Sodium Bicarbonate: Origins, Science, and the Road Ahead
Historical Development
Long before modern chemistry gave sodium bicarbonate its official name, people tinkered with natural mineral sources for everything from cleaning wounds to easing digestive problems. In the late 18th century, French chemist Nicolas Leblanc stumbled on a way to produce “soda ash” from table salt, paving the road for sodium carbonate and, soon after, sodium bicarbonate. The Solvay Process from the 1860s took center stage in industry, using salt brine, limestone, and ammonia to crank out bicarbonate in bulk. Drawn to its gentle touch compared to caustic soda and lye, bakers, medics, and homemakers across continents grew to rely on it. The story of sodium bicarbonate runs alongside advances in chemistry and industry, proving that sometimes, very simple molecules can change how people work and live.
Product Overview
Most people know sodium bicarbonate as baking soda, a white, crystalline powder stashed in countless pantries and tucked behind the scenes in industrial plants. Though it might look unremarkable, this compound serves countless purposes: leavening baked goods, controlling pH in pools, slowing fires underground, soothing heartburn, and scrubbing away stains. Producers, from chemical giants to family-run suppliers, aim for purity and stable performance. Raw minerals or industrial byproducts serve as inputs, but the finished product lands on store shelves or in bulk containers as a reliable, everyday staple.
Physical & Chemical Properties
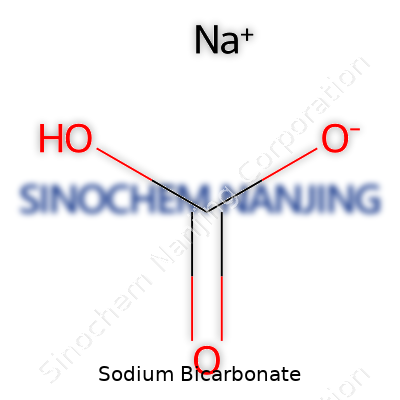
Sodium bicarbonate, with the formula NaHCO3, appears as a fine white powder or crystalline material. It does not smell and feels gritty between the fingers, much like salt. Storing it in a dry environment keeps it caking-free. It starts breaking down at temperatures above 50°C, and by 270°C it decomposes to sodium carbonate, water, and carbon dioxide. The slightly alkaline nature (pH about 8.3 in water) gives it the power to soften water, neutralize acids, and gently buffer solutions. Though stable, it reacts when combined with acids or high heat. The solubility runs about 9g per 100mL water at room temperature, which matters for food preparation or cleaning agents where consistency counts.
Technical Specifications & Labeling
Commercial sodium bicarbonate usually comes with a purity of 99% or higher, especially for food or pharmaceutical use. Technical grades allow a little leeway for minor impurities, usually capped at levels set by regulatory bodies like the US Food and Drug Administration, the European Food Safety Authority, or national equivalents. Labels show chemical formula, grade, batch number, and manufacturing date, plus hazard warnings for industrial-size shipments. Food packages must state “Baking Soda” or “Sodium Bicarbonate” plainly, along with net weight, intended uses, and in many countries, nutritional information as a food additive (E500(ii)). For pharmaceuticals, firms guarantee consistent chemical identity and absence of contaminants such as heavy metals and unwelcome carbonate. On the industrial side, bulk shipments travel in lined bags, drums, or silos with clear tagging and safety sheets in tow.
Preparation Method
Today’s large-scale sodium bicarbonate production relies on the Solvay process. Salty water pumped from wells mixes with ammonia gas and carbon dioxide. This technique yields solid sodium bicarbonate, which filters out, and the process loops on thanks to resource recycling. After drying and purification, the product reaches the needed standard for each application. Specialty sodium bicarbonate for high-purity pharmaceutical or analytical use often relies on extra filtration or recrystallization, stripping away even traces of unwanted ions. Adjusting the process flow allows manufacturers to tweak granule size and bulk density for specific uses, from tablet making to powder blending.
Chemical Reactions & Modifications
Mix sodium bicarbonate with acids—vinegar, lemon juice, or hydrochloric acid—and a fizzing reaction begins. The compound breaks apart, unleashing carbon dioxide, water, and a leftover salt. This behavior underpins its use in baking and fire extinguishers. Strong heat or concentrated bases also cause it to break down, forming sodium carbonate, steam, and CO2. Chemists tweak the molecule for advanced uses: surface coatings in tablet formulations, encapsulation for slow-release fertilizers, or even blending with other salts for custom blends in water treatment. In recent years, research labs explored grafting the bicarbonate anion onto polymer backbones, seeking out new catalysts or capture agents for carbon dioxide.
