Getting to Know Sodium Bifluoride: More Than Just a Chemical Compound
Historical Beginnings: A Legacy Forged in Industry
Sodium bifluoride has never really caught the public eye the way some chemicals have, but its story ties closely to some core shifts in industry and technology. Early chemists stumbled upon it while sorting out the uses for fluorine-based substances, realizing sodium bifluoride could bridge the gap between reactivity and practical utility. Over the decades, the demand from glass etching, cleaning products, and metallurgy paved a steady road for this salt. By the mid-1900s, factories depended on it to handle tough cleaning jobs and treat metals—functions that would have seemed nearly impossible with gentler solutions. As technology moved forward, sodium bifluoride tagged along, adapting to new challenges in manufacturing and research.
Peeling Back the Chemical Curtain
Anyone who works with sodium bifluoride gets to know its quirks. The substance appears as a white, crystalline powder, with a sharp, biting taste if anyone were reckless enough to try it—a bad idea, given how toxic it can be. Its molecular formula, NaHF2, reveals that its composition is a tight marriage between sodium, hydrogen, and two fluorine atoms. Unlike many simple salts, sodium bifluoride packs an acidic punch, easily releasing hydrogen fluoride when wet or heated. In the lab, even a passing whiff can bring a sharp sting to the nose, a clear signal that this isn’t just another salt to handle with bare hands. In water, it dissolves quickly, breaking into ions that make cleaning and metalwork possible. Its reactivity attracts chemists, but the same properties also demand respect from anyone handling it.
Technical Landscape and the Trouble with Labels
Anyone shopping for sodium bifluoride soon realizes that not every bag is made equal. Purity levels matter, especially in industries where contamination means scrapping entire batches of product. Labels usually list at least 98% purity for technical-grade material, with moisture content and particle size playing important roles in how the product behaves during tasks like etching or cleaning. Keeping that fine white powder dry matters—a clump here or a leak there could spell headaches or danger. Regulators don’t take chances with sodium bifluoride, spelling out labeling protocols that mark it as hazardous. Beyond the numbers, workers in storage rooms and shipping docks have stories about careful handling etched in memory, reminding everyone that a misplaced scoop can lead to nasty burns or chemical releases.
Making Sodium Bifluoride: Not Your Garden-Variety Recipe
Factories don’t churn out sodium bifluoride by accident; they work through reactions between sodium carbonate or sodium hydroxide and hydrofluoric acid. Handling hydrofluoric acid is tough even for seasoned chemical engineers. One slip, and the consequences can be life-threatening. Operators must measure reactants precisely and control temperature at every stage. After the reaction, the solution gets concentrated and then cooled to cast out the crystalline sodium bifluoride. Leftover gases and solutions require careful neutralization and disposal—another reminder that chemical manufacturing rarely leaves room for shortcuts or mistakes.
Chemical Flexibility: What Happens in the Beaker Doesn’t Stay There
Everyone who has worked in a modest-sized chemical lab knows that sodium bifluoride never sits quietly on the shelf. Add a bit of water or acid, and it pops out hydrofluoric acid, useful in etching glassware or creating new fluorine reagents. It plays a middleman in making other fluorides, breaking apart or building new bonds, depending on what gets tossed into the beaker. Combine it with strong bases, and sodium fluoride forms, pushing the reactivity in new directions. Scientists eyeing new reaction pathways often reach for sodium bifluoride to nudge things along because it can donate fluoride ions far more gently than raw hydrofluoric acid, making it safer and easier to manage in controlled settings.
Name Game: Synonyms in the Chemical World
Sodium bifluoride has picked up several aliases as it moved through labs and markets: sodium hydrogen difluoride, acid sodium fluoride, and even sodium acid fluoride show up in older textbooks and supplier lists. Depending on country and context, you might find it listed under an international registry number or classified differently by regulatory groups. These subtleties matter once rules about shipping, storage, and worker safety come into play—a lesson learned quickly by anyone juggling both domestic and import product paperwork.
