Sodium Arsenite: Chemical, History, and the Way Forward
Historical Development
Sodium arsenite holds a legacy as old as industrial chemistry itself. In the 19th century, factories, tanneries, and agricultural operations leaned heavily on arsenites as preservatives and pesticides. Early pharmacists even employed arsenical compounds in “tonic” formulations despite warnings about their dangers. Through the years, direct water pollution, environmental neglect, and industrial accidents shaped how society treats and studies sodium arsenite. Workers in textile factories and insecticide plants in the early 1900s often faced chronic poisoning due to lax controls, only fueling more stringent laws and industrial hygiene practices as the chemical age matured.
Product Overview
You run into sodium arsenite as a pale, crystalline powder or dissolved in solution. The chemical arises from reacting arsenic trioxide with sodium carbonate or sodium hydroxide, leaving a compound with real bite for both life and landscape. Beyond its murky legacy as a pesticide and wood preservative, sodium arsenite figures in labs as a reagent, stabilizer, and even as a model toxicant in research projects—an unfortunate testament to its staying power across applications.
Physical & Chemical Properties
Crystalline sodium arsenite takes a colorless or faintly colored form with a distinctly astringent taste. People in labs recognize it readily for its solubility in water and strong alkalinity. Volatility isn’t part of its nature, but its interaction with acids quickly liberates highly toxic arsine gas. This compound dissolves with speed, which partly explains the severity of environmental spills and groundwater contamination. On a molecular level, sodium arsenite builds on the AsO2- anion, which features a planar structure that pulls in water, affecting storage and handling. The molecular formula reads NaAsO2 for the most common hydrated forms.
Technical Specifications & Labeling
Proper labels on sodium arsenite remain a must. The chemical registers under several identifiers, including CAS number 7784-46-5. In the United States, federal agencies like OSHA, EPA, and the Department of Transportation wrap the compound in layers of regulation. Danger symbols for acute toxicity dominate its labeling, and safety data sheets go deep on first aid, handling precautions, reactive hazards, and spill containment. Containers almost always note UN Number 1685, hazard codes, and clear warnings for skin, respiratory, and environmental exposure.
Preparation Method
Manufacturing sodium arsenite demands precision. Start with arsenic trioxide, a notorious byproduct of copper and lead smelting. By adding sodium hydroxide, a simple acid-base reaction takes place, yielding sodium arsenite in solution with water as a by-product. Heating, filtration, and then crystallization leave a purified end product. Neither step is gentle—highly toxic fumes and corrosive reactants make every fragment of the process a workplace hazard, underscoring why plant operators rely on advanced air handling and sealed equipment today.
Chemical Reactions & Modifications
Sodium arsenite functions as a reducing agent in several organic transformations. Researchers see its value in shifting oxidation states, enabling those classic Marsh and Gutzeit tests for arsenic detection. Acids act fast on it, driving the release of stink-laden arsine gas. Chlorine, bromine, or oxidizing agents transform sodium arsenite with brisk oxidation to less soluble sodium arsenate, a pivot environmental chemists use in clean-up scenarios. Despite its age, the compound remains a workhorse in various synthesis and analytical pathways.
Synonyms & Product Names
Sodium arsenite crops up under a crowd of names, which can obscure hazards if not tracked closely. Chemists often refer to it as “disodium arsenite”, “arsenous acid, sodium salt”, or simply “NaAsO2”. Trade names pop up in older pesticide guides, carrying brand identities, but modern inventory relies on standardized nomenclature due to global regulation. Different names can signal formulation differences—liquid, crystalline, technical grade—so keeping a close eye on product data wades off costly or dangerous mix-ups.
Safety & Operational Standards
No shortcut exists for the safe management of sodium arsenite. Regulatory agencies demand glove use, goggles, and ventilation, all backed by strict workplace exposure limits. Personal experience in chemical labs reminds one how a single whiff or skin contact can cause burning, nausea, or worse—long-term exposure drives up rates of lung, bladder, and skin cancers. Rescue protocols, spill kits, and secure disposal routes keep workplaces safe, but only if people follow them rigorously. Workers train with dummy compounds and simulation gear before ever handling the real thing.
