Sodium Azide: Navigating History, Science, and the Road Ahead
Historical Development
Once used primarily as a laboratory curiosity, sodium azide has slipped quietly through the back doors of both industrial chemistry and safety engineering. The early 20th century saw it introduced by German scientists, who managed to isolate it while experimenting with hydrazine compounds. Years rolled forward, and researchers noticed its lively tendency to decompose into pure nitrogen gas and sodium metal. This energetic reaction soon landed sodium azide a controversial but vital role in propelling the technology behind automobile airbags. For a while, it seemed incredible that a lab chemical first tinkered with by European chemists would soon become a silent, invisible life-saver embedded in every modern car. Yet that’s how progress works—chemicals often gain a second, unexpected life in the hands of sharp engineers and cautious risk-takers.
Product Overview
Sodium azide is a fine white powder, often found packaged in drums lined with plastic to keep moisture at bay. It doesn't really draw attention until its unique talent is put to use: explosive gas generation. Simple and compact, the powder transforms under the press of a charge, filling airbags in split seconds, a feat most chemicals can only dream of matching for speed and reliability. Long before this use took off, sodium azide stayed in the shadows as a reagent for chemical synthesis, a preservative for biological samples, and a starting point in making other energetic materials.
Physical & Chemical Properties
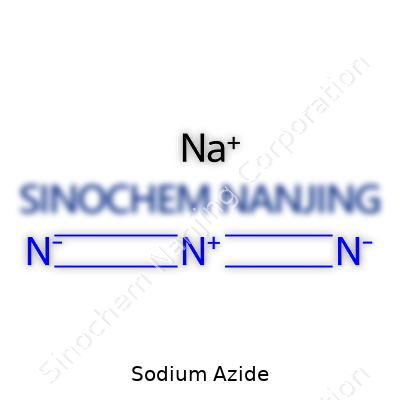
Sodium azide is a white, crystalline solid with a surprisingly simple formula—NaN₃. The crystals dissolve well in water; the solution usually remains colorless and has a faint musty odor. At room temperature, this compound remains stable, but heating beyond about 300°C flips a chemical switch and, through a simple decomposition, nitrogen gas erupts from the solid phase with startling force. What stands out most for chemists is this balance of stability and volatility—the trick is knowing just how much energy has to be pumped in before sodium azide breaks down.
Technical Specifications & Labeling
Anyone who’s handled sodium azide in a lab knows about its clear hazard labels—those bright diamonds warning of acute toxicity and its shocking ability to explode if treated carelessly. Labels avoid being decorative: details like purity, percentage of water content, and storage temperature get listed right next to the red warning symbols. Industry and academia both push for precise lot numbers and dates of manufacture, since keeping track of shelf life and degradation is just as important as knowing what’s inside the container. This isn’t just red tape; mishandling sodium azide over the years has caused accidents, pushing regulators to demand transparency and traceability through exhaustive labeling practices.
Preparation Method
Making sodium azide isn’t for the faint of heart. Traditional preparation starts with sodium nitrate and hydrazine sulfate. After a round of careful mixing and controlled reactions, the end result is filtered and crystallized from solution. Every chemist I’ve known who’s attempted this synthesis speaks about it with a mix of pride and fear. The process produces hazardous byproducts and requires proper ventilation, chemical-resistant gloves, and constant monitoring. Scale-up efforts in industry pay just as much attention to air flow, waste management, and the risk that impurities might coax out unwanted sensitivity in the final product.
Chemical Reactions & Modifications
Sodium azide dances around a few core reactions. It breaks down neatly to give nitrogen gas and sodium upon heating or impact. In the hands of synthetic chemists, it serves as a key source of the azide ion—useful for “click chemistry” and forming complex structures familiar to drug developers. Sometimes, labs tweak the azide itself, exchanging the sodium ion for heavier metals, producing explosive derivatives that fuel sky-high pyrotechnics or specialized detonators. Despite the creative chemical gymnastics, most modifications must consider safety first. A safer road involves keeping sodium azide under cold storage, sealed, and away from shock, friction, and incompatible chemicals like acids (which can release toxic, volatile hydrazoic acid gas).