Staying Safe Means Staying Sharp
Dealing with sodium bifluoride isn’t for the careless or the unlucky. Even brief skin contact can cause deep burns, and inhaling dust lands someone in the emergency room. Proper gloves, full-face shields, and fume hoods aren’t overkill—they are routine. Any lab tech or plant worker worth their salt takes the safety briefings seriously because the stories of accidents are never far from memory. Spills call for prompt cleanup with plenty of ventilation and neutralizing agents ready to go, usually calcium-based for neutralizing potential hydrofluoric acid release. Storing this powder in sealed, corrosion-resistant containers in cool, dry conditions helps reduce risk. Ongoing training, clear signage, and strict incident reporting form the backbone of responsible handling. Many lives depend on these standards holding up, every shift, every day.
Where It Shows Up: The Reach of Sodium Bifluoride
Sodium bifluoride’s mainday job is tough cleaning—think tile, porcelain, or removing rust from steel. Platers and metal finishers see it as a silent partner in making surfaces shine. Etching factories depend on it for both glass and metal. It helps keep automotive radiators clear from internal build-up. The oil industry leans on it for extracting challenging deposits from well bores, allowing equipment to last longer and production to run smoother. Researchers and sometimes municipal water plant operators use tightly controlled amounts in water treatment or making specialty fluorine compounds, although regulated for safety’s sake. These applications only scratch the surface—almost every time someone needs the dissolving power of concentrated fluorides but wants more manageable handling than raw hydrofluoric acid, sodium bifluoride sits on the shortlist.
Glimpses from the Lab: Research and Development
Chemists in my circle swap stories about sodium bifluoride’s problems and promise. Some highlight its role as a safer way to introduce fluoride into organic molecules during synthesis, cutting down on the dangers of direct hydrofluoric acid. Others explore its use for more eco-friendly cleaning formulations. Labs keep poking at its reactivity and stability, hoping to find combinations that squeeze more performance and less hazard out of each batch. In surface coating and pipeline maintenance, new ways to blend or encapsulate sodium bifluoride aim to reduce workplace exposure without forfeiting cleaning strength. Environmental risk assessments and computational chemistry models have started to redraw limits and unlock alternatives for safe, targeted applications, pointing toward more efficient and sustainable use in the long term.
Learning the Hard Way: Toxicity and Health Impacts
Anyone who has worked in industrial cleaning or glass etching soon learns to respect sodium bifluoride’s darker side. Short-term contact causes acute burns, and it doesn’t take much dust in the air to spark breathing trouble. The fluoride ion, once inside the body, binds up calcium, causing deep tissue damage and sometimes affecting the heart. Medical literature and incident reports collect evidence of accidents, making it clear that ignoring basic precautions can have lifelong consequences. Poison control centers and hospital staff know that quick action can save lives, but prevention remains the only sure bet. Regulations keep tightening as more research confirms how quickly this chemical can overcome even healthy workers; regular medical surveillance and workplace monitoring have become a cornerstone of responsible chemical management.
Shifting the Field: What’s Ahead for Sodium Bifluoride
What’s next for sodium bifluoride depends on how industries adapt to tighter health and environmental standards. Cleaning and etching demand isn’t fading, but researchers and manufacturers keep searching for ways to balance effectiveness against health risk. Mechanical alternatives, greener chemical substitutes, and automation stand to reduce direct human contact. Companies bet on closed systems and better ventilation to keep exposure low. Meanwhile, new applications in energy and material science tempt researchers, provided safety hurdles can be cleared. The chemical’s core properties stay relevant, but those using it carry a growing responsibility—industry tradition gives way to stricter oversight, and new generations of workers learn from the stories that came before them.
What is Sodium Bifluoride used for?
Behind the Label: What Sodium Bifluoride Brings to Industry
Anyone who's spent time looking at industrial labels has probably noticed one chemical that pops up in some unexpected places: sodium bifluoride. It shows up in cleaning products, glass etching, and water treatment facilities. In these workplaces, the chemical’s bite makes it useful, but also calls for respect.
Sodium Bifluoride in Cleaning and Laundry Products
Many folks working in janitorial services or laundromats have crossed paths with sodium bifluoride without realizing it. The substance kicks stains and rust to the curb in industrial laundries. That stubborn brown ring on bathtubs or that rust on pipes? Some restroom cleaners rely on sodium bifluoride to loosen and dissolve these marks fast. A product that cuts grime quickly boosts productivity for cleaning crews who don’t have time to scrub all day.
My time helping manage a gym exposed me to next-level maintenance routines. Locker rooms never stayed spotless for long, and folks brought plenty of minerals in with sweaty workout gear. Cleaners with sodium bifluoride, handled carefully, made tackling mineral buildup much faster. This meant less downtime and fewer complaints about dirty showers.
