Sodium Bromate: Past, Present, and Future
Historical Development
Sodium bromate’s journey stretches back to days when chemists tried to understand the patterns behind elemental behavior. In the 19th century, researchers in Europe found bromine’s value in a range of chemical processes, and through those early efforts, sodium bromate stepped onto the scene. Over time, manufacturers recognized that sodium bromate’s oxidizing power worked better than many other agents, so the chemical popped up across industries from gold extraction to textiles. The way it captured attention shows how chemistry evolves through hands-on experimentation and constant comparison with what’s already in use.
Product Overview
Sodium bromate appears as a colorless, odorless crystal that can dissolve in water, leaving behind a salty, slightly bitter flavor. Its formula, NaBrO3, hides a structure packed with three oxygen atoms bonded to one bromine, tying together both oxygen’s attraction for electrons and bromine’s reactive punch. Chemists often choose sodium bromate for its role as an oxidizer, pushing other substances to give up electrons during industrial and laboratory work. This is a material that never sits idle on the shelf once engineers and researchers understand its strengths.
Physical & Chemical Properties
Sodium bromate features all the markers of a robust oxidant: a strong white crystalline structure, a melting point above 350°C, and the ability to pull electrons away from other compounds efficiently. Its solubility in water stands out, allowing quick mixing for processes that need even distribution. Given the right partners, sodium bromate can react quickly, creating bromine or oxygen gas, both of which impact the environment and require careful handling. This energy stored within each molecule makes sodium bromate both useful and something to treat with serious respect in the lab or plant.
Technical Specifications & Labeling
Manufacturers detail sodium bromate’s purity, often above 98% for technical grade material, and flag up any trace contaminants like sodium bromide or heavy metals. Packaging usually involves robust containers—plastic or metal drums—each with hazard labeling that warns about oxidative risk, requirements for ventilated storage, and recommendations for personal protective equipment. Transport involves details about UN numbers and compatibility with other goods, giving shippers and handlers clear instructions from warehouse to end user. Overlook any of these steps and the result could spell danger.
Preparation Method
Industrially, sodium bromate comes from bubbling chlorine gas through a sodium bromide solution. The reaction takes place in large-scale, stirred tanks where the chlorine and bromide ions combine to form sodium bromate and sodium chloride as a byproduct. The process depends on maintaining precise temperature and pH, managing gas flows, and ensuring the waste stream doesn’t damage the environment. Some labs go smaller scale, relying on electrolysis with platinum electrodes, but the principle stays the same: control conditions to steer the outcome safely.
Chemical Reactions & Modifications
In chemistry classrooms and industry, sodium bromate acts as a catalyst for change. It fuels redox reactions, oxidizing organic or inorganic compounds. For example, in dyeing processes, it helps set colors into fabric fibers by enhancing fastness and preventing fading. Mix sodium bromate with sulfur-based reducing agents, and the resulting reaction produces heat and potentially hazardous gases. Sometimes, chemical engineers modify sodium bromate’s structure by introducing other oxidizers or reducing conditions, transforming it into bromine gas for water purification or disinfection. These reactivities push scientists to look for new applications, while strict protocols aim to reduce environmental impact.
Synonyms & Product Names
You might hear sodium bromate called bromic acid, sodium salt, or simply sodium bromate(V). In the marketplace, suppliers use unique catalog codes or trade names, but regulations require the core chemical name and formula on every label. These synonyms help cross-border trade and international collaboration stay on track, but for safety and compliance, sticking to clearly recognized nomenclature remains crucial.
Safety & Operational Standards
The hazards around sodium bromate keep regulators and workplace safety teams on alert. As a strong oxidizer, it increases fire risk if spilled on organic material, so companies adopt strict material segregation and ventilation rules. Gloves, goggles, and fume hoods form the baseline, but many plants stretch further—using automated systems to limit human exposure and digital monitoring of storage temperature and humidity. Emergency plans must address accidental releases, focusing on spill containment, neutralization, and immediate medical response if exposure happens. These safety standards evolve, reflecting lessons from past incidents and ongoing research updates.
Application Area
Sodium bromate shows up across diverse industries. In mining, it helps extract gold by breaking chemical bonds in ores, boosting recovery rates that make projects profitable or accessible. Textile companies depend on it as a bleaching and dye-fixing agent, chasing brighter colors and longer-lasting fabrics. Some drinking water treatment plants experimented with sodium bromate for disinfection, but worries about byproduct toxicity have steered many utilities to other agents. Laboratory researchers still value it for redox titrations or as a component in chemical oscillation reactions, enriching science education and analytical chemistry.