Synonyms & Product Names
Sodium azide doesn’t confuse many with its naming. You’ll hear people call it by its chemical formula, NaN₃, just as often as “Laboratory Preservative Number 1” in some teaching labs. Sometimes, suppliers label it “Sodazide” or wrap it under “Preservative 30” for specialty applications. The name might shift, but in every context, the risk warnings and applications point to the same field of energetic, reactive chemistry.
Safety & Operational Standards
Everyone who handles sodium azide gets drilled on the dangers. It’s known for quick, violent decomposition, especially in solid state or as a dust. Ingenuity in chemistry often collides with the cold reality of risk, so labs and factories stash sodium azide in climate-controlled rooms, locked away from heat or physical shock. Spillage control gets real emphasis, as the dust should never meet heavy metals or acid wastes, unless someone feels like courting a disaster. Government agencies audit the workspaces, demanding written protocols and detailed records of every gram purchased, used, or discarded. Workers in the airbag industry, for example, train on what to do if an errant spark shows up near a pile of stored azide—fire suppression goes hand-in-hand with explosion mitigation, not just for the safety of the crew, but for the homes and neighborhoods downwind.
Application Area
Sodium azide finds its leading role in vehicle airbags, where ingenuity meets necessity. The moment a car crashes, a sensor fires, current surges, and sodium azide transforms, unleashing a cloud of nitrogen gas that inflates the protective bag in less than 50 milliseconds. That same rapid gas release put sodium azide into other fields, from rocket propellants for tiny satellites to gas-phase preservative systems for certain medical machines. In the lab, it’s a go-to for inhibiting unwanted bacterial growth in biological samples—nothing halts metabolism quite like a poison that jams cellular respiration. Some researchers keep sodium azide handy for introducing azide groups into organic molecules, useful for new drug formulations or tracking biological processes.
Research & Development
Research tends to flow in cycles—new ways of making sodium azide more safely, discovering replacements that work just as well for airbags without the toxicity, or finding ways to recover or recycle unused material. A fair amount of effort goes into developing catalysts that help create sodium azide at lower temperatures or with higher yields, sparing both energy and cost. Labs investigating bio-orthogonal chemistry keep sodium azide front and center, since it performs well during “click” reactions, snapping molecular pieces together without interfering with sensitive biological matter. Academic and commercial teams race to balance performance with safety, which often means hunting for alternative compounds or fresh approaches to decomposition and disposal.
Toxicity Research
Sodium azide doesn’t hide its dangers. The compound blocks cytochrome oxidase in cells, which leads to rapid systemic shutdown in animals, including humans. Acute exposure can lower blood pressure, damage nervous and cardiovascular tissue, and result in organ failure at doses not much higher than you might accidentally inhale in a cloud of dust. On a gut level, this hits hard—long before toxicology studies mapped out the exact mechanism, older chemists told stories of colleagues who blacked out or suffered from headaches after a careless spill. Modern research drills into mitigation, antidote development, and quick response guidelines. Wastewater plants hate the stuff; just a few milligrams kill off the bacteria that process sewage, so industrial disposal practices tighten year by year. Researchers have measured the compound’s environmental half-life and push for better neutralization protocols to avoid accidental poisoning via water runoff.
Future Prospects
Chemists often debate where sodium azide fits into the next phase of safety technology. Newer airbag systems experiment with tetrazoles and other safer azide-free compounds, but none quite hit the cost-effectiveness and instantaneous gas release sodium azide brings to the table. At the same time, chemical firms and green engineering projects keep searching for cleaner synthesis routes, reduced-waste processing, and improved recycling. Environmental groups push for a world in which smart design and advanced materials hand off the baton from sodium azide without sacrificing public safety. For now, it remains an uneasy hero in life-saving technology, a perilous yet valuable staple of modern science and engineering. As research deepens and regulations tighten, the real challenge is finding replacements that leave sodium azide’s dangers—and its unique advantages—safely in the past.
What is sodium azide used for?