Essential for Glass Etching and Metalwork
Glass manufacturers rely on sodium bifluoride for a reason: it attacks silica in glass. Artists use it to create frosted finishes, business owners use it to make logos and patterns. In metal shops, it’s part of the pickling solution that strips away oxides before welding or finishing. A metalworker doesn’t want old rust or mill scale fighting against paint or welds. Sodium bifluoride makes this prep work smoother.
These benefits come at a cost. Poor ventilation, skin contact, or casual attitudes about protective gear lead to burns, breathing trouble, or worse. Industry accidents, some blocked from public view, show the toll that comes from skipping real safety measures. In my circle, one story sticks: a maintenance crew in a small plant ignored glove and goggle rules. Chemical splashes led to several hospital visits. Simple lessons remind us that regulations and training exist for a reason.
Role in Water Treatment
You might not think about what keeps pool water clear or tap water safe, but sodium bifluoride quietly plays its part. It acts as a source of fluoride ions. In measured amounts, this prevents tooth decay and keeps bacterial growth down. Too much, and water operators invite criticism or worse, sick consumers. History shows the balance is delicate—towns have faced scrutiny over fluoridated water, especially when communities don’t feel informed about what’s going in their pipes.
Safe Handling and Moving Forward
As with many chemicals, sodium bifluoride demands caution. The basics matter—gloves, masks, careful storage, clear labels. Schools and employers bear responsibility to teach real handling protocols, not just hand out a sheet and hope for the best. I’ve seen teams forced to improvise when safety equipment was missing or ignored because “that’s how it’s always been.” Culture shifts only stick when management backs up talk with real support, from updated gear to honest conversations about near-misses.
Some industries now look for greener swaps, including citric acid or less reactive agents. Change often meets resistance because proven processes are tough to replace. It’s up to manufacturers and regulators to keep pushing, investing in safer alternatives, and listening to workers on the front lines who know these products best. Until safer options take hold, trust and training remain the strongest shield.
Is Sodium Bifluoride hazardous to health?
Understanding Sodium Bifluoride
Sodium bifluoride shows up in places many folks never think about. Used to clean metal, treat water, and etch glass, it earns its keep in industry. In the lab, just a little can get a big job done. But the real question is what price comes with its use, especially in terms of health.
Exposure Risks and What Science Says
Sodium bifluoride isn’t a household name, but its hazards shouldn’t be underestimated. The compound releases hydrogen fluoride gas when touched by water or acids. That gas, even in small doses, stings the eyes and can damage lungs—no one wants to breathe it. Looking at the science, studies from trusted sources like the CDC and NIOSH warn workers to take it very seriously. Skin contact leads to burns and blisters. Inhaling dust or fumes may bring on coughing, shortness of breath, and, in worst cases, life-threatening lung injury. Swallowing the substance can trigger nausea, vomiting, abdominal pain, and in severe poisonings, heart rhythm problems or worse.
Fluorides generally affect the body by binding calcium. Low calcium means nerve and muscle trouble—not to mention lasting damage to bone if exposures drag on. Anyone who's spent time in a laboratory or industrial plant can point to real stories of coworkers burned by careless handling or hurt by cutting corners on safety gear.
Workplace Safety Gaps
Despite known dangers, sodium bifluoride turns up in more than heavy industry. Some cleaning products mix it in for scale removal. Every year, reports come from hospitals of folks injured cleaning furnaces, HVAC coils, or bathrooms, often because of poor labeling or a lack of protective equipment. In factories, tight training and rules save lives, but smaller operations might miss the mark. Not all workers read safety data sheets or get proper gloves, goggles, and masks. Tight budgets often mean outdated procedures linger.
Fixing What’s Broken
Across communities, people can cut risk by handling sodium bifluoride with smart precautions. Employers should rethink calling safety gear optional. Goggles, chemical-resistant gloves, and ventilation systems make a difference. It helps to walk through the process step-by-step, training every new hire as if their life depends on it—because sometimes, it does. Clear labels and direct warnings on bottles and bags cut down on tragic mix-ups in homes and workplaces.
I’ve seen firsthand how a missing label or a shortcut during night-shift cleanups can leave someone in the ER. Emergency procedures matter. First responders should keep calcium gluconate gel stocked and know how to use it, as it helps counteract burns and systemic problems from fluoride exposure. Local hazmat teams could partner with clinics on practice drills. Communities near factories may want real-time air monitoring—not just an alarm for show, but one with teeth, tied to quick community alerts.