Application Area
Legacy applications painted sodium arsenite as a pesticide for tree trunks and insect infestations, plus as a wood preservative against rot and boring insects. Those uses faded after mounting evidence of water contamination and soil toxicity. In research, it finds a place today in testing antioxidant defense in cells, probing enzyme activity, or acting as a benchmark in toxicology studies. Environmental agencies track its fingerprints in contaminated groundwater, leveraging that data to push new remediation techniques or tighter discharge standards.
Research & Development
The story of sodium arsenite research stretches from pesticide impact studies to the molecular roots of toxicity. I have seen toxicologists apply it to cultured cells to map out how arsenic compounds disrupt proteins or incite DNA damage. Environmental chemists keep searching for rapid detection kits and more cost-effective remediation strategies—hoping to spot minute traces in soils or industrial outflows before the hazards bloom into a crisis. Breakthroughs in analytical chemistry regularly draw on sodium arsenite as a calibration or challenge material, maintaining the compound’s presence in high-tech labs.
Toxicity Research
Years of data paint a clear picture: sodium arsenite ranks as a potent carcinogen, mutagen, and acute toxin. Researchers confirm that even low levels trigger chronic diseases, ranging from vascular damage to neurological disorders. Its legacy in rural groundwater contamination in large swaths of Asia tells a sobering story. People and animals exposed over time suffer skin lesions, cancers, and developmental damage. Every step of modern research underlines the urgency of replacing or neutralizing arsenic compounds as quickly as science allows.
Future Prospects
Awareness of sodium arsenite’s dangers reshapes its place in industry and science. Regulatory pressure, public health mandates, and green chemistry innovations all push for non-arsenic alternatives in wood treatment, pest management, and laboratory processes. Environmental scientists focus on tighter monitoring, new filter technologies, and ways to permanently lock arsenic away from the food chain. As remediation methods improve—with better filtration, chelation, and bioremediation—society could see landscapes and communities bounce back from arsenic’s harsh legacy. The future trend pushes for maximum control, safer substitutes, and a robust stewardship ethos in every field where sodium arsenite ever played a role.
What is Sodium Arsenite used for?
Understanding Sodium Arsenite’s Role
Sodium arsenite stirs up all kinds of debates in science and industry. Brought up in chemistry classes, it comes with a dangerous reputation. This chemical, based on arsenic, is toxic to people and animals. Even so, it still finds work—mainly in places that don’t show up on most people’s radar.
Pesticide Practices in Agriculture
Older farming textbooks mention sodium arsenite as a popular choice for weed and insect control. It attacks weeds and fungus with a strong hand, wiping out what farmers don’t want in fruit orchards and cotton fields. While this made for healthier-looking crops, heavy use risked contaminating the soil and water. I grew up around farming towns, and old-timers still talk about “bad water” days where these poisons crept into wells, forcing everyone to haul water from far away or get sick. The world’s gotten wise to the health hazards, and sodium arsenite often lands on banned lists in many countries, including the US, but it hasn’t vanished everywhere.
Wood Preservation and Industrial Uses
Timber yards once used sodium arsenite to keep wood safe from termites and rot. Walk through certain sheds that never get torn down, and you might smell a weird tang in the damp corners—that’s chemical-soaked wood refusing to break down. Old railroad ties and poles that outlast the original builders sometimes owe their survival to this compound.
On the industrial side, sodium arsenite helps out in chemistry labs. Researchers use it to make other arsenic-based chemicals, and sometimes, companies tap into its harshness for glass making and pigment production. Because it’s dangerous and gets absorbed through skin or lungs, workers need layers of protection—goggles, gloves, and strict training. No shortcuts allowed when handling this stuff.