Synonyms & Product Names
Walk through a supermarket, hardware store, or laboratory, and sodium bicarbonate reveals itself under many aliases. Baking Soda stands as the everyday household favorite, while industrial and technical circles still call it Bicarbonate of Soda. E500(ii) shows up on ingredient lists in Europe. Other product names in specialty trades include Sodium Hydrogen Carbonate and, rarely, Nahcolite—coined after a natural mineral source. Some cleaning and health labels highlight “Alkaline Powder” or “Acid Neutralizer,” which, though less precise, call attention to its key properties. On the supply chain, names like USP Sodium Bicarbonate or Food Grade NaHCO3 clearly signal the intended use and underlying quality standards.
Safety & Operational Standards
Few chemicals bridge food, medicine, and heavy industry quite like sodium bicarbonate. Regulators do not treat it as a hazardous chemical under usual conditions. In contact with skin, it rarely causes irritation, and accidental ingestion in reasonable doses poses little risk. But no one should overlook particle exposure; large amounts can irritate the eyes, mouth, or lungs. Industrial workers handle it with gloves, masks, and dust control. Safety Data Sheets spell out procedures for spills, fire, or long-term storage. Storage tanks, bins, and packaging must stay dry—contact with moisture triggers lumping and chemical breakdown. In water or effluent treatment, legal limits on sodium and carbonate discharge still apply, so environmental controls earn attention. Food and pharma standards demand traceability from batch to shelf, with regular checks for heavy metals and microbial contamination.
Application Area
Most people bump into sodium bicarbonate through baking or cleaning, but its real reach goes much further. Bakers depend on it for fluffy cakes or biscuits, thanks to the steady carbon dioxide gas it produces with heat or acid. Hospitals stock sodium bicarbonate as an injectable life-saving buffer against acidosis. Firefighters use it in dry chemical fire extinguishers—its reaction chokes off flames. Homeowners, janitors, and laundry workers value it as a gentle scouring powder, odor remover, and water softener. Farmers dust it on crops to stop mildew or mix it with feed as a pH adjuster. Municipal waterworks dose it to keep pipes corrosion-free. Dentists bake it into toothpastes as a mild abrasive and acid neutralizer. Laboratories rely on it as an easy-to-measure alkaline buffer. In animal care, it balances rumen pH for healthier livestock. Swimming pool operators dump it into cloudy water, dialing in the desired alkalinity with simple tests and a scoop straight from the bag.
Research & Development
Recent science stretches sodium bicarbonate into new territory. The carbon capture industry eyes it for affordable CO2 scrubbing solutions, hoping to plug it into pilot plants and, down the road, city-scale emissions control. Pharmaceutical firms experiment with custom-engineered granules to deliver fast-acting or slow-release tablet doses for emergency IVs and at-home remedies. In environmental protection, scientists track its power to limit heavy metal uptake in soils or treat acid mine drainage, cutting the cost and toxicity of water cleanup. Medical research revisits its role in managing chronic kidney disease, new cancer therapies, and shock recovery, balancing the upsides of alkalinization with known risks. Biotech startups poke at sodium bicarbonate’s buffering action to control pH in microbe and cell cultures. In sport, endurance athletes look for performance edges by dosing “bicarb” under medical guidance, pushing the boundaries with strict oversight around safe dosing and individual responses.
Toxicity Research
Toxicology studies place sodium bicarbonate in the “generally recognized as safe” camp for food and medicine, but only when folks stick to labeled dosages. Swallowed in large amounts or injected incorrectly, it throws off the body's salt balance, driving up blood sodium and causing metabolic alkalosis. That risk looms large in elderly patients or those with kidney or heart trouble. Occupational exposure to dust causes sneezing, coughing, and rarely, mild eye or skin irritation. Environmental experts checked its movement in soil and water; under most situations, bicarbonate breaks down quickly, posing little persistent risk. Still, aquatic life does not tolerate massive doses, so major spills into waterways need prompt cleanup. The safety conversation now extends to chronic low-level exposure, especially for workers in low-ventilation plants or those handling the pure powder near food or pharmaceutical lines.