The Science Behind Its Use
Mention sodium azide, and most folks remember high school chemistry. It's the white powder nobody wanted to spill. In reality, sodium azide pops up in more places than most people realize. This compound carries a lot of punch for its size. Cars wouldn’t be nearly as safe without it. Sodium azide creates the explosive gas that fills airbags the instant an impact occurs. Roughly every modern airbag system relies on that fast chemical reaction, which converts sodium azide into nitrogen gas—just what’s needed to create the cushion between passengers and dashboards. A single gram can generate a large volume of pressurized gas in milliseconds. The gas doesn’t burn you, but the process can get hot, and the byproducts pose their own risks.
Factories don’t just use sodium azide for airbags. In the lab, sodium azide helps make pharmaceuticals. Scientists tap into its ability to release nitrogen atoms, which makes it a handy ingredient for some antibiotics or blood pressure medications. It even assists researchers working with DNA, helping break it into smaller fragments. In hospitals, azide plays a smaller but important role as a preservative in some diagnostic kits—preventing bacteria from ruining test results.
The Risks Outweigh The Benefits Without Training
Growing up with a teacher who doubled as a volunteer firefighter, I always heard stories about lab mishaps. Sodium azide is notorious for being toxic. Even tiny amounts can cause big trouble. The gas it releases can disorient, cause headaches, or worse if inhaled. Spills in car assembly lines or laboratories could threaten workers if not handled properly. It reacts violently when mixed with heavy metals, risking dangerous explosions.
Statistics show that emergency rooms deal with accidental sodium azide exposure more often than you’d think. Between 1989 and 2018, the CDC tracked over 20 cases of workplace exposures, some fatal. Poison control experts rank it just below cyanide on the danger scale. Most folks at home or in smaller businesses should never come close to sodium azide, but online shopping has made hazardous chemicals easier to get than ever.
Safety Is Key—And So Is Oversight
Realistically, keeping the public safe means teaching those who handle sodium azide what not to do. That training can’t be just a few slides or a quick chat. Factories with airbag lines train staff regularly and invest in protective gear. Labs need clearly posted protocols for storage, handling, and cleanup. Rules around shipment and secure storage help keep it out of the wrong hands. Hospitals keep it locked up too, with staff double-checking labels so it doesn’t end up in the wrong place.
The internet gave easy access to chemicals years ago, but responsible sellers vet buyers. Online marketplaces need to keep vetting stricter, flagging suspicious purchases to regulators. Community outreach and education offer an added layer—reminding the average person just how easy it is for things to go wrong with a few grams of the wrong substance.
Practical Tips Make The Difference
Knowledge protects more than regulations alone. Training drills, quick access to eye wash or safety showers, and proper chemical labeling save lives. If you work near sodium azide, knowing how to react within seconds—the habits you pick up from repeated practice—will make all the difference. Taking its risks seriously means fewer headlines, fewer accidents, and more lives saved before they even know they’re at risk.
Is sodium azide dangerous or toxic?
Understanding What’s at Stake
I’ve seen a lot of buzz lately about sodium azide, especially with its role in car airbags and sometimes turning up in the news when it ends up in places it never should. Many folks don’t recognize the name, but anyone who’s ever popped an airbag in a crash has relied on this compound's explosive power. For all its usefulness, this isn’t something to take lightly or treat like table salt.
Why Sodium Azide Matters
Sodium azide gets put to work in airbags because it converts quickly into nitrogen gas, creating a rapid inflation that can save lives. In research labs, it's used to stop bacterial growth or as a chemical reagent. Despite those solid uses, everything flips when this chemical is handled carelessly.
Direct contact with sodium azide can burn the skin or damage the eyes. Swallow a small amount, and risk sharp drops in blood pressure, headaches, vomiting, and even coma. My time in science taught me just how cautious you need to be. Fact: According to CDC data, exposure can kill at a dose of just a few hundred milligrams — less than a teaspoon. Toxicity doesn’t care about good intentions.
History and Call for Respect
There’s a tragic history here. Accidental poisonings make headlines every few years. In hospitals, tiny amounts of commercial sodium azide sometimes ended up in wrong places, causing severe illness and even death. One reason this happens: sodium azide dissolves easily in water and can’t be seen or tasted. Once it’s inside the body, it disrupts cell function, something most people never see coming.
The numbers reinforce the problem. According to Poison Control, there were over 100 incidents in one decade involving this compound worldwide. Most incidents involved accidental lab exposure. Still, each one highlights how unforgiving this chemical can be outside safe, controlled settings.
