time to time, it gets attention for good reason. People like me who spent their college years hunched over glassware know this substance mainly as an oxidizing agent in chemical labs. In organic chemistry, it gives chemists a way to spot alcohols in unknown compounds during classic tests, clearing up mystery mixtures that can’t get identified by smell or color. Put a suspicious substance in a test tube, add sodium cerium nitrate, watch for a color change—that’s a direct answer no fancy spectrometer needed.
Vital Tool for Eye Care
In eye clinics and hospitals, sodium cerium nitrate has carved out a useful space. Used in some eye drops, its job includes cleaning corneal surfaces, breaking down foreign material better than saline alone. It’s about providing relief when simple solutions fall short, especially with chemical injuries—those incidents happen more often than people think, and the faster someone gets the right rinse, the better their odds of keeping healthy vision. This is a tool that eye specialists rely on for emergencies as well as routine care.
Glass Polishing—A Surprisingly High-Tech Application
Most folks don’t realize how much work goes into making lenses and phone screens flawless. The factories producing them won’t get far without a chemical like sodium cerium nitrate. It smooths and polishes glass, removing fine scratches without damaging the precision shape. The process isn’t as simple as rubbing with a cloth—it involves chemical reactions that clean up what machines leave behind. Polishing compounds with this rare-earth salt help keep everything from your eyeglasses to telescope mirrors crystal clear. Polishing glass used to require more elbow grease and more basic abrasive materials. Since this salt entered the scene, finished products look sharper and last longer.
Environmental and Safety Factors
Like many strong chemicals, sodium cerium nitrate calls for care during disposal. Companies can’t dump water contaminated with it straight down the drain without risking trouble with regulators. It doesn’t belong in local water systems, and even in small amounts, the build-up doesn’t help wildlife or people. There’s responsibility here for anyone—lab tech, factory worker, hospital staff—to follow safety instructions. From my days working in a chemistry storeroom, labeling and secure storage made all the difference in staying accident-free.
Pursuing Safer, Smarter Solutions
More sustainable practices in manufacturing often come down to careful inventory tracking, safer waste disposal, and hunting down new polishing or cleaning compounds with gentler environmental footprints. Hospitals and factories that train people properly and follow evidence-based procedures improve not just patient outcomes and product quality, but also protect their staff and surrounding communities. Technology keeps evolving, and staying informed means better decisions about which chemicals get used, and how they get handled from first use to final disposal.
Is Sodium Cerium Nitrate safe to handle?
Real Safety Matters: Sodium Cerium Nitrate in the Lab
Sometimes people hear about rare-sounding chemicals and picture something right out of a science fiction movie—glowing liquids or clouds of weird fumes. Sodium cerium nitrate isn’t radioactive or out of the ordinary in that sense, but living with it on lab shelves has shown me some basics everyone handling chemicals should respect. This compound has a legitimate place in industry and research. In some labs, it helps analyze trace elements or runs in glass-polishing processes. The big question isn’t whether it has a purpose, but whether using it comes with invisible risks.
Looking Beyond the Name: What Happens with Contact
Holding a jar of sodium cerium nitrate feels no different from other powders, yet it’s not the sort of thing I’d toss around. There’s a simple reason: this chemical’s hazards come from its ability to irritate skin, eyes, and the lungs. Some colleagues got lazy on a Friday afternoon once and wiped up a small spill using paper towels, no gloves. Both ended up with nasty red patches on their hands. The safety sheet makes it clear—direct contact can burn, especially if you touch your face later. Some people act tough about these warnings, but that’s how trips to medical get logged.
Dust comes next. If you pour this powder or use it in solution, you can’t always see airborne particles. It only takes one accidental gust near a beaker to catch a bit of dust in your eye or inhale something, then regret it. Studies show respiratory irritation can hit surprisingly quickly, even if you aren’t prone to allergies.
Long-Term Risks and Regulation
People sometimes ask, “Will this stuff give me cancer?” There’s no huge database pointing to carcinogenic effects for sodium cerium nitrate itself. The science doesn’t rub shoulders with asbestos or lead, but repeated exposure never turns out well in the chemical world. The Environmental Protection Agency lays out clear storage and handling protocols. You’ll find this compound on most safety data sheets, with the same best practices you’d see for similar oxidizers: eye protection, gloves, proper ventilation, good labeling. These aren’t just hoops labs jump through; they come from actual incidents logged over years.