Future Prospects
The world keeps finding new ways to use sodium bicarbonate. Clean energy projects tap it for carbon capture, hoping to draw down emissions at every scale, from power plants to indoor air scrubbers. Battery makers and tech startups research its role in sodium-ion and lithium-ion storage, hunting for green, cheap materials. Scientists probe its impact on gut health and chronic disease, aiming for tailored dietary supplements or advanced therapies. Water utilities want new blends and custom granules for cost-effective, large-scale water treatment. Industrial cleaning firms see promise in bicarbonate blasting, offering a safer, less abrasive alternative for cleaning engines, monuments, and delicate equipment. Regulatory agencies monitor environmental and workplace exposure, updating standards to reflect new evidence. The old, simple “baking soda” of the past keeps adjusting, surprising even the chemical insiders who thought they knew all its tricks.
What is sodium bicarbonate used for?
Baking, Cleaning, and Health All Lean on This Classic Household Ingredient
Sodium bicarbonate, most folks call it baking soda, has a long reputation as the handyman of the household. My grandmother swore by it, whether she was whipping up biscuits or scrubbing stains out of my baseball uniform. Some stories get passed through generations for good reason—baking soda shows up everywhere, quietly solving problems that pop up in kitchens, bathrooms, and even garages.
A Reliable Leavening Agent
Baking soda goes into banana bread and pancakes. It reacts with acids like buttermilk or lemon juice, helping dough rise and get fluffy. Baked goods wouldn’t feel right without it; cakes stay dense without some way to produce carbon dioxide bubbles in the oven. This little white powder turns everyday recipes into family favorites. According to the American Chemical Society, baking soda’s chemical reaction gives off carbon dioxide gas, which forms bubbles and gives that “just right” crumb in things like cookies and muffins.
A Workhorse for Cleaning Jobs
This stuff isn’t just for the kitchen. Baking soda blends into a paste with water and scrubs away burnt food on pans and rings in the tub. Its gentle abrasiveness protects surfaces you care about, like countertops and sinks, without scratching finishes. My own kitchen sink gets a regular dose when nothing else lifts stubborn stains after a long night of cooking. The Environmental Protection Agency notes that sodium bicarbonate, as part of homemade cleaning mixtures, reduces reliance on commercial chemicals—cutting down on both harsh fumes and plastic waste.
Freshening and Deodorizing
Pretty much every fridge in America holds an open box of baking soda tucked in a corner. Even as a kid, I noticed the difference; strong odors from onions and leftovers just fade. That box traps smells, keeping them from spreading to milk or fruit. Years later, I see it in laundry rooms and sneakers, where baking soda handles smells that detergent alone can’t crack.
Supporting Health at Home and in Hospitals
Doctors rely on sodium bicarbonate too. Emergency rooms use it for treating certain types of overdoses and severe acid buildup in the blood. At home, some folks turn to it for heartburn, mixing a bit in water for quick relief from stomach acid. Yet, it’s best to use caution—overusing it can upset natural balances in the body. Speaking personally, I trust this more as a kitchen staple and a cleaner than as a go-to medicine, and I always check with a professional before trying any home remedy.
Fire Safety and Pool Care
Kitchens sometimes deal with grease fires, and baking soda smothers flames better than water, which spreads grease around. I once saw a neighbor save his stove thanks to a quick shake of it. Pool owners add sodium bicarbonate to bring pH in line, protecting plumbing and skin. These uses often fly under the radar but carry real weight for home safety and comfort.
Easy, Affordable, and Trusted
Baking soda stands tall because it’s cheap, versatile, and easy to find. A box costs less than a coffee, yet keeps homes cleaner, safer, and food tastier. The world is full of fancy cleaners and complicated fixes, but sometimes simple answers—like a scoop of sodium bicarbonate—really are best. My own shelves never go without it, and judging by most kitchen cabinets I’ve seen, I’m not alone.
Is sodium bicarbonate safe for consumption?
Looking at the Everyday Ingredient
Most pantries hold a box of baking soda. Few stop and ask if it's safe to use in baking, cleaning, or even dealing with an upset stomach. Sodium bicarbonate gets handed down through generations as a remedy for heartburn, a cleaning tool, and a staple for baked goods. It looks innocent, but people should look closer at what regular use means for health.
Sodium Bicarbonate’s Role in the Body
Sodium bicarbonate turns up in the medicine cabinet and the kitchen. The body actually produces a form of bicarbonate to support acid-base balance, especially in the blood. This helps neutralize excess acid, which lies at the root of why people reach for baking soda to ease indigestion. In small quantities, sprinkling a bit into a glass of water won’t harm most adults. The FDA even recognizes it as generally safe in food.
How Much Is Too Much?
