Sodium Butoxide: Practical Insights into a Classic Alkoxide
A Journey from Lab Curiosity to Useful Reagent
Sodium butoxide stands out in the world of alkoxides for reliable performance, crisp reactivity, and solid versatility. Back in the early days of synthetic organic chemistry, researchers noticed sodium metal’s strong reaction with primary alcohols and tried their hands with butanol. The result, sodium butoxide, stepped onto the scene around the turn of the nineteenth century as chemists demanded tools for creating new bonds and exploring different molecular architectures. Industrial adoption came once researchers found ways to produce it on a larger scale, and today it has a regular presence in the labs of both academia and industry. This reagent now often fuels the backbone of key research, from pharmaceutical synthesis to new materials.
At a Glance: Product Overview and Reliable Properties
What sets sodium butoxide apart isn’t flash or novelty but trusty dependability. Looking at it in the bottle, you see a white or off-white powder, sometimes granular, sometimes a bit waxy. It dissolves in butanol and other organic solvents, holding up well in tightly sealed bottles. Leave it out in humid air, and it starts reacting with moisture, so storage jars always stay closed unless in use. Its melting point lands above many common laboratory alkoxides, and it gives off a distinctly strong, almost medicinal odor. Sodium butoxide is basic, with a pKa of its conjugate acid (butanol) high enough to deprotonate a decent range of active methylene compounds, yet not as aggressive as sodium tert-butoxide. This makes it a solid all-rounder in synthesis where extreme aggression from the base is not required.
Looking at Labeling and Technical Specifications
A sodium butoxide bottle generally carries information about purity (standard is often above 95 percent), residual sodium hydroxide content, and water content. Too much moisture spells trouble for sensitive knock-on reactions. Laboratory-grade sodium butoxide usually lists butanol residue and contaminant levels. These figures end up mattering when making pharmaceuticals or electronics intermediates, as impurities can chain-react or deactivate precious catalysts. For any researcher paying attention to detail, clear and accurate numbers on the label really make the difference between a straightforward workflow and frustrating reruns.
Preparation: Classic yet Robust
Open any synthetic procedures compilation, and you’ll find a section on sodium alkoxides. Sodium butoxide comes to life by reacting pure butanol with freshly cut sodium metal under a nitrogen or argon atmosphere. The prototypical recipe means dropping sodium chunks into butanol, gently heating the mix, and waiting for gentle bubbling of hydrogen to subside. Filtering the final solution and removing butanol under vacuum leaves the solid alkoxide. It’s hardly changed since the times of early organic chemists—sometimes classical methods stick for a reason. It's risky business without proper lab technique, as sodium metal reacts violently with water. Industrial plants use enclosed systems, minimizing human risk while scaling batches to hundreds of kilos at a time.
Real Chemistry: What Sodium Butoxide Makes Possible
What’s special about sodium butoxide is how it helps chemists build carbon-carbon bonds, prepare ethers by Williamson synthesis, and drive elimination reactions. Its moderate bulkiness assists in deprotonating certain molecules without always going too far into elimination or side reactions. It plays a key role in the preparation of β-keto esters and other intermediates. By fine-tuning conditions—solvent, temperature, and concentration—one can harness its basicity for specific transformations. Sodium butoxide enables regioselective reactions in aromatic substitutions and aids in transesterification processes where weaker bases would fail to drive the reaction to completion. In more specialized uses, its sodium ion can be exchanged for transition metals, leading to tailored catalysts. Organic chemists rely on these properties to solve stubborn synthetic challenges.
Friends and Names in Other Contexts
Sodium n-butoxide or sodium 1-butanolate often appear as alternative names for this alkoxide. In scientific and technical circles, the term “sodium butylate” crops up as well, though that can cause confusion with the methylate and ethylate analogs. CAS numbers and other identifiers help reduce miscommunication, especially when working across international teams who use different shorthand. No matter what label sits on the bottle, the chemistry stays predictable, as long as the formula matches up.