On the policy side, government regulators should keep sodium bifluoride and its relatives on inspection lists. Regular reviews and updated exposure limits can push companies to do better. Industry watchdogs and unions can back up workers if employers drag their feet. Sharing public health data keeps everyone honest, sparking improvements that move beyond suggestions to real protection.
How should Sodium Bifluoride be stored?
Understanding the Hazards
Sodium bifluoride looks quiet sitting in a bag, but someone who’s actually handled chemicals in a lab or warehouse knows that looks can mislead. Used for metal cleaning, water fluoridation, and even insecticides, this compound packs a punch. A tiny spill eats through skin. Breathing in dust triggers coughing and a sore chest. Nothing good comes from letting sodium bifluoride get where it shouldn’t be.
The Right Storage Spot Isn’t a Luxury
I worked in a plant where sodium bifluoride shared shelf space with detergents and other cleaning powders. Just one careless employee opened a bag near a damp spot, and suddenly we had a hard, clumpy mass that vented acidic fumes all week. This stuff loves water—once it mixes, it forms hydrogen fluoride gas, which can burn lungs and eyes. Keeping it away from moisture looks simple on paper but demands vigilance day in and day out.
Keep Out Water, Keep Out Trouble
Dry storage stands as rule number one. Humid aisles or rooms with leaky pipes bring trouble. Anyone who’s worked through a chemical cleanup knows how terrifying it gets once moisture sneaks in. Sodium bifluoride belongs inside tightly sealed, corrosion-resistant containers. No cardboard. Plastic or polyethylene works much better, and steel bins with good liners will suffice if checked for leaks.
Every tub or drum needs a clear label. The label shouldn’t just read “Dangerous” but spell out the acid gas risk, the threat to skin, and the poison hazard. Labels that fade or peel away can cause confusion, so better to have spares and train staff to replace worn signs.
Safe Placement: Not All Spaces Are Equal
No one with real experience wants sodium bifluoride anywhere near food, animal feed, or drinking water supplies. Storage rooms should sit far from kitchen spaces, break rooms, or any area where water gets used. Over the years, I saw what happens when lazy storage puts chemicals on a utility room shelf—pipes burst, and suddenly cross-contamination is a real risk.
Facilities should dedicate specific shelving or a chemical cabinet designed to resist acids for sodium bifluoride. Keep it low to the ground, since dropping a bag from height can kick up clouds of hazardous dust. Don’t stack heavy items above it either. If there’s a spill, reaching over other products adds risk.
Avoid Bad Combinations
Store incompatible chemicals far apart. Sodium bifluoride and anything alkaline—a simple bleach or household ammonia—can trigger violent reactions. Experienced handlers know not to lean acid powders against oxidizers or metals either. Sorting chemicals by hazard class may sound like overkill until you see a small warehouse fire become a major disaster because someone mixed storage zones.
Training and Emergency Plans Save Lives
All the best storage means little if staff don’t understand what they’re up against. Long hours and routine jobs leave room for mistakes, so refreshers matter. Everyone handling sodium bifluoride should know the closest eyewash station, vented storage rules, and how to use gloves and masks. I’ve seen well-meaning employees skip gear because they think one scoop won’t hurt them.
Routine checks for corrosion, punctures, and leaks save headaches down the road. Emergency plans must be more than a poster on the wall. Drills help build the habits that pay off when something really goes wrong.
The Stakes Go Beyond Compliance
Nobody forgets their first chemical burn or the smell of acid in the air. Sodium bifluoride’s risks don’t only threaten workers—they can spread through pipes, floors, and ventilation. Good storage protects both staff and neighborhoods. The right setup isn’t just ticking off regulations; it’s building trust and keeping people out of harm’s way.
What precautions should be taken when handling Sodium Bifluoride?
Knowing the Risks Before You Start
Sodium bifluoride might look like just another white crystalline powder, but it packs a punch if you treat it carelessly. The chemical works its way into a lot of cleaning products, water treatment solutions, and even metal surface preparations. From what I’ve seen in real workplaces, a jar of sodium bifluoride is anything but harmless. It reacts with moisture to create hydrofluoric acid, which doesn’t just burn—it seeps right through the skin and attacks bones. Shortcuts and guesswork have no place when you handle it.