Practical Ways Forward
Education remains the best defense against dangerous mishandling. In my lab, nobody touched sodium azide without gloves, face shield, and a fume hood. We had strict labels, locked cabinets, and clear protocols. For industries, every crew member needs up-to-date training and easy access to emergency wash stations. It shouldn’t take a tragedy to revisit safety plans.
Disposal needs special attention. Pouring it down the drain or tossing it with trash brings real risk, including creating explosive byproducts if it meets heavy metals or acids. The EPA recommends full-on hazardous waste procedures — anything less just gambles with water safety and public health.
The Big Picture
Sodium azide is a good example of science and industry walking a risky line. Useful, but only in the right hands and with respect for just how potent it can be. No shortcut replaces training, awareness, and serious safety measures. With these in place, the benefits can continue, and the risks can be managed, but the lessons come from real-world experience and vigilance, not from a label on a bottle.
How should sodium azide be stored and handled?
Getting Real with Sodium Azide
Some chemicals carry risk that few people recognize until a close call brings it home. Sodium azide is one of those. I’ve worked in labs where a pinch went a long way—energetic, toxic, ready to change form if treated wrong. It only takes a quick read through a chemical safety board report to realize how things can go sideways in routine work. Sodium azide gets a spot on every lab’s list of substances to respect.
Strict Guidelines for Storage
Sodium azide calls for more than a labeled bottle on the reagent shelf. It thrives in dry, cool spaces, away from sunlight and humidity. Moisture should never get near it since water can unleash hydrazoic acid fumes, which corrode lungs and metal pipes. Flammable materials never share a locker with sodium azide; oxidizers, acids, and even some plastics can set off reactions. Stainless steel and some specific plastics work for containers, but metals like copper or lead turn risky fast as they react to produce explosive metal azides. Storage away from drainage and sewer connections changes the outcome if a spill comes up; sodium azide and plumbing don’t mix, as pipes might slowly accumulate shock-sensitive byproducts.
In my own lab days, a minimalist approach helped—never stock up more than necessary. Small vials mean easier control. A cabinet marked for poisons, with good ventilation, offers far more than just another locked shelf. Regular inventory checks keep forgotten stashes from aging and degrading, which brings hidden dangers.
Handling Basics the Safe Way
People get used to their routines, but the routine with sodium azide needs thoughtful care each time. Lab coats, chemical goggles, and nitrile gloves come standard. Disposable spatulas reduce the risk of cross-contamination. Any open handling happens in a fume hood, not at an open bench, to save your lungs from toxic dust or vapor. Colleagues get a quick heads-up—the more eyes on safety, the better.
I remember a colleague who wiped down a bench and landed in the hospital after not checking for residue—one slip, and sodium azide does not give second chances. Open-source accidents illustrate the same point: label everything, train every newcomer, and rehearse spill cleanup. Spills get absorbed with inert materials and secured for hazardous waste pickup, never poured down the drain. Emergency eyewash and showers matter more with sodium azide around.
Smart Disposal Strategies
Old or unused sodium azide needs proper disposal; landfill or sink disposal risks both immediate and long-term harm. Partnering with a licensed chemical disposal company avoids accidents down the line. Containers must be leak-proof and labeled with both name and hazard. Waste managers can tell stories about unmarked containers found years later—unseen risks waiting for the unwary.
Why Strong Controls Matter
No sense in leaving safety by chance where sodium azide works. Training, clear labeling, smart purchasing, and strict inventory do more than meet regulations; they protect every person in the building. I learned early on: respect makes all the difference, and good habits shape safe labs. Missteps prove the need for discipline and knowledge. Sharing these lessons keeps dangerous surprises out of routine work, which saves lives.
What should I do in case of sodium azide spills or exposure?
Sodium Azide’s Dangers Aren’t Just On Paper
Sodium azide shows up in some places you might not expect: car airbags, research labs, and certain chemical manufacturing sites. Its notoriety comes from its poisonous, explosive character. Skin contact or inhaling powder dust doesn’t just cause mild irritation—it can bring on headaches, breathing problems, low blood pressure, and in serious cases, deadly effects. Toxicity ramps up fast, even in small amounts. These facts hit home when labs scramble during an accident; things escalate more quickly than most people anticipate.