Safety and Operational Good Sense
Talking about sodium butoxide means talking about safety. It’s both caustic and reactive with moisture, producing butanol and sodium hydroxide, as well as heat. Splashing it on skin or eyes sends folks straight to the safety shower or eyewash. Fume hoods and dry atmospheres aren’t negotiable. Sodium butoxide also reacts with acids, so cross-contamination on messy benchtops causes problems. Professional users always keep the material tightly sealed and never pipette by mouth—common sense, but good to reiterate for new users. Lab veterans have stories of what happens if containers are left open under humid conditions—solid cakes form and lose potency. Facilities focused on industrial-scale use adopt closed-system engineering controls to reduce spills and airborne dust.
Where It Makes a Mark: Broad Applications
Over the years, sodium butoxide found a productive niche in pharmaceuticals, fine chemicals, and polymer derivatization. It’s a favorite for alkylating agents and base-catalyzed condensations. Pharmaceuticals count on the consistency sodium butoxide brings for synthesizing intermediates where enolate rearrangements or controlled elimination steps must run smoothly. Resin producers and advanced plastics manufacturers count on its capacity for ring-opening polymerizations, especially with monomers sensitive to stronger or more nucleophilic bases. In research labs, sodium butoxide finds a home both on shelves for routine applications and as part of ambitious multi-step syntheses. Universities regularly include it in advanced organic chemistry curricula to help students understand both power and limits of base strength.
Research, Development, and Tweaks in Chemistry
Research groups continue to push sodium butoxide’s chemistry in new directions. Some teams investigate its role as a promoter for cross-coupling reactions or as a starting point for new catalyst systems where the balance between nucleophilicity and basicity matters more than brute force. Others probe its impacts on selectivity in asymmetric synthesis, especially in preparing privileged motifs for pharmaceuticals. Modifications, such as combinations with crown ethers or cryptands, allow sodium butoxide to function in less polar solvents or even for phase-transfer catalysis. It isn’t a material that breaks the rules, but each year, published research uncovers new tricks where its steady hand brings tangible value.
Watching for Hazards: Toxicity and Regulation
As much as sodium butoxide is useful, its hazards deserve respect and thoughtful attention. Accidental ingestion or inhalation can cause severe burns, and contact with eyes risks permanent injury. The breakdown products—sodium hydroxide and butanol—bring their own toxicological profiles, both as irritants and as potential industrial contaminants. Regulatory agencies require clear labeling, restricted workplace exposures, and prompt disposal of spills. Safety data for sodium butoxide points to the importance of working under dry conditions, wearing gloves and goggles, and handling spills with caution. Waste disposal protocols demand neutralization and dilution before landfill or incineration. The chemical does not persist in the environment, but accidental release can still harm aquatic systems due to rapid changes in pH and organics load.
Looking Ahead: The Future of Sodium Butoxide Chemistry
The prospects for sodium butoxide remain robust. Sustainability concerns and green chemistry principles drive researchers to re-imagine how base-promoted reactions operate. Areas like biobased plastic synthesis and environmental catalysis stand to benefit from this reagent. As synthetic demands shift toward more selective, atom-efficient transformations, sodium butoxide’s measured strength may offer an edge over less predictable alternatives. Its ease of preparation from renewable butanol may also play a role in greener supply chains. Drug discovery chemists keep an eye on new applications, particularly when modifications to classics like sodium butoxide can help avoid costly purification or hazardous waste. As chemistry finds new frontiers, this tried-and-true reagent stands ready, familiar and adaptable, for the next wave of discovery.
What is Sodium Butoxide used for?
A Chemical with Quiet Power
Most people never think about chemicals like sodium butoxide. While the name doesn't exactly roll off the tongue, it’s a real workhorse in the world of chemistry. I remember my early days in a university research lab, and we kept a jar of sodium butoxide locked away in the chemical store room. Safety goggles on, gloves snapped tight, and not a whiff of the stuff—handling it always meant we meant business. This compound isn’t flashy, but it has a big impact, especially for folks working with complex molecules.
Getting Down to Business: Why It Matters
Sodium butoxide usually shows up in the toolbox of organic chemists. This chemical moves reactions forward, especially in the creation of medicines and advanced materials. It acts as a strong base, which means it helps pull hydrogen atoms off molecules. This sounds geeky, but it’s a lot like having a crowbar that can gently pry apart tough bonds, letting scientists stitch molecules together in creative and useful ways.