Research & Development
Chemists keep exploring sodium bromate’s behavior—looking for better yields, lower-cost processes, and safer handling protocols. Some teams investigate how tiny traces could function in pharmaceutical synthesis or specialty polymers, hoping to tap redox potential while minimizing residue. Research into automated monitoring systems tries to catch leaks or accidental mixing events before danger builds. Universities and private labs both chase new catalysts that could outperform sodium bromate in terms of reactivity while easing safety concerns, but so far, few replacements match its unique blend of power and reliability.
Toxicity Research
Concerns about sodium bromate’s health effects push scientific inquiry. Animal studies highlight toxicity at relatively low doses, showing links to kidney damage, hearing loss, and—in severe cases—cancer. Some health agencies flag sodium bromate as a possible carcinogen, which restricts its use in food and tighter control in industrial settings. Workplace studies continue to track exposure levels, aiming for better benchmarks in air quality and biological monitoring. Investing in technical fixes can reduce risk, but the need for active oversight stays strong, especially in regions where regulatory standards lag behind scientific findings.
Future Prospects
The future of sodium bromate depends on balancing risk with irreplaceable utility. Automation could shrink exposure, while environmental monitoring limits waste, but strict regulations will likely hold firm as new data emerges. Some research trends focus on green chemistry approaches—developing alternative oxidizers or process tweaks to minimize sodium bromate’s footprint. Demand may shift as more industries chase sustainability and substitute less hazardous materials, but few chemicals deliver the same performance in specific tasks. For companies and researchers, the next chapter will require adaptability, careful record-keeping, and an open mind to changing science and public expectations.
What is Sodium Bromate used for?
The Bread You Eat and Chemicals You See
Sodium bromate pops up in some places you wouldn’t expect, and once you know its uses, you start to wonder why such a strong chemical ends up in stuff as familiar as bread. Bakers once used sodium bromate to help dough rise higher and produce bread with a softer texture. This practice grew out of the push for perfect industrial loaves, and it spread through bakeries all over the world, especially in the last century. The compound acts as an oxidizing agent, strengthening the dough and making bread seem fresher and fluffier for longer stretches.
Over the years, scientists discovered that sodium bromate can stick around in the finished loaf. Health officials flagged this as a concern because studies linked the compound to an increased cancer risk in animal testing. As more food safety watchdogs started publishing research, countries like the United States set limits on its use, and others such as the European Union and Canada banned it outright in food. Despite these rules, you might still find sodium bromate in foods in countries without strong regulation, especially where factory bread is the norm. It’s an example of how chemical shortcuts in food manufacturing often come with trade-offs for health.
From Bleach to Dyes: Industrial Roles
Plenty of people only know sodium bromate from food stories, but its biggest footprint comes from industrial chemistry. Textile factories depend on sodium bromate to help dyes attach to fabrics, especially during color-fixing processes. It works fast and delivers consistent results, which became the selling point as clothing production scaled up. Water treatment plants sometimes rely on sodium bromate during chemical disinfection. Anyone who’s studied municipal water systems knows that controlling by-products becomes a full-time job. Even tiny traces of bromate in drinking water have triggered health warnings, most notably in cases where ozone treatment breaks down and bromide atoms sneak in.
Lab Work and Gold Mining
Chemistry labs use sodium bromate as a strong oxidizer in research and analysis. In gold mining, the compound plays a role in extracting gold from ores. Mining companies chase efficiency, and sodium bromate lets them recover more metal with less hassle. That means less digging but more chemicals handling, which often leads to controversies about environmental safety and the legacy of chemical waste. Gold extraction with sodium bromate hasn’t replaced older cyanide processes everywhere, but the mining industry keeps exploring new approaches that trade one risk for another.
Public Health, Policy, and Possible Solutions
Many countries revisit chemicals like sodium bromate whenever new information comes in. Food policy experts argue for stronger labeling on bread and other processed foods. Most shoppers wouldn’t choose a product with a potential carcinogen spelled out on the package. Public health campaigns push manufacturers to switch to safer dough improvers, such as ascorbic acid (vitamin C), which can boost bread texture without the health baggage.
On the industrial side, smarter treatment protocols in water and textile factories help keep bromate contamination out of drinking water and rivers. Some water districts invested in better monitoring and developed systems to hold plant operators accountable. Industry-wide shifts often start with new technology: more efficient oxidizers, cleaner chemical reactions, or porous materials that filter out unwanted by-products. These aren’t quick fixes, but every step to limit sodium bromate means cleaner water, safer food, and less risk for people who work in factories or live near them.