Tough PPE Isn’t Optional
No one forgets the sting of a chemical burn, especially if they’ve experienced one firsthand. I watched a co-worker ease up on his personal protection gear once, thinking he knew his way around the chemical. He ended up in the emergency room with a deep, unseen burn. Eyes, hands, and skin deserve top-of-the-line gear: chemical-resistant gloves, splash-proof goggles, and a long-sleeved lab coat. Rubber boots and a face shield come next if the job risks splashing or releases dust into the air.
Ventilation Can Save Your Lungs
Inhaling sodium bifluoride dust can irritate the lungs, throat, and nose. Relying on open windows or fans won’t do much. Real ventilation means working in a fume hood or a proper exhaust system that sends fumes directly outside. People often underestimate the risks and trust in basic airflow. A cough or throat tickle might feel minor until it leads to bigger breathing problems.
Safe Handling Requires Training, Not Just Warning Labels
Most injuries happen because someone assumes training is just a formality. I’ve sat through countless safety meetings—some inspiring, some sleep-inducing—but the good ones share real stories and hands-on demos. Getting people to practice with mock spills or cold drills helps them stay sharp if things go sideways. It’s not about memorizing rules from a safety data sheet, but actually knowing how to use an eye-wash station in a hurry or neutralize a small spill without panic.
Storage Makes a Difference
Keeping sodium bifluoride away from anything wet is simple advice, but it keeps getting ignored. One time in a shared storeroom, a leaking pipe overhead nearly caused a disaster. Store it in airtight plastic containers, high up and away from acids or reactive chemicals. Keeping it locked means only folks with a clear head and real training get near it. No one should leave it near open shelves or break rooms no matter how short the stop.
Emergency Plans Go Beyond Paperwork
When an accident happens, fumbling for the right response wastes precious seconds. At one site, we ran monthly emergency checks—spills, splashes, and all. Running through the steps as if the threat was real built muscle memory. Eyewash stations, showers, and neutralizing agents like calcium gluconate gel need checking, restocking, and clear access at all times. Regular practice has saved more than one person from long-term injury.
Personal Accountability and Looking Out For Others
The buddy system saves lives. If someone looks tired, distracted, or awkward in their gear, it’s better to call it out than hope for the best. No job gets done faster or better if someone gets hurt along the way. Trust means reminding each other of the rules and walking through each step without rushing. In the long run, paying attention to each other—and respecting sodium bifluoride’s risks— keeps everyone safer, healthier, and able to go home at the end of the shift.
What is the difference between Sodium Bifluoride and Sodium Fluoride?
Understanding the Chemistry
People often assume sodium bifluoride and sodium fluoride are interchangeable because of their similar names, but the chemistry tells a different story. Sodium fluoride comes with the formula NaF. It acts pretty straightforwardly: one sodium atom, one fluoride atom, and it dissolves smoothly in water. Sodium bifluoride, written as NaHF2, includes an extra hydrogen atom caught up with the fluoride. That little difference changes how the chemical behaves and where it gets used.
Where Each One Shows Up
Sodium fluoride feels familiar to many thanks to its connection with dental care. Toothpaste manufacturers use it for its cavity-fighting abilities. Municipal water systems add it to prevent tooth decay—a practice that kicked off in the last century and now covers millions of people in the United States. The Centers for Disease Control and Prevention highlights the public health benefit: fewer cavities, healthier mouths, and lower dental costs for families.
Sodium bifluoride doesn’t share that household fame. Its main gig happens in industry, especially on metal surfaces. Factories use it to clean or etch glass, remove rust from steel, or as a chemical intermediate for products like aluminum. When sodium bifluoride meets water, it gives off hydrofluoric acid—a nasty substance that eats through metal and glass alike. That means you find sodium bifluoride in factories and workshops, handled carefully with gloves and ventilation, not in kitchen cabinets or public waterworks.
Handling and Safety
Sodium fluoride, while not risk-free, gets shepherded into controlled amounts in food-grade products. Swallowing large doses can turn dangerous, but fluoride poisoning from toothpaste or tap water rarely happens. The U.S. Environmental Protection Agency and Food and Drug Administration both watch over how much sodium fluoride ends up in consumable products, and scientific reviews keep policy updated.