Public Health Concerns
The risks attached to sodium arsenite can’t be ignored. Even tiny exposures over time could spark headaches, stomach cramps, and skin problems, with bigger doses causing far worse. The World Health Organization, CDC, and EPA point to links between long-term arsenic exposure and cancers of the skin, lungs, and bladder. Families living near old farming ground still find arsenic traces in their soil and produce. Regular testing, cleanup, and safety warnings help lessen the dangers, but nobody can undo decades of careless dumping.
Better rules and stronger enforcement make a real difference. Disposal rules today won’t let a barrel of sodium arsenite get buried behind a shed. Workers require chemical training and medical checks. Everyday folks have more protection now than years ago, and scientific research keeps coming up with alternatives that don’t poison the earth or anyone living on it.
Looking for Safer Alternatives
Researchers and eco-focused companies are always on the hunt for gentler ways to protect crops or preserve wood. Biological pest controls, safe herbicides, and heat treatments get more attention than ever in agriculture and building. Community awareness and simple caution help out, too—anyone who stumbles on an old container knows to call in professionals. In the end, society keeps learning from the long shadow left by chemicals like sodium arsenite, figuring out better, safer paths forward.
Is Sodium Arsenite hazardous or toxic?
Looking Closer at What Sodium Arsenite Does
Sodium arsenite stands out for all the wrong reasons. Used mostly in laboratories, wood preservation, or as a pesticide, this chemical often flies under the radar unless something goes wrong. The trouble comes from its easy ability to poison living things, including humans, pets, and wildlife. One small mistake with storage or handling, and lives may hang in the balance.
Why Worry About Toxicity?
I’ve seen people talk about “toxicity” as if it’s just a warning label someone in an office slaps onto a bottle. That barely scratches the surface. Sodium arsenite packs a punch because it carries arsenic, a well-known carcinogen. The U.S. National Toxicology Program calls arsenic a proven cause of cancer. The World Health Organization places it high on their list of chemicals best kept away from drinking water and food.
Even low doses, if swallowed, inhaled, or absorbed through the skin, spell trouble. Symptoms start with headaches, stomach cramps, confusion, and vomiting. The real threat appears over time—arteries harden, nerves suffer, and cancer rates shoot up. Long-term exposure through water or air in a contaminated area can cause irreversible harm. In places where sodium arsenite has escaped into rivers or soil, the community feels it in rising illness rates and lost productivity.
Accidents Happen—But Some Risks Aren’t Worth Taking
You don’t have to look far into history to find arsenic horror stories. Workers in wood treatment plants once faced daily doses of sodium arsenite dust. Without good protection, rates of skin and lung cancer spiked. Towns near mines or factories saw arsenic leach into the groundwater. Wells poisoned generations of families.
Some people dismiss fears about chemicals, claiming regulations keep everything safe. My experience says otherwise. Regulations set a bar, but they can’t undo damage when someone cuts corners or skips the right gear. I’ve seen old labs and storage rooms where bottles leak or labels flake away. Contaminated soil around factories still poses a silent threat decades after the plant shuts down.
Better Ways Forward
We’ve got options, and they start with transparency. Communities deserve to know when sodium arsenite rests anywhere near their water or soil. Companies hold the responsibility to teach workers real-world dangers, not just point out a Material Safety Data Sheet. Using safer substances wherever possible also makes sense. Innovations in wood treatment and pest control have given us less toxic alternatives that work well without carrying the same lifelong risks.
Anyone who handles sodium arsenite should use gloves, goggles, and a properly-fitted mask every single time. Safe cleanup matters, too. Properly sealed containers, clear labels, and honest disposal practices prevent future headaches. Emergency plans mean little if workers aren’t involved or drills don’t happen. Feeling safe shouldn’t require luck—it should be baked into the system.
Relying on Facts and Experience
Science and history provide all the proof needed. Sodium arsenite is more than just “hazardous”—it’s an urgent public health concern. Real protection means learning from hard lessons and refusing to accept shortcuts. Informed communities and workplaces lead to better choices, safer families, and fewer regrets.
How should Sodium Arsenite be stored?