Is Sodium Bromate safe to handle?
Taking a Closer Look at What We’re Dealing With
Sodium bromate gets a reputation for helping out in dyeing textiles and treating water, but its safety record deserves a lot of attention. My own experience working in chemical labs reminds me how easily folks forget the risks when focused on getting the job done. Reach for the wrong bottle once, and you remember the consequences for life.
Touching or breathing in sodium bromate isn’t like handling table salt. The crystals can produce serious problems for skin, eyes, and lungs. Direct contact burns. Even a small spill forces a scramble for the nearest eyewash station. Lab workers I know tell stories about colleagues who skipped gloves once, only to spend the rest of the day chasing a burning, stinging pain.
Research has put this substance under the microscope for years. Studies in rats link sodium bromate exposure with cancer and kidney problems. The World Health Organization flagged it as a possible human carcinogen. Nobody ends up with these studies on their mind in the middle of a busy shift, but those long-term risks hang over every careless moment in a production plant or classroom.
Why It Still Ends Up in Industry
Factories and water plants keep sodium bromate around because it gets results. It pushes dye deep into fibers, creates permanent colors, and knocks out some tough contaminants in drinking water and pools. The problem comes from the thin line workers must walk between performance and personal risk. Any job with chemicals always brings up questions about the balance between output and health.
These dangers bring regulation. In the European Union, only trained professionals can handle this chemical. The US Occupational Safety and Health Administration tells employers to give staff respirators, splash-proof goggles, and strict training. You may find sodium bromate locked behind extra barriers, with a stack of warning signs. In some labs, sensors watch the air for traces of dust or vapor.
Lessons from Experience
I learned to stop and check labels after a coworker threw what looked like simple white powder in a waste bin. Hours later, a faint chemical smell set off alarms. No one remembered who tossed it, because sodium bromate sits in containers next to more harmless substances. Routine moments create risks. Skipping protective clothing – because “it’s just a quick transfer” – happens too often.
Training helps, but culture keeps people safe. Workplaces that make safety part of daily conversation do a better job of protecting employees. I’ve never seen a place that skipped regular drills, assigned mentors, and encouraged questions go more than a few weeks without an accident.
Better Ways Moving Forward
Reducing risk means more than running through the same checklist before every task. Employers can invest in equipment that limits exposure, like sealed handling systems and smarter ventilation. Switching to less dangerous alternatives where possible cuts out trouble before it starts. Workers should push supervisors when they notice gaps in safety, not wait for the next staff meeting.
Using sodium bromate isn’t just about following guidelines. Every time someone reaches for the bottle, they face a decision that goes home with them. The real question isn’t just about the safety of the chemical, but about the habits and systems built to keep people out of the ER.
What is the chemical formula of Sodium Bromate?
A Straight Answer: NaBrO3
Sodium bromate's formula is NaBrO3. For lots of us, that string of letters and numbers doesn’t jump off the page, but in the world of chemistry and public health, it matters for some practical reasons. Sodium gives the compound its basic characteristics, bromine supplies the backbone, and oxygen rounds it out. That’s the heart of NaBrO3.
Where Sodium Bromate Puts Down Roots
Back in my time as a lab tech, this compound popped up every time someone mentioned water treatment or bleaching. Sodium bromate comes in as a strong oxidizer. In practice, it gets tossed into the mix for processes that need reliable and calculated chemical changes — not too wild, not too tame. Textile companies like it for its ability to bleach without pushing fibers too hard. Municipalities test for it in drinking water, wary of its potential to form as a byproduct when using ozone to clean supplies.
Globally, this compound shows up in a few more roles than many expect. Hair salons in the past leaned on sodium bromate for perms, chasing that well-known “springy” look. Today, stricter safety rules have shifted those habits, but you still see echoes of those decisions in regulations and health guides.
Risks and Realities
The reasons scientists and governments pay attention to sodium bromate come down to public health. Drinking water sometimes picks up trace amounts through treatment processes. The International Agency for Research on Cancer has flagged sodium bromate as a possible human carcinogen. Japan set strict exposure limits decades ago, and Europe keeps its use tightly controlled. In the United States, agencies like the EPA keep a close watch, publishing regular updates about the quality of municipal water, part of a wider effort to keep communities safe.