Sodium bifluoride brings much greater risk. Accidents can cause skin burns and severe respiratory problems. Hydrofluoric acid produced during its use penetrates tissue, causing pain and cell damage beyond where you’d expect. Hospitals treat chemical burns from it with calcium compounds to stop it from moving into bones and the heart. Workers exposed to sodium bifluoride get specialized training; personal stories from industry veterans often mention the critical safety drills and emergency wash stations nearby.
The Big Picture: Choosing the Right Compound
Each of these compounds serves a clear purpose. Research backs up sodium fluoride’s benefits for public dental health. The American Dental Association points to lower cavity rates in communities using fluoride. But putting sodium bifluoride into consumer-facing products would invite disaster. Businesses need strict rules and staff training for anything that involves bifluoride.
Mistaking one for the other doesn’t just lead to bad science; it invites real risk. Families, schools, and city officials picking products should check labels and safety data. Manufacturers benefit from consistent audits and regular staff education. New solutions in chemical containment and smart packaging can further reduce accidental exposure.
Making informed decisions about these chemicals isn’t just about chemistry—it’s about protecting lives and supporting health in our communities.
| Names | |
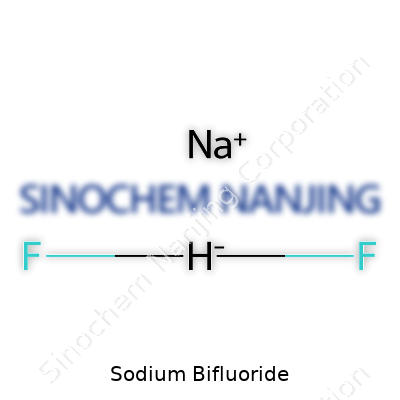
| Preferred IUPAC name | Sodium difluoride |
| Other names |
Sodium hydrogen fluoride Sodium acid fluoride |
| Pronunciation | /ˌsəʊdiəm baɪˈflʊəraɪd/ |
| Identifiers | |
| CAS Number | 1333-83-1 |
| 3D model (JSmol) | |
| Beilstein Reference | 3566858 |
| ChEBI | CHEBI:91250 |
| ChEMBL | CHEMBL1376 |
| ChemSpider | 69238 |
| DrugBank | DB11410 |
| ECHA InfoCard | 200-578-6 |
| EC Number | 231-663-8 |
| Gmelin Reference | 37760 |
| KEGG | C14045 |
| MeSH | D012972 |
| PubChem CID | 24557 |
| RTECS number | The RTECS number of Sodium Bifluoride is "WB0350000". |
| UNII | XE2P230B8Y |
| UN number | UN1813 |
| Properties | |
| Chemical formula | NaHF2 |
| Molar mass | 62.004 g/mol |
| Appearance | White crystalline solid |
| Odor | odorless |
| Density | 2.47 g/cm³ |
| Solubility in water | 29.2 g/100 mL (20 °C) |
| log P | -2.0 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 3.5 |
| Basicity (pKb) | 10.8 |
| Magnetic susceptibility (χ) | -49.0e-6 cm³/mol |
| Refractive index (nD) | 1.377 |
| Viscosity | Viscous liquid |
| Dipole moment | 6.1 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 57.70 J·K⁻¹·mol⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -694.06 kJ mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -749.7 kJ/mol |
| Pharmacology | |
| ATC code | V03AB16 |
| Hazards | |
| Main hazards | Toxic if swallowed, causes severe skin burns and eye damage, harmful if inhaled. |
| GHS labelling | GHS05, GHS06 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H314: Causes severe skin burns and eye damage. |
| Precautionary statements | P260, P264, P270, P271, P273, P280, P301+P330+P331, P302+P352, P304+P340, P305+P351+P338, P310, P312, P321, P330, P363, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-1-Acidity |
| Autoignition temperature | 515 °C (959 °F; 788 K) |
| Lethal dose or concentration | LD50 (oral, rat): 175 mg/kg |
| LD50 (median dose) | LD50 (median dose): 175 mg/kg (oral, rat) |
| NIOSH | NIOSH: WB6450000 |
| PEL (Permissible) | 2.5 mg/m³ |
| REL (Recommended) | 6 mg/m³ |
| IDLH (Immediate danger) | 30 mg/m3 |
| Related compounds | |
| Related compounds |
Ammonium bifluoride Potassium bifluoride Sodium fluoride Hydrofluoric acid |