Lessons from Real-World Experience
Chemical safety instruction used to bore me to tears. All those rules and labels seemed like overkill. Then during my own undergrad years, a friend splashed a nitrate in their eye and spent hours at the campus clinic. The stories about burns aren't just anecdotes for newcomers—they’re warnings that even “safe” handling can turn ugly if you ignore basic prep. The difference between a routine day and an ER visit comes down to the effort put into goggles, gloves, and checking the fume hood’s flow.
Staying Safe Isn’t Complicated
Lab work faces enough pressure without taking shortcuts. Sodium cerium nitrate won’t leap out of its bottle, but its risks are real. Ventilation, eye protection, gloves, and washing up afterward cut those risks way down. Secure storage, careful labeling, and clear spill protocols go further. Chemical safety works like seatbelts—useless if ignored, lifesaving in a pinch.
So, is sodium cerium nitrate safe? It’s only as safe as the mindset of the person handling it. With routine care and a respect for protocols, trouble stays at bay. Getting too casual or treating any lab chemical like dried soup mix never pays off.
What are the storage requirements for Sodium Cerium Nitrate?
Why Care About Storage?
Sodium cerium nitrate sits on the shelf in many labs and factories, but not everyone stops to think about how easy it is to ruin a whole batch with careless storage. Unlike table salt or sugar, this chemical brings a set of quirks. If you store it wrong, you face clumping, annoying impurities, or worse—potential safety hazards. Everyone who’s had to write an incident report because someone skipped storage rules knows how much hassle that piles onto a busy day. Avoiding those headaches starts with respecting a few key facts about this compound.
Moisture Is the Enemy
This material grabs water from the air. Leave an open bottle out, and you’ll find a soggy mess by morning, with the contents sometimes fusing into one big lump. Some labs report that a single humid afternoon spoiled a whole batch. That means you need airtight containers—preferably glass or HDPE with a reliable seal. Toss in a desiccant canister and you shrink the risk of hydration, which can throw off weighing and dissolving later. Most manufacturers call for storage in a cool, dry spot, temperatures around room temperature. Even in air conditioning, moisture sneaks in, so proper sealing trumps everything else.
Sunlight and Heat Speed Up Decay
Sodium cerium nitrate can break down if left on a sunny windowsill. Direct UV light slowly messes with its oxidation state. Over the months, that adds up to unreliable results in sensitive applications. Old-timers always put this stuff on a low shelf, away from sunlight and distance from heaters. Any high heat storage—think right next to a furnace, or under a steam pipe—can push the compound past its prime quickly. Temperatures over 30°C increase degradation risks and might even trigger slow release of nitrate fumes. Keeping it cool and shaded isn’t just good practice; it supports safety and product consistency.
The Trouble With Cross-Contamination
Open a jar of sodium cerium nitrate on a crowded benchtop and tiny slips happen. Powders cross-contaminate easily, especially in busy environments. Airborne dust from spilled samples can end up miles away (anyone who’s done a proper lab wipe-down knows what hides behind those reagent shelves). Trained workers label everything and store chemicals apart. Some labs keep sodium cerium nitrate in a dedicated chemical cabinet with other rare earth salts. That small step keeps things cleaner and avoids accidental mixing with acids or organics, which could cause unwanted reactions or extra work cleaning up.
Why Trust Actually Matters Here
Patients, researchers, and quality control officers rely on chemicals acting as promised. If storage cuts into the accuracy of sodium cerium nitrate, it isn’t a victimless inconvenience. In fields ranging from optics to catalysis, batch failures or unpredictable outcomes cost time and sometimes reputation. Mishandling even small amounts makes the difference between a reliable experiment and repeat failures. Not all mistakes look big at the start, but over time, small storage slip-ups snowball.
Practical Steps that Work
Set aside one seal-tight bottle—never scoop from the original container. Use a spoon or spatula reserved just for this compound. Regularly check for moisture damage, discoloration, or clumps. Rotate stock so old bottles don’t decay at the back of the cabinet. If you store chemicals in bulk, add an extra desiccant pack to each bin. Don’t store sodium cerium nitrate near acids or flammable solvents. Routine checks and a quick review of storage practices every few months keep things safe and reliable. Mistakes cost more than a little time; they eat away at trust in everything produced by the lab or plant.
What is the chemical formula of Sodium Cerium Nitrate?