Problems tend to show up when someone goes overboard. It's easy to assume that if a little is good, more should be better. That doesn’t hold true with sodium bicarbonate. Each teaspoon packs more sodium than some folks realize—about 1,259mg. If you have high blood pressure or heart trouble, that much sodium at once can push blood pressure higher or strain kidneys. I’ve known relatives who used baking soda to settle their stomachs and ended up bloated and uncomfortable.
Medical cases warn us about the risks. Swallowing large amounts can disrupt the blood’s acid-base balance, leading to metabolic alkalosis. This condition makes muscle cramps, confusion, and twitchiness show up, and severe cases send people to the hospital. The risk goes up for the elderly or those with kidney issues, since their bodies struggle to get rid of excess sodium and bicarbonate.
Baking and Cooking Uses
In most kitchens, sodium bicarbonate gets measured in teaspoons. Baked goods like cakes, cookies, and pancakes rely on it for fluffy texture. This type of use never causes trouble, since the actual chemical reaction uses up the sodium bicarbonate—there’s less impact on sodium intake by the time the muffins cool. The only kitchen risk comes from a handful of recipes that call for way too much baking soda, which can leave an unpleasant taste and add too much sodium.
Using Baking Soda Safely for Health
Doctors sometimes recommend it for specific problems. For example, it neutralizes acid in the stomach, easing some ulcers or kidney conditions. In these situations, the dose matters. No one should self-medicate without checking with a doctor, because risks stack up quickly if you take it for longer than a few days.
I’ve seen friends make a quick paste for bug bites, clean coffee stains off mugs, and even freshen the fridge—all without concern. For cleaning, it’s safe. For eating or drinking, moderation rules. Cutting back on extra salt elsewhere in the diet can help balance total sodium intake. Anyone with health concerns should talk with a doctor before reaching for that orange box.
Looking Forward: Safer Habits
Sodium bicarbonate works wonders when used thoughtfully. Reading labels and understanding serving sizes makes a difference, especially in the age of processed foods. Many know it can settle a queasy belly, but few realize how easily habits can turn into trouble. Sometimes the simplest kitchen staple deserves a moment’s pause and a little math before it goes in the cup or pot.
Can sodium bicarbonate be used for cleaning?
Looking Beyond the Kitchen
Most pantries hide a box of sodium bicarbonate, often labeled as baking soda. It's not just for fluffy cakes or settling an upset stomach. For decades, plenty of homes have reached for this white powder to tackle just about any cleaning job. Growing up, my mother would pour baking soda down our kitchen sink and chase it with vinegar. We’d listen for that fizz, then chase it down with boiling water. Old pipes ran clearer, not just because a commercial promised it, but because we trusted what we saw in our own home.
How Does Baking Soda Clean?
Sodium bicarbonate can tackle messes because it’s slightly abrasive and mild enough for most surfaces. This makes it useful on grimy stovetops, stained mugs, and even tiled bathroom floors. Mixed with water, it becomes a gentle scrubbing paste. Science backs this up: the gritty texture scrapes off grime, and the alkaline pH interrupts the stickiness of oily stains. That means less elbow grease and no harsh chemicals lingering in the air or on your hands.
Comparing Baking Soda to Commercial Products
With so many cleaning sprays on the market, it’s easy to overlook baking soda. Commercial products can include fragrances, foaming agents, and strong solvents. These can irritate sensitive skin and pollute indoor air. The Environmental Working Group has flagged concerns about synthetic cleaners, especially for kids and pets. Baking soda keeps things simple. It doesn’t cover up dirt with a candy-like scent or leave chemical deposits behind. There’s a reason schools and hospitals recommend it for safe, nontoxic surface cleaning.
Real-World Success Stories
Anyone who ever owned a coffee machine knows stains collect over time. My own has fended off brown rings for years with nothing more than a tablespoon of baking soda and hot water. Carpets, too, benefit from a light dusting; let it sit, then vacuum it up to help eliminate odors. The science supports this—research out of the University of Oxford points to sodium bicarbonate’s odor-neutralizing power against stubborn smells from pets, socks, or old food spills.
Limits and Responsible Use
Sodium bicarbonate isn’t perfect. It won’t fight bacteria with the same punch as bleach, nor can it dissolve all mineral buildup from hard water. Certain surfaces, such as waxed wood and aluminum, can develop damage or discoloration after frequent scrubbing. For deep kitchen sanitizing or killing viruses, professional products often rely on EPA registration for proven germ-fighting action, which baking soda does not hold. Crowdsourced tips from cleaning forums often overlook this, so it pays to know what each job demands.