Understanding the Risks
Long before anyone picks up a bottle labeled “sodium arsenite,” the dangers are well-known. Sodium arsenite carries a heavy reputation for toxicity, and even small mistakes in storing this chemical have led to real harm. In labs and factories across the world, a moment of carelessness has poisoned water, injured workers, or sparked investigations. It isn’t dramatic to say that an ounce of attention can mean the difference between routine safety and disaster.
Simple Steps, Serious Protection
Nobody wants a spill to cost someone their health or life, so strict storage protocols aren’t negotiable. Sodium arsenite should always go in a secure chemical storage cabinet. That cabinet must lock, not just to keep out casual snoopers but also to stay out of the hands of janitorial staff and newcomers unfamiliar with its dangers. The best storage areas stay dry and cool. Heat, sunlight, and moisture break down containers, or worse, spark a chain reaction that could release toxic fumes.
Too often, someone can end up storing chemicals alphabetically or for convenience. That approach backfires when one substance, like sodium arsenite, can react violently with oxidizers, acids, or other incompatible chemicals. Segregation on dedicated shelves, with clear labels that wear well over time, helps prevent these deadly mix-ups. Years spent in research labs have shown that a well-organized chemical cabinet—a few extra minutes spent sorting bottles—prevents headaches and safety incidents down the line.
Ventilation and Containment—Non-Negotiables
Stale air leads to dangerous buildups. Sometimes, the smell of a chemical can give away a problem, but sodium arsenite doesn’t always warn before causing harm. Proper ventilation is critical. Fume hoods and extraction systems take away airborne particles or vapors, lowering risks for everyone in the workspace. Chemical safety experts have documented cases where a poorly ventilated room turned a small spill into a health emergency.
Containers do plenty of heavy lifting. Sodium arsenite belongs in strong, corrosion-resistant bottles with tightly fitting lids. Glass or high-quality plastic will do the job, but damaged or unmarked containers invite confusion or leaks. Inspections by trained staff—monthly or quarterly—not only spot defects but also keep outdated chemicals out of circulation. A fresh set of eyes on an old shelf can pick up that missing label or chipped lid just in time.
Training and Emergency Readiness
Training helps people know what to do long before a crisis arrives. Lessons learned in chemical hygiene training stick. In practice, clear rules make everyone’s life safer—from the seasoned technician to the new recruit. Written protocols, checked during walkthroughs, keep old habits from slipping through the cracks.
Nearby spill kits, stocked with gloves, protective goggles, and neutralizing agents, show a workplace takes threats seriously. Fast response limits exposure. Working for years in shared spaces, it’s become clear that teams who drill emergency procedures suffer fewer mishaps. A written plan, posted where everyone can see, saves critical seconds when things go wrong.
Accountability and Oversight
Real oversight means someone checks not just the inventory but also the condition and organization of the entire chemical storage setup. This check links to regulatory compliance. Global health agencies, from the CDC to the WHO, outline strict guidelines for handling toxins like sodium arsenite. Regular audits don’t just tick boxes—they offer a chance to learn from mistakes and improve every year.
What precautions should be taken while handling Sodium Arsenite?
Understanding the Risks
Sodium arsenite shows up as a white, odorless powder or in a crystalline form, and it's long been used in labs, wood preservation, and even as a pesticide in the past. Most folks have heard arsenic isn’t harmless—sodium arsenite takes that reputation up a notch due to how easily it can enter the body. Skin contact, accidental ingestion, and inhaling the dust risk real and lasting harm. Exposure can lead to skin lesions, neurological issues, and increases the risk for cancers, especially if contact drags on or repeats. The World Health Organization, CDC, and numerous researchers link even tiny exposures to negative health effects. I once watched a coworker learn this the hard way—he worked an afternoon without real protective gear, and just that short exposure left him sick for a week.