Pharmaceutical companies use this stuff to build more effective drugs. Someone designing a painkiller or a cancer medicine may need to shift parts of a molecule around, flipping one atom for another. A strong base like sodium butoxide makes that swap possible, setting the stage for the next step in a long and winding chemical journey.
Beyond the Lab: Real-World Uses
This chemical is not just for science fairs or graduate students. The paint and coatings industry depends on sodium butoxide for specialty polymers. Everyday objects like resistant paints or scratch-proof plastics impress us with their looks and durability, but what impresses me most is the chemistry behind them. Companies pump out gallons of tailored polymers only because chemicals like this make the synthesis possible.
Fuel producers, especially in the biodiesel field, turn to sodium butoxide when they convert plant oils into usable fuel. The process—transesterification—uses sodium butoxide to strip away unwanted chunks and rework molecules into something that burns clean. Having seen the push toward sustainable energy with my own eyes, I know how every step forward matters, and chemicals like this one are quietly nudging things in the right direction.
Health, Safety, and Environmental Questions
Every tool comes with a catch, and sodium butoxide is no different. This stuff doesn’t exactly cooperate if it gets on your skin or into your eyes. Back in the lab, anything flammable or caustic demanded respect. The chemical reacts violently with water and can release butanol, itself not a friend to lungs or wildlife. Factories and research centers use engineered ventilation systems, and they train staff for spills. The safety protocols weren’t just red tape—they were vital.
A big push is happening for greener chemistry. Researchers are searching for alternatives that do the same job but create less waste or soften their environmental footprint. In the meantime, careful disposal and best-practice safety keep risk down.
Looking Forward
Sodium butoxide keeps the wheels turning—sometimes literally, as with biodiesel. Its main interests lie in making new medicines, better plastics, and safer fuels possible. It isn’t glamorous, but I’ve seen firsthand how much depends on tools like this. Respect for the chemical, and the people who use it wisely, goes a long way in ensuring progress and safety keep pace.
Is Sodium Butoxide hazardous or toxic?
Understanding What Sodium Butoxide Really Is
Sodium butoxide sounds unfamiliar to most folks outside a chemistry lab. This compound, made from sodium and butanol, finds use in making pharmaceuticals and other specialty chemicals. Its real purpose comes into play during chemical reactions that need a strong base. Even though it doesn’t show up in household goods, sodium butoxide occasionally pops up in news about chemical safety and toxic spills.
Sizing Up the Hazard
Chemists know sodium butoxide doesn’t play nicely with water or skin. Drop a bit on your hand, and you’ll feel a burning sting, not just a simple itch. Most of us have handled degreasers that warn about skin irritation, but sodium butoxide takes that a notch higher. I’ve learned from years of working in labs that not wearing gloves around chemicals like this can quickly turn a normal workday into a trip to the first-aid station. It acts as a strong base; touching it for even a short time damages the skin, possibly leading to serious burns. Inhaling dust or vapors from this compound irritates the nose, throat, and lungs, sometimes causing coughing or difficulty breathing. Eye contact brings an extremely painful experience and can cause lasting damage.
Is It Toxic?
Talking about toxicity, it’s not the kind of chemical someone would label as instantly fatal unless someone ingests a large dose. Most issues relate to its ability to disrupt cells and tissues it comes in contact with. Lab tests on animals show moderate oral toxicity. If sodium butoxide makes its way into the digestive system, it causes nausea, vomiting, stomach pain, and tissue damage along the tract. That’s a far cry from table salt or the butanol used to make it. More indirect toxicity appears when it reacts with water, releasing heat and sometimes producing flammable gases; accidental releases or improper mixing in factories have caused fires or explosions in rare cases. Most occupational safety guidelines treat sodium butoxide as a material demanding special handling and thoughtful disposal.
Environment, Community, and Worker Safety
Spills from facilities storing or transporting sodium butoxide threaten water sources because of its strong reactivity with water. It poisons aquatic life by raising the pH and disrupting biological processes. Once, during a safety audit, attention to secondary containment around chemical storage tanks impressed upon me how a single overlooked valve could put entire streams at risk. The lesson stuck: chemicals don’t just stay where we put them, especially those as reactive as sodium butoxide.