Immediate Actions: No Room for Hesitation
Nobody gets ready for a spill by daydreaming about textbook protocols. The air in the room thickens with urgency. Grab a proper respirator with filters rated for toxic dust and gases. Use nitrile gloves and eye shields. Close off the area—don’t let more folks in. This isn’t about overreacting; sodium azide can release fatal gas if it reacts with water or acid. I’ve seen colleagues freeze up in the moment, and hesitation adds risk.
If the skin gets dusted or splashed, move straight to a sink, peel off contaminated clothes, and rinse the skin for 15 minutes. Anyone exposed to the eyes must flush with water for at least 15 minutes. Move outdoors to fresh air if dust clouds rise. Call emergency medical help right away, even if the exposed person claims to feel fine. Sodium azide doesn’t always show its worst effects at first.
Cleaning Up: Shortcuts Can Cost Lives
It feels inconvenient to vent the building and wait for a trained hazardous materials crew, but skipping steps can turn a mistake into a tragedy. Standard vacuum cleaners, brooms, or mops only spread dust into the air where it enters lungs. Specialized spill kits and absorbent materials help scoop powder carefully, keeping personal safety gear on the whole time. Never send azide down a drain—it can react with pipes and create explosive compounds. Dispose of contaminated clothing and cleanup materials following local hazardous waste rules, no exceptions.
Prevention: Training Over Assumptions
I’ve worked in science classrooms where nobody reviewed spill response for months, then watched panic break out at a tiny incident. Regular safety drills matter more than fancy equipment. Hang up clear signage near storage sites, and make safety showers and eyewash stations easy to find. Lock chemicals in sealed, labeled containers far from acids and water sources. Rely on your team—never handle sodium azide solo.
Proper chemical inventory systems keep surprises to a minimum. If a labmate stores sodium azide somewhere new, everyone should know before the next shift starts. Even veterans trip up without reminders; constant vigilance beats heroic rescue stories.
Looking Forward: Pushing for Safer Alternatives
Some institutions phase out sodium azide, swapping in safer chemicals for lab work and switching to next-generation airbag inflators in vehicles. That makes sense. Industry should keep searching for alternatives so fewer people risk exposure in the first place. Everyone deserves a safe workplace, no matter how rare large-scale spills seem.
Sodium azide insists on respect. Handling it takes serious planning, honest safety culture, and tough decisions about when to call in the experts. Every incident becomes a wake-up call that echoes through a lab or workshop for months. Preparation, courage to speak up, and good routines help everyone walk out healthy at the end of the day.
What are the proper disposal methods for sodium azide?
Why Disposing of Sodium Azide Matters
Sodium azide is a chemical that turns up in a lot of unexpected places, from vehicle airbags to some research labs. It’s cheap, effective, but it’s also dangerous if handled the wrong way. I remember working in a lab where a tiny, forgotten bottle of sodium azide in a dusty cabinet made everyone nervous. There’s good reason for that. If sodium azide finds its way into acid or metal plumbing, it can create explosive or toxic conditions.
This chemical can quickly become a public health problem. Small amounts dropped down the drain may seem innocent, but sodium azide forms hydrazoic acid in water, which is volatile—and potentially lethal. The drain runs into larger pipes, and that’s where things get hazardous. In my experience, nobody wants to be the reason the chemistry department gets evacuated.
Safe Storage as the First Step
Let’s talk about storage first. Keeping sodium azide dry and away from acids or heavy metals already cuts down most risks. Always label it clearly, and store it with compatible chemicals. Leaving it forgotten in a fridge or unmarked jar is asking for trouble, as I learned from a close call with some unlabelled containers.
How to Dispose of Sodium Azide Safely
The Environmental Protection Agency (EPA) shows the way for chemical disposal. The safest response is to ask a licensed chemical waste contractor to pick it up. Most universities and pharmaceutical labs use professional hazardous waste services. This keeps risk to a minimum and removes uncertainty. In my own lab days, calling professionals saved a lot of headache.