Putting Safety Gear Front and Center
Before opening a container, eye protection and gloves need to come out. Standard cotton gloves won’t give enough assurance—nitrile or neoprene work better. Long-sleeved lab coats help keep dust off the skin. Respirators fitted with filters tested for particulates and chemicals block invisible dust from entering the lungs. Goggles provide a shield in case powder flakes up. Workers should never handle sodium arsenite alone—another person nearby means fast help in case of an accident. This careful approach isn’t just about avoiding fines, it’s about keeping people healthy and confident that they’ll go home safe.
Work Habits That Make a Difference
It’s all too easy to get comfortable—leaving open containers on the bench, skipping a hand wash because it’s just a “quick measurement.” But small mistakes build up. Only open or transfer sodium arsenite in a fume hood or ventilated area. This keeps dust and fumes away from the breathing zone. Keep the workspace free from clutter; loose paper and old gloves can collect powder and become a hidden hazard. If something spills, don’t reach for the broom. Wet methods—like damp paper towels—contain the substance instead of spreading it. I’ve seen folks try to shake off powdery gloves over a trash can, and that’s exactly how workplace contamination starts. Always double-bag waste and label it clearly for hazardous disposal.
Health Checks and Training: Continuing Responsibility
Relying on a one-time safety briefing isn’t enough. Regular safety drills and refreshers keep good habits sharp. Personal monitoring, like urine or blood tests, picks up early warning signs before anyone notices symptoms. A trusted building manager or safety officer should check storage and signage regularly. Solutions don’t end at the lab or industrial floor—public education matters as well. In smaller or older facilities, sodium arsenite can still turn up in forgotten corners. Ongoing surveys help stop unintentional exposure.
Reducing Risk with Good Planning
Clear labeling and up-to-date safety data sheets give everyone a fighting chance to act wisely. Spills and mistakes happen, but emergency response plans—real, practical, and tested—focus the team and reduce panic. Handwashing stations, emergency eye washes, and showers placed nearby make all the difference in a bad moment. People who feel empowered to ask questions and stop unsafe work signal a site where health truly comes first. Open conversations about risks, new findings, and close calls keep the spirit of shared responsibility alive, and that’s what turns rules into lasting safety.
What is the chemical formula and appearance of Sodium Arsenite?
Chemical Formula and Structure
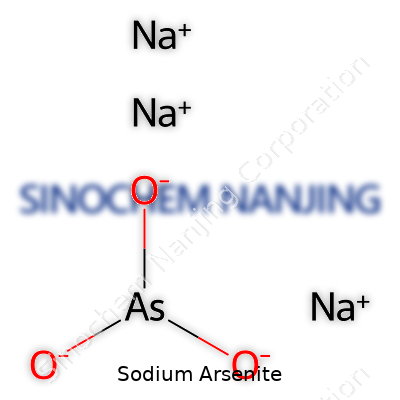
Sodium arsenite stands out with the formula NaAsO2. Created by reacting arsenic trioxide with sodium hydroxide, the compound contains sodium, arsenic, and oxygen. Looking at its molecular structure, sodium bonds with arsenic that connects to two oxygen atoms, forming an inorganic salt. The arrangement reveals a lot about its reactivity and reasons authorities keep a close eye on its use.
Physical Appearance
People working in labs instantly recognize sodium arsenite from its appearance. The commercial forms usually turn up as colorless to slightly grayish solids or as a white powder. Sometimes, it appears as tiny crystalline granules—think of sugar dust that has a duller hue and an off-white, gray edge. Its sharp, acrid smell offers a hint of danger; nobody mistakes it for household powder. Solubility in water remains high; when mixing, it dissolves swiftly and creates a clear solution, showing just how responsive the compound feels even at room temperature.
Why Knowing This Matters
Sodium arsenite doesn't just fill a page in a textbook. It's part of daily life in fields like metallurgy, pest control, and even glass manufacturing. Some old-school wood preservation methods used this compound, but modern regulations now limit these uses sharply due to the compound’s toxicity. The arsenic ion inside the molecule raises immediate health flags. Arsenic, throughout history, has gained a dark reputation. Prolonged contact with sodium arsenite can harm skin, eyes, lungs, and even cause systemic poisoning.