People working with or around this compound deserve clear training and reliable protective equipment. Companies supply respirators, chemical splash goggles, and proper gloves, based on OSHA and CDC recommendations. These measures aren’t optional; workplace accidents involving sodium butoxide happen mostly during transfer or mixing, especially if safety steps get skipped.
Finding Solutions and Practical Steps
It makes sense for manufacturers to install real-time detection systems for leaks. Regular safety drills sharpen emergency response, especially for less-experienced workers who might hesitate during an alarm. Local fire departments and environmental agencies stay in the loop through regular reporting. Communities near factories benefit from transparent information and clear emergency action plans.
Better labeling and public outreach lower the risk of accidental misuse during transportation. Responsible companies train drivers and warehouse staff not just in general chemical safety, but in the risks tied to this specific compound. These small steps add up; they prevent spills, avoid workplace injuries, and protect the communities living and working alongside the chemicals that keep industry running.
What is the chemical formula of Sodium Butoxide?
Chemical Formula and What It Looks Like
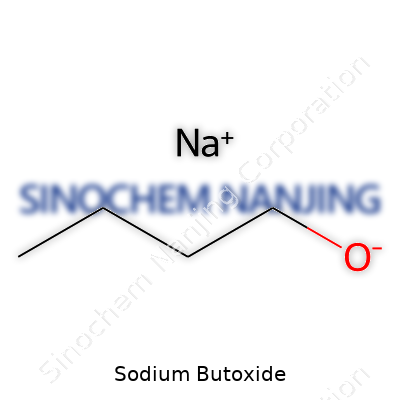
Sodium butoxide shows up in labs and manufacturing hubs, recognized by its formula C4H9ONa. The structure looks simple on paper: a butyl group (C4H9) connected to an oxygen atom, which links up with a sodium ion. Some people refer to it as sodium n-butoxide, sticking closely to the straight-chain version. The formula says a lot about how this compound reacts and where it gets put to use, which tends to fly under the radar for those outside chemistry circles.
Practical Purposes: Why This Stuff Shows Up
Sodium butoxide pops up in organic synthesis. It acts as a strong base, pushing reactions along that won’t budge without a little muscle. For chemists making esters, ethers, or even tinkering with pharmaceuticals, this compound clears hurdles that slow things down. I spent my college days wrestling with reaction yields, and sodium butoxide always made its mark where gentler compounds couldn’t get the job done. Working on the bench, you get used to its signature smell and the quick, exothermic splash when adding it to solvents.
Sourcing and Handling: Safety Isn’t Guesswork
This stuff isn’t bought at the corner store. Labs usually take delivery as a solid, white powder. It reacts eagerly with moisture, so chemists keep it under dry nitrogen, locked tight. Folks who half-close a bottle lid wind up with lumpy, clumped sodium butoxide—an expensive mistake. Gloves and goggles aren’t for show, since it reacts pretty fiercely if it contacts water or your skin. People sometimes think that only acids burn, but this alkaline powder can deliver some real damage to tissue. Growing up around my dad’s old chemical supply, I heard all kinds of cautionary tales about ignoring basic safety.
Why Accuracy with the Formula Matters
Getting the chemical formula right matters in the real world. C4H9ONa isn’t just trivia—it dictates how much you weigh out, how you predict results, and even how you dispose of leftover material. One decimal point or atom off, and the next steps in a process turn into guesswork. I remember a summer internship where a typo led to an entire wasted batch. Maybe that sounds minor, but mislabeling sodium butoxide could bring bigger trouble. Folks trust published data, from academic papers to chemical suppliers, and one error can ripple through many projects down the line.
Risks and Solutions: Building a Safer Lab
Mistakes with sodium butoxide can get serious. Accidental contact with moisture starts a chain reaction, sometimes releasing butanol gas and caustic sodium hydroxide. If the storage fails, ruined batches can tie up workflow and leave labs scrambling. One local company started using digital inventory and labeling, reducing wrong picks. Regular staff training, even for seasoned techs, helps everyone remember just how sensitive some compounds prove to be. Chemistry demands respect, not just curiosity, and sodium butoxide keeps its lessons close at hand for those who handle it. Getting the formula right is just the beginning; aware handling and respect for those dry stashes keep everyone safe and work on track.