Balancing Tradition and Innovation
Rather than picking one cleaner for every job, it makes sense to match the solution to the problem. Sodium bicarbonate has a place on the shelf because it works well for daily cleaning, scrubbing, and reducing smells without strong scents or chemical residue. In kitchens with kids or pets, or for folks worried about allergies, it’s become a preferred choice. Each time I reach for that familiar orange box, I recall generations leaning on simple chemistry for honest results.
What are the side effects of sodium bicarbonate?
The Real Story Behind the Powder in the Pantry
Sodium bicarbonate has always been right there in the kitchen, ready for baking, cleaning, or easing the sting of heartburn. It seems like a harmless white powder. Still, using it as a quick fix or home remedy can bring some side effects that fly under the radar. Some of these issues surprised me the first time I reached for the box during a bout of reflux, thinking nothing could be safer than a spoonful of baking soda in water.
Digestive Tract Takes the Hit
Swallowing sodium bicarbonate sets off changes in the stomach. It neutralizes stomach acid fast, but that relief doesn’t always last. I’ve noticed people feel bloated or gassy after taking it. The fizzing in a glass also happens inside the gut, which leads to burping and, sometimes, abdominal pain. Folks who use it often to tame acidity can see it backfire, since the body tries to make even more acid in response, making heartburn stick around.
Salt in the Bloodstream
Each teaspoon packs a big dose of sodium. Too much sodium raises blood pressure and strains the heart and kidneys. I spoke with a neighbor who ended up in the ER after using baking soda as a laxative; his blood sodium shot up, causing confusion and muscle twitching. The risks climb for older adults, folks with kidney disease, or people taking certain medicines. Prescription meds might not work as planned if sodium bicarbonate changes the stomach’s pH, throwing off absorption.
Metabolic Alkalosis
Doctors see metabolic alkalosis most often in people who use large amounts of baking soda. The body turns too alkaline, and symptoms can sneak up: shakiness, muscle cramps, and even trouble breathing. I’ve seen how electrolyte levels, especially potassium, drop too low in these cases, leading to heart rhythm problems. Athletes sometimes use sodium bicarbonate for workouts, but even they risk this dangerous side effect if dosing goes overboard.
Interactions Affecting Longevity and Health
People taking certain meds—diuretics, aspirin, or lithium—might not realize mixing in extra sodium bicarbonate throws off how their medicine works. I remember reading research in The American Journal of Emergency Medicine: some patients landed in the hospital with symptoms traced to these kinds of interactions. Not every bottle or website warns about this, making it easy to overlook.
Who Faces the Highest Risk?
Children should not take sodium bicarbonate without a doctor’s okay; their kidneys can’t always get rid of extra sodium the way an adult’s can. Pregnant women and seniors often deal with blood pressure swings or fluid buildup more acutely, so even modest doses can push them into dangerous territory.
Looking for Safer Solutions
Instead of reaching for the baking soda box at the first sign of stomach troubles, some turn to lifestyle changes—skipping late-night meals and limiting acidic or spicy foods helps me avoid heartburn. For people dealing with acid reflux, medical consultation brings safer, targeted medicine options. Staying hydrated, eating balanced meals, and monitoring salt intake matters far more than adding a dash of powder for a quick fix.
Final Thoughts
A sprinkle of sodium bicarbonate in a recipe or to scrub a coffee mug won’t cause harm. When it becomes a regular part of self-care, especially without guidance, the risks stack up fast. Always talk with a healthcare professional before turning to it for more than an occasional mild fix.
How should sodium bicarbonate be stored?
Keeping Baking Soda Fresh at Home
Every kitchen seems to have that familiar orange box in the cupboard or refrigerator. I remember my grandmother would swear by tossing a box in the fridge to soak up odors, while also using it for baking and cleaning. But more than once, I saw lumps forming or powder caking in damp corners, and that’s not what you want in a baking ingredient.
Baking soda—sodium bicarbonate—is a household staple. Clumping and contamination can ruin it. Keeping it dry tops the list. Moisture breaks down the fine powder, causing it to clump. That not only makes measuring a headache but can also wreck a recipe in the kitchen. The answer lies in simple storage solutions. An airtight container with a tight-fitting lid keeps moisture out. Glass jars with rubber seals do the trick, as do sturdy food-grade plastic tubs. Factory packaging isn’t built to last. Moving powder to a sealed secondary container makes sense.