Anyone who works with oxidizing chemicals learns the routines fast. Keep supplies dry, don’t mix with organics on a whim, never try shortcuts. Schools teach this to budding chemists on day one, right after the periodic table lesson. The memory of one hasty mix-up in a school lab still sticks with me — a small pile of sodium bromate, carelessly placed near a bit of sugar, made for a charred lesson in what not to do. It’s a good way to get your point across about risk.
Better Solutions and Smarter Choices
Chemists and engineers stay on the lookout for new ways to lower the odds of sodium bromate contaminating water. Some towns use alternate disinfectants, turning to ultraviolet light or tweaking O3 dosing so the byproducts stay lower. Textiles rely more on hydrogen peroxide or other oxygen-based bleaches that don’t produce the same residues. Careful monitoring, modern testing, and a transparent approach to water systems all play into a longer-term solution.
People have a right to know what’s in their tap water, and what tools are part of its story. A formula scribbled on a label—NaBrO3—tells a bigger tale about public health, chemistry in the real world, and the balance between progress and caution.
How should Sodium Bromate be stored?
Understanding the Risks
Most people won’t find sodium bromate in their kitchens, but it has a real place in industrial plants, labs, and water treatment facilities. What stands out to me, working around chemicals for years, is how much trouble can come when folks overlook storage details. Sodium bromate carries some genuine hazards: it’s a strong oxidizer and reacts with many everyday materials. Accidents don’t only hurt people—they also stir up lawsuits, fines, and sometimes permanent damage to buildings or the local environment.
Why the Storage Space Matters
Many assume the right building is enough. All it takes is a humid area or leaky roof, though, and trouble follows fast. Moisture gives sodium bromate a foothold to start breaking down or mixing with unwanted materials. The fumes or powder in the air can irritate eyes, lungs, or skin. Direct sunlight changes the game, causing the chemical to become unstable, sometimes even sparking fires if touched by organic matter.
Picking the Right Containers
Metal drums often look strong, but sodium bromate eats away at most metals or causes corrosion over time. Experience tells me plastics with high chemical resistance, like HDPE or polypropylene, stay safe longer and keep leaks off the floor. Tight-fitting, well-labeled lids mark the difference between a controlled environment and a disaster waiting to happen. Labels with hazard symbols help new workers spot danger at a glance, too.
Distance Makes a Difference
Sodium bromate stays much safer away from fuel, oil, acids, and paper. I’ve watched operators learn this lesson the hard way, scrambling after an off-the-shelf cleaning product touched oxidizers and kicked off a fire. A separate, locked area—one with real airflow and no glass containers—keeps chemicals apart. Shelving should stay below eye level, since spills from above can splash and burn.
The Perks of Good Ventilation
Old storerooms with stale air trap fumes, which stack up over time and ruin health. Pulmonologists have linked prolonged exposure to oxidizer dust with chronic respiratory problems. Effective ventilation systems, checked by a responsible manager, mean fewer headaches and a safer work environment all around. Fans alone won’t help if the room seals up tight, so regular checks matter most.
Training Saves Lives
Training goes beyond a single safety talk. There’s no beating routine drills and spot-checks to see what people actually remember. If someone spills sodium bromate, a well-trained hand grabs gloves, eye protection, and a dust mask before cleaning up. Dry sweepers and vacuums work better than wet mops, which can cause more reactions and spread contamination.
Tools for the Job
Spill kits belong right in the storage area, not tucked away three hallways down. Eye-wash stations, running water, and emergency showers help if contact happens. Fire extinguishers, especially ones marked for chemical fires (not just water-based), play a huge role in shutting down small blazes before they spread.
Policy and Oversight
Chemicals don’t respect emotions—they respond to clear rules and reliable routines. Regular inspections, written logs, and clear accountability keep the space under control. OSHA and local fire marshals hand out guidance for good reason: following these rules reduces injuries and helps everyone go home safely at the end of the shift.
What are the hazards associated with Sodium Bromate?
What’s Really in That White Powder?
Sodium bromate pops up in industries from textile dyeing to hair perms. Its chemical thumbprint—NaBrO₃—gives it the power to oxidize and bleach. Just because it works well in the plant or the salon doesn’t mean it gets a free pass in everyday life. For most people, the real risks don’t come into view until things go sideways. The industrial world learned these lessons the hard way.
Exposure Isn’t Just a Factory Problem
Most of us don’t run across sodium bromate at home. Folks who work in dye houses or hair salons, on the other hand, might touch it daily. The compound’s fine powder slips into the air or clings to surfaces. Without strong ventilation or protective gear, it can find its way onto skin or into lungs. This is where risk starts to turn into real harm.