How should Sodium Butoxide be stored?
A Real Threat Hiding in Plain Sight
Sodium butoxide sounds like a harmless chemical pulled from a dusty high school textbook, but anyone who’s come into contact with it behind laboratory doors knows it doesn’t forgive carelessness. The stuff burns in air. A splash of moisture sparks heat and releases fumes that can burn skin, lungs, and eyes. If mixed unknowingly with acids or water, you’ve got a recipe for disaster—fire, gas, and instant regret. This isn’t the sort of material you drop off on an average supply shelf with the rest of your reagents.
The Container Matters
Too many accidents start with sloppy storage. Sodium butoxide eats through thin plastics. Glass bottles with tight caps block out the air and keep the chemical steady—if they’re sealed well, the shelf life stretches, and the risk drops. The cap must remain closed except when measuring it out. Years back, I watched a colleague ignore this rule. Some moisture crept in; the cap fused to the glass, and the bottle started leaking white powder days later. Lesson learned, for everyone in that lab.
Metal cans seem sturdy at first glance, but this chemical can chew up weak alloys and leave cracks invisible until liquid spills out. Stick with high-quality glass, the kind that has a solid record resisting caustic materials. Good housekeeping in the lab means everyone knows to check for the appropriate hazard label showing corrosive and flammable warnings. It takes one wrong grab in an emergency to turn a small spill into a full-blown incident.
Beyond the Bottle: Where and How You Store It
Temperature swings play havoc with sodium butoxide. High heat can trigger fires or make vapors leak through seals. Find a cool, ventilated spot far away from any source of water. During summer, I’ve seen labs with box fans running on a backup power line just to keep storage from climbing above room temperature. Damp basements, sunlit window ledges, over-packed chemical cabinets—the risk multiplies with each shortcut taken.
Every single time the bottle comes off the shelf, dry gloves and goggles go on first. Even the dust settled inside the cap can set off a nasty burn. Good practice means one person signs out the bottle, measures quickly, closes it, then returns it to the same shelf. Skipping steps out of routine or boredom leaves others picking up the pieces. I’ve seen the hour wasted by a cleanup team after a careless tech let a tiny residue combine with condensation.
Why Good Storage Tactics Matter Today
Labs and workshops keep growing, adding new chemicals each year. With sodium butoxide in particular, letting new employees handle storage or transport without hands-on training puts everyone at risk. Emergency drills and hazard signage save more than face—they protect reputations, budgets, and lives. It’s common sense, but common sense comes from lived experience and mistakes that get remembered.
Keep storage cool, dry, and well ventilated. Lock up incompatible chemicals elsewhere. Double-check container condition and labeling every week. Small habits make for safe work over the long run. No industry standard or regulatory checklist beats the value of a lab culture that genuinely respects chemical hazards. My experience shows that cutting corners after a few uneventful months can invite a problem that lasts much longer.
What precautions should be taken when handling Sodium Butoxide?
Getting Familiar with Sodium Butoxide
Sodium butoxide finds a home in chemical synthesis. Labs and factories use it to push reactions forward. Though the name rolls off the tongue, its effects stick around. Skin burns, eye damage, and lung irritation come quickly if you treat it carelessly. This chemical reacts strongly with water and moisture in the air. That means a splash on the floor or open bottle can cause a mess bigger than anyone wants to deal with.
Recognizing Hazards
A fine powder like sodium butoxide quickly spreads through the air if not handled with control. Even touching it with unprotected hands can lead to serious skin burns. If the dust finds its way into the eyes or the mouth, you're looking at some painful consequences. I remember seeing a rushed clean-up end with a colleague getting medical attention for a splash – a lesson that sticks with you.
The dust can also catch fire in the right conditions. Small particles mixing with air in the lab or factory create a risk for explosions, not just fires. Even brief exposure to the dust can strain the lungs, leading to coughing or shortness of breath. These dangers make proper handling more than just a checklist.