Having spent time working in an industrial chemistry environment, I learned how even a trace of sodium arsenite puts safety protocols on high alert. Chemists suit up with thick gloves, goggles, and breathing masks far before measuring or pouring. Training covers more than just wearing the gear; it drills in what happens if you miss a step. One exposure incident quickly becomes a health emergency, not just for one worker but for those nearby.
Toxicity and Human Impact
Many sources, including the International Agency for Research on Cancer (IARC), classify sodium arsenite as carcinogenic. Research confirms that exposure increases the risk of skin, lung, and other internal organ cancers. Water-soluble properties make accidental introduction into water supplies a public health crisis. A town in Bangladesh once experienced mass arsenic poisoning, not from sodium arsenite only, but the incident highlighted how easily compounds with high arsenic content can disrupt communities.
Reducing the Risks
Strict storage and handling practices require sealed, labeled containers and full personal protective equipment. Spill response means instant isolation of the area and careful neutralization or cleanup. Disposal always follows hazardous waste protocols, separating this arsenical compound from general waste streams.
Switching to less toxic alternatives for applications such as wood preservation or pesticides cuts exposure risk. Regulatory boards, including the EPA, continue to restrict and monitor its legitimate uses. In workplaces, continuous training, air monitoring, and medical surveillance programs help prevent tragic incidents.
Final Thought
Sodium arsenite’s formula and appearance alone never tell its full story. It represents the lines where science, industry, and public safety intersect. Understanding this compound—down to how it looks on the lab bench—reminds us why chemistry always needs respect and vigilance, not just curiosity.
| Names | |
| Preferred IUPAC name | disodium arsenite |
| Other names |
Arsenious acid, sodium salt Sodium metaarsenite Sodium arsenite (NaAsO2) Sodium arsenous acid Sodium arsenite solution |
| Pronunciation | /ˈsəʊdiəm ɑːˈsɛnaɪt/ |
| Identifiers | |
| CAS Number | 7784-46-5 |
| Beilstein Reference | 4038907 |
| ChEBI | CHEBI:28665 |
| ChEMBL | CHEMBL135907 |
| ChemSpider | 21506 |
| DrugBank | DB01394 |
| ECHA InfoCard | 100.058.006 |
| EC Number | 215-481-4 |
| Gmelin Reference | 68277 |
| KEGG | C13923 |
| MeSH | D013785 |
| PubChem CID | 5359597 |
| RTECS number | CN6475000 |
| UNII | 7U1EEY6H5Q |
| UN number | UN1871 |
| Properties | |
| Chemical formula | NaAsO2 |
| Molar mass | 129.91 g/mol |
| Appearance | White solid |
| Odor | Odorless |
| Density | 2.5 g/cm³ |
| Solubility in water | Soluble |
| log P | -4.1 |
| Acidity (pKa) | pKa ~ 9.0 |
| Basicity (pKb) | 8.0 |
| Magnetic susceptibility (χ) | -26.8e-6 cm³/mol |
| Refractive index (nD) | 1.520 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 146.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -703.4 kJ/mol |
| Pharmacology | |
| ATC code | V03AB03 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; causes severe skin burns and eye damage; may cause cancer; very toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS05, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H300 + H310 + H330, H372, H410 |
| Precautionary statements | P201, P202, P260, P264, P270, P273, P280, P301+P310, P302+P352, P304+P340, P308+P313, P310, P321, P330, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-3-W |
| Lethal dose or concentration | LD50 oral rat 15 mg/kg |
| LD50 (median dose) | 15 mg/kg (oral, rat) |
| NIOSH | CY1400000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Sodium Arsenite: 0.01 mg/m³ |
| REL (Recommended) | 10 mg/m3 |
| IDLH (Immediate danger) | 5 mg/m3 |
| Related compounds | |
| Related compounds |
Arsenic trioxide Sodium arsenate Potassium arsenite Arsenous acid |