Dealing with Bulk Purchases
Some folks like to buy in bulk for cleaning, pool maintenance, or even feeding backyard chickens. Bulk bags and cardboard boxes don’t hold up once opened. The moment air gets in, stray humidity follows. After purchasing a large amount, it helps to split it into several smaller, airtight containers. Mark the date on the container so it’s easy to track how long it’s been sitting on the shelf. The FDA says sodium bicarbonate has a long shelf life—often years if stored well—but direct sunlight speeds up degradation, so a cool, shaded place adds extra insurance.
Why Avoid Certain Storage Spots?
Under the sink seems handy, but that’s ground zero for sneaky leaks and extra humidity. The fridge absorbs odors, but the open box method contaminates the powder for other uses. Kitchens grow warm during cooking, and temperature swings encourage condensation inside containers. Repeated exposure breaks down the consistency and potency. I have found cabinets high up, away from steam and splashes, keep things pristine for much longer. A dark pantry shelf beats a sunny windowsill every time.
Preventing Contamination
Baking soda acts like a sponge, soaking up smells and even tastes around it. Storing it near spices, coffee, or strong-smelling cleaning agents gives it a strange flavor twist. Nobody wants cinnamon-scented biscuits unless it’s on purpose. Food-grade material makes a difference. Reusing an old pickles jar without a thorough wash leaves a faint tang behind. Cleanliness counts if the powder is going into anything edible.
Using Science and Common Sense
Sodium bicarbonate remains stable at room temperature in a dry environment. Testing freshness isn’t hard—a teaspoon in a bit of vinegar should fizz right away. If it barely bubbles, that’s a warning sign. Older powder in homemade fire extinguishers or pool maintenance may still work, but baking calls for the fresh stuff.
So many pantry mishaps can be dodged by treating baking soda like other ingredients—protect from light, air, and moisture. A little care in storage saves money, reduces waste, and keeps baking success within reach. Anyone who counts on consistent results—from cooks to chemistry teachers—should take those extra steps to keep sodium bicarbonate in top condition.
| Names | |
| Preferred IUPAC name | Sodium hydrogencarbonate |
| Other names |
Baking Soda Bicarbonate of Soda Sodium Hydrogen Carbonate E500 Bread Soda |
| Pronunciation | /ˌsəʊ.di.əm baɪˈkɑː.bə.neɪt/ |
| Identifiers | |
| CAS Number | 144-55-8 |
| 3D model (JSmol) | `data:Molecule Na+ C(=O)[O-]` |
| Beilstein Reference | Beilstein Reference: 35354 |
| ChEBI | CHEBI:32139 |
| ChEMBL | CHEMBL1357 |
| ChemSpider | 5749 |
| DrugBank | DB01390 |
| ECHA InfoCard | ECHA InfoCard: 029-003-00-5 |
| EC Number | 011-005-00-2 |
| Gmelin Reference | 63519 |
| KEGG | C00288 |
| MeSH | D015928 |
| PubChem CID | 516892 |
| RTECS number | VZ0950000 |
| UNII | FK37T614FG |
| UN number | UN3077 |
| Properties | |
| Chemical formula | NaHCO3 |
| Molar mass | 84.006 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 2.20 g/cm³ |
| Solubility in water | 7.8 g/100 mL (20 °C) |
| log P | -4.7 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 6.3 |
| Basicity (pKb) | pKb = 7.7 |
| Magnetic susceptibility (χ) | 'Magnetic susceptibility (χ) = -49.0 × 10⁻⁶ cm³/mol' |
| Refractive index (nD) | 1.380 |
| Dipole moment | 1.41 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 102.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −947.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -947.7 kJ/mol |
| Pharmacology | |
| ATC code | A02AA02 |
| Hazards | |
| Main hazards | Not considered hazardous under normal conditions. |
| GHS labelling | **"Not a hazardous substance or mixture according to the Globally Harmonized System (GHS)"** |
| Pictograms | GHS07 |
| Signal word | No Signal Word |
| Hazard statements | Not a hazardous substance or mixture. |
| Precautionary statements | P264, P270, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 0-0-0 |
| Lethal dose or concentration | LD50 (oral, rat): 4220 mg/kg |
| LD50 (median dose) | LD50 (median dose): 4220 mg/kg (oral, rat) |
| NIOSH | VZ0950000 |
| PEL (Permissible) | 15 mg/m3 |
| REL (Recommended) | 1.0 g |
| Related compounds | |
| Related compounds |
Sodium carbonate Sodium hydroxide Sodium chloride Potassium bicarbonate Calcium carbonate Ammonium bicarbonate |