Sensible Protective Measures
Lab coats and nitrile gloves make a good start. I can't count the times I've seen gloves save someone's skin, literally. Safety goggles are just as important. Sodium butoxide splashes do not forgive. Full-face shields give even more protection—worth it on busy days.
Using a chemical fume hood helps keep dust and fumes from spreading. Closing containers tightly after use can stop moisture from creeping in. Spills or splashes should get attention right away, not next time someone has a free minute. I always keep a large bucket of sand for spill control nearby, and it has paid off more than once.
Working with a buddy, especially when handling large amounts, brings extra safety. A second pair of eyes can catch mistakes, and in an emergency, quick help matters. Washing hands thoroughly after working with chemicals feels like a small step, but it’s one that gives peace of mind as you leave the lab or workshop.
Planning for Emergency Situations
Spills and splashes need fast action. Plenty of water at an eyewash station and a safety shower nearby can mean the difference between mild irritation and a bad injury. It’s not just about having these stations nearby—everyone needs to know how to use them. Fire extinguishers rated for chemical fires should always be within arm’s reach when sodium butoxide is open.
Training new team members, even the most experienced chemists, helps everyone stay sharp. I’ve seen training sessions save lab time in the long run by cutting down on accidents and lost product. Strong labeling and clear instructions make sure nobody grabs the wrong container in a rush.
Products Used Before and After Handling
Working on a cleanable, chemical-resistant bench or floor surface makes clean-up safer and faster. Soap made for heavy chemicals cuts through any leftover residue on hands and surfaces. I also keep vinegar nearby for neutralizing small splashes; the reaction bubbles show it’s working fast.
Sharpening Best Practices
Small, careful habits make a big impact. Opening the smallest bottle needed, writing down what gets used, and checking expiration dates all help stop accidents before they start. Care for yourself and your coworkers builds a culture where everyone looks out for each other. That kind of environment turns chemical handling from a daily risk into a task you trust yourself—and your team—to tackle.
| Names | |
| Preferred IUPAC name | sodium butanolate |
| Other names |
Sodium butanolate Sodium n-butoxide Butyl alcohol, sodium salt Sodium 1-butoxide |
| Pronunciation | /ˈsəʊdiəm bjuːˈtɒksaɪd/ |
| Identifiers | |
| CAS Number | 137-30-4 |
| 3D model (JSmol) | `C[O-].[Na+]` |
| Beilstein Reference | 1699791 |
| ChEBI | CHEBI:40873 |
| ChEMBL | CHEMBL135757 |
| ChemSpider | 5464279 |
| DrugBank | DB11272 |
| ECHA InfoCard | 100.032.214 |
| EC Number | 243-783-5 |
| Gmelin Reference | 1362156 |
| KEGG | C18606 |
| MeSH | Sodium Butoxide |
| PubChem CID | 8655 |
| RTECS number | WH7000000 |
| UNII | JN7Q7W6G1I |
| UN number | UN2812 |
| Properties | |
| Chemical formula | C4H9NaO |
| Molar mass | 96.09 g/mol |
| Appearance | White to yellowish powder |
| Odor | Alcohol-like |
| Density | 0.94 g/mL at 25 °C |
| Solubility in water | Reacts with water |
| log P | -0.89 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 15.5 |
| Basicity (pKb) | 2.5 |
| Magnetic susceptibility (χ) | -52.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.394 |
| Dipole moment | 1.66 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 235.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -368.5 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02,GHS05 |
| Signal word | Danger |
| Hazard statements | H261, H314, H302 |
| Precautionary statements | P210, P261, P264, P280, P301+P312, P305+P351+P338, P330, P337+P313, P370+P378, P403+P233, P501 |
| NFPA 704 (fire diamond) | 2-3-1-W |
| Autoignition temperature | 370°C |
| Lethal dose or concentration | LD₅₀ Oral Rat: 2,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2,000 mg/kg (rat, oral) |
| NIOSH | ID6500000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 mg/m3 |
| Related compounds | |
| Related compounds |
Sodium ethoxide Potassium butoxide Potassium methoxide Sodium methoxide Sodium tert-butoxide |