Clearing Up Chemical Confusion: Formula Matters
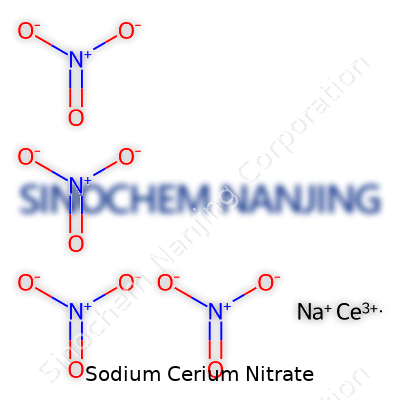
Sodium Cerium Nitrate often pops up in scientific labs, electronics research, and glass polishing facilities. That odd mix of uses reflects its unique make-up. In chemistry, a clear formula spells out exactly what people work with. The formula for Sodium Cerium Nitrate is NaCe(NO3)4.
Some chemistry classes toss this formula around without much context—just another mouthful among dozens. Yet, the details pack a real punch in fields that rely on getting proportions right every time. In my schooling, folks sometimes mixed up the sodium and cerium. That’s an expensive mistake in a college lab, let alone in a manufacturing setting. Knowing this isn’t just book learning: this knowledge keeps operations precise, which can save both time and money. The difference between working with cerium(III) and cerium(IV) compounds illustrates how chemistry formulas act as critical roadmaps, not just labels.
Why This Stuff Shows Up Everywhere
Sodium Cerium Nitrate’s formula matters because each atom pulls weight in how the compound performs. Cerium stands out with its ability to shift between different oxidation states—plus, it’s one of the rare earth elements most people outside chemistry circles never think about. In this compound, it rides along in the +4 state. Add in sodium, a common stabilizer, and four nitrate groups, and the result packs serious oxidizing power.
This combination shows up in real-world settings. Glass polishers love it for buffing out scratches so deep you’d swear they’d never come out. Electricians and researchers use it to grow single crystals for electronics. In personal experience, I watched a classmate waste an afternoon fighting cloudy glass, only to discover the problem came from picking the wrong cerium source. The right chemical formula delivers results—no substitutes.
Getting the Facts Straight: Avoiding Costly Errors
Misreading a chemical formula in industry can mean big headaches. Picture a technician ordering Ce(NO3)4 without sodium, hoping to save money or streamline a purchase. Once production halts because the material behaves differently, the error becomes obvious—too late to fix cheaply. It pays to double-check. Not just for safety, but for consistent quality.
Reliable public databases and supplier sheets post the formula as NaCe(NO3)4. Government and academic sources—including Ulrich Müller’s “Inorganic Structural Chemistry”—confirm this identification. Journals in applied chemistry highlight the exact formula during studies of industrial glass polishing and advanced ceramics work. That’s how trust gets built: through documentation and repeated testing.
Looking Ahead: Encouraging Smarter Use
Schools and companies could step up their approach to teaching formulas. Adding examples, emphasizing hands-on practice, and connecting formulas with everyday mistakes makes chemistry less abstract. On the manufacturing side, clear labeling and staff retraining help prevent incorrect orders and lost hours.
When chemical formulas come off the page and land in a toolbox, it changes the way workers, students, and researchers solve problems. Using NaCe(NO3)4 as more than a trivia answer means fewer errors and more innovation. In a world stuffed with brands and trade names, staying grounded in the real formula keeps science exact—and that’s what propels real progress.
How should I dispose of Sodium Cerium Nitrate?
Talking Straight About Chemical Disposal
Lots of folks working in labs, especially those just diving into chemistry, eventually bump into bottles labeled “Sodium Cerium Nitrate.” Maybe you’re cleaning out a high school stockroom, or maybe your garage is closer to a DIY chemistry corner. Let’s be real: figuring out what to do with leftover chemicals rarely gets spelled out in plain English.
I’ve dealt with old chemical stores in schools and small labs a few times. I’ve seen people shrug and pour things down the drain, thinking a rinse with water makes it all go away. But sodium cerium nitrate is an oxidizing agent, posing real risks to plumbing, sewer workers, and downstream aquatic systems. Pouring it down the drain, especially without neutralization, can leach into groundwater. I once saw an entire row of plants along a schoolyard wither because some old nitrate compound got dumped nearby. It taught me quick: the environment keeps the tab on our shortcuts.
What’s in This Stuff, Anyway?