Neutralization can work for small amounts, but this is a job for trained chemists only. The typical process turns sodium azide into harmless nitrogen gas and sodium hydroxide by reaction with nitrous acid. But this reaction releases gases, and if done badly, it shoots out all sorts of toxic byproducts. Fume hoods, full personal protective equipment, and spill kits aren’t optional—they’re required. Sometimes, someone thinks using bleach is safer, but mixing bleach with sodium azide can create deadly gases.
Flushing down the drain, burning, or tossing it in the trash takes the problem from your hands and gives it to someone else. In my city, the utility workers have faced pipe explosions from improper disposal. That’s a strong lesson to avoid shortcuts.
Preventing Problems Before They Start
Training and awareness go a long way. New researchers or students must learn what makes sodium azide a high-risk chemical. Keeping inventories up-to-date stops old stock from being ignored. In some workplaces, audits help catch forgotten containers before they turn into a problem.
Laws and safety guidelines keep everyone safer. Following local, state, and federal rules reduces health risks for sanitation workers and first responders. Institutions that don’t enforce strict chemical protocols end up paying for it sooner or later, sometimes with serious injuries.
Better Solutions for Chemical Waste
Innovation in disposal is starting to make a difference. Some companies develop less hazardous alternatives to sodium azide or push for sealed, single-use systems. Until those take over, responsible management and professional disposal remain the only reliable answers.
Anyone handling sodium azide holds a bit of responsibility for public safety. Careful handling, up-to-date training, and quick reporting make a world of difference. The best solution is a community of educated users who look out for each other and respect the danger in that small bottle on the shelf.
| Names | |
| Preferred IUPAC name | Sodium azide |
| Other names |
Azide Sodide Sodium salt of hydrazoic acid Sodum azium NaN3 |
| Pronunciation | /ˈsəʊdiəm əˈzaɪd/ |
| Identifiers | |
| CAS Number | 26628-22-8 |
| Beilstein Reference | 358715 |
| ChEBI | CHEBI:26219 |
| ChEMBL | CHEMBL1367 |
| ChemSpider | 14252 |
| DrugBank | DB00778 |
| ECHA InfoCard | 100.011.134 |
| EC Number | 011-004-00-7 |
| Gmelin Reference | 604887 |
| KEGG | C01701 |
| MeSH | D019653 |
| PubChem CID | 33557 |
| RTECS number | VY8050000 |
| UNII | N3Z6X3HED1 |
| UN number | UN1687 |
| Properties | |
| Chemical formula | NaN₃ |
| Molar mass | 65.009 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | D 1.85 g/cm3 |
| Solubility in water | Soluble |
| log P | “-2.0” |
| Vapor pressure | Negligible |
| Acidity (pKa) | pKa ≈ 4.5 |
| Basicity (pKb) | 8.9 |
| Magnetic susceptibility (χ) | Paramagnetic |
| Refractive index (nD) | 1.513 |
| Viscosity | 20 cP (20°C) |
| Dipole moment | 1.85 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 149.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -21.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -22.7 kJ/mol |
| Pharmacology | |
| ATC code | V03AB38 |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled; contact with acids liberates very toxic gas; explosive under certain conditions; very toxic to aquatic life. |
| GHS labelling | GHS02, GHS03, GHS06, GHS09 |
| Pictograms | GHS01,GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H300 + H310 + H330: Fatal if swallowed, in contact with skin or if inhaled. H372: Causes damage to organs through prolonged or repeated exposure. H400: Very toxic to aquatic life. |
| Precautionary statements | P210, P220, P223, P264, P273, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P310, P312, P330, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-0-W |
| Autoignition temperature | 250 °C (482 °F) |
| Explosive limits | Explosive limits: 0.7–59% |
| Lethal dose or concentration | LD₅₀ (oral, rat): 27 mg/kg |
| LD50 (median dose) | LD50 (median dose): 27 mg/kg (oral, rat) |
| NIOSH | NA0166 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Sodium Azide: "0.3 mg/m³ (as NaN₃), ceiling |
| REL (Recommended) | 10 mg/m³ |
| IDLH (Immediate danger) | APF = 50 mg/m³ |
| Related compounds | |
| Related compounds |
Hydrazoic acid Lead azide Potassium azide |