Health Risks Don’t Hide for Long
Swallowing even a small amount can send someone to the ER. The World Health Organization calls sodium bromate a probable carcinogen. Lab tests tie it to cancers and kidney damage, and stories from poisoning cases talk of vomiting, abdominal pain, and hearing loss. Workers sometimes come away with skin rashes or breathing trouble.
Environmental Fallout
Factories that flush sodium bromate waste into local streams end up hurting more than just fish. This compound lingers in water and doesn’t break down without a fight. Studies find it harms aquatic life and may seep into groundwater. Reports from the EPA and regional water agencies show how even low levels spark new rules and cleanup costs.
Why Do We Still Use It?
The textile industry leans on sodium bromate for color fixing. Hair stylists get the curls their customers want using formulas with this chemical. There are alternatives, but sometimes old habits or costs mean businesses stick to what they know. Some countries—including members of the European Union—are phasing it out or banning it from food and cosmetics.
Protecting Health in the Real World
From my time around labs and industrial settings, the biggest danger stems from taking shortcuts. Gloves, goggles, and masks make a difference. Training matters just as much—people protect themselves better if they know what they’re dealing with. Still, regulations only reach so far. Governments keep track of contamination at water treatment plants and monitor levels in consumer goods. Without regular inspections and real consequences for unsafe practices, these rules end up as paperwork nobody reads.
What Would Real Change Look Like?
Cutting down on sodium bromate starts with tougher substitution rules. Industries need clear incentives to ditch this chemical for something safer. Research drives this process forward. Lab teams around the world look for greener oxidizers and better water treatment methods. Wastewater systems in Japan, for example, reduce bromate by tapping advanced filtration and ozone removal. These aren’t just technical fixes—they protect communities and workers at the same time.
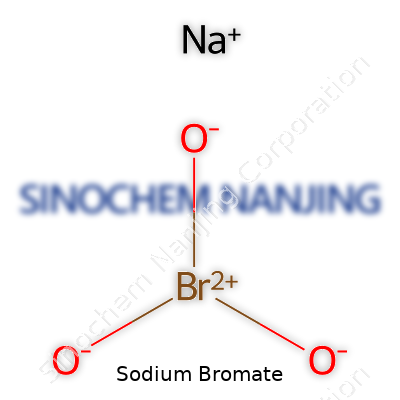
| Names | |
| Preferred IUPAC name | Sodium bromate |
| Other names |
Bromic acid, sodium salt Sodium bromate(V) Sodium bromate, Reagent Bromate de sodium |
| Pronunciation | /ˈsəʊdiəm ˈbrəʊmeɪt/ |
| Identifiers | |
| CAS Number | 7789-38-0 |
| Beilstein Reference | 1711351 |
| ChEBI | CHEBI:78050 |
| ChEMBL | CHEMBL1357 |
| ChemSpider | 53656 |
| DrugBank | DB11090 |
| ECHA InfoCard | 100.960.189 |
| EC Number | 231-889-6 |
| Gmelin Reference | 42174 |
| KEGG | C18635 |
| MeSH | D013252 |
| PubChem CID | 23668172 |
| RTECS number | EF8775000 |
| UNII | OI29X68OJD |
| UN number | 1494 |
| Properties | |
| Chemical formula | NaBrO3 |
| Molar mass | 150.89 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 3.34 g/cm³ |
| Solubility in water | Soluble |
| log P | -2.37 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 9.0 |
| Basicity (pKb) | 8.69 |
| Magnetic susceptibility (χ) | -47.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.510 |
| Viscosity | 1500 cP (20°C) |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 137.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −480.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -359.5 kJ/mol |
| Pharmacology | |
| ATC code | V03AB16 |
| Hazards | |
| Main hazards | Oxidizing, harmful if swallowed, causes serious eye damage |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS03,GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H271, H301, H318, H373 |
| Precautionary statements | H260, H272, H302, H318, P210, P220, P221, P280, P305+P351+P338, P370+P378, P301+P312, P310, P501 |
| NFPA 704 (fire diamond) | 3-0-2-OX |
| Autoignition temperature | > 400°C |
| Lethal dose or concentration | LD50 oral rat 316 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 316 mg/kg |
| NIOSH | STY68000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 15 mg/m³ |
| IDLH (Immediate danger) | 40 mg/m3 |
| Related compounds | |
| Related compounds |
Sodium bromide Potassium bromate Potassium bromide |