Sodium cerium nitrate mixes two metals—sodium and cerium—with nitrate, which means you’re handling something with both reactivity and toxicity. It’s used in analytical chemistry. Not fertilizer, not food—nobody wants this drifting into community water supplies. The Environmental Protection Agency (EPA) and local hazardous waste programs never list this chemical among their “safe for drain” compounds. That’s a warning, not a suggestion.
Real-World Disposal—No Shortcuts
Every year, thousands of tons of chemical waste turns up in landfills because people imagine that trash bins make things vanish. Anyone who’s sorted through chemical disposal day at college knows how messy this gets—paint cans with god-knows-what still inside, containers of crystalline orange nitrates. Even a few grams of a compound like sodium cerium nitrate can start a fire if mixed with organic trash. Forget about regular trash bins—these risk not just minor accidents, but fires or toxic smoke right in a disposal truck.
So, what do the pros do? They work with hazardous waste collection programs. Many cities have days when households can drop off old chemicals—these collections funnel that waste to certified handlers. For businesses or labs, the law says hazardous waste gets labeled, logged, and picked up by licensed contractors. The reason? Improper disposal can bring fines, lawsuits, and more important, soil and water contamination that doesn’t just vanish. If you’re unsure, never just guess. Local agencies and universities often post chemical lists and have hotlines for disposal guidance. One quick phone call has saved me from embarrassing and costly mistakes.
Lessons From Practice
Anyone who’s mopped up a beaker spill or pulled a bottle off a dusty shelf knows chemistry isn’t just theory. If you find yourself with sodium cerium nitrate, treat the job like you’re handling a loaded power tool—you don’t “just toss it” anywhere you please. If a school’s chemical hygiene officer or safety officer is in place, let them know. If you’re on your own, look up your city’s hazardous waste collection dates. Always label the container clearly and make sure lids are secure. If the chemical looks crusty or the label is peeling, double-bag it before handing it off.
Everything we flush or toss away floats downstream, literally and metaphorically. Choosing the right disposal saves rivers, drinking water, and sometimes just keeps the plumber’s day a little less exciting. Respect the stuff, ask for expert support, and let real hazardous waste handlers close out the story.
| Names | |
| Preferred IUPAC name | sodium;cerium(4+);dinitrate |
| Other names |
Cerium(III) sodium nitrate Cerium sodium nitrate Sodium cerous nitrate |
| Pronunciation | /ˈsəʊdiəm ˈsɪəriəm ˈnaɪtreɪt/ |
| Identifiers | |
| CAS Number | 10102-17-7 |
| Beilstein Reference | 3529783 |
| ChEBI | CHEBI:91283 |
| ChEMBL | CHEMBL1201648 |
| ChemSpider | 12678714 |
| DrugBank | DB09210 |
| ECHA InfoCard | 100.011.783 |
| EC Number | 234-900-1 |
| Gmelin Reference | 10803 |
| KEGG | C18636 |
| MeSH | Sodium Cerium Nitrate |
| PubChem CID | 159629 |
| RTECS number | VV8510000 |
| UNII | KFQ5T35V7J |
| UN number | UN3264 |
| Properties | |
| Chemical formula | Na₃Ce(NO₃)₆ |
| Molar mass | 568.01 g/mol |
| Appearance | Light yellow powder |
| Odor | Odorless |
| Density | 2.15 g/cm3 |
| Solubility in water | Soluble in water |
| log P | -4.68 |
| Basicity (pKb) | 7.8 |
| Magnetic susceptibility (χ) | +2250.0e-6 cm³/mol |
| Refractive index (nD) | 1.565 |
| Viscosity | Viscous liquid |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 253.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1648.6 kJ/mol |
| Pharmacology | |
| ATC code | V03AB33 |
| Hazards | |
| Main hazards | Oxidizing, harmful if swallowed, causes serious eye irritation, may cause respiratory irritation |
| GHS labelling | GHS05, GHS07, GHS08 |
| Pictograms | GHS05,GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H272, H315, H319, H335 |
| Precautionary statements | P261, P280, P304+P340, P305+P351+P338, P312 |
| NFPA 704 (fire diamond) | 2-0-0-OX |
| Lethal dose or concentration | LD50 Oral - rat - 4,200 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 4300 mg/kg |
| NIOSH | Not established |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Sodium Cerium Nitrate: Not established |
| REL (Recommended) | 500 mg |
| IDLH (Immediate danger) | Not listed. |
| Related compounds | |
| Related compounds |
Cerium(IV) sulfate Cerium(IV) oxide Ammonium cerium(IV) nitrate |

